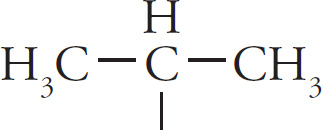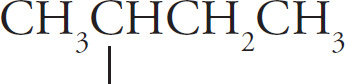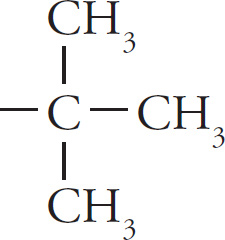MCAT Organic Chemistry Review: For MCAT 2015 (Graduate School Test Preparation) - P.J. Alaimo, Ph.D. 2015
Organic Chemistry Fundamentals
3.1 BACKGROUND AND INTRODUCTION
This section covers the fundamentals of nomenclature in organic chemistry. Although this section will require memorization as your primary study technique, it is in your best interest to be comfortable reading, hearing, and using this terminology. Although most of the terminology that appears on the MCAT is IUPAC, some common nomenclature is also used.
Basic Nomenclature
In the case of an all-carbon containing ring, these are preceded by the prefix cyclo-. Hence, a six-membered ring containing all −CH2− units is called cyclohexane.
|
Nomenclature For Substituents |
|
Substituent |
Name |
−CH3 |
methyl |
−CH2CH3 |
ethyl |
− CH2CH2CH3 |
propyl |
|
isopropyl |
− CH2CH2CH2CH3 |
butyl (or n-butyl) |
|
sec-butyl |
|
tert-butyl (or t-butyl) |
Common Functional Groups
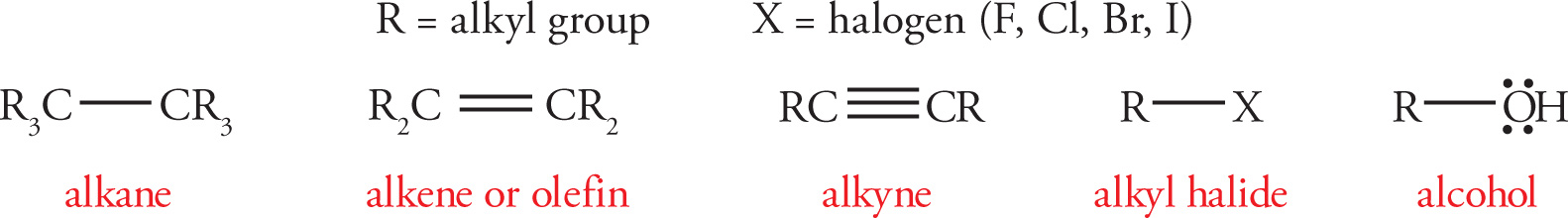
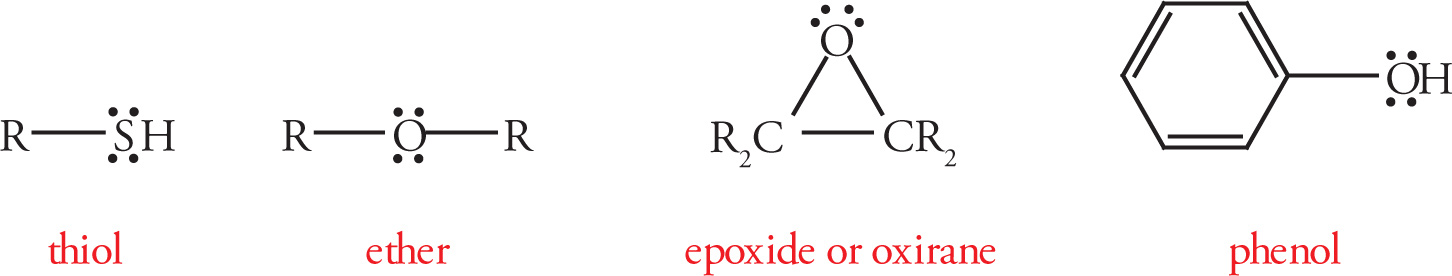
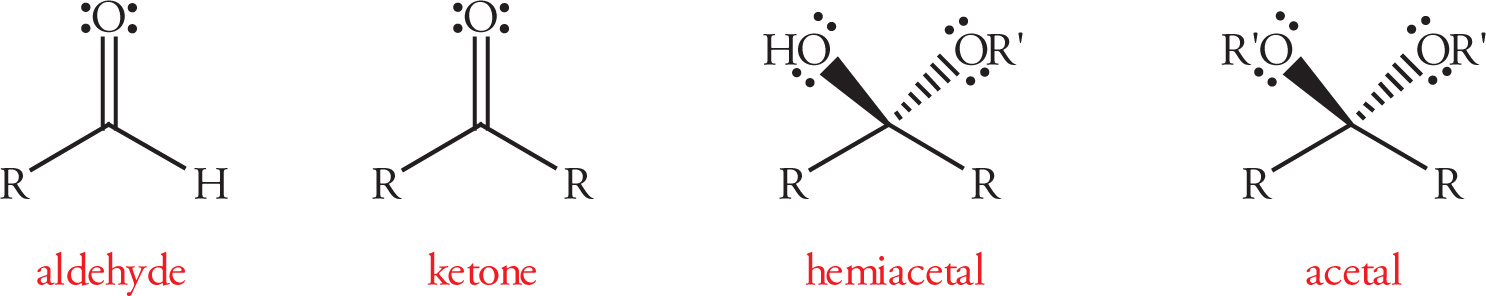
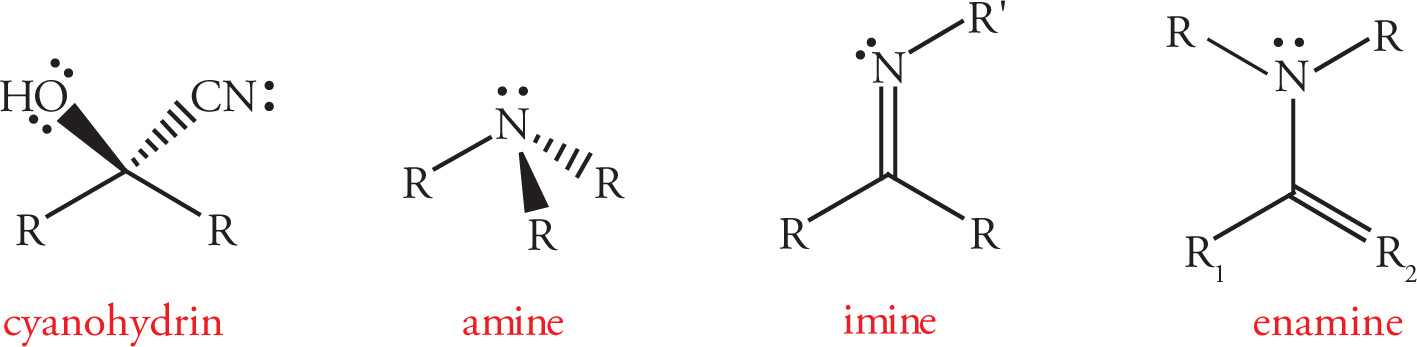
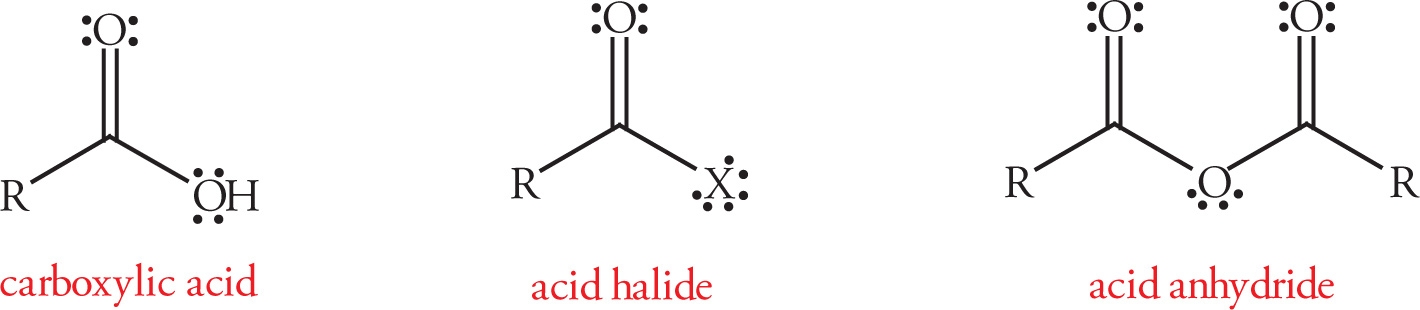
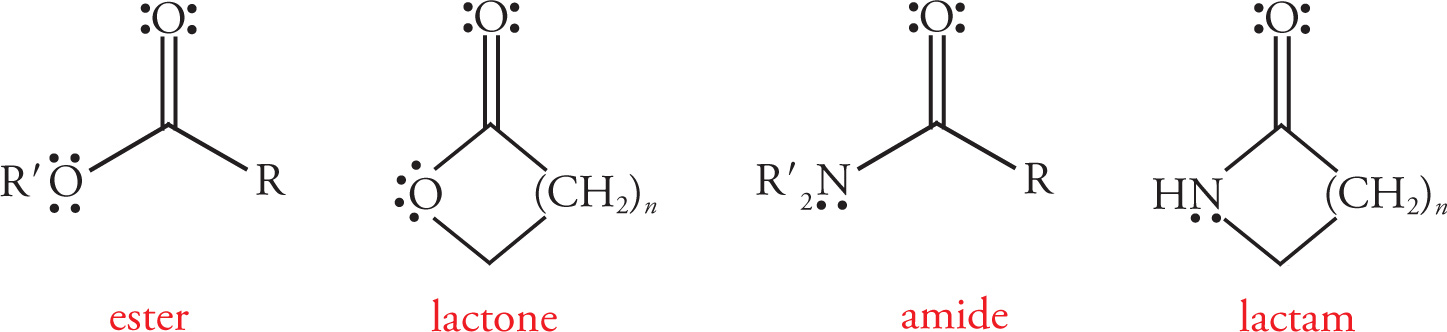
3.2 ABBREVIATED LINE STRUCTURES
