MCAT General Chemistry Review
Chapter 2: The Periodic Table
2.2 Types of Elements
When we consider the trends of chemical reactivity and physical properties together, we can begin to identify groups of elements with similar characteristics. These larger collections are divided into three categories: metals, nonmetals, and metalloids (also called semimetals).
METALS
Metals are found on the left side and in the middle of the Periodic Table. They include the active metals, the transition metals, and the lanthanide and actinide series of elements. Metals are lustrous (shiny) solids, except for mercury, which is a liquid under standard conditions. They generally have high melting points and densities, but there are exceptions, such as lithium, which has a density about half that of water. Metals have the ability to be deformed without breaking; the ability of metal to be hammered into shapes is called malleability, and its ability to be pulled or drawn into wires is called ductility. At the atomic level, a metal is defined by a low effective nuclear charge, low electronegativity (high electropositivity), large atomic radius, small ionic radius, and low ionization energy. All of these characteristics are manifestations of the ability of metals to easily give up electrons.
Many of the transition metals (Group B elements) have two or more oxidation states (charges when forming bonds with other atoms). Because the valence electrons of all metals are only loosely held to their atoms, they are free to move, which makes metals good conductors of heat and electricity. The valence electrons of the active metals are found in the s subshell; those of the transition metals are found in the d subshell; and those of the lanthanide and actinide series elements are in the f subshell. Some transition metals—copper, nickel, silver, gold, palladium, and platinum—are relatively nonreactive, a property that makes them ideal for the production of coins and jewelry.
KEY CONCEPT
Alkali and alkaline earth metals are both metallic in nature because they easily lose electrons from the s subshell of their valence shells.
An example of a metal is shown in Figure 2.1 with an indium wire. The wire exhibits luster, malleability, and ductility. It is used as a wire because it also exhibits good heat and electrical conductivity.
 Figure 2.1. Indium (In) Metal Wire
Figure 2.1. Indium (In) Metal Wire
NONMETALS
Nonmetals are found predominantly on the upper right side of the Periodic Table. Nonmetals are generally brittle in the solid state and show little or no metallic luster. They have high ionization energies, electron affinities, and electronegativities, as well as small atomic radii and large ionic radii. They are usually poor conductors of heat and electricity. All of these characteristics are manifestations of the inability of nonmetals to easily give up electrons. Nonmetals are less unified in their chemical and physical properties than the metals.
Carbon, shown in Figure 2.2, is a stereotypical nonmetal that retains a solid structure but is brittle, nonlustrous, and generally a poor conductor of heat and electricity.
 Figure 2.2. Activated Charcoal, Composed of the Nonmetal Carbon (C)
Figure 2.2. Activated Charcoal, Composed of the Nonmetal Carbon (C)
METALLOIDS
Separating the metals and nonmetals are a stair-step group of elements called the metalloids. The metalloids are also called semimetals because they share some characteristics with both metals and nonmetals. The electronegativities and ionization energies of the metalloids lie between those of metals and nonmetals. Their physical properties—densities, melting points, and boiling points—vary widely and can be combinations of metallic and nonmetallic characteristics. For example, silicon (Si) has a metallic luster but is brittle and a poor conductor. The reactivities of the metalloids are dependent on the elements with which they are reacting. Boron (B), for example, behaves like a nonmetal when reacting with sodium (Na) and like a metal when reacting with fluorine (F). The elements classified as metalloids form a “staircase” on the Periodic Table and include boron, silicon, germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), polonium (Po), and astatine (At). While there is debate over polonium and astatine’s status as metalloids, most sources (including the MCAT) will label them as such. Figure 2.3 color-codes the major classifications of elements on the Periodic Table.
