MCAT General Chemistry Review
Chapter 12: Electrochemistry
12.1 Electrochemical Cells
Electrochemical cells are contained systems in which oxidation–reduction reactions occur. There are three fundamental types of electrochemical cells: galvanic cells (also known as voltaic cells), electrolytic cells, and concentration cells. In addition, there are specific commercial cells such as Ni–Cd batteries through which we can understand these fundamental models.
Galvanic cells and concentration cells house spontaneous reactions, whereas electrolytic cells contain nonspontaneous reactions. Remember that spontaneity is indicated by the change in Gibbs free energy, ΔG. All three types contain electrodes where oxidation and reduction take place. For all electrochemical cells, the electrode where oxidation occurs is called the anode, and the electrode where reduction occurs is called the cathode. Other descriptors of electrochemical cells include the electromotive force (emf), which corresponds to the voltage or electrical potential difference of the cell. If the emf is positive, the cell is able to release energy (ΔG < 0), which means it is spontaneous. If the emf is negative, the cell must absorb energy (ΔG > 0), which means it is nonspontaneous.
MNEMONIC
Electrodes in a electrochemical cell: AN OX and a RED CAT
The anode is the site of oxidation; reduction occurs at the cathode.
Furthermore, we can also state that, for all electrochemical cells, the movement of electrons is from anode to cathode, and the current (I) runs from cathode to anode. This point can be a point of confusion among students. In physics, it is typical to state that current is the direction of flow of a positive charge through a circuit; this model was first proposed by Ben Franklin and continues to be used among physicists. Modern chemists are interested in the flow of electrons, but may discuss the current (a theoretical flow of positive charge) as a proxy for the flow of electrons; the current and the flow of electrons are always of equal magnitude but in opposite directions.
KEY CONCEPT
Electrons move through an electrochemical cell opposite to the flow of current (I).
Last, it is important to note that all batteries are influenced by temperature changes. For instance, lead–acid batteries in cars, like most galvanic cells, tend to fail most in cold weather. The thermodynamic reasons behind this will be discussed later in this chapter.
GALVANIC (VOLTAIC) CELLS
All of the nonrechargeable batteries you own are galvanic cells, also called voltaic cells. Accordingly, because household batteries are used to supply energy to a flashlight or remote control, the reactions in these cells must be spontaneous. This means that the reaction’s free energy is decreasing (ΔG < 0) as the cell releases energy to the environment. By extension, if the free energy change is negative for these cells, their electromotive force (Ecell) must be positive; the free energy change and electromotive force always have opposite signs.
REAL WORLD
Galvanic cells are commonly used as batteries; to be worthwhile (that is, producing energy to power some device or appliance), these batteries must be spontaneous!
Let’s examine the inner workings of a galvanic (voltaic) cell. Two electrodes of distinct chemical identity are placed in separate compartments, which are called half-cells. The two electrodes are connected to each other by a conductive material, such as a copper wire. Along the wire, there may be other various components of a circuit, such as resistors or capacitors, but for now, we’ll focus on the battery itself.
Surrounding each of the electrodes is an aqueous electrolyte solution composed of cations and anions. As shown in the Daniell cell illustrated in Figure 12.1, the cations in the two half-cell solutions can be of the same element as the respective metal electrode. Connecting the two solutions is a structure called a salt bridge, which consists of an inert salt. When the electrodes are connected to each other by a conductive material, charge will begin to flow as the result of an oxidation–reduction reaction that is taking place between the two half-cells. The redox reaction in a galvanic cell is spontaneous, and therefore the change in Gibbs free energy for the reaction is negative (ΔG < 0). As the spontaneous reaction proceeds toward equilibrium, the movement of electrons results in a conversion of electrical potential energy into kinetic energy. By separating the reduction and oxidation half-reactions into two compartments, we are able to harness this energy and use it to do work by connecting various electrical devices into the circuit between the two electrodes.