The prevalence of carbon-hydrogen (C—H) bonds in organic chemistry has led chemists to use an abbreviated drawing system, merely for convenience. Just imagine having to draw every C—H bond for a large molecule like a steroid or polymer! Abbreviated line structures use only a few simple rules:
1. Carbons are represented simply as vertices.
2. C—H bonds are not drawn.
3. Hydrogens bonded to any atom other than carbon must be shown.
To illustrate rules 1 and 2, pentane can be represented using the full Lewis structure,
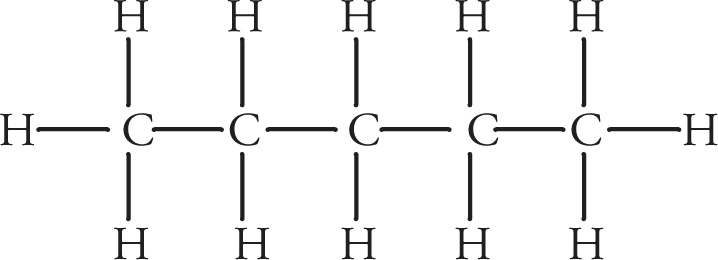
or using the abbreviated line structure.
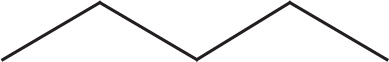
Although C—H bonds are not drawn, the number of hydrogens required to complete carbon’s valency are assumed. To clarify this, let’s look more closely at the abbreviated line structure of pentane:
These three carbon atoms are each bonded to two other carbon atoms. In order to complete carbon’s valency, we assume there are two hydrogens bonded to each of these carbons.
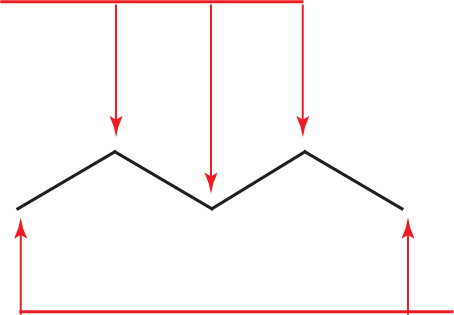
These two carbon atoms are each bonded to one other carbon atom. In order to complete carbon’s valency, we assume there are three hydrogens bonded to each of these carbons.
This must be correct, because if we draw out all of the hydrogens in pentane, we get the full Lewis structure shown above.