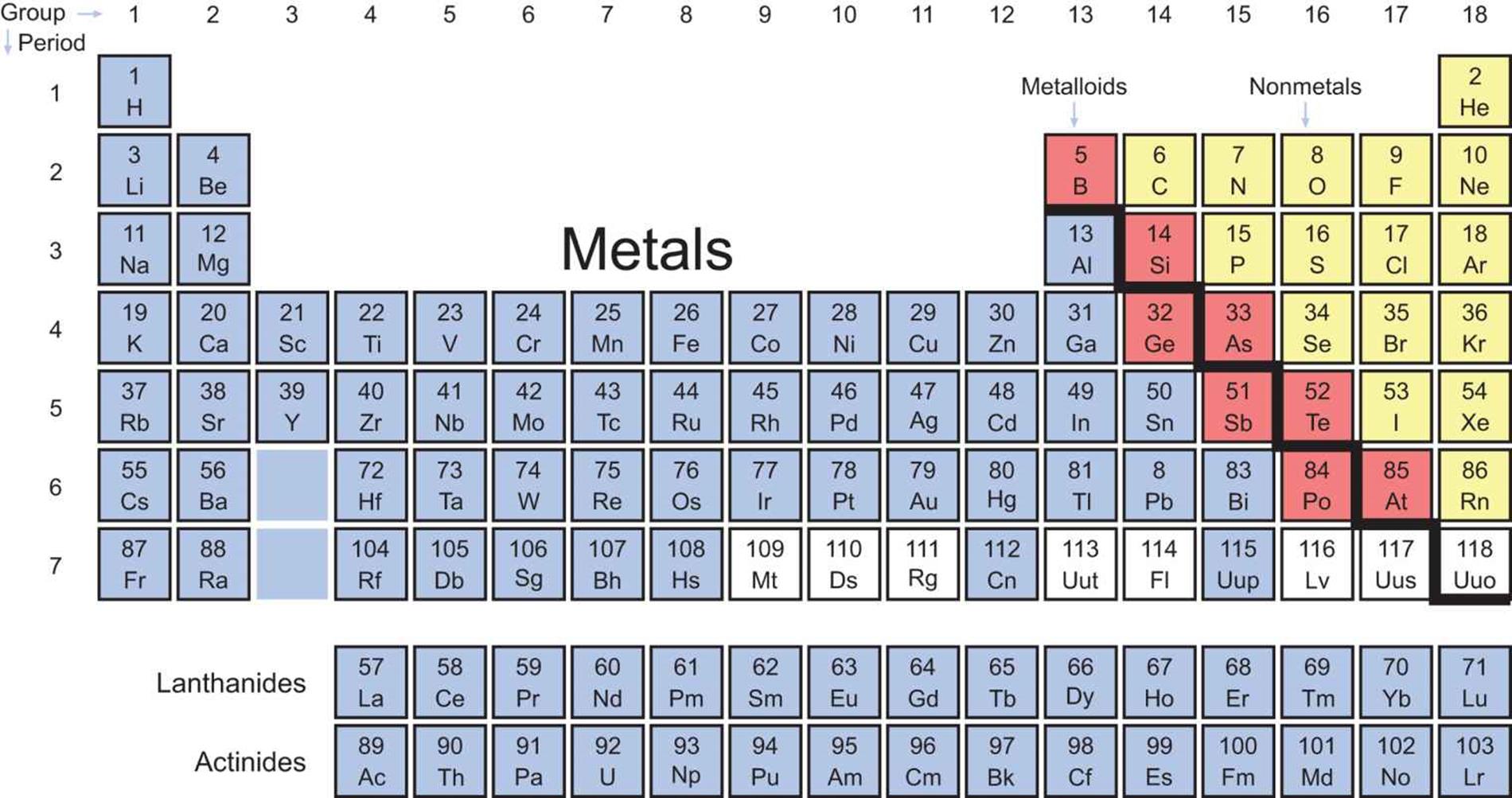 Figure 2.3. Periodic Table, Coded by Element Type
Figure 2.3. Periodic Table, Coded by Element Type
REAL WORLD
Metalloids share some properties with metals and others with nonmetals. For instance, metalloids make good semiconductors due to their partial conductivity of electricity.
MCAT Concept Check 2.2:
Before you move on, assess your understanding of the material with these questions.
1. Based on their location in the Periodic Table, identify a few elements that likely possess the following properties:
· Luster:
· Poor conductivity of heat and electricity:
· Good conductivity but brittle:
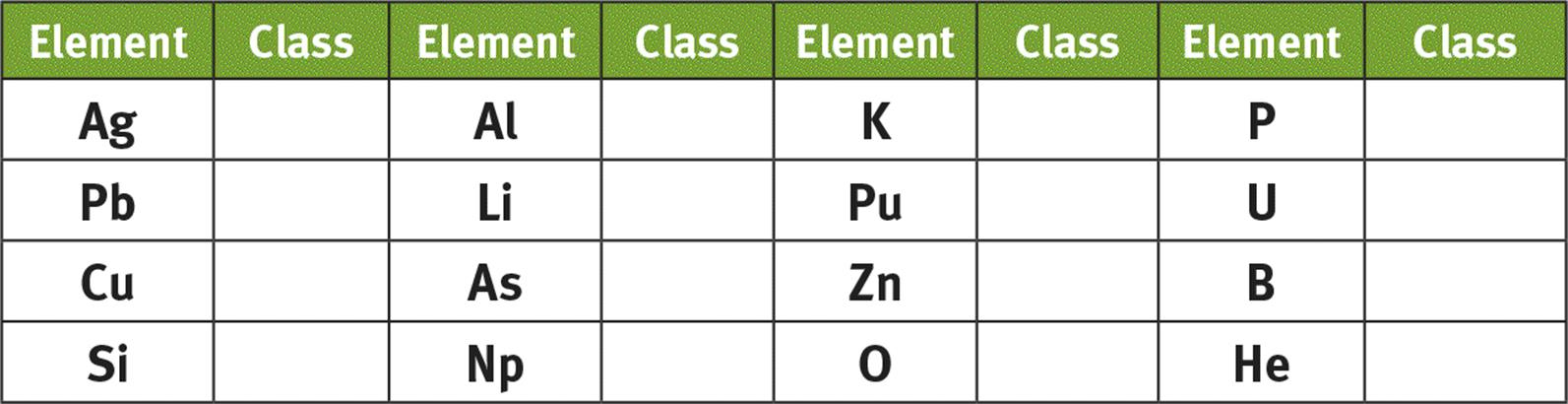
2. Classify the following elements as metals (M), nonmetals (NM), or metalloids (MO):