MCAT General Chemistry Review
Chapter 9: Solutions
Conclusion
Our review of solution chemistry has provided an opportunity for us to consider the nature of solutions, solutes, and solvents, and the interactions between solutes and solvents in the formation of solutions. We reviewed solubility and the rules that reflect the solubility of common compounds in water. The different ways of expressing the amount of solute in solution were identified, and examples were given for each unit of concentration, including percent composition, mole fraction, molarity, molality, and normality. Next, we reviewed the thermodynamic principles of solution equilibria and defined unsaturated, saturated, and supersaturated solutions with respect to ion product (IP) and solubility product constant (Ksp). Subsequently, we discussed the common ion effect from the perspective of Le Châtelier’s principle for a solution at equilibrium. And finally, we examined the colligative properties of solutions and the mathematics that govern them. The colligative properties—vapor pressure depression, boiling point elevation, freezing point depression, and osmotic pressure—are physical properties of solutions that depend on the concentration of dissolved particles but not on their chemical identity.
Concept Summary
Nature of Solutions
· Solutions are homogenous mixtures composed of two or more substances.
o They combine to form a single phase, generally the liquid phase.
o Solvent particles surround solute particles via electrostatic interactions in a process called solvation or dissolution.
o Aqueous solutions are most important for the MCAT; solvation in water can also be called hydration.
o Most dissolutions are endothermic, although the dissolution of gas into liquid is exothermic.
· Solubility is the maximum amount of a solute that can be dissolved in a given solvent at a given temperature; it is often expressed as molar solubility—the molarity of the solute at saturation.
· Complex ions or coordination compounds are composed of metallic ions bound to various neutral compounds and anions, referred to as ligands.
o Formation of complex ions increases the solubility of otherwise insoluble ions (the opposite of the common ion effect).
o The process of forming a complex ion involves electron pair donors and electron pair acceptors such as those seen in coordinate covalent bonding.
Concentration
· There are many ways of expressing concentration.
o Percent composition by mass (mass of solute per mass of solution times 100%) is used for aqueous solutions and solid-in-solid solutions.
o The mole fraction (moles of solute per total moles) is used for calculating vapor pressure depression and partial pressures of gases in a system.
o Molarity (moles of solute per liters of solution) is the most common unit for concentration and is used for rate laws, the law of mass action, osmotic pressure, pH and pOH, and the Nernst equation.
o Molality (moles of solute per kilograms of solvent) is used for boiling point elevation and freezing point depression.
o Normality (number of equivalents per liters of solution) is the molarity of the species of interest and is used for acid–base and oxidation–reduction reactions.
Solution Equilibria
· Saturated solutions are in equilibrium at that particular temperature.
· The solubility product constant (Ksp) is simply the equilibrium constant for a dissolution reaction.
· Comparison of the ion product (IP) to Ksp determines the level of saturation and behavior of the solution:
o IP < Ksp: the solution is unsaturated, and if more solute is added, it will dissolve
o IP = Ksp: the solution is saturated (at equilibrium), and there will be no change in concentrations
o IP > Ksp: the solution is supersaturated, and a precipitate will form
· Formation of a complex ion in solution greatly increases solubility.
o The formation or stability constant (Kf) is the equilibrium constant for complex formation. Its value is usually much greater than Ksp.
o The formation of a complex increases the solubility of other salts containing the same ions because it uses up the products of those dissolution reactions, shifting the equilibrium to the right (the opposite of the common ion effect).
o The common ion effect decreases the solubility of a compound in a solution that already contains one of the ions in the compound. The presence of that ion in solution shifts the dissolution reaction to the left, decreasing its dissociation.
Colligative Properties
· Colligative properties are physical properties of solutions that depend on the concentration of dissolved particles but not on their chemical identity.
· Vapor pressure depression follows Raoult’s law.
o The presence of other solutes decreases the evaporation rate of a solvent without affecting its condensation rate, thus decreasing its vapor pressure.
o Vapor pressure depression also explains boiling point elevation—as the vapor pressure decreases, the temperature (energy) required to boil the liquid must be raised.