To illustrate rule 3, consider dimethyl amine:
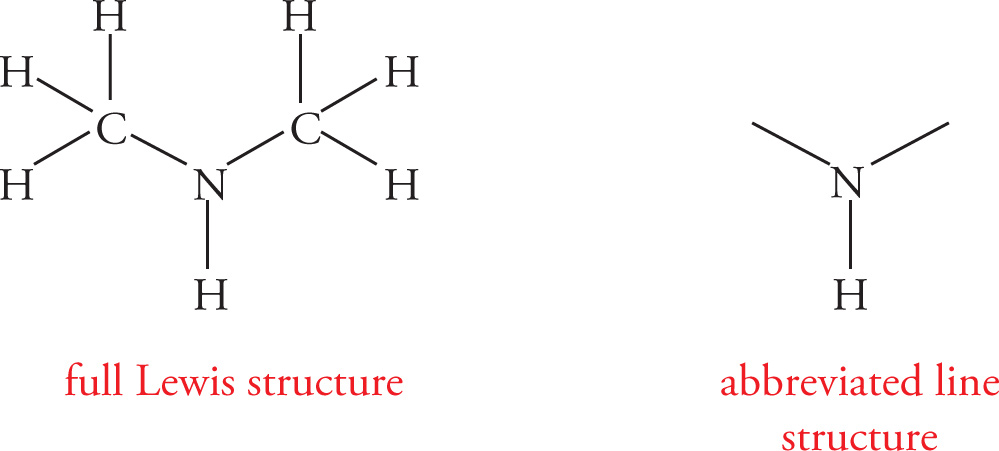
Remember that hydrogens bonded to carbon can be assumed (the methyl groups in dimethyl amine, for example), but hydrogens bonded to any other atom must be shown. Lone pairs of electrons are often omitted.
Example 3-1: Translate each of the following Lewis structures into an abbreviated line structure:
(a) 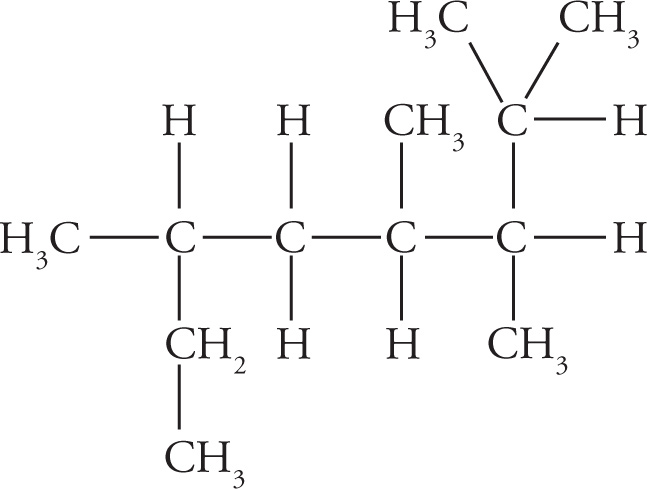
(b) 
(c) 
(d) 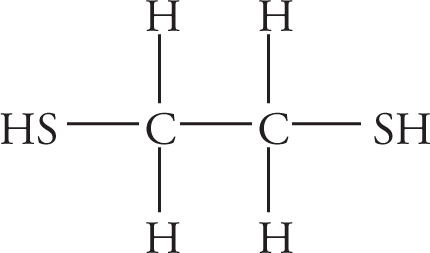
Solution:
(a) 
(b) 
(c) 
(d) 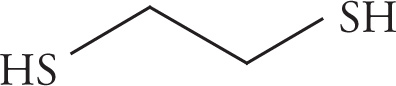
Example 3-2: Translate each of the following abbreviated line structures into a Lewis structure:
(a) 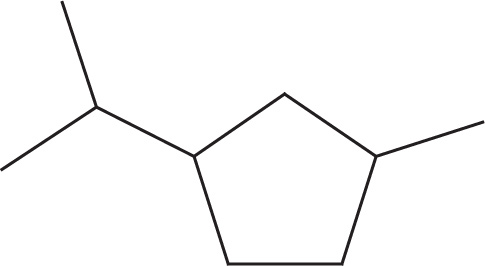