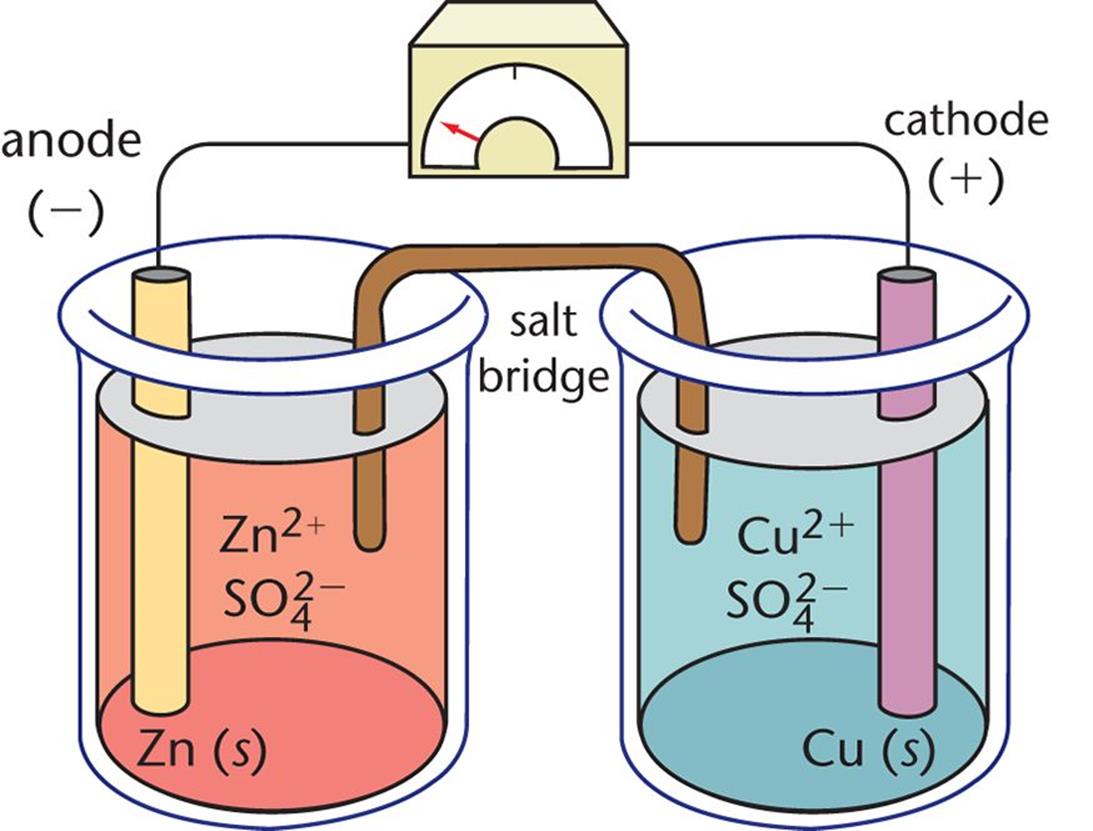 Figure 12.1. Daniell Cell In this galvanic cell, zinc is the anode and copper is the cathode; each electrode is bathed in an electrolyte solution containing its cation and sulfate.
Figure 12.1. Daniell Cell In this galvanic cell, zinc is the anode and copper is the cathode; each electrode is bathed in an electrolyte solution containing its cation and sulfate.
In the Daniell cell, a zinc electrode is placed in an aqueous ZnSO4 solution, and a copper electrode is placed in an aqueous CuSO4 solution. The anode of this cell is the zinc bar where Zn (s) is oxidized to Zn2+ (aq). The cathode is the copper bar, and it is the site of the reduction of Cu2+ (aq) to Cu (s). The half-cell reactions are written as follows:
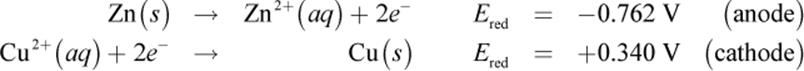
The net reaction is
|
Zn (s) + Cu2+ (aq) → Zn2+ (aq) + Cu (s) |
Ecell = +1.102 V |
We will discuss the calculation of cell potential in the next section. For now, appreciate that the calculation can be accomplished by knowing each half-reaction. If the two half-cells were not separated, the Cu2+ ions would react directly with the zinc bar, and no useful electrical work would be done. Because the solutions and electrodes are physically separated, they must be connected by a conductive material to complete the circuit.
However, if only a wire were provided for this electron flow, the reaction would soon stop because an excess positive charge would build up on the anode, and an excess negative charge would build up on the cathode. Eventually, the excessive charge accumulation would provide a countervoltage large enough to prevent the oxidation–reduction reaction from taking place, and the current would cease. This charge gradient is dissipated by the presence of a salt bridge, which permits the exchange of cations and anions. The salt bridge contains an inert electrolyte, usually KCl or NH4NO3, which contains ions that will not react with the electrodes or with the ions in solution. While the anions from the salt bridge (Cl–) diffuse into the solution on the anode side (ZnSO4) to balance out the charge of the newly created Zn2+ ions, the cations of the salt bridge (K+) flow into the solution on the cathode side (CuSO4) to balance out the charge of the sulfate ions left in solution when the Cu2+ ions are reduced to Cu and precipitate onto the electrode. This precipitation process onto the cathode itself can also be called plating or galvanization.
KEY CONCEPT
The purpose of the salt bridge is to exchange anions and cations to balance, or dissipate, newly generated charges.
During the course of the reaction, electrons flow from the zinc anode through the wire and to the copper cathode. A voltmeter can be connected to measure this electromotive force. As mentioned earlier, the anions (Cl–) flow externally from the salt bridge into the ZnSO4, and the cations (K+) flow externally from the salt bridge into the CuSO4. This flow depletes the salt bridge and, along with the finite quantity of Cu2+ in the solution, accounts for the relatively short lifespan of the cell.