· Freezing point depression and boiling point elevation are shifts in the phase equilibria dependent on the molality of the solution.
· Osmotic pressure is primarily dependent on the molarity of the solution.
· For solutes that dissociate, the van ’t Hoff factor (i) is used in freezing point depression, boiling point elevation, and osmotic pressure calculations.
Answers to Concept Checks
· 9.1
1. Solvation refers to the breaking of intermolecular forces between solute particles and between solvent particles, with formation of intermolecular forces between solute and solvent particles. In an aqueous solution, water is the solvent.
2. Solubility is the amount of solute contained in a solvent. Saturation refers to the maximum solubility of a compound at a given temperature; one cannot dissolve any more of the solute just by adding more at this temperature.
3. Solubility of solids can be increased by increasing temperature. Solubility of gases can be increased by decreasing temperature or increasing the partial pressure of the gas above the solvent (Henry’s law).
4. Group I metals, ammonium, nitrate, and acetate salts are always soluble.
· 9.2
1. 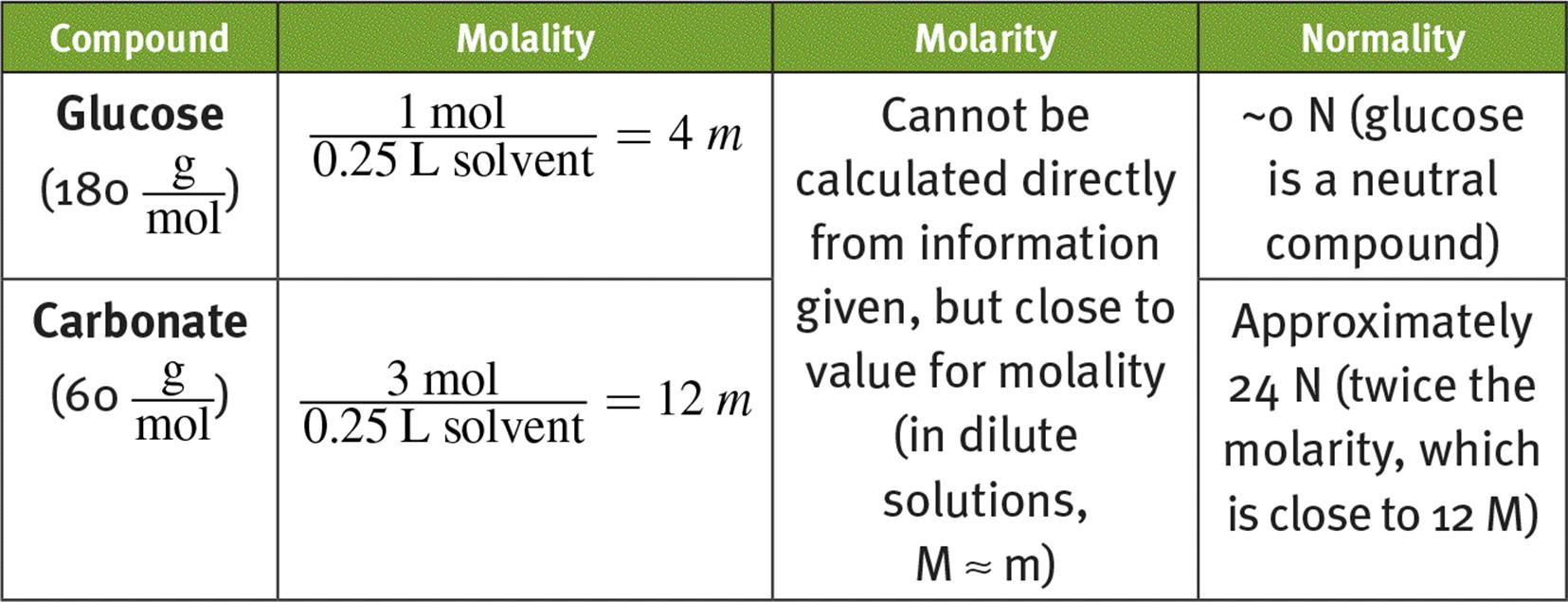
2. 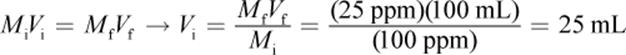
Thus, start with 25 mL of the stock solution and add 75 mL pure water to get 100 mL of solution with 25 ppm Cl2.