(b) 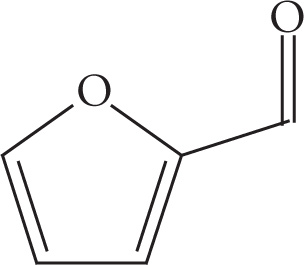
(c) 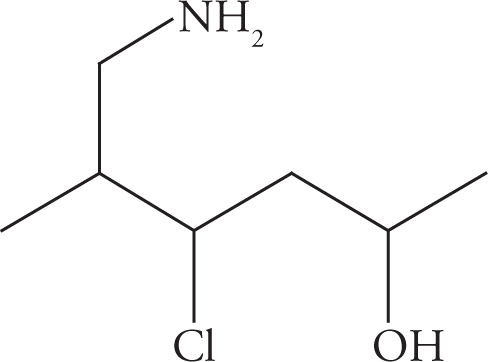
(d) 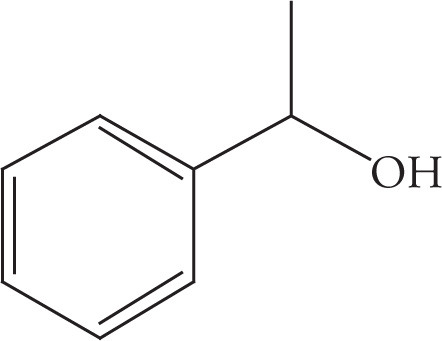
Solution:
(a) 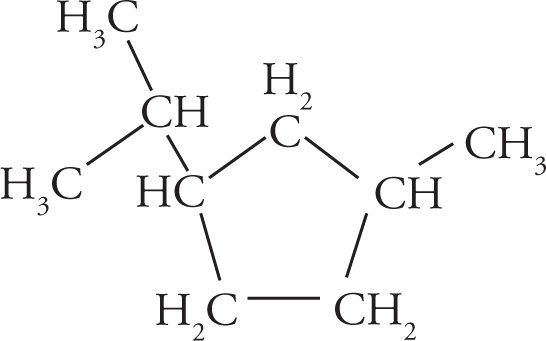
(b) 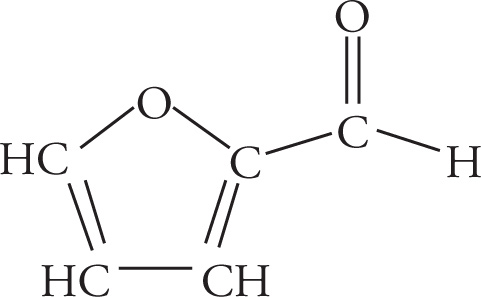
(c) 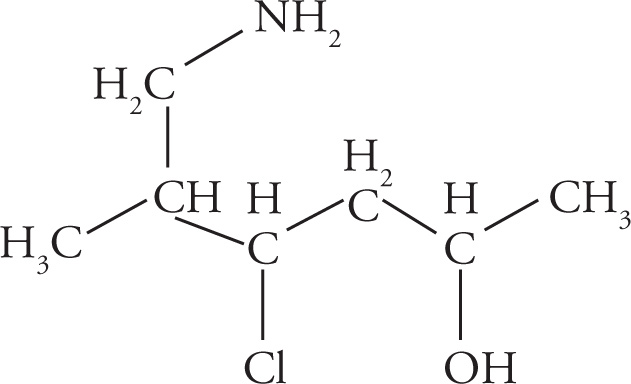
(d) 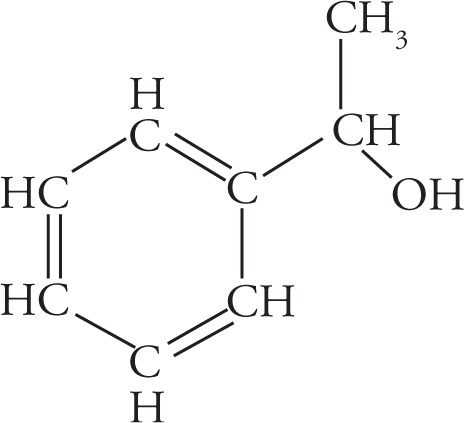
3.3 NOMENCLATURE OF ALKANES
Alkanes are named by a set of simple rules. One particular alkane (shown below) will be used to illustrate this process:
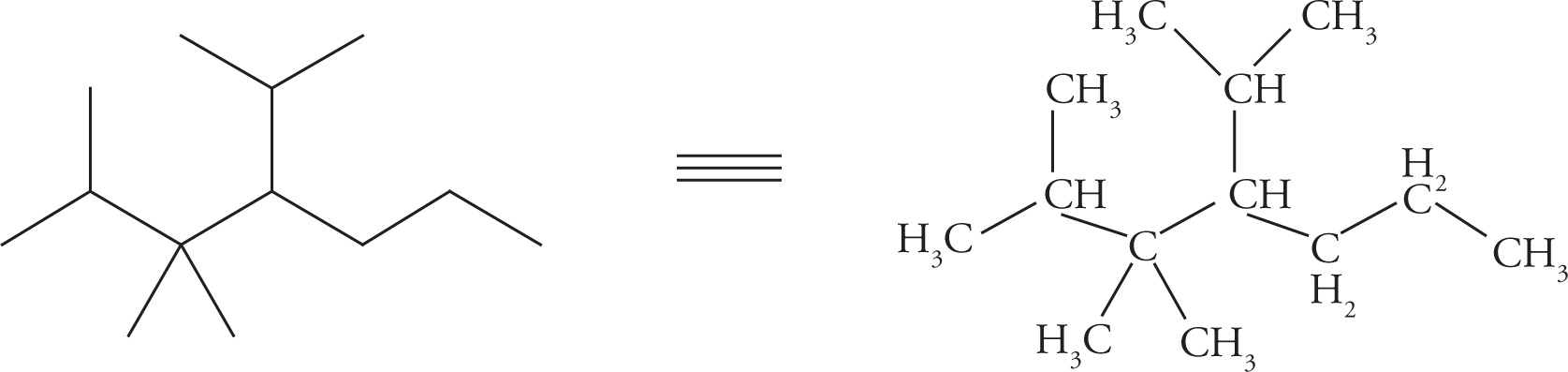
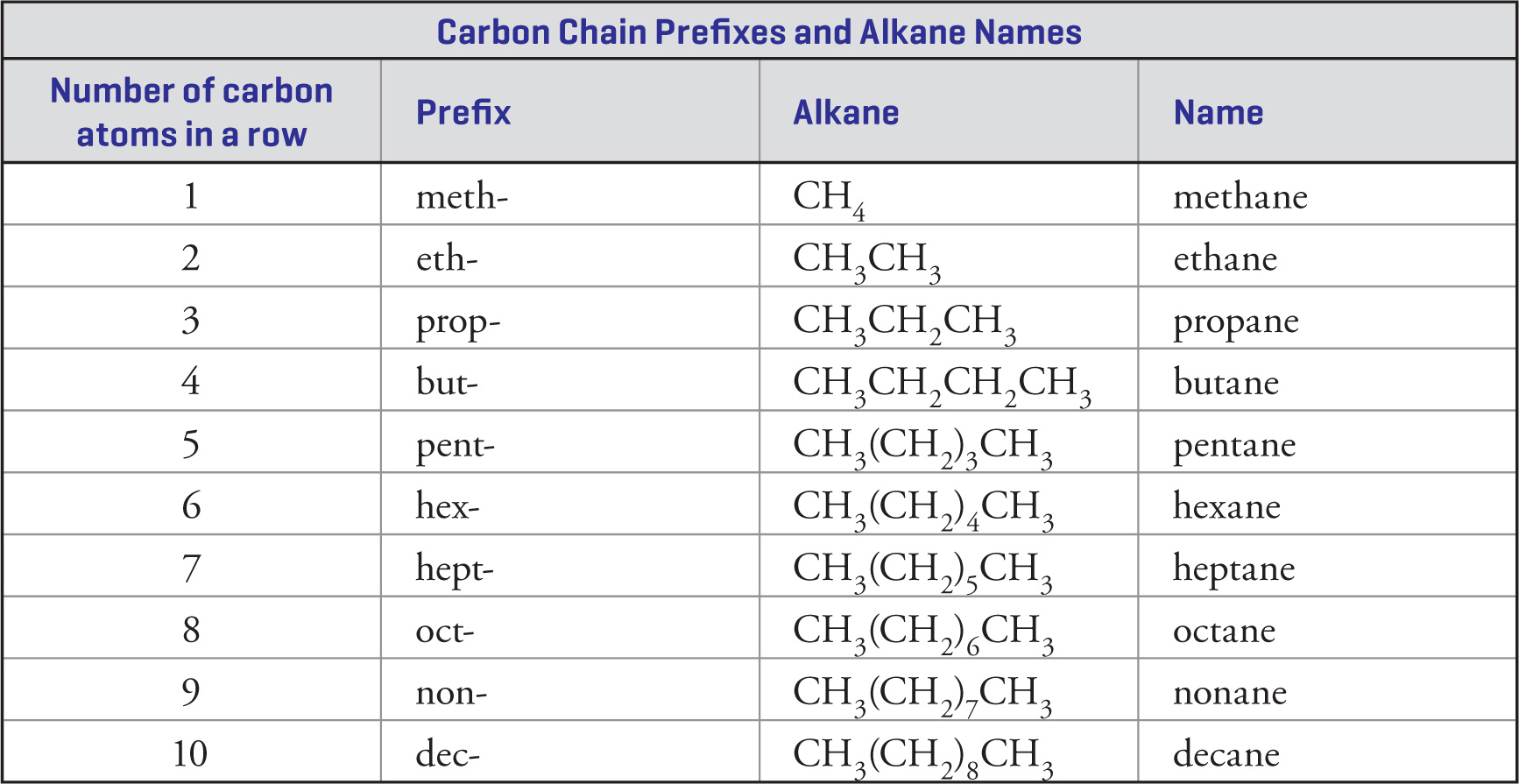
1. Identify the longest continuous carbon chain. The names of these chains are given in the first table in this chapter (“Carbon Chain Prefixes and Alkane Names”).