MNEMONIC
Electron flow in an electrochemical cell: A → C (order in the alphabet)
Electrons flow from anode to cathode in all types of electrochemical cells.
A cell diagram is a shorthand notation representing the reactions in an electrochemical cell. A cell diagram for the Daniell cell is as follows:
Zn (s) | Zn2+ (1 M) || Cu2+ (1 M) | Cu (s)
The following rules are used in constructing a cell diagram:
1. The reactants and products are always listed from left to right in this form: anode | anode sol’n (concentration) || cathode sol’n (concentration) | cathode
2. A single vertical line indicates a phase boundary.
3. A double vertical line indicates the presence of a salt bridge or some other type of barrier.
MCAT EXPERTISE
Recognize and understand the shorthand cell notation for electrochemical cells on Test Day. Passages frequently use this format rather than spelling out which reactions take place at the anode and cathode.
ELECTROLYTIC CELLS
When comparing and contrasting galvanic and electrolytic cells, it is important to keep straight what remains consistent between the two types of cells and what differs. All types of electrochemical cells have a reduction reaction occurring at the cathode, an oxidation reaction occurring at the anode, a current flowing from cathode to anode, and electron flow from anode to cathode. However, electrolytic cells, in almost all of their characteristics and behavior, are otherwise the opposite of galvanic cells. Whereas galvanic cells house spontaneous oxidation–reduction reactions, which generate electrical energy, electrolytic cells house nonspontaneous reactions, which require the input of energy to proceed. Therefore, the change in free energy for an electrolytic cell is positive. This type of oxidation–reduction reaction driven by an external voltage source is calledelectrolysis, in which chemical compounds are decomposed. For example, electrolytic cells can be used to drive the nonspontaneous decomposition of water into oxygen and hydrogen gas. Another example, the electrolysis of molten NaCl, is illustrated in Figure 12.2.
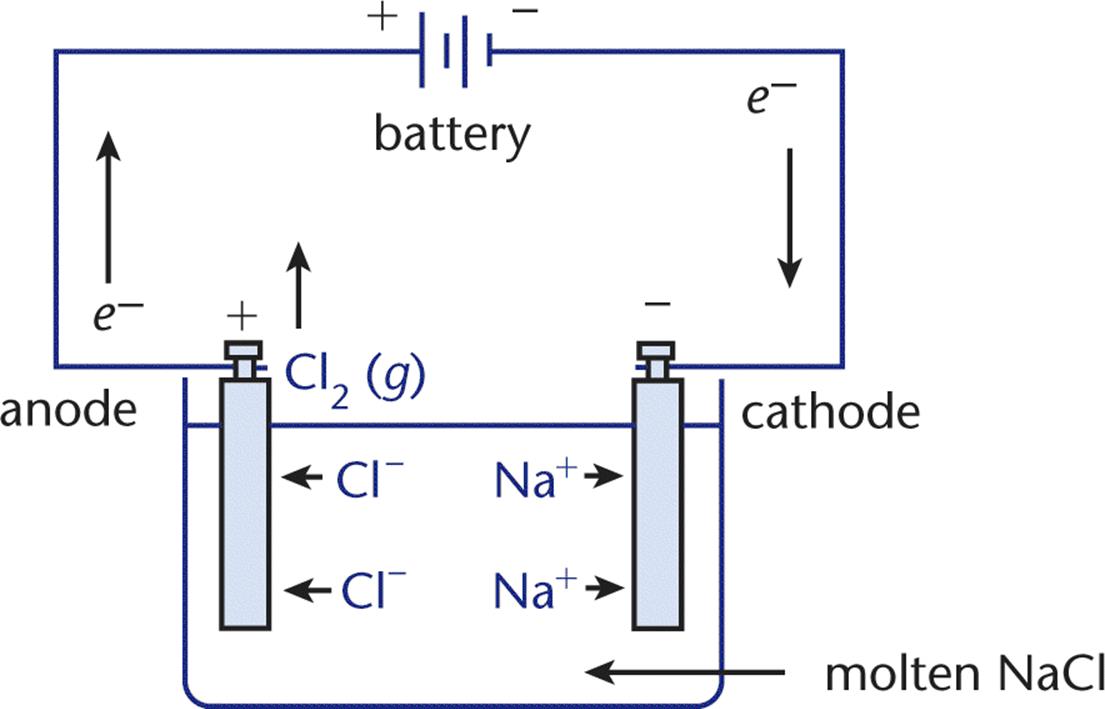 Figure 12.2. Electrolysis of Molten NaCl
Figure 12.2. Electrolysis of Molten NaCl
KEY CONCEPT
Because electrolysis is nonspontaneous, the electrode (anode or cathode) can consist of any material so long as it can resist the high temperatures and corrosion of the process.
