MCAT General Chemistry Review
Chapter 10: Acids and Bases
10.1 Definitions
Over the last century, chemists have used different definitions to identify compounds as acids or bases. Three definitions emerged, with each more inclusive than the former one.
ARRHENIUS
The first and most specific definition of an acid or base is the Arrhenius definition. An Arrhenius acid will dissociate to form an excess of H+ in solution, and an Arrhenius base will dissociate to form an excess of OH− in solution. These behaviors are generally limited to aqueous acids and bases. Arrhenius acids and bases are easily identified; acids contain H at the beginning of their formula (HCl, HNO3, H2SO4, and so on) and bases contain OH at the end of their formula (NaOH, Ca(OH)2, Fe(OH)3, and so on).
MCAT EXPERTISE
Any mention of the Arrhenius definition on Test Day will likely be in comparison to other definitions of acids. The Arrhenius definition is by far the most restrictive; the Brønsted–Lowry and Lewis definitions predominate on the MCAT.
BRØNSTED–LOWRY
A more inclusive definition of acids and bases was proposed independently by Johannes Brønsted and Thomas Lowry in 1923. A Brønsted–Lowry acid is a species that donates hydrogen ions (H+), while a Brønsted–Lowry base is a species that accepts them.
The advantage of this definition over Arrhenius’s is that it is not limited to aqueous solutions. For example, OH−, NH3, and F− are all Brønsted–Lowry bases because each has the ability to accept a hydrogen ion. However, neither NH3 nor F− can be classified as Arrhenius bases because they do not dissociate to produce an excess of OH− ions in aqueous solutions. According to both of these definitions, there is only one way for a species to be an acid: producing hydrogen ions. The only differences between the two definitions for acids are the requirement of an aqueous medium in the Arrhenius definition and the acidity of water. In the Arrhenius definition, water is not considered