The longest chain in the compound above is a 7-carbon chain, which is called heptane. (This chain is shown below, outlined by dashed lines.)
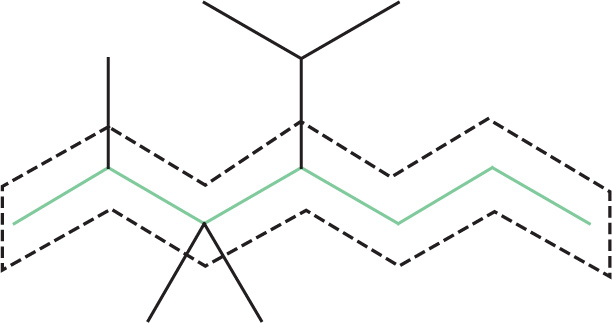
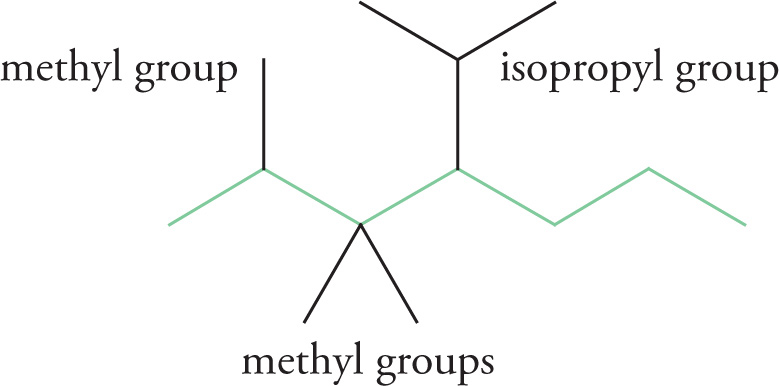
2. Identify any substituents on this chain. The names of some common hydrocarbon substituents are given in the second table in this chapter (“Nomenclature for Substituents”).
There are four substituents in this example: three methyl groups and one isopropyl group.
3. Number the carbons of the main chain such that the substituents are on the carbons with lower numbers.
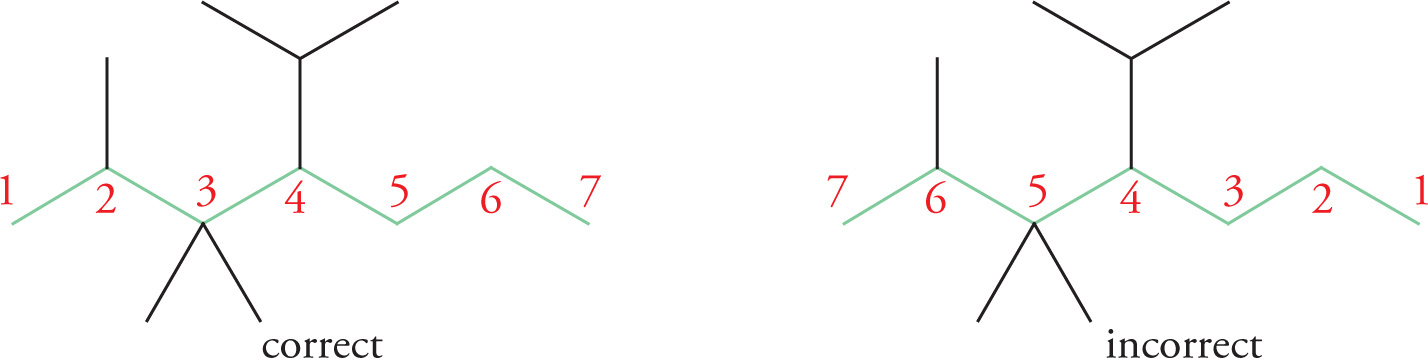
Now each substituent can be associated with the carbon atom to which it’s attached:
2 — methyl
3 — methyl
3 — methyl
4 — isopropyl
4. Identical substituents are grouped together; the prefixes di-, tri-, tetra-, and penta- are used to denote how many there are, and their carbon numbers are separated by a comma.
In this case we have

5. Alphabetize the substituents, ignoring the prefixes di-, tri-, etc. and n-, sec-, tert-, and separate numbers from words by a hyphen and numbers from numbers by a comma. Note that “iso” is not a prefix but is part of the name of the substituent, so it is NOT ignored when alphabetizing.
The complete name for our molecule is therefore 4-isopropyl-2,3,3-trimethylheptane.