In this electrolytic cell, molten NaCl is decomposed into Cl2 (g) and Na (l). The external voltage source—a battery—supplies energy sufficient to drive the oxidation–reduction reaction in the direction that is thermodynamically unfavorable (nonspontaneous).
In this example, Na+ ions migrate toward the cathode, where they are reduced to Na (l). At the same time, Cl– ions migrate toward the anode, where they are oxidized to Cl2 (g). Notice that the half-reactions do not need to be separated into different compartments; this is because the desired reaction is nonspontaneous. Note that sodium is a liquid at the temperature of molten NaCl; it is also less dense than the molten salt and, thus, is easily removed as it floats to the top of the reaction vessel.
This cell is used in industry as the major means of sodium and chlorine production. You may wonder why one would do so much work to obtain pure sodium and chlorine. Remember that these elements are never found naturally in their elemental form because they are so reactive. Thus, to use elemental sodium or chlorine gas in a reaction, it must be manufactured through processes such as these.
Michael Faraday was the first to define certain quantitative principles governing the behavior of electrolytic cells. He theorized that the amount of chemical change induced in an electrolytic cell is directly proportional to the number of moles of electrons that are exchanged during the oxidation–reduction reaction. The number of moles exchanged can be determined from the balanced half-reaction. In general, for a reaction that involves the transfer of n electrons per atom M,
Mn+ + n e– → M (s)
Equation 12.1
KEY CONCEPT
Faraday’s laws state that the liberation of gas, and deposition of elements, on electrodes is directly proportional to the number of electrons being transferred during the oxidation–reduction reaction. Here, normality or gram equivalent weight is used. These observations are proxy measurements of the amount of current flowing in a circuit.
According to this equation, one mole of metal M (s) will logically be produced if n moles of electrons are supplied to one mole of Mn+. Additionally, the number of moles of electrons needed to produce a certain amount of M (s) can now be related to the measurable electrical property of charge. One electron carries a charge of 1.6 × 10–19 coulombs (C). The charge carried by one mole of electrons can be calculated by multiplying this number by Avogadro’s number, as follows:
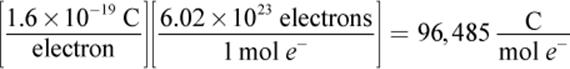
This number is called the Faraday constant, and one faraday (F) is equivalent to the amount of charge contained in one mole of electrons (1 F = 96,485 C) or one equivalent. On the MCAT, you should round up this number to ![]() to make calculations more manageable.
to make calculations more manageable.
KEY CONCEPT
One faraday (F) is equivalent to the amount of charge contained in one mole of electrons (1 F = 96,485 C).
The electrodeposition equation summarizes this process and helps determine the number of moles of element being deposited on a plate:
![]()
Equation 12.2
where mol M is the amount of metal ion being deposited at a specific electrode, I is current, t is time, n is the number of electron equivalents for a specific metal ion, and F is the Faraday constant.
MNEMONIC
Electrodeposition equation: Calculating Moles of Metal, It is Not Fun.
![]()
Example:
What mass of copper will be deposited in a Daniell cell if a current of 2 A flows through the cell for 3 hours?
Solution:
We will use the equation ![]()
A Daniell cell uses a copper electrode in copper sulfate (CuSO4) solution. Because the oxidation state of copper in solution is +2, n = 2. Now we can plug into the equation.

Then, we must determine the actual mass of copper being deposited. 0.1 mol Cu should have a mass of 6.35 g because the molar mass of copper is ![]() (actual = 7.11 g).
(actual = 7.11 g).
CONCENTRATION CELLS
A concentration cell is a special type of galvanic cell. Like all galvanic cells, it contains two half-cells connected by a conductive material, allowing a spontaneous oxidation–reduction reaction to proceed, which generates a current and delivers energy. The distinguishing characteristic of a concentration cell is in its design: the electrodes are chemically identical. For example, if both electrodes are copper metal, they have the same reduction potential. Therefore, current is generated as a function of a concentration gradient established between the two solutions surrounding the electrodes. The concentration gradient results in a potential difference between the two compartments and drives the movement of electrons in the direction that results in equilibration of the ion gradient. The current will stop when the concentrations of ionic species in the half-cells are equal. This implies that the voltage (V) or electromotive force of a concentration cell is zero when the concentrations are equal; the voltage, as a function of concentrations, can be calculated using the Nernst equation.
In a biological system, a concentration cell is best represented by the cell membrane of a neuron, as shown in Figure 12.3. Sodium and potassium cations, and chlorine anions, are exchanged as needed to produce an electrical potential. The actual value depends on both the concentrations and charges of the ions. In this way, a resting membrane potential (Vm) can be maintained. Disturbances of the resting membrane potential, if sufficiently large, may stimulate the firing of an action potential.