an acid—it does not produce an excess of H+ in solution. Water is, on the other hand, a Brønsted–Lowry acid because it is able to donate a proton to other species. Most acid–base chemistry reactions on the MCAT will involve the transfer of hydrogen ions in accordance with the Brønsted–Lowry definitions.
MCAT EXPERTISE
Every Arrhenius acid (or base) can also be classified as a Brønsted–Lowry acid (or base). Every Brønsted–Lowry acid (or base) can also be classified as a Lewis acid (or base). This logic does not always work the other way (for example, NH3 is a Brønsted–Lowry base, but not an Arrhenius base).
Brønsted–Lowry acids and bases always occur in pairs because the definitions require the transfer of a proton from the acid to the base. These are conjugate acid–base pairs, as described in the next section. For example, in the autoionization of water, H3O+ is the conjugate acid and OH− is the conjugate base, as shown in Figure 10.1.
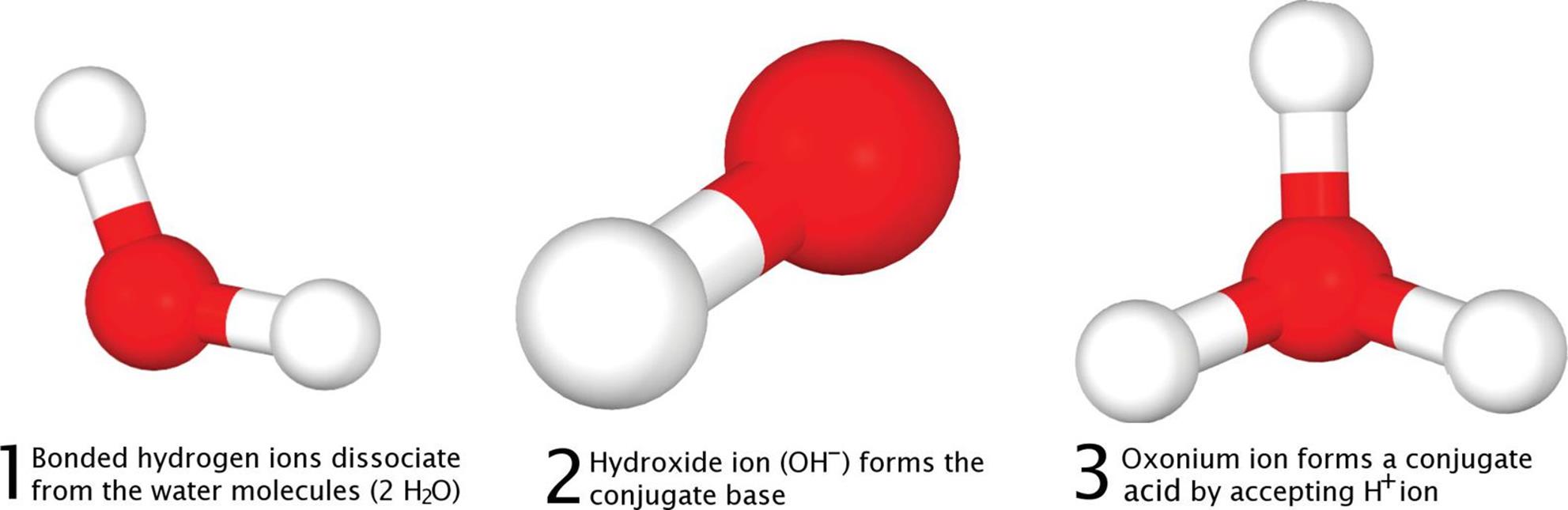 Figure 10.1. Autoionization of Water into Its Conjugate Acid and Conjugate Base The hydroxide ion is the conjugate base; the oxonium (hydronium) ion is the conjugate acid. H2O (l) + H2O (l) ⇌ H3O+ (aq) + OH− (aq)
Figure 10.1. Autoionization of Water into Its Conjugate Acid and Conjugate Base The hydroxide ion is the conjugate base; the oxonium (hydronium) ion is the conjugate acid. H2O (l) + H2O (l) ⇌ H3O+ (aq) + OH− (aq)
LEWIS
At approximately the same time as Brønsted and Lowry’s publications, Gilbert Lewis also proposed a definition for acids and bases. A Lewis acid is defined as an electron pair acceptor, and a Lewis base is defined as an electron pair donor, as shown in Figure 10.2. The electron pair being donated is a lone pair and is not involved in any other bonds.
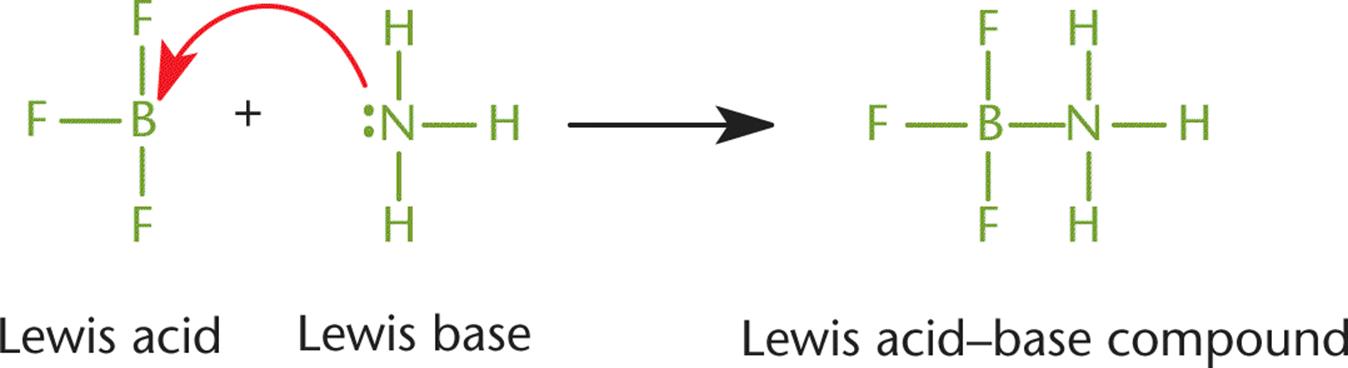 Figure 10.2. Lewis Acid–Base Chemistry Boron trifluoride serves as the Lewis acid, accepting a lone pair. Ammonia serves as a Lewis base, donating a lone pair.
Figure 10.2. Lewis Acid–Base Chemistry Boron trifluoride serves as the Lewis acid, accepting a lone pair. Ammonia serves as a Lewis base, donating a lone pair.
MNEMONIC
A Lewis acid is an acceptor of electron pairs.
On the MCAT, Lewis acid–base chemistry appears with many names. The underlying idea is that one species pushes a lone pair to form a bond with another. This same chemistry can be called coordinate covalent bond formation (discussed in Chapter 3 of MCAT General Chemistry Review), complex ion formation (discussed in Chapter 9 of MCAT General Chemistry Review), or nucleophile–electrophile interactions (discussed in Chapter 4 of MCAT Organic Chemistry Review).
There is an intuitive approach to understanding the differences in the definitions we have discussed so far. The Lewis definition relies on a behavior that is not vastly different from the Brønsted–Lowry interactions—the only difference is the focus. For Brønsted–Lowry acids and bases, we follow the exchange of the hydrogen ion (H+), which is essentially a naked proton. In the Lewis definition, the focus of the reaction is no longer on the proton, but instead the electrons forming the coordinate covalent bond. This difference can be seen using curved arrows, as shown in Figure 10.3.
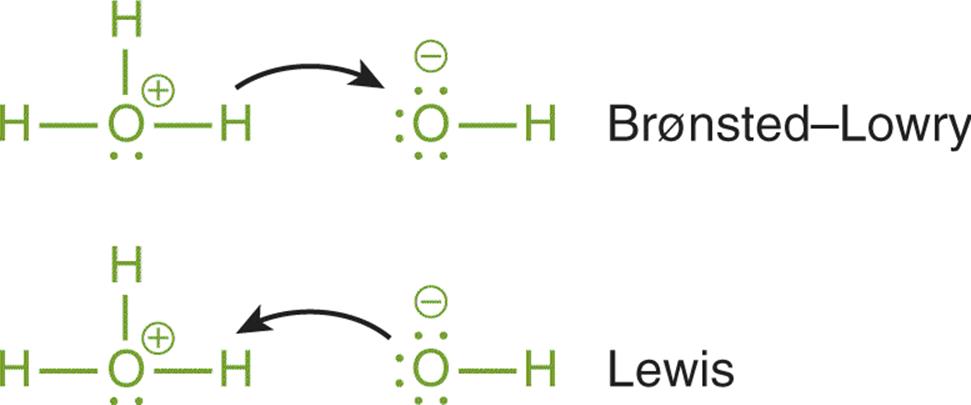 Figure 10.3. Comparison of Brønsted–Lowry and Lewis Definitions of Acids and Bases In the Brønsted–Lowry definition, the focus is on the transfer of the proton. In the Lewis definition, the focus is on the attack of the Lewis acid (electrophile) by the lone pair of the Lewis base (nucleophile).
Figure 10.3. Comparison of Brønsted–Lowry and Lewis Definitions of Acids and Bases In the Brønsted–Lowry definition, the focus is on the transfer of the proton. In the Lewis definition, the focus is on the attack of the Lewis acid (electrophile) by the lone pair of the Lewis base (nucleophile).
Note that the Lewis definition is the most inclusive: every Arrhenius acid is also a Brønsted–Lowry acid, and every Brønsted–Lowry acid is also a Lewis acid (and likewise for bases). However, the converse is not necessarily true. The Lewis definition encompasses some species not included within the Brønsted–Lowry definition; for example, BF3 and AlCl3 are species that can each accept an electron pair, which qualifies them as Lewis acids, but they lack a hydrogen ion to donate, disqualifying them as both Arrhenius and Brønsted–Lowry acids.
On the MCAT, you may encounter Lewis acids in the context of organic chemistry reactions because Lewis acids are often used as catalysts.
AMPHOTERIC SPECIES
An amphoteric species is one that reacts like an acid in a basic environment and like a base in an acidic environment. In the Brønsted–Lowry sense, an amphoteric species can either gain or lose a proton, making it amphiprotic as well. On the MCAT, water is the most common example. When water reacts with a base, it behaves as an acid:
H2O + B− ⇌ HB + OH−
When water reacts with an acid, it behaves as a base:
HA + H2O ⇌ H3O+ + A−
The partially dissociated conjugate base of a polyvalent acid is usually amphoteric. For example, HSO4− can either gain a proton to form H2SO4 or lose a proton to form SO42−). The hydroxides of certain metals (such as Al, Zn, Pb, and Cr) are also amphoteric. Furthermore, species that can act as both oxidizing and reducing agents are often considered to be amphoteric as well because by accepting or donating electron pairs, they act as Lewis acids or bases, respectively.
Complex amphoteric molecules include amino acids that have a zwitterion intermediate with both cationic and anionic character, as shown in Figure 10.4. Such species are discussed in great detail in Chapter 1 of MCAT Biochemistry Review.
 Figure 10.4. Amino Acid Zwitterions Are Complex Amphoteric Species The amino group can release a proton (acid) and the carboxylate group can accept a proton (base).
Figure 10.4. Amino Acid Zwitterions Are Complex Amphoteric Species The amino group can release a proton (acid) and the carboxylate group can accept a proton (base).
KEY CONCEPT
Water, amino acids, and partially deprotonated polyprotic acids such as bicarbonate and bisulfate are common examples of amphoteric and amphiprotic substances. Metal oxides and hydroxides are also considered amphoteric but not necessarily amphiprotic because they do not give off protons.
ACID–BASE NOMENCLATURE
The names of most acids are related to the names of their parent anions (the anion that combines with H+ to form the acid). Acids formed from anions with names that end in –ide have the prefix hydro– and the ending –ic.
|
F− |
Fluoride |
HF |
Hydrofluoric acid |
|
Cl− |
Chloride |
HCl |
Hydrochloric acid |
|
Br− |
Bromide |
HBr |
Hydrobromic acid |
Acids formed from oxyanions are called oxyacids. If the anion ends in –ite (less oxygen), then the acid will end with –ous acid. If the anion ends in –ate (more oxygen), then the acid will end with –ic acid. Prefixes in the names of the anions are retained. Some common examples include the following:
|
ClO− |
Hypochlorite |
HClO |
Hypochlorous acid |
|
ClO2− |
Chlorite |
HClO2 |
Chlorous acid |
|
ClO3− |
Chlorate |
HClO3 |
Chloric acid |
|
ClO4− |
Perchlorate |
HClO4 |
Perchloric acid |
|
NO2− |
Nitrite |
HNO2 |
Nitrous acid |
|
NO3− |
Nitrate |
HNO3 |
Nitric acid |
|
CO32− |
Carbonate |
H2CO3 |
Carbonic acid |
|
SO42− |
Sulfate |
H2SO4 |
Sulfuric acid |
|
PO43− |
Phosphate |
H3PO4 |
Phosphoric acid |
|
BO33− |
Borate |
H3BO3 |
Boric acid |
|
CrO42− |
Chromate |
H2CrO4 |
Chromic acid |
|
CH3COO− |
Acetate |
CH3COOH |
Acetic acid |
MCAT EXPERTISE
There are some exceptions to the nomenclature rules. For instance, MnO4− is called permanganate even though there are no MnO3− or MnO2− ions.
MCAT Concept Check 10.1:
Before you move on, assess your understanding of the material with these questions.
1. Compare and contrast the three definitions for acids and bases:
|
Definition |
Acid |
Base |
|
Arrhenius |
||
|
Brønsted-Lowry |
||
|
Lewis |
2. Utilizing Arrhenius acid naming trends, predict the acid formula and name for the following anions:
|
Anion |
Acid Formula |
Acid Name |
|
MnO4− |
||
|
Titanate (TiO32−) |
||
|
I− |
||
|
IO4− |
3. Identify which reactants are amphoteric species in the following reactions. For those species, determine if the compound is also amphiprotic.
|
Reaction |
Amphoteric Reactant |
Amphiprotic? |
|
HCO3− + HBr → H2CO3 + Br− |
||
|
3 HCl + Al(OH)3 → AlCl3 + 3 H2O |
||
|
2 HBr + ZnO → ZnBr2 + H2O |