3. 
· 9.3
1. Ksp = [Ni2+][OH−]2 = (5.2 × 10−6 M)(10.4 × 10−6 M)2 ≈ (5 × 10−6)(100 × 10−12) = 500 × 10−18 = 5 × 10−16 (actual = 5.6 × 10−16).
2.
|
[Ba2+] |
Ion Product |
Behavior of Solution |
|
0.5 M |
(0.5 M)(1 M)2 = 0.5 |
0.5 > 5.0 × 10−3 → precipitation |
|
0.1 M |
(0.1 M)(0.2 M)2 = 4.0 × 10−3 |
4.0 × 10−3 < 5.0 × 10−3 → dissolution |
|
0.05 M |
(0.005 M)(0.1 M)2 = 5.0 × 10−4 |
5.0 × 10−4 < 5.0 × 10−3 → dissolution |
3. Note that the concentration of hydroxide is double that of barium, assuming Ba(OH)2 was the only salt added to solution. While there will be a very small contribution of hydroxide from the autoionization of water, this amount is negligible compared to the values given in the question.
3. Ksp = [Fe2+][OH−]2 = (x)(0.1 + 2x)2. x is almost always negligible in addition or subtraction, so the math simplifies to Ksp = (x)(0.1)2. Thus, 1.8 × 10−15 = 0.01x. x = molar solubility = 1.8 × 10−13.
· 9.4
1. Colligative properties are those that depend on the amount of solute present, but not the actual identity of the solute particles. Examples include vapor pressure depression, boiling point elevation, freezing point depression, and osmotic pressure.
2. Molarity (M) and molality (m) are nearly equal at room temperature. This is only because 1 L solution is approximately equal to 1 kg solvent for dilute solutions (the denominators of the molarity and molality equations, respectively). For other solvents, molarity and molality differ significantly because their densities are not ![]() like water.
like water.
3. 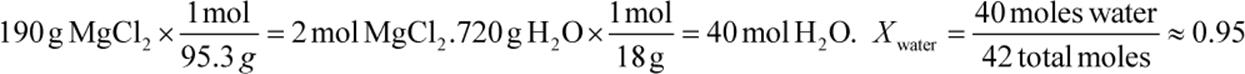
Pwater = Xwater P°water = (0.95)(3.2) ≈ 3.0 kPa (actual = 3.05 kPa)
4. 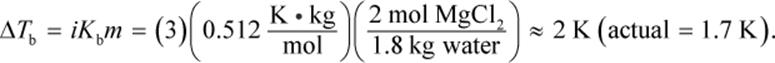
The new boiling point will be 373 + 2 = 375 K.
5. 
Equations to Remember
(9.1) Percent composition by mass:
![]()
(9.2) Mole fraction:

(9.3) Molarity:
![]()
(9.4) Molality:
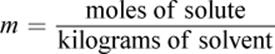
(9.5) Dilution formula: MiVi = MfVf
(9.6) Solubility product constant: Ksp = [An+]m[Bm−]n
(9.7) Ion product: IP = [An+]m[Bm−]n
(9.8) Raoult’s law (vapor pressure depression): PA = XA P°A
(9.9) Boiling point elevation: ΔTb = iKbm
(9.10) Freezing point depression: ΔTf = iKfm
(9.11) Osmotic pressure: ∏ = iMRT
Shared Concepts
· Biology Chapter 10
o Homeostasis
· General Chemistry Chapter 3
o Bonding and Chemical Interactions
· General Chemistry Chapter 6
o Equilibrium
· General Chemistry Chapter 7
o Thermochemistry
· General Chemistry Chapter 10
o Acids and Bases
· General Chemistry Chapter 12
o Electrochemistry