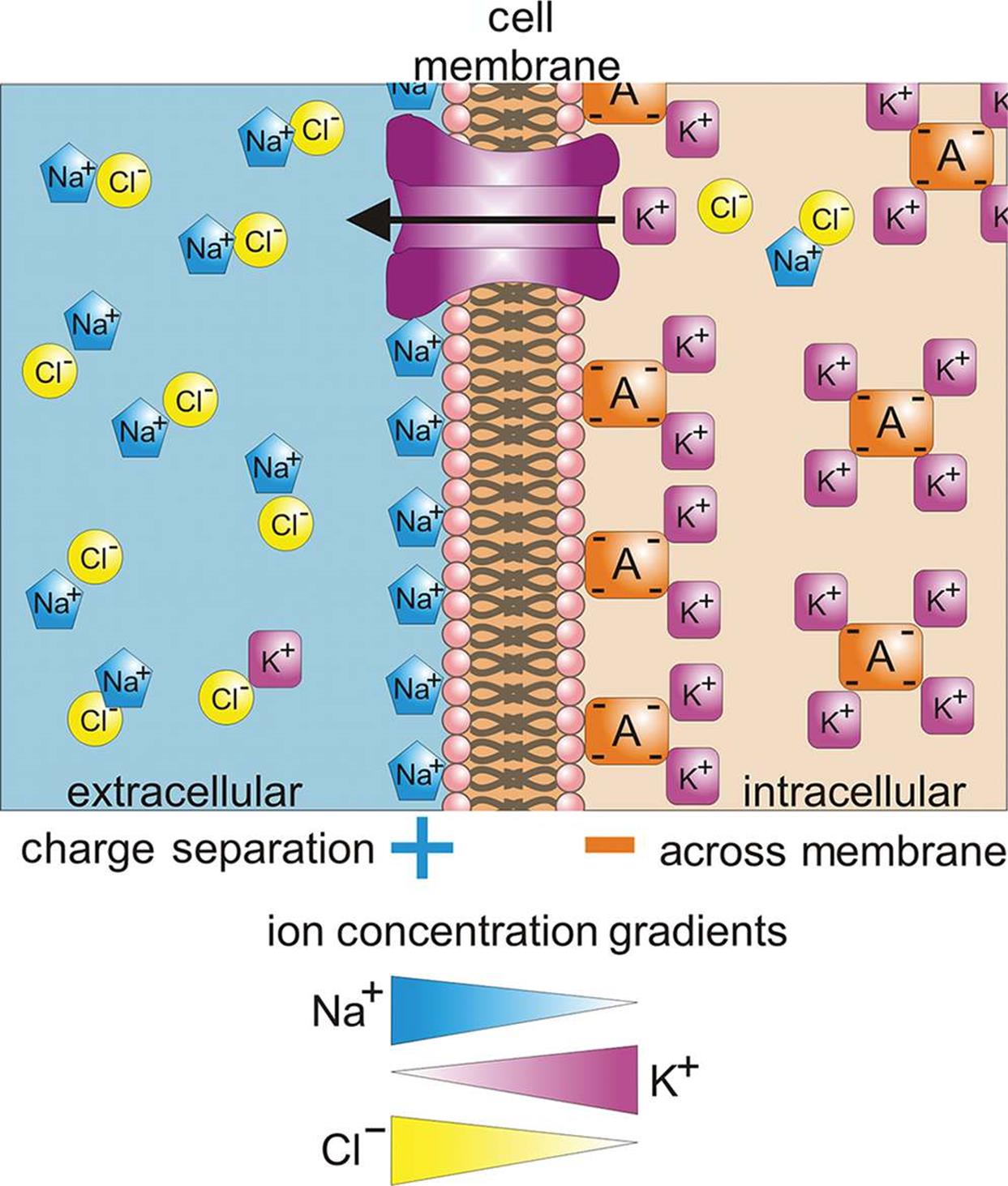 Figure 12.3. The Cell Membrane as an Example of a Concentration Cell The electrochemical gradient created by separation of ions across the cell membrane is analogous to a cell with two electrodes composed of the same material.
Figure 12.3. The Cell Membrane as an Example of a Concentration Cell The electrochemical gradient created by separation of ions across the cell membrane is analogous to a cell with two electrodes composed of the same material.
BRIDGE
The maintenance of a resting membrane potential is discussed in Chapter 8 of MCAT Biochemistry Review. The conduction of an action potential is discussed in Chapter 4 of MCAT Biology Review. The transfer of ions and electrons during an action potential produces biochemical work.
RECHARGEABLE CELLS
A rechargeable cell or rechargeable battery is one that can function as both a galvanic and electrolytic cell.