Let’s do another example and find the name of this molecule:
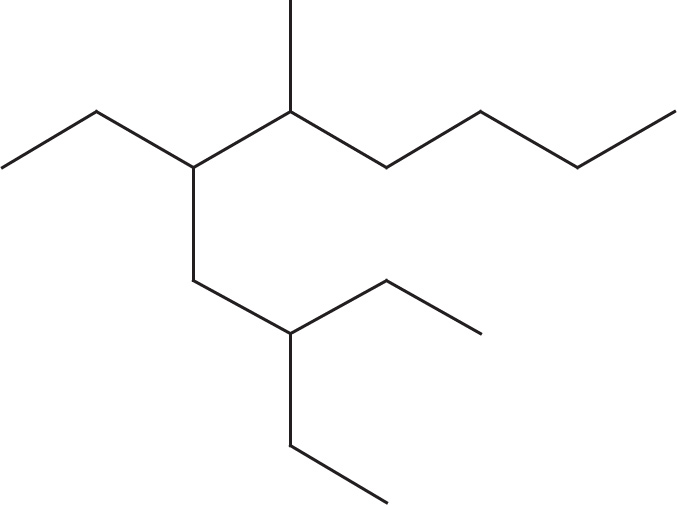
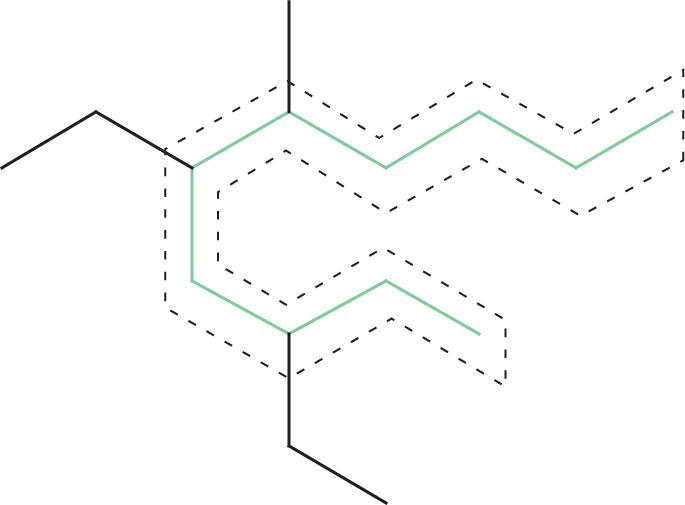
1. The longest continuous carbon chain is a 10-carbon chain, called decane.
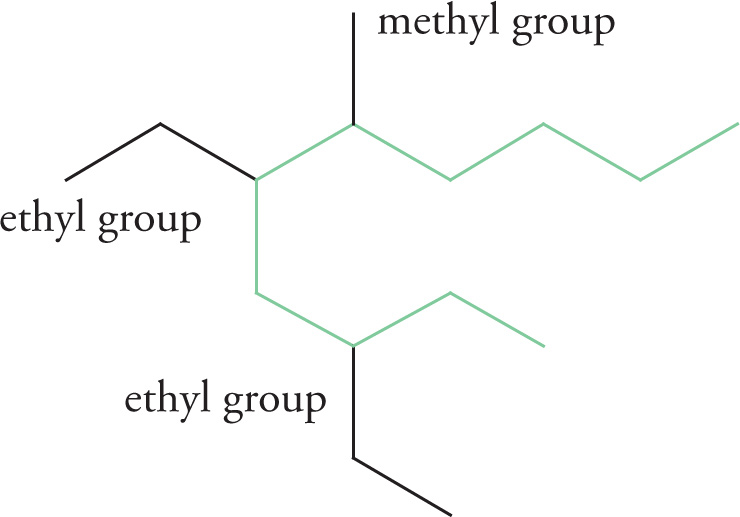
2. There are three substituents on this chain: two ethyl groups and a methyl group.
3. The correct numbering of the carbons in the main chain is as follows:
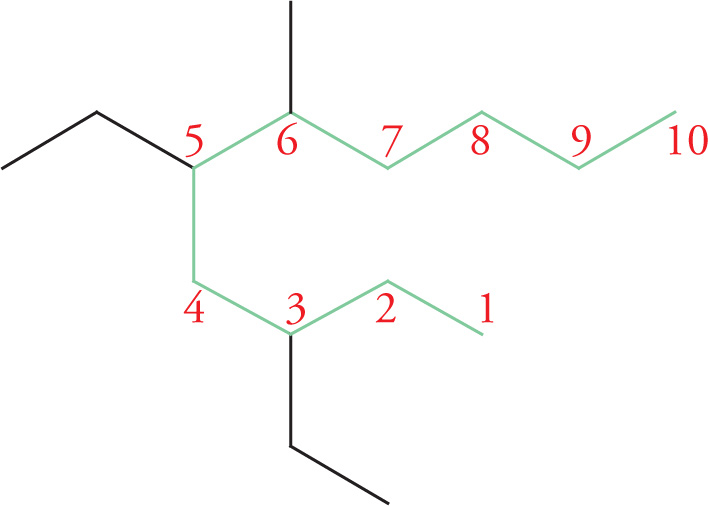
4. The substituents are now identified as:
3,5-diethyl
6-methyl
5. The complete name of the molecule is therefore 3,5-diethyl-6-methyldecane.
Example 3-3: Name each of the following alkanes:
(a) 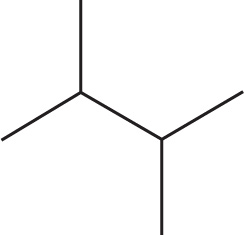
(b) 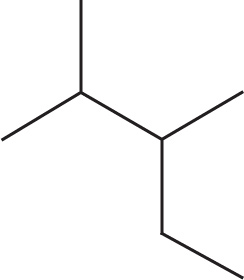
(c) 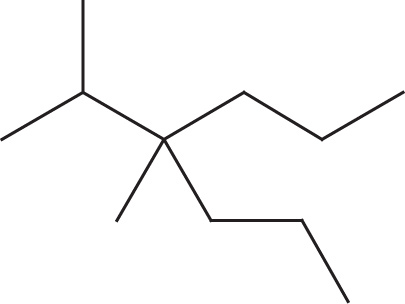
(d) 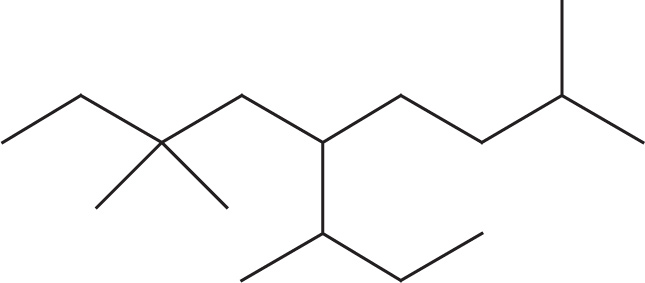
(e) 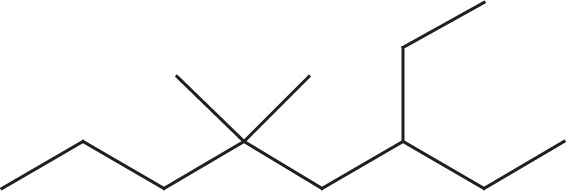
Solution:
(a) 2,3-dimethylbutane
(b) 2,3-dimethylpentane
(c) 4-isopropyl-4-methylheptane
(d) 5-sec-butyl-2,7,7-trimethylnonane
(e) 3-ethyl-5,5-dimethyloctane
3.4 NOMENCLATURE OF HALOALKANES
Alkanes with halogen (F, Cl, Br, I) substituents follow the same set of rules as simple alkanes. Halogens are named using these prefixes:
|
Halogen |
Prefix |
fluorine |
fluoro- |
chlorine |
chloro- |
bromine |
bromo- |
iodine |
iodo- |
By applying the same rules as for naming simple alkanes, verify the following names:
Structure |
Name |
|
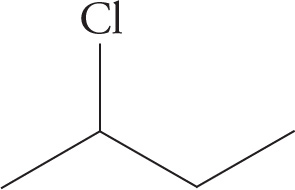
2-chlorobutane |
|
2-chloro- 1 -fluoro-4-methylpentane |
|
2,2-dibromo-3-iodo-4-methylhexane |
Example 3-4: Name each of the following haloalkanes:
(a) 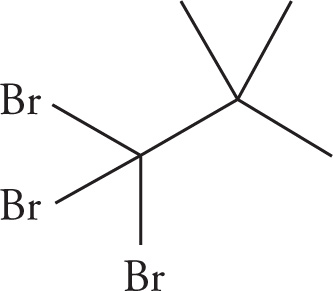
(b) 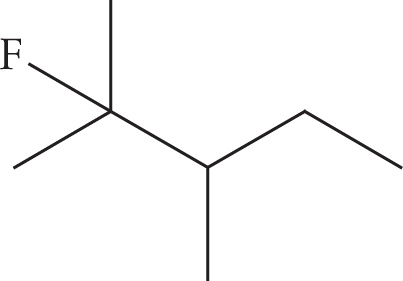
(c) 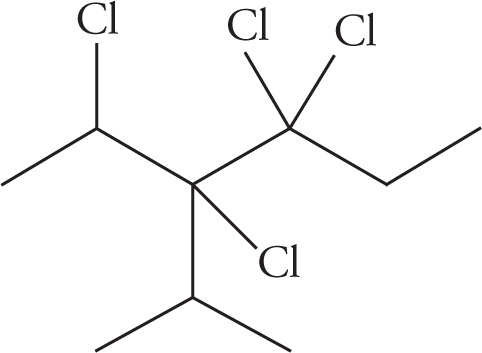
(d) 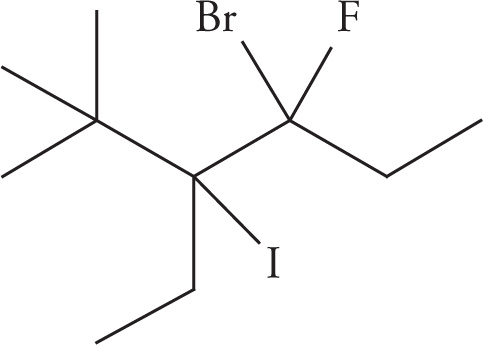
Solution:
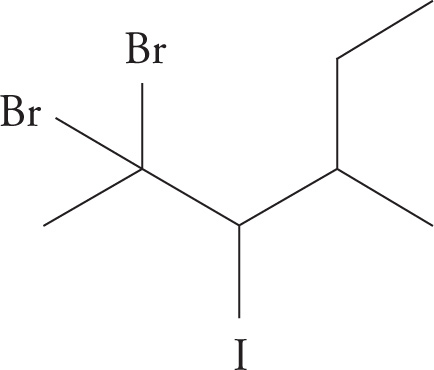
(a) 1,1,1-tribromo-2,2-dimethylpropane
(b) 2-fluoro-2,3-dimethylpentane
(c) 2,3,4,4-tetrachloro-3-isopropylhexane
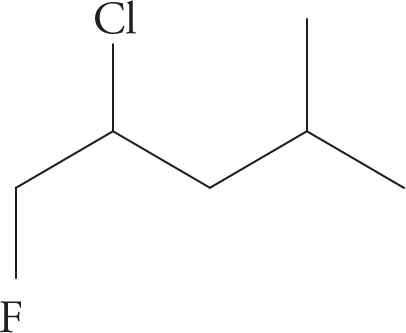
(d) 4-bromo-3-ethyl-4-fluoro-3-iodo-2,2-dimethylhexane
Example 3-5: For each name, draw the structure:
(a) 3-chloro-2,2-dimethylbutane
(b) 3-bromo-4-chloro-5,5-diethylnonane
(c) 2,3-dibromo-1,1-diiodopropane
(d) 3,4-difluoro-2,2,3-trimethylpentane
Solution:
(a) 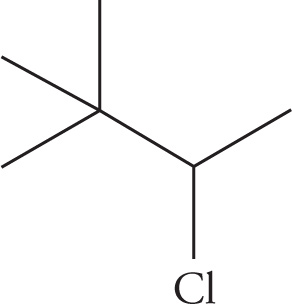
(b) 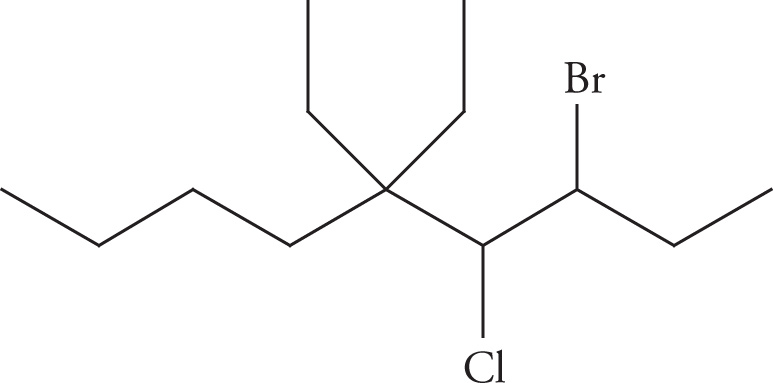
(c) 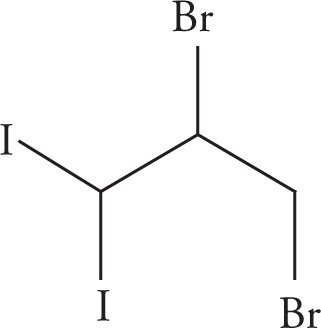
(d) 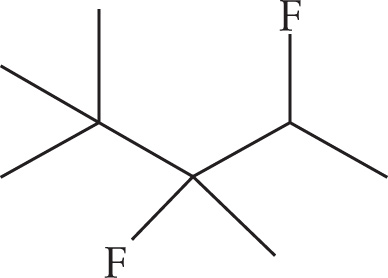
3.5 NOMENCLATURE OF ALCOHOLS
Alcohols also follow many of the same nomenclature rules as alkanes. Hydroxyl groups (−OH), however, are typically denoted by a suffix to the main alkyl chain. The table of straight-chain alcohols given below shows that to denote a hydroxyl group, the suffix −ol replaces the last −e in the name of the alkane.
|
Alkanes |
|
Structure |
Name |
CH4 |
methane |
CH3CH3 |
ethane |
CH3CH2CH3 |
propane |
CH3CH2CH2CH3 |
butane |
|
Alcohols |
|
Structure |
Name |
CH3OH |
methanol |
CH3CH2OH |
ethanol |
CH3CH2CH2OH |
propanol |
CH3CH2CH2CH2OH |
butanol |
When the position of the hydroxyl group needs to be specified, the number is placed after the name of the longest carbon chain and before the −ol suffix, separated by hyphens. For example:
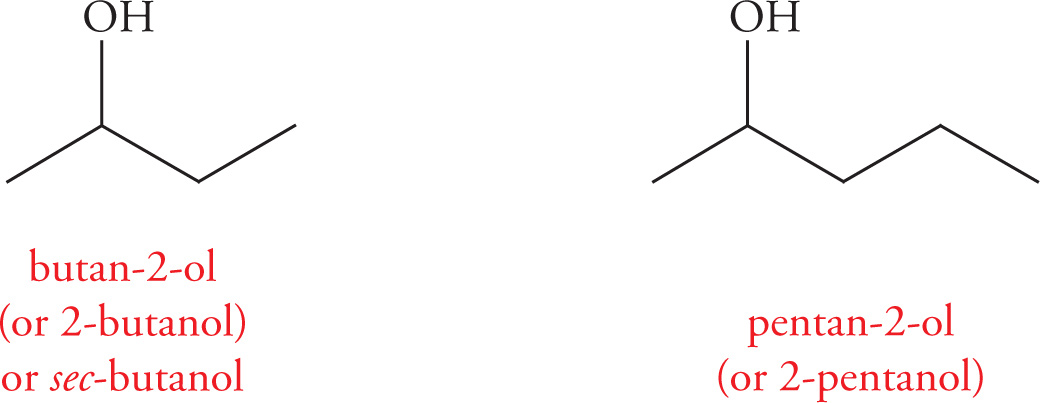
Priorities are assigned (the way the main carbon chain is numbered) to give the lowest number to the hydroxyl group. For example:
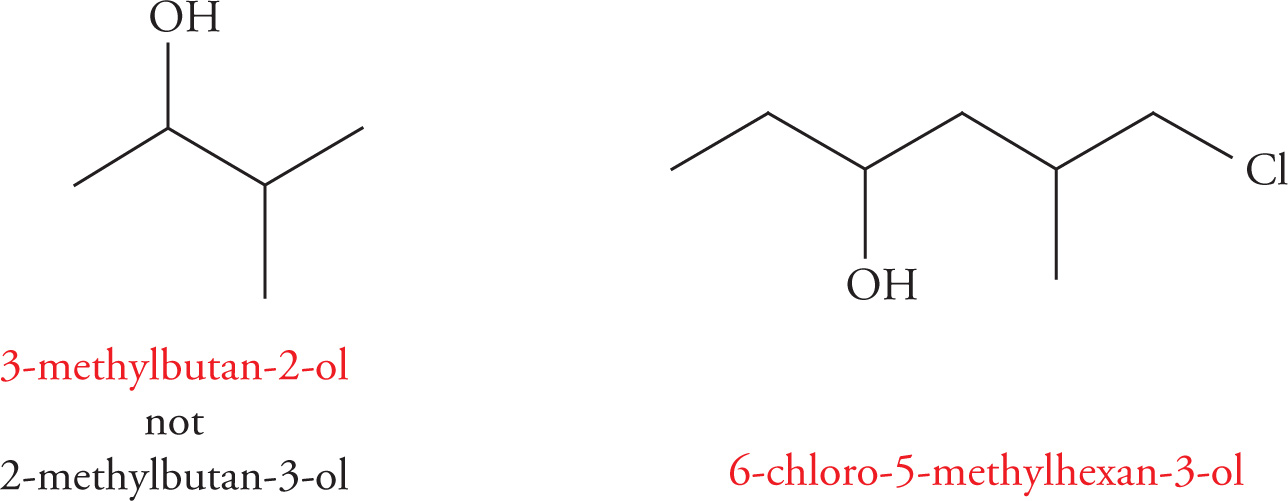
Example 3-6: Name each of the following molecules:
(a) 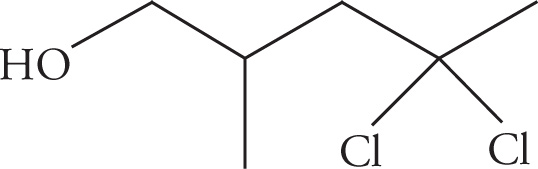
(b) 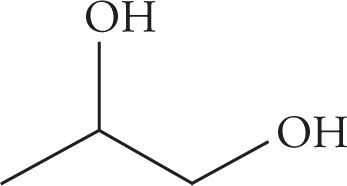
(c) 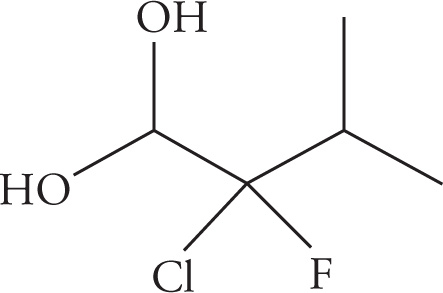
(d) 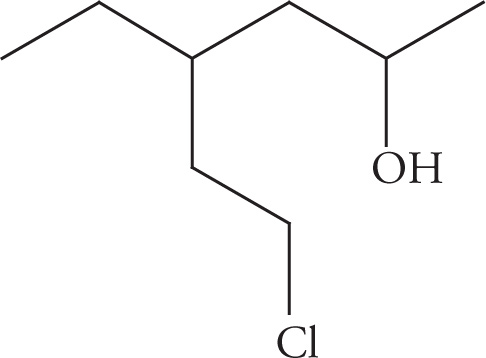
Solution:
(a) 4,4-dichloro-2-methylpentanol (the “-1-” is assumed if no number is given)
(b) propan-1,2-diol (or 1,2-propandiol)
(c) 2-chloro-2-fluoro-3-methylbutan-1,1-diol
(d) 6-chloro-4-ethylhexan-2-ol
Other organic functional groups have small nuances to their nomenclature, but this introduction to nomenclature should allow you to interpret chemical names on the MCAT.