Lead–Acid Batteries
A lead–acid battery, also known as a lead storage battery, is a specific type of rechargeable battery. As a voltaic cell, when fully charged, it consists of two half-cells—a Pb anode and a porous PbO2 cathode, connected by a conductive material (concentrated 4 M H2SO4). When fully discharged, it consists of two PbSO4 electroplated lead electrodes with a dilute concentration of H2SO4, as shown in Figure 12.4.
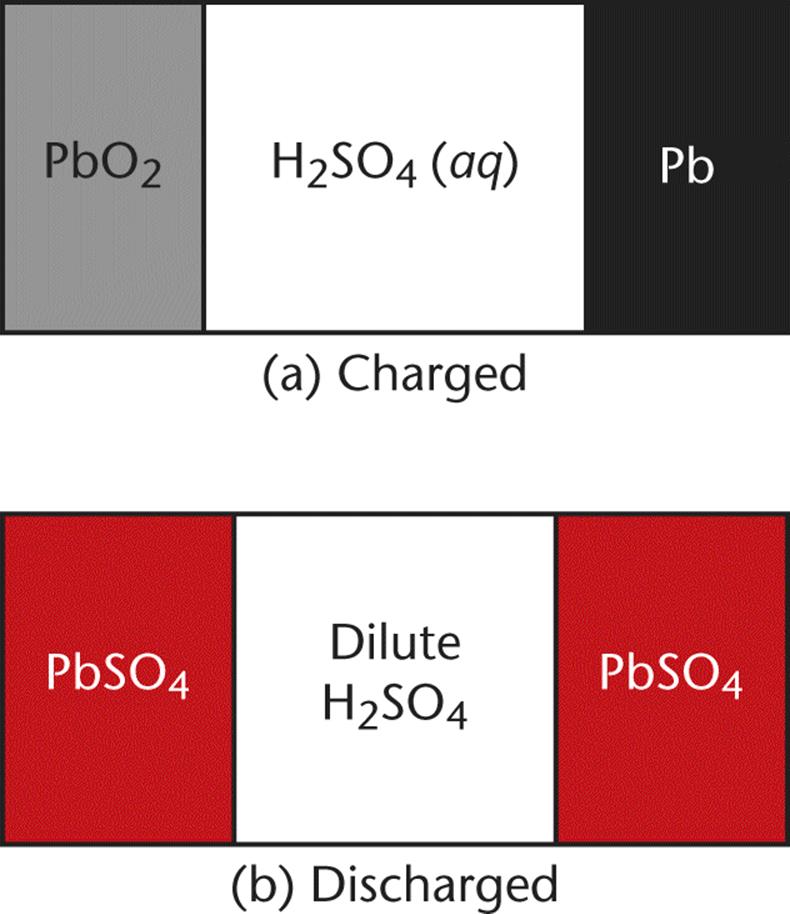 Figure 12.4. Lead–Acid Battery When charged (a), the cell contains a Pb anode and PbO2 cathode; when discharged (b), both electrodes are coated with lead sulfate.
Figure 12.4. Lead–Acid Battery When charged (a), the cell contains a Pb anode and PbO2 cathode; when discharged (b), both electrodes are coated with lead sulfate.
The oxidation half-reaction at the lead (negative) anode is:
|
Pb (s) + HSO4− (aq) → PbSO4 (s) + H+ (aq) + 2 e− |
E°red = −0.356 V |
The reduction half-reaction at the lead(IV) oxide (positive) cathode is:
|
PbO2 (s) + SO42− (aq) + 4 H+ + 2 e− → PbSO4 (s) + 2 H2O |
E°red = 1.685 V |
Both half-reactions cause the electrodes to plate with lead sulfate (PbSO4) and dilute the acid electrolyte when discharging. The lead anode is negatively charged and attracts the anionic bisulfate. The lead(IV) oxide cathode is a bit more complicated. This electrode is porous, which allows the electrolyte (sulfuric acid) to solvate the cathode into lead and oxide ions. Then, the hydrogen ions in solution react with the oxide ions to produce water, and the remaining sulfate ions react with the lead to produce the electroplated lead sulfate.
Overall, the net equation for a discharging lead–acid battery is:
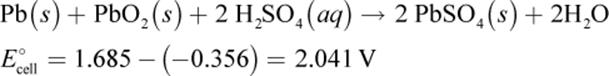
When charging, the lead–acid cell is part of an electrolytic circuit. These equations and electrode charge designations are the opposite because an external source reverses the electroplating process and concentrates the acid solution—this external source is very evident when one uses jumper cables to restart a car.
Lead–acid batteries, as compared to other cells, have some of the lowest energy-to-weight ratios (otherwise known as energy density). Energy density is a measure of a battery’s ability to produce power as a function of its weight. Lead–acid batteries, therefore, require a heavier amount of battery material to produce a certain output as compared to other batteries.
Nickel–Cadmium Batteries
Nickel–cadmium batteries are also rechargeable cells. They consist of two half-cells made of solid cadmium (the anode) and nickel(III) oxide-hydroxide (the cathode) connected by a conductive material, typically potassium hydroxide (KOH). Most of us are familiar with AA and AAA cells made of Ni–Cd materials, inside of which the electrodes are layered and wrapped around in a cylinder, as shown in Figure 12.5.
 Figure 12.5. A Nickel–Cadmium Battery (1) Metal casing, (2) salt bridge, (3) NiO(OH) cathode, (4) Cd anode.
Figure 12.5. A Nickel–Cadmium Battery (1) Metal casing, (2) salt bridge, (3) NiO(OH) cathode, (4) Cd anode.
The oxidation half-reaction at the cadmium (negative) anode is:
|
Cd (s) + 2 OH− (aq) → Cd(OH)2 (s) + 2 e− |
E°red = −0.86 V |
The reduction half-reaction at the nickel oxide–hydroxide (positive) cathode is:
|
2 NiO(OH) (s) + 2 H2O + 2 e− → 2 Ni(OH)2 (s) + 2 OH− |
E°red = 0.49 V |
Both half-reactions cause the electrodes to plate with their respective products. Overall, the net equation for a Ni–Cd battery is
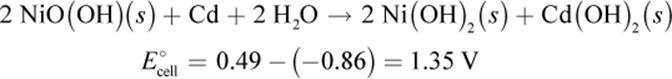
As in our previous example, charging reverses the electrolytic cell potentials. Some Ni–Cd designs are vented for this reason to allow for the release of built up hydrogen and oxygen gas during electrolysis.
Ni–Cd batteries have a higher energy density than lead–acid batteries. The electrochemistry of the Ni–Cd half-reactions also tends to provide higher surge current. Surge currents are periods of large current (amperage) early in the discharge cycle. This is preferable in appliances such as remote controls that demand rapid responses. It is important to note that modern Ni–Cd batteries have largely been replaced by more efficient nickel–metal hydride (NiMH) batteries. These newer batteries have more energy density, are more cost effective, and are significantly less toxic. As the name suggests, in lieu of a pure metal anode, a metal hydride is used instead.
ELECTRODE CHARGE DESIGNATIONS
In a galvanic cell, current is spontaneously generated as electrons are released by the oxidized species at the anode and travel through the conductive material to the cathode, where reduction takes place. Because the anode of a galvanic cell is the source of electrons, it is considered the negative electrode; the cathode is considered the positive electrode, as shown in Figure 12.1 previously. Electrons, therefore, move from negative (low electrical potential) to positive (high electrical potential), while the current—the flow of positive charge—is from positive (high electrical potential) to negative (low electrical potential).
Conversely, the anode of an electrolytic cell is considered positive because it is attached to the positive pole of the external voltage source and attracts anions from the solution. The cathode of an electrolytic cell is considered negative because it is attached to the negative pole of the external voltage source and attracts cations from the solution.
KEY CONCEPT
In a galvanic cell, the anode is negative and the cathode is positive. In an electrolytic cell, the anode is positive and the cathode is negative. This is because an external source is used to reverse the charge of an electrolytic cell. However, in both types of cells, reduction occurs at the cathode, and oxidation occurs at the anode; cations are attracted to the cathode, and anions are attracted to the anode.
In spite of this difference in designating charge (sign), oxidation always takes place at the anode and reduction always takes place at the cathode in both types of cells; electrons always flow through the wire from the anode to the cathode and current flows from cathode to anode. Finally, note that—regardless of its charge designation—the cathode always attracts cations and the anode always attracts anions. In the Daniell cell, for example, the electrons created at the anode by the oxidation of elemental zinc travel through the wire to the copper half-cell. There, they attract copper(II) cations to the cathode, resulting in the reduction of the copper ions to elemental copper, and drawing cations out of the salt bridge into the compartment. The anode, having lost electrons, attracts anions from the salt bridge at the same time that zinc(II) ions formed by the oxidation process dissolve away from the anode.
REAL WORLD
Recognize that in any system in which batteries are placed, it is important to line up cathodes and anodes. Electronics tend to have (+) and (–) designations to line up the electrodes—think jumper cables, television remotes, and button batteries in watches.
This is an important rule to understand not only for electrochemistry in the Chemical and Physical Foundations of Biological Systems section on Test Day, but also for electrophoresis in both this section and Biological and Biochemical Foundations of Living Systems. Isoelectric focusing is a technique used to separate amino acids or polypeptides based on their isoelectric points (pI). The positively charged amino acids (protonated at the solution’s pH) will migrate toward the cathode; negatively charged amino acids (deprotonated at the solution’s pH) will migrate toward the anode. The technique of isoelectric focusing is discussed in detail in Chapter 3 of MCAT Biochemistry Review.
MNEMONIC
Anions are attracted to the anode. Cations are attracted to the cathode. This is true regardless of the type of cell (galvanic, electrolytic, or concentration cells).
MCAT Concept Check 12.1:
Before you move on, assess your understanding of the material with these questions.
1. Circle which electrode each of the following statements describes in a galvanic (voltaic) cell:
|
· Site of oxidation: |
Anode |
Cathode |
|
· Electrons flow toward it: |
Anode |
Cathode |
|
· Current flows toward it: |
Anode |
Cathode |
|
· Has (–) designation: |
Anode |
Cathode |
|
· Attracts cations: |
Anode |
Cathode |
2. Circle which electrode each of the following statements describes in an electrolytic cell:
|
· Site of oxidation: |
Anode |
Cathode |
|
· Electrons flow toward it: |
Anode |
Cathode |
|
· Current flows toward it: |
Anode |
Cathode |
|
· Has (–) designation: |
Anode |
Cathode |
|
· Attracts cations: |
Anode |
Cathode |
3. Write the cell diagram for the discharging state of a lead–acid battery:
4. Which type of cell has a positive ΔG? A positive Ecell?
· Positive ΔG:
· Positive Ecell:
5. How much current is required to produce 0.23 kg Na from a molten NaCl electrolytic cell that runs for 30 hours? Assume the cell is 100% efficient.
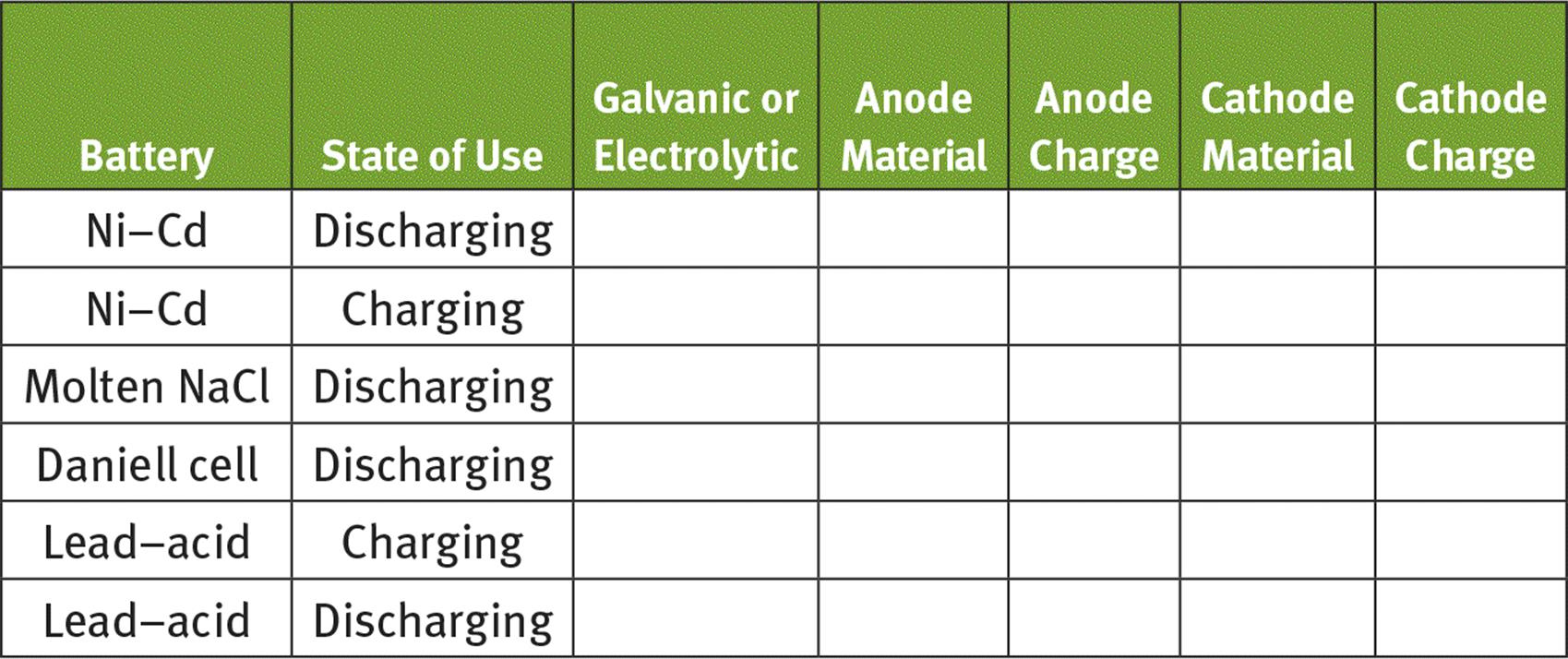
6. Fill in the following chart to summarize electrode charge designations of batteries we have analyzed: