MCAT General Chemistry Review - Steven A. Leduc 2015
Acids and Bases
11.1 DEFINITIONS
There are three different definitions of acids and bases.
Arrhenius Acids and Bases
Arrhenius gave us the most straightforward definitions of acids and bases, though they are the oldest and narrowest definitions so they are the least commonly used:
Acids ionize in water to produce hydrogen (H+) ions.
Bases ionize in water to produce hydroxide (OH−) ions.
For example, HCl is an acid,
HCl → H+ + Cl−
and NaOH is a base:
NaOH → Na+ + OH−
It’s important to remember that H+ does not exist by itself. Rather, it will combine with a molecule of water to give H3O+. However, for purposes of the MCAT, it doesn’t matter which of the two you use: H+ or H3O+.
Brønsted-Lowry Acids and Bases
Brønsted and Lowry offered the following definitions:
Acids are proton (H+) donors.
Bases are proton (H+) acceptors.
This definition of an acid is essentially the same idea as put forth by Arrhenius. The subtlety is apparent in their definition of a base. A Brønsted-Lowry base is a substance that is capable of accepting a proton. While hydroxide ions qualify as Brønsted-Lowry bases, many other compounds fit this definition as well.
If we consider the reversible reaction below:
H2CO3 + H2O ![]() H3O+ + HCO3−
H3O+ + HCO3−
then according to the Brønsted-Lowry definition, H2CO3 and H3O+ are acids; HCO3− and H2O are bases. The Arrhenius and Brønsted-Lowry definitions of acid and bases are the most important ones for MCAT General Chemistry.
Lewis Acids and Bases
Lewis’s definitions of acids and bases are the broadest of all:
Lewis acids are electron-pair acceptors.
Lewis bases are electron-pair donors.
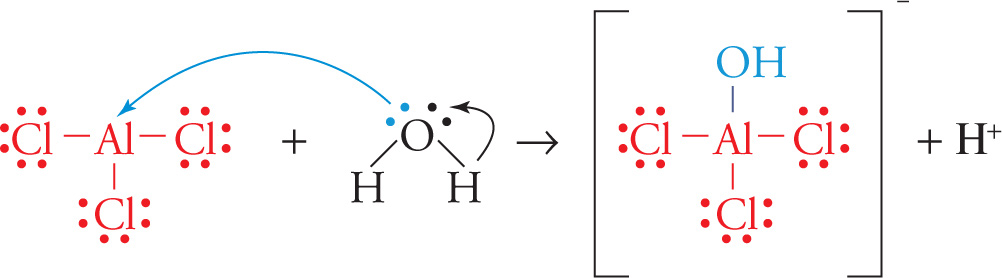
If we consider the reversible reaction below:
AlCl3 + H2O ![]() (AlCl3OH)− + H+
(AlCl3OH)− + H+
then according to the Lewis definition, AlCl3 and H+ are acids because they accept electron pairs; H2O and (AlCl3OH)− are bases because they donate electron pairs. Lewis acid/base reactions frequently result in the formation of coordinate covalent bonds, as discussed in Chapter 5. For example, in the reaction above, water acts as a Lewis base since it donates both of the electrons involved in the coordinate covalent bond between OH− and AlCl3. AlCl3 acts as a Lewis acid, since it accepts the electrons involved in this bond.
11.2 CONJUGATE ACIDS AND BASES
When a Brønsted-Lowry acid donates an H+, the remaining structure is called the conjugate base of the acid. Likewise, when a Brønsted-Lowry base bonds with an H+ in solution, this new species is called the conjugate acid of the base. To illustrate these definitions, consider this reaction:
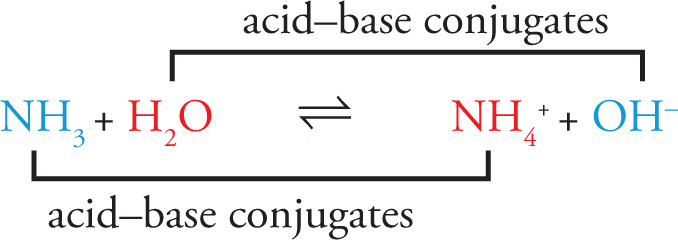
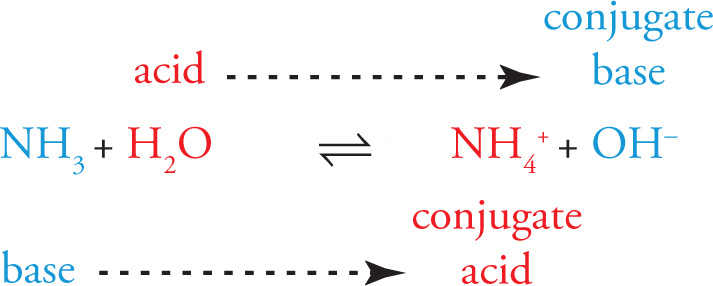
Considering only the forward direction, NH3 is the base and H2O is the acid. The products are the conjugate acid and conjugate base of the reactants: NH4+ is the conjugate acid of NH3, and OH− is the conjugate base of H2O:
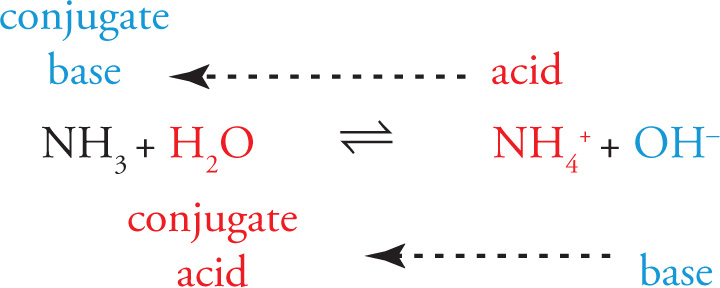
Now consider the reverse reaction in which NH4+ is the acid and OH− is the base. The conjugates are the same as for the forward reaction: NH3 is the conjugate base of NH4+, and H2O is the conjugate acid of OH−:
The difference between a Brønsted-Lowry acid and its conjugate base is that the base is missing an H+. The difference between a Brønsted-Lowry base and its conjugate acid is that the acid has an extra H+.
forming conjugates:
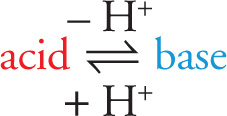
Example 11-1: Which one of the following can behave as a Brønsted-Lowry acid but not a Lewis acid?
A) CF4
B) NaAlCl4
C) HF
D) Br2
Solution: A Brønsted-Lowry acid donates an H+, while a Lewis acid accepts a pair of electrons. Since a Brønsted-Lowry acid must have an H in the first place, only choice C can be the answer.
Example 11-2: What is the conjugate base of HBrO (hypobromous acid)?
A) H+
B) H2BrO2
C) H2BrO+
D) BrO−
Solution: To form the conjugate base of an acid, simply remove an H+. Therefore, the conjugate base of HBrO is BrO−, choice D.
11.3 THE STRENGTHS OF ACIDS AND BASES
Brønsted-Lowry acids can be placed into two big categories: strong and weak. Whether an acid is strong or weak depends on how completely it ionizes in water. A strong acid is one that dissociates completely (or very nearly so) in water; hydrochloric acid, HCl, is an example:
HCl(aq) + H2O(l) → H3O+(aq) + Cl−(aq)
This reaction goes essentially to completion.
On the other hand, hydrofluoric acid, HF, is an example of a weak acid, since its dissociation in water,
HF(aq) + H2O(l) ![]() H3O+(aq) + F−(aq)
H3O+(aq) + F−(aq)
does not go to completion; most of the HF remains undissociated.
If we use HA to denote a generic acid, its dissociation in water has the form
HA(aq) + H2O(l) ![]() H3O+(aq) + A−(aq)
H3O+(aq) + A−(aq)
The strength of the acid is directly related to how much the products are favored over the reactants. The equilibrium expression for this reaction is
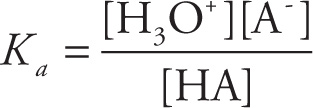
This is written as Ka, rather than Keq, to emphasize that this is the equilibrium expression for an acid-dissociation reaction. In fact, Ka is known as the acid-ionization (or acid-dissociation) constant of the acid (HA). If Ka > 1, then the products are favored, and we say the acid is strong; if Ka < 1 then the reactants are favored and the acid is weak. We can also rank the relative strengths of acids by comparing their Ka values: The larger the Ka value, the stronger the acid; the smaller the Ka value, the weaker the acid.
The acids for which Ka > 1—the strong acids—are so few that you should memorize them:
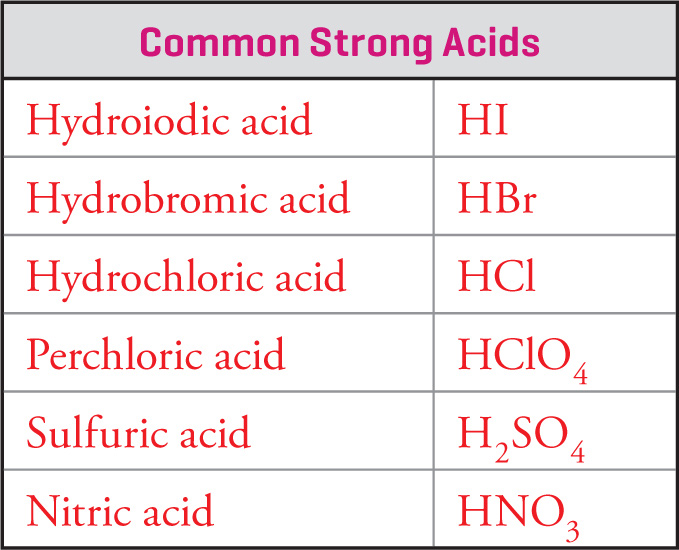
The values of Ka for these acids are so large that most tables of acid ionization constants don’t even list them. On the MCAT, you may assume that any acid that’s not in this list is a weak acid. (Other acids that fit the definition of strong are so uncommon that it’s very unlikely they’d appear on the test. For example, HClO3 has a pKa of —1, and could be considered strong, but it is definitely one of the weaker strong acids and is not likely to appear on the MCAT.)
Example 11-3: In a 1 M aqueous solution of boric acid (H3BO3, Ka = 5.8 × 10−10), which of the following species will be present in solution in the greatest quantity?
A) H3BO3
B) H2BO3−
C) HBO32—
D) H3O+
Solution: The equilibrium here is H3BO3(aq) + H2O(l) ![]() H3O+(aq) + H2BO3−(aq). Boric acid is a weak acid (it’s not on the list of strong acids), so the equilibrium lies to the left (also, notice how small its Ka value is). So, there’ll be very few H3O+ or H2BO3− ions in solution but plenty of undissociated H3BO3. The answer is A.
H3O+(aq) + H2BO3−(aq). Boric acid is a weak acid (it’s not on the list of strong acids), so the equilibrium lies to the left (also, notice how small its Ka value is). So, there’ll be very few H3O+ or H2BO3− ions in solution but plenty of undissociated H3BO3. The answer is A.
Example 11-4: Of the following, which statement best explains why HF is a weak acid, but HCl, HBr, and HI are strong acids?
A) F has a greater ionization energy than Cl, Br, or I.
B) F has a larger radius than Cl, Br, or I.
C) F− has a larger radius than Cl−, Br−, I−.
D) F− has a smaller radius than Cl−, Br−, I−.
Solution: F is smaller than Cl, Br, or I (eliminating choices B and C). Ionization energy is associated with forming a cation from a neutral atom, and has no bearing here. Choice D is therefore correct. The more stable an acid’s conjugate base is, the stronger the acid. Larger anions are better able to spread out their negative charge, making them more stable. HF is the weakest of the H-X acids because it has the least stable conjugate base due to its size.
Example 11-5: Of the following acids, which one would dissociate to the greatest extent (in water)?
A) HCN (hydrocyanic acid), Ka = 6.2 × 10−10
B) HNCO (cyanic acid), Ka = 3.3 × 10−4
C) HClO (hypochlorous acid), Ka = 2.9 × 10−8
D) HBrO (hypobromous acid), Ka = 2.2 × 10−9
Solution: The acid that would dissociate to the greatest extent would have the greatest Ka value. Of the choices given, HNCO (choice B) has the greatest Ka value.
We can apply the same ideas as above to identify strong and weak bases. If we use B to denote a generic base, its dissolution in water has the form
B(aq) + H2O(l) ![]() HB+(aq) + OH−(aq)
HB+(aq) + OH−(aq)
The strength of the base is directly related to how much the products are favored over the reactants. If we write the equilibrium constant for this reaction, we get
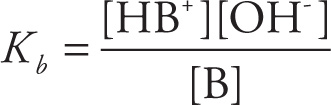
This is written as Kb, rather than Keq, to emphasize that this is the equilibrium expression for a base-dissociation reaction. In fact, Kb is known as the base-ionization (or base-dissociation) constant. We can rank the relative strengths of bases by comparing their Kb values: The larger the Kb value, the stronger the base; the smaller the Kb value, the weaker the base.
For the MCAT and general chemistry, you should know about the following strong bases that may be used in aqueous solutions:
|
Common Strong Bases |
Group 1 hydroxides (For example, NaOH) |
Group 1 oxides (For example, Li2O) |
Some group 2 hydroxides (Ba(OH)2, Sr(OH)2, Ca(OH)2) |
Metal amides (For example, NaNH2) |
Weak bases include ammonia (NH3) and amines, as well as the conjugate bases of many weak acids, as we’ll discuss on the following page.
The Relative Strengths of Conjugate Acid-Base Pairs
Let’s once again look at the dissociation of HCl in water:
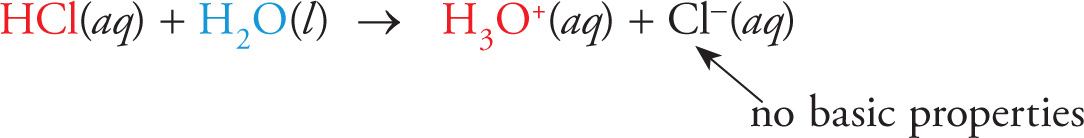
The chloride ion (Cl−) is the conjugate base of HCl. Since this reaction goes to completion, there must be no reverse reaction. Therefore, Cl− has no tendency to accept a proton and thus does not act as a base. The conjugate base of a strong acid has no basic properties in water.
On the other hand, hydrofluoric acid, HF, is a weak acid since its dissociation is not complete:
HF(aq) + H2O(l) ![]() H3O+(aq) + F−(aq)
H3O+(aq) + F−(aq)
Since the reverse reaction does take place to a significant extent, the conjugate base of HF, the fluoride ion, F−, does have some tendency to accept a proton, and so behaves as a weak base. The conjugate base of a weak acid is a weak base.
In fact, the weaker the acid, the more the reverse reaction is favored, and the stronger its conjugate base. For example, hydrocyanic acid (HCN) has a Ka value of about 5 × 10−10, which is much smaller than that of hydrofluoric acid (Ka ≈ 7 × 10−4). Therefore, the conjugate base of HCN, the cyanide ion, CN−, is a stronger base than F−.
The same ideas can be applied to bases:
1) The conjugate acid of a strong base has no acidic properties in water. For example, the conjugate acid of LiOH is Li+, which does not act as an acid in water.
2) The conjugate acid of a weak base is a weak acid (and the weaker the base, the stronger the conjugate acid). For example, the conjugate acid of NH3 is NH4+, which is a weak acid.
Example 11-6: Of the following anions, which is the strongest base?
A) I−
B) CN−
C) NO3−
D) Br−
Solution: Here’s another way to ask the same question: Which of the following anions has the weakest conjugate acid? Since HI, HNO3, and HBr are all strong acids, while HCN is a weak acid, CN− (choice B) has the weakest conjugate acid, and is thus the strongest base.
Example 11-7: Of the following, which acid has the weakest conjugate base?
A) HClO4
B) HCOOH
C) H3PO4
D) H2CO3
Solution: Here’s another way to ask the same question: Which of the following acids is the strongest? Thought about this way, the answer’s easy. Perchloric acid, choice A, is the only strong acid in the list.
Amphoteric Substances
Take a look at the dissociation of carbonic acid (H2CO3), a weak acid:
H2CO3(aq) + H2O(l) ![]() H3O+(aq) + HCO3−(aq) (Ka = 4.5 × 10−7)
H3O+(aq) + HCO3−(aq) (Ka = 4.5 × 10−7)
The conjugate base of carbonic acid is HCO3−, which also has an ionizable proton. Carbonic acid is said to be polyprotic, because it has more than one proton to donate.
Let’s look at how the conjugate base of carbonic acid dissociates:
HCO3−(aq) + H2O(l) ![]() H3O+(aq) + CO32−(aq) (Ka = 4.8 × 10−11)
H3O+(aq) + CO32−(aq) (Ka = 4.8 × 10−11)
In the first reaction, HCO3− acts as a base, but in the second reaction it acts as an acid. Whenever a substance can act as either an acid or a base, we say that it is amphoteric. The conjugate base of a weak polyprotic acid is always amphoteric, because it can either donate or accept another proton. Also notice that HCO3− is a weaker acid than H2CO3; in general, every time a polyprotic acid donates a proton, the resulting species will be a weaker acid than its predecessor.
11.4 THE ION-PRODUCT CONSTANT OF WATER
Water is amphoteric. It reacts with itself in a Brønsted-Lowry acid-base reaction, one molecule acting as the acid, the other as the base:
H2O(l) + H2O(l) ![]() H3O+(aq) + OH−(aq)
H3O+(aq) + OH−(aq)
This is called the autoionization (or self-ionization) of water. The equilibrium expression is
Kw = [H3O+][OH−]
This is written as Kw, rather than Keq, to emphasize that this is the equilibrium expression for the autoionization of water; Kw is known as the ion-product constant of water. Only a very small fraction of the water molecules will undergo this reaction, and it’s known that at 25°C,
Kw = 1.0 × 10−14
(Like all other equilibrium constants, Kw varies with temperature; it increases as the temperature increases. However, because 25°C is so common, this is the value you should memorize.) Since the number of H3O+ ions in pure water will be equal to the number of OH− ions, if we call each of their concentrations x, then x2 = Kw, which gives x = 1 × 10−7. That is, the concentration of both types of ions in pure water is 1 × 10−7 M. (In addition, Kw is constant at a given temperature, regardless of the H3O+ concentration.)
If the introduction of an acid increases the concentration of H3O+ ions, then the equilibrium is disturbed, and the reverse reaction is favored, decreasing the concentration of OH− ions. Similarly, if the introduction of a base increases the concentration of OH− ions, then the equilibrium is again disturbed; the reverse reaction is favored, decreasing the concentration of H3O+ ions. However, in either case, the product of [H3O+] and [OH−] will remain equal to Kw.
For example, suppose we add 0.002 moles of HCl to water to create a 1-liter solution. Since the dissociation of HCl goes to completion (it’s a strong acid), it will create 0.002 moles of H3O+ ions, so [H3O+] = 0.002 M. Since H3O+ concentration has been increased, we expect the OH− concentration to decrease, which it does:
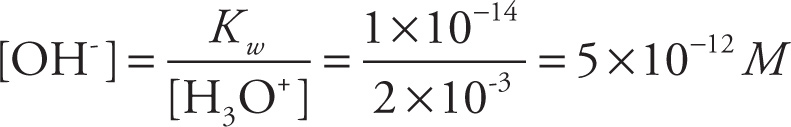
11.5 pH
The pH scale measures the concentration of H+ (or H3O+) ions in a solution. Because the molarity of H+ tends to be quite small and can vary over many orders of magnitude, the pH scale is logarithmic:
pH = − log[H+]
This formula implies that [H+] = 10−pH. Since [H+] = 10−7 M in pure water, the pH of water is 7. At 25°C, this defines a pH neutral solution. If [H+] is greater than 10−7 M, then the pH will be less than 7, and the solution is said to be acidic. If [H+] is less than 10−7 M, the pH will be greater than 7, and the solution is basic (or alkaline). Notice that a low pH means a high [H+] and the solution is acidic; a high pH means a low [H+] and the solution is basic.
pH > 7 |
basic solution |
pH = 7 |
neutral solution |
pH < 7 |
acidic solution |
The range of the pH scale for most solutions falls between 0 and 14, but some strong acids and bases extend the scale past this range. For example, a 10 M solution of HCl will fully dissociate into H+ and Cl−. Therefore, the [H+] = 10 M, and the pH = −1.
An alternate measurement expresses the acidity or basicity in terms of the hydroxide ion concentration, [OH−], by using pOH. The same formula applies for hydroxide ions as for hydrogen ions.
pOH = − log[OH−]
This formula implies that [OH−] = 10−pOH.
Acids and bases are inversely related: the greater the concentration of H+ ions, the lower the concentration of OH− ions, and vice versa. Since [H+][OH−] = 10−14 at 25°C, the values of pH and pOH satisfy a special relationship at 25°C:
pH + pOH = 14
So, if you know the pOH of a solution, you can find the pH, and vice versa. For example, if the pH of a solution is 5, then the pOH must be 9. If the pOH of a solution is 2, then the pH must be 12.
On the MCAT, it will be helpful to be able to figure out the pH even in cases where the H+ concentration isn’t exactly equal to the whole-number power of 10. In general, if y is a number between 1 and 10, and you’re told that [H+] = y × 10−n (where n is a whole number) then the pH will be between (n — 1) and n. For example, if [H+] = 6.2 × 10−5, then the pH is between 4 and 5.
Relationships Between Conjugates
pKa and pKb
The definitions of pH and pOH both involved the opposite of a logarithm. In general, “p” of something is equal to the —log of that something. Therefore, the following definitions won’t be surprising:
pKa = −log Ka
pKb = −log Kb
Because H+ concentrations are generally very small and can vary over such a wide range, the pH scale gives us more convenient numbers to work with. The same is true for pKa and pKb. Remember that the larger the Ka value, the stronger the acid. Since “p” means “take the negative log of…,” the lower the pKa value, the stronger the acid. For example, acetic acid (CH3COOH) has a Ka of 1.75 × 10−5, and hypochlorous acid (HClO) has a Ka of 2.9 × 10−8. Since the Ka of acetic acid is larger than that of hypochlorous acid, we know this means that more molecules of acetic acid than hypochlorous acid will dissociate into ions in aqueous solution. In other words, acetic acid is stronger than hypochlorous acid. The pKa of acetic acid is 4.8, and the pKa of hypochlorous acid is 7.5. The acid with the lower pKa value is the stronger acid. The same logic applies to pKb: the lower the pKb value, the stronger the base.
Ka and Kb
Let’s now look at the relationship between the Ka and the Kb for an acid-base conjugate pair by working through an example question. Let Ka be the acid-dissociation constant for formic acid (HCOOH) and let Kb stand for the base-dissociation constant of its conjugate base (the formate ion, HCOO−). If Ka is equal to 5.6 × 10−11, what is Ka × Kb?
The equilibrium for the dissociation of HCOOH is
HCOOH(aq) + H2O(l) ![]() H3O+(aq) + HCOO−(aq)
H3O+(aq) + HCOO−(aq)
so
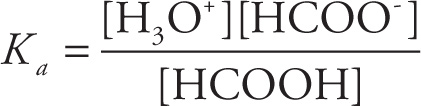
The equilibrium for the dissociation of HCOO− is
HCOO−(aq) + H2O(l) ![]() HCOOH(aq) + OH−(aq)
HCOOH(aq) + OH−(aq)
so
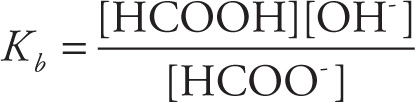
Therefore,
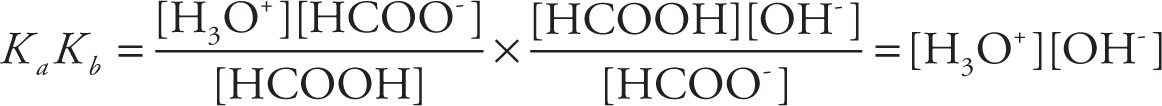
We now immediately recognize this product as Kw, the ion-product constant of water, whose value (at 25°C) is 1 × 10−14.
This calculation wasn’t special for HCOOH; we can see that the same thing will happen for any acid and its conjugate base. So, for any acid-base conjugate pair, we’ll have
KaKb = Kw = 1 × 10−14
This gives us a way to quantitatively relate the strength of an acid and its conjugate base. For example, the value of Ka for HF is about 7 × 10−4; therefore, the value of Kb for its conjugate base, F−, is about 1.4 × 10−11. For HCN, Ka ≈ 5 × 10−10, so Kb for CN− is 2 × 10−5.
It also follows from our definitions and logarithm algebra that for an acid-base conjugate pair at 25°C, we’ll have
pKa + pKb = 14
Example 11-8: Of the following liquids, which one contains the lowest concentration of H3O+ ions?
A) Lemon juice (pH = 2.3)
B) Blood (pH = 7.4)
C) Seawater (pH = 8.5)
D) Coffee (pH = 5.1)
Solution: Since pH = — log [H3O+], we know that [H3O+] = 1/10pH. This fraction is smallest when the pH is greatest. Of the choices given, seawater (choice C) has the highest pH.
Example 11-9: What is the pH of a solution at 25°C whose hydroxide ion concentration is 1 × 10−4 M?
Solution: Since pOH = −log[OH−], we know that pOH = 4. Therefore, the pH is 10.
Example 11-10: Orange juice has a pH of 3.5. What is its [H+]?
Solution: Because pH = −log[H+], we know that [H+] = 10−pH. For orange juice, then, we have [H+] = 10−3.5 = 100.5—4 = 100.5 × 10−4 = ![]() × 10−4 ≈ 3.2 × 10−4 M.
× 10−4 ≈ 3.2 × 10−4 M.
Example 11-11: If 99% of the H3O+ ions are removed from a solution whose pH was originally 3, what will be its new pH?
Solution: If 99% of the H3O+ ions are removed, then only 1% remain. This means that the number of H3O+ ions is now only 1/100 of the original. If [H3O+] is decreased by a factor of 100, then the pH is increased by 2—to pH 5 in this case—since log 100 = 2.
Example 11-12: Given that the self-ionization of water is endothermic, what is the value of the sum pH + pOH at 50°C?
H2O(l) + H2O(l) ![]() H3O+(aq) + OH−(aq)
H3O+(aq) + OH−(aq)
A) Less than 14
B) Equal to 14
C) Greater than 14
D) Cannot determine from the information given
Solution: This is a Le Châtelier’s principle question in disguise. Imagine we start at equilibrium at 25°C; which way would the self-ionization reaction shift if we increase the temperature to 50°C? Since the question tells us this reaction is endothermic, we can consider heat as one of the reactants, and therefore an increase in temperature would cause the system to shift to the right. Shifting to the right means that at equilibrium, [H+] and [OH−] will increase. So pH and pOH will both be lower than 7 at 50°C, and the sum of pH and pOH will be less than 14 at 50°C. Choice A is the correct answer.
pH Calculations
For Strong Acids
Strong acids dissociate completely, so the hydrogen ion concentration will be the same as the concentration of the acid. That means that you can calculate the pH directly from the molarity of the solution. For example, a 0.01 M solution of HCl will have [H+] = 0.01 M and pH = 2.
For Weak Acids
Weak acids come to equilibrium with their dissociated ions. In fact, for a weak acid at equilibrium, the concentration of undissociated acid will be much greater than the concentration of hydrogen ion. To get the pH of a weak acid solution, you need to use the equilibrium expression.
Let’s say you add 0.2 mol of HCN (hydrocyanic acid, a weak acid) to water to create a 1-liter solution, and you want to find the pH. Initially, [HCN] = 0.2 M, and none of it has dissociated. If x moles of HCN are dissociated at equilibrium, then the equilibrium concentration of HCN is 0.2 — x. Now, since each molecule of HCN dissociates into one H+ ion and one CN− ion, if x moles of HCN have dissociated, there’ll be x moles of H+ and x moles of CN−:

(Actually, the initial concentration of H+ is 10−7 M, but it’s so small that it can be neglected for this calculation.) Our goal is to find x, because once we know [H+], we’ll know the pH. So, we set up the equilibrium expression:
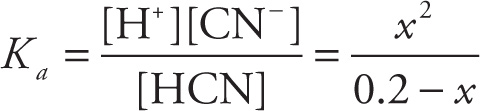
It’s known that the value of Ka for HCN is 4.9 × 10−10. Because the Ka is so small, not that much of the HCN is going to dissociate. (This assumption, that x added to or subtracted from a number is negligible, is always a good one when K < 10−4 [the usual case found on the MCAT].) That is, we can assume that x is going to be a very small number, insignificant compared to 0.2; therefore, the value (0.2 — x) is almost exactly the same as 0.2. By substituting 0.2 for (0.2 — x), we can solve the equation above for x:
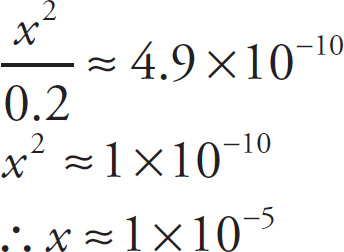
Since [H+] is approximately 1 × 10−5 M, the pH is about 5.
We simplified the computation by assuming that the concentration of hydrogen ion [H+] was insignificant compared to the concentration of undissociated acid [HCN]. Since it turned out that [H+] ≈ 10−5 M, which is much less than [HCN] = 0.2 M, our assumption was valid. On the MCAT, you should always simplify the math wherever possible.
Example 11-13: If 0.7 mol of benzoic acid (C6H5COOH, Ka = 6.6 × 10−5) is added to water to create a 1-liter solution, what will be the pH?
Solution: Initially [C6H5COOH] = 0.7 M, and none of it has dissociated. If x moles of C6H5COOH are dissociated at equilibrium, then the equilibrium concentration of C6H5COOH is 0.7 — x. Now, since each molecule of C6H5COOH dissociates into one H+ ion and one C6H5COO− ion, if x moles of C6H5COOH have dissociated, there’ll be x moles of H+ and x moles of C6H5COO−:
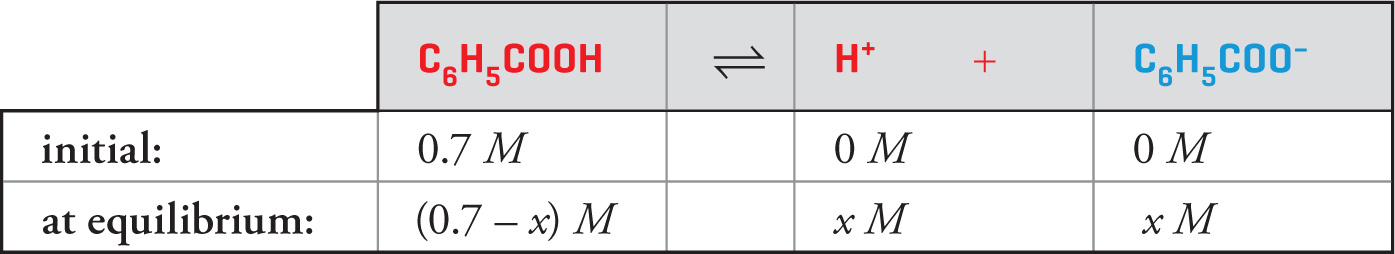
(Again, the initial concentration of H+ is 10−7 M, but it’s so small that it can be neglected.) Our goal is to find x, because once we know [H+], we’ll know the pH. So, we set up the equilibrium expression:
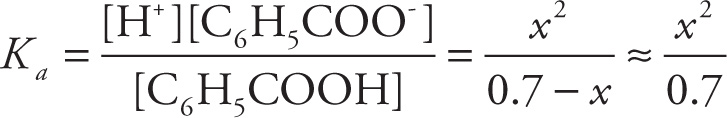
and then solve the equation for x:
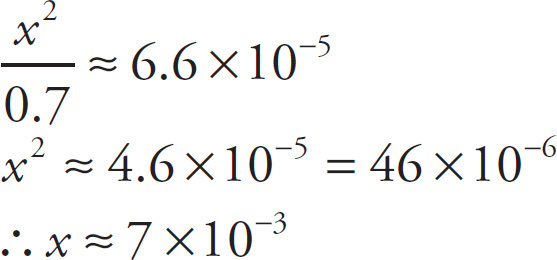
Since [H+] is approximately 7 × 10−3 M ≈ 10−2 M, the pH is a little more than 2, say 2.2.
11.6 NEUTRALIZATION REACTIONS
When an acid and a base are combined, they will react in what is called a neutralization reaction. Oftentimes this reaction will produce a salt and water. Here’s an example:

This type of reaction takes place when, for example, you take an antacid to relieve excess stomach acid. The antacid is a weak base, usually carbonate, that reacts in the stomach to neutralize acid.
If a strong acid and strong base react (as in the example above), the resulting solution will be pH neutral (which is why we call it a neutralization reaction). However, if the reaction involves a weak acid or weak base, the resulting solution will generally not be pH neutral.
No matter how weak an acid or base is, when mixed with an equimolar amount of a strong base or acid, we can expect complete neutralization. It has been found experimentally that all neutralizations have the same exothermic “heat of neutralization,” the energy released from the reaction that is the same for all neutralizations: H+ + OH− → H2O.
As you can see from the reaction above, equal molar amounts of HCl and NaOH are needed to complete the neutralization. To determine just how much base (B) to add to an acidic solution (or how much acid (A) to add to a basic solution) in order to cause complete neutralization, we just use the following formula:
a × [A] × VA = b × [B] × VB
where a is the number of acidic hydrogens per formula unit and b is a constant that tells us how many H3O+ ions the base can accept.
For example, let’s calculate how much 0.1 M NaOH solution is needed to neutralize 40 mL of a 0.3 M HCl solution:
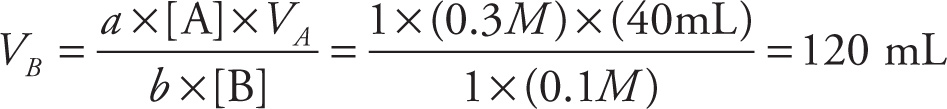
Example 11-14: Binary mixtures of equal moles of which of the following acid-base combinations will lead to a complete (99+%) neutralization reaction?
I. HCl and NaOH
II. HF and NH3
III. HNO3 and NaHCO3
A) I only
B) I and II only
C) II and III only
D) I, II, and III
Solution: Remember, regardless of the strengths of the acids and bases, all neutralization reactions go to completion. Choice D is the correct answer.
11.7 HYDROLYSIS OF SALTS
A salt is an ionic compound, consisting of a cation and an anion. In water, the salt dissociates into ions, and depending on how these ions react with water, the resulting solution will be either acidic, basic, or pH neutral. To make the prediction, we notice that there are essentially two possibilities for both the cation and the anion in a salt:

Whether the salt solution will be acidic, basic, or pH neutral depends on which combination of possibilities (four total) from the diagram above applies. The reaction of a substance—such as a salt or an ion—with water is called a hydrolysis reaction, a more general use of the term since the water molecule may not be split. Let’s look at some examples.
If we dissolve NaCl in water, Na+ and Cl− ions go into solution. Na+ ions are Group 1 ions and do not react with water. Since Cl− is the conjugate base of a strong acid (HCl), it also doesn’t react with water. These ions just become hydrated (surrounded by water molecules). Therefore, the solution will be pH neutral.
How about NH4Cl? In solution it will break into NH4+ and Cl−. The ammonium ion is a stronger acid than water (it’s the conjugate acid of NH3, a weak base), and Cl− will not react with water. As a result, a solution of this salt will be acidic and have a pH less than 7, and NH4Cl is called an acidic salt.
Now let’s consider sodium acetate, Na(CH3COO). In solution it will break into Na+ and CH3COO−. Na+ is a Group 1 cation and does not react with water. However, CH3COO− is a stronger base than water since it’s the conjugate base of acetic acid (CH3COOH), a weak acid. Therefore, a solution of the salt will be basic and have a pH greater than 7, and NaCH3COO is a basic salt.
Finally, let’s consider NH4CN. In solution it will break into NH4+ and CN−. NH4+ is a stronger acid than water, and CN− is a stronger base than water (it’s the conjugate base of HCN, a weak acid). So, which one wins? In a case like this, we need to know the Ka value for the reaction of the cation with water with the Kb value for the reaction of the anion with water and compare these values. Since
NH4+(aq) + H2O(l) ![]() NH3(aq) + H3O+(aq) (Ka = 6.3 × 10−10)
NH3(aq) + H3O+(aq) (Ka = 6.3 × 10−10)
CN−(aq) + H2O(l) ![]() HCN(aq) + OH−(aq) (Kb = 1.6 × 10−5)
HCN(aq) + OH−(aq) (Kb = 1.6 × 10−5)
we see that in this case Kb of CN− > Ka of NH4+, so the forward reaction of the second reaction will dominate the forward reaction of the first reaction, and the solution will be basic.
Example 11-15: Which of the following salts will produce a basic solution when added to pure water?
A) KCl
B) NaClO
C) NH4Cl
D) MgBr2
Solution: NaClO (choice B) will dissociate into Na+ and ClO−. Na+ is a Group 1 cation, so it has no effect on the pH. However, ClO−, the hypochlorite ion, is the conjugate base of a weak acid, HClO (hypochlorous acid). Therefore, the solution will be basic. The salt in choices A will have no effect on the pH, and the salts in choice C and D will leave the solution acidic (since NH4+ is the conjugate acid of a weak base, NH3, and Mg2+ will react with water to form the weak base Mg(OH)2).
Example 11-16: Which of the following is an acidic salt?
A) KNO3
B) SrCl2
C) CuCl2
D) Ba(CH3COO)2
Solution: Cu2+ is a stronger acid than water, so CuCl2 (choice C) is an acidic salt. The salts in choices A and B are neither acidic nor basic, and choice D is a basic salt.
11.8 BUFFER SOLUTIONS
A buffer is a solution that resists changing pH when a small amount of acid or base is added. The buffering capacity comes from the presence of a weak acid and its conjugate base (or a weak base and its conjugate acid) in roughly equal concentrations.
One type of buffer is made from a weak acid and a salt of its conjugate base. To illustrate how a buffer works, let’s look at a specific example and add 0.1 mol of acetic acid (CH3COOH) and 0.1 mol of sodium acetate (NaCH3COO) to water to obtain a 1-liter solution. Since acetic acid is a weak acid (Ka = 1.75 × 10−5), it will partially dissociate to give some acetate (CH3COO−) ions. However, the salt is soluble and will dissociate completely to give plenty of acetate ions. The addition of this common ion will shift the acid dissociation to the left, so the equilibrium concentrations of undissociated acetic acid molecules and acetate ions will be essentially equal to their initial concentrations, 0.1 M.
CH3COOH + H2O ![]() H3O+ + CH3COO−
H3O+ + CH3COO−
Since buffer solutions are designed to resist changes in pH, let’s first figure out the pH of this solution. Writing the expression for the equilibrium constant gives
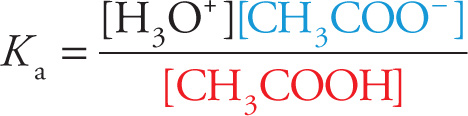
which we can solve for [H3O+]:

Since the equilibrium concentrations of both CH3COOH and CH3COO− are 0.1 M, this equation tells us that
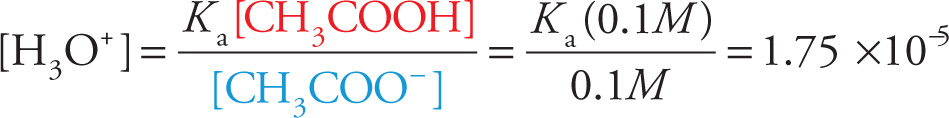
and pH = —log[H3O+], so
pH = −log(1.75 × 10−5)
pH = 4.76
Okay, now let’s see what happens if we add a little bit of strong acid—HCl, for example. If we add, say, 0.005 mol of HCl, it will dissociate completely in solution into 0.005 mol of H+ ions and 0.005 mol of Cl− ions. The Cl− ions will have no effect on the equilibrium, but the added H+ (or H3O+) ions will. Adding a product shifts the equilibrium to the left, and the acetate ions react with the additional H3O+ ions to produce additional acetic acid molecules. As a result, the concentration of acetate ions will drop by 0.005, from 0.1 M to 0.095 M; the concentration of acetic acid will increase by 0.005, from 0.1 M to 0.105 M. Let’s now use Equation (1) above to find the new pH:
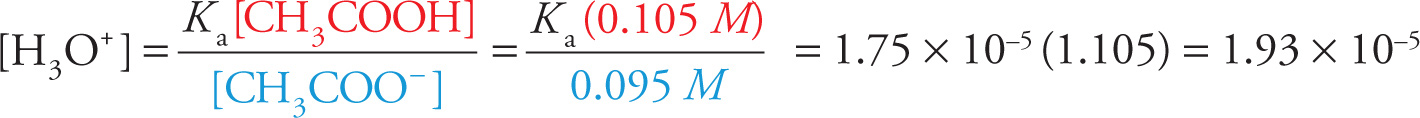
and
pH = −log(1.93 × 10−5)
pH = 4.71
Notice that the pH dropped from 4.76 to 4.71, a decrease of just 0.05. If we had added this HCl to a liter of pure water, the pH would have dropped from 7 to 2.3, a much larger decrease! The buffer solution we created was effective at resisting a large drop in pH because it had enough base (in the form of acetate ions in this case) to neutralize the added acid.
Now let’s see what happens if we add a little bit of strong base—KOH, for example. If we add, say, 0.005 mol of KOH, it will dissociate completely in solution into 0.005 mol of K+ ions and 0.005 mol of OH− ions. The K+ ions will have no effect, but the added OH− ions will shift the equilibrium to the right, since they’ll react with acetic acid molecules to produce more acetate ions (CH3COOH + OH− → CH3COO− + H2O). As a result, the concentration of acetic acid will drop by 0.005, from 0.1 M to 0.095 M; the concentration of acetate ions will increase by 0.005, from 0.1 M to 0.105 M. Let’s again use Equation (1) above to find the new pH:
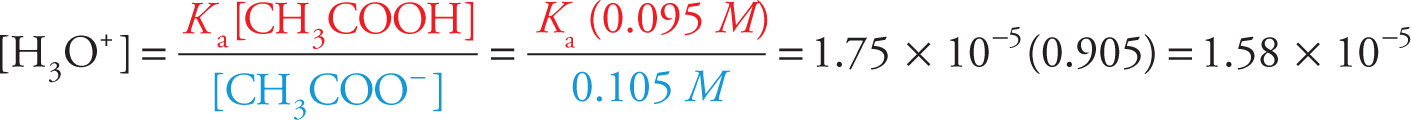
and
pH = −log(1.58 × 10−5)
pH = 4.80
Notice that the pH increased from 4.76 to 4.80, an increase of just 0.04. If we had added this KOH to a liter of pure water, the pH would have increased from 7 to 11.7, a much larger increase! The buffer solution we created was effective at resisting a large rise in pH because it had enough acid to neutralize the added base.
If we generalize Equation (1) to any buffer solution containing a weak acid and a salt of its conjugate base, we get [H3O+] = Ka([weak acid]/[conjugate base]). Taking the −log of both sides give us the
Henderson-Hasselbalch Equation (for acid)
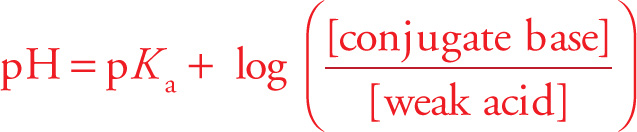
To design a buffer solution, we choose a weak acid whose pKa is as close to the desired pH as possible. An ideal buffer would have [weak acid] = [conjugate base], so pH = pKa. If no weak acid has the exact pKa needed, just adjust the initial concentrations of the weak acid and conjugate base accordingly.
We can also design a buffer solution by choosing a weak base (and a salt of its conjugate acid) such that the pKb value of the base is as close to the desired pOH as possible. The version of the Henderson-Hasselbalch equation in this situation looks like this:
Henderson-Hasselbalch Equation (for base)
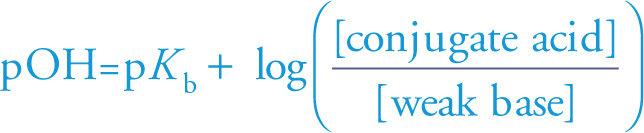
Example 11-17: Which of the following compounds could be added to a solution of HCN to create a buffer?
A) HNO3
B) CaCl2
C) NaCN
D) KOH
Solution: HCN is a weak acid, so we’d look for a salt of its conjugate base, CN−. Choice C, NaCN, is such a salt.
Example 11-18: As hydrogen ions are added to an acidic buffer solution, what happens to the concentrations of undissociated acid and conjugate base?
Solution: The conjugate base, A−, reacts with the added H+ to form HA, so the conjugate base decreases and the undissociated acid increases.
Example 11-19: As hydrogen ions are added to an alkaline buffer solution, what happens to the concentrations of base and conjugate acid?
Solution: The base, B, reacts with the added H+ to form HB+, so the base decreases and the conjugate acid increases.
11.9 INDICATORS
An indicator is a weak acid that undergoes a color change when it’s converted to its conjugate base. Let HA denote a generic indicator. In its non-ionized form, it has a particular color, which we’ll call color #1. When it has donated a proton to become its conjugate base, A−, it has a different color, which we’ll call color #2.
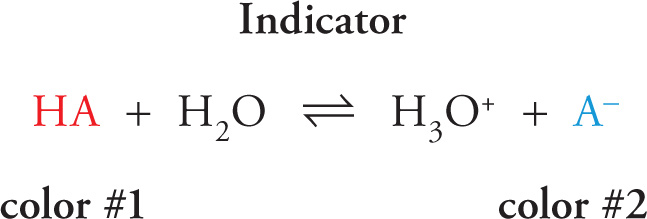
Under what conditions would an indicator change its color? What if an indicator were added to an acidic solution—that is, one whose pH were quite low due to a high concentration of H3O+ ions? Then according to Le Châtelier, the indicator’s equilibrium would shift to the left, and the indicator would display color #1. Conversely, if the indicator were added to a basic solution (that is, one with plenty of OH− ions), the amount of H3O+ would decrease, and the indicator’s equilibrium would be shifted to the right, causing it to display color #2. We can make this discussion a little more precise.
Take the expression for the indicator’s equilibrium constant, Ka = [H3O+][A−]/[HA] and easily rearrange it into
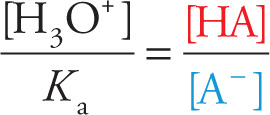
Written this way, we can see that
• If [H3O+] >> Ka, then [HA] >> [A−], so we’d see color #1.
• If [H3O+] ≈ Ka, then [HA] ≈ [A−], so we’d see a mix of colors #1 & #2.
• If [H3O+] << Ka, then [HA] << [A−], so we’d see color #2.
Note that the indicator changes color within a fairly short pH range, about 2 units:
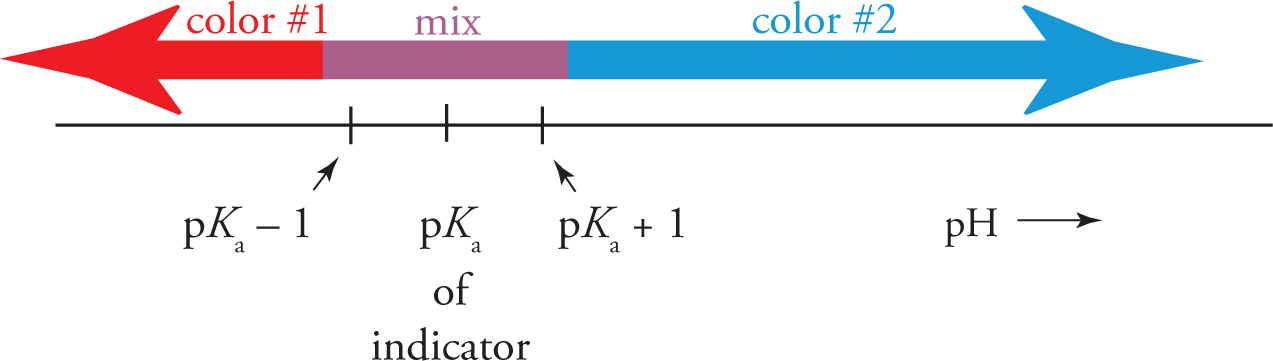
Therefore, if we want our indicator to be useful, we need to select one whose pKa value is convenient for our purposes. For example, phenolphthalein is an indicator with a pKa value of about 9.0. When added to a solution whose pH is less than 8, it remains colorless. However, if the solution’s pH is above 10, it will turn red. (For 8 < pH < 10, the solution will be pink.) Thus, phenolphthalein can be used to differentiate between a solution whose pH is, say, 7 from one whose pH is 11. However, the indicator methyl orange could not distinguish between two such solutions: It would be yellow at pH 7 and yellow at pH 11. Methyl orange has a pKa of about 3.8, so it changes color around pH 4.
Note: The pKa ± 1 range for an indicator’s color change is convenient and typical, but it’s not a hard-and-fast rule. Some indicators (like methyl orange) have a color-change range of only 1.2 (rather than 2) pH units. Also, some indicators have more than just two colors. Polyprotic indicators, like thymol blue and bromocesol green, can change color more than once, and can therefore exhibit more than two distinct colors.
11.10 ACID-BASE TITRATIONS
An acid-base titration is an experimental technique used to determine the identity of an unknown weak acid (or weak base) by determining its pKa (or pKb). Titrations can also be used to determine the concentration of any acid or base solution (whether it be known or unknown). The procedure consists of adding a strong acid (or a strong base) of known identity and concentration—the titrant—to a solution containing the unknown base (or acid). (One never titrates an acid with an acid or a base with a base.) While the titrant is added in small, discrete amounts, the pH of the solution is recorded (with a pH meter).
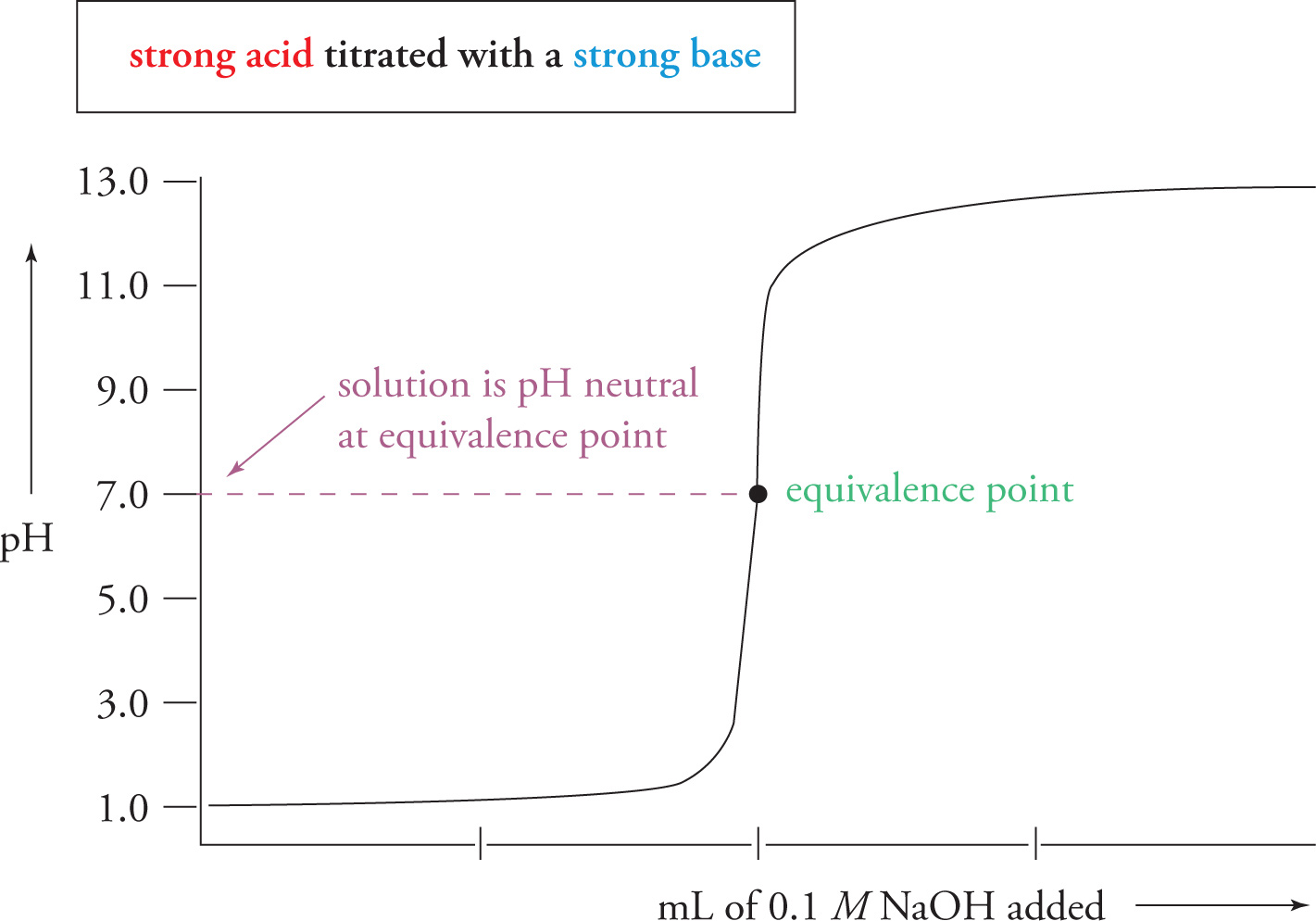
If we plot the data points (the pH value vs. the volume of titrant added), we obtain a graph called a titration curve. Let’s consider a specific example: the titration of HF (a weak acid) with NaOH (a strong base).
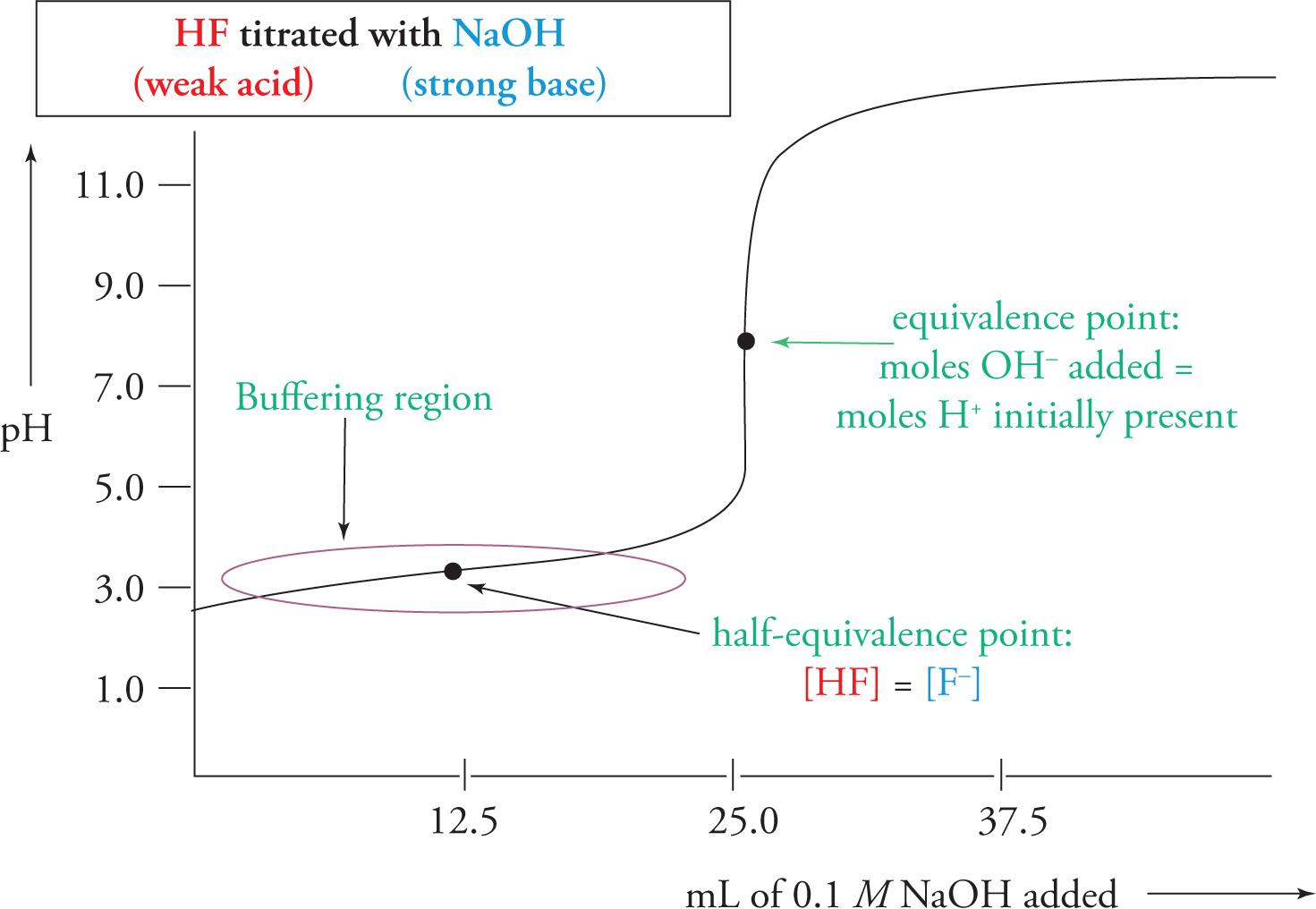
When the amount of titrant added is 0, the pH is of course just the pH of the original, pure solution of HF. Then, as NaOH is added, an equivalent amount of HF will be neutralized according to the reaction
NaOH + HF → Na+ + F− + H2O
As HF is neutralized, the pH will increase. But from the titration curve, we can see that the pH is certainly not increasing very rapidly as we add the first 20 or so mL of NaOH. This should tell you that at the beginning of this titration the solution is behaving as a buffer. As HF is being converted into F−, we are forming a solution that contains a weak acid and its conjugate base. This section of the titration curve, where the pH changes very gradually, is called the buffering domain (or buffering region).
Now, as the experiment continues, the solution suddenly loses its buffering capability and the pH increases dramatically. At some point during this drastic increase, all HF is neutralized and no acid remains in solution. Every new molecule of OH− that is added remains in solution. Therefore, the pH continues to increase rapidly until the OH− concentration in solution is not that much different from the NaOH concentration in the titrant. From here on, the pH doesn’t change very much and the curve levels off.
There is a point during the drastic pH increase at which just enough NaOH has been added to completely neutralize all the HF. This is called the acid-base equivalence point. At this point, we simply have Na+ ions and F− ions in solution. Note that the solution should be basic here. In fact, from what we know about the behavior of conjugates, we can state the following facts about the equivalence point of different titrations:
• For a weak acid (titrated with a strong base), the equivalence point will occur at a pH > 7.
• For a weak base (titrated with a strong acid), the equivalence point will occur at a pH < 7.
• For a strong acid (titrated with a strong base) or for a strong base (titrated with a strong acid), the equivalence point will occur at pH = 7.
Therefore, by just looking at the pH at the equivalence point of our titration, we can tell whether the acid (or base) we were titrating was weak or strong.
Recall the purpose of this titration experiment: to determine the pKa (or pKb) of the unknown weak acid (or weak base). From the titration curve, determine the volume of titrant added at the equivalence point; call it Vat equiv. A key question is this: What’s in solution when the volume of added titrant is 1/2 Vat equiv? Let’s return to our titration of HF by NaOH. We can read from its titration curve that Vat equiv = 25 mL. When the amount of NaOH added was 1/2 Vat equiv = 12.5 mL, the solution consisted of equal concentrations of HF and F−, i.e., enough NaOH was added to convert 1/2 of the HF to F−. (After all, when the amount of titrant added was twice as much, Vat equiv = 25 mL, all of the HF had been converted to F−. So naturally, when 1/2 as much was added, only 1/2 was converted.) Therefore, at this point—called the half-equivalence point—we have
[HF]at half-equiv = [F−]at half-equiv
The Henderson-Hasselbalch equation then tells us that
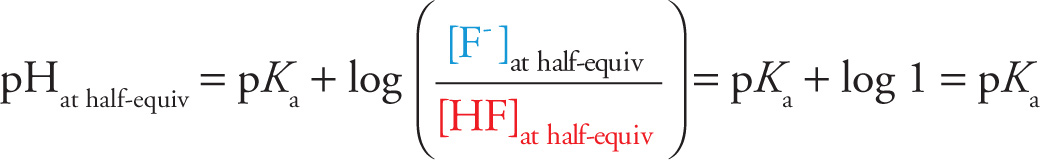
The pKa of HF equals the pH at the half-equivalence point. For our curve, we see that this occurs around pH 3.2, so we conclude that the pKa of HF is about 3.2.
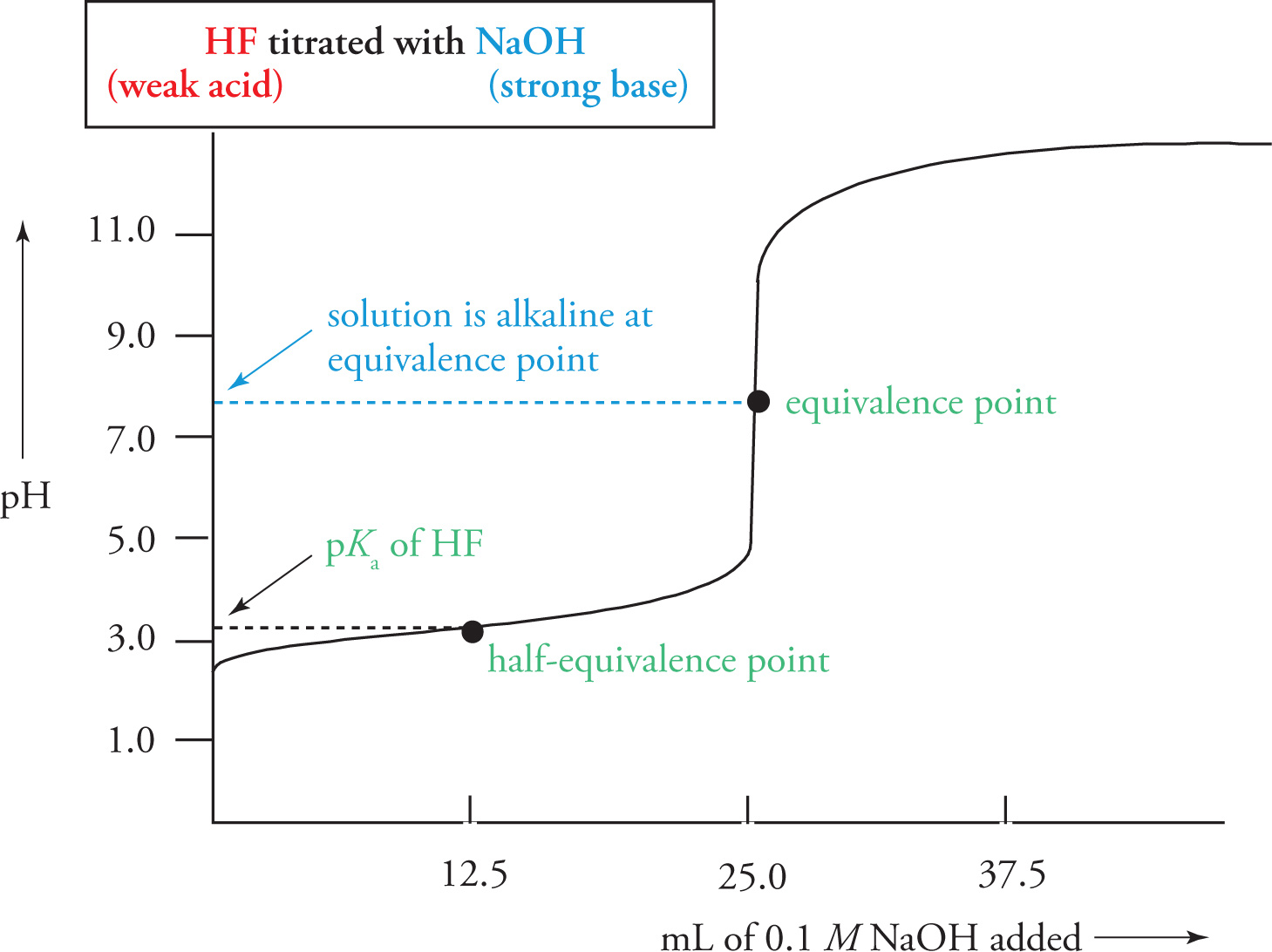
Compare the sample titration curves for a weak base titrated with a strong acid to the one for a weak acid titrated with a strong base (like the one we just looked at). Note the pH at the equivalence point (relative to pH 7) for each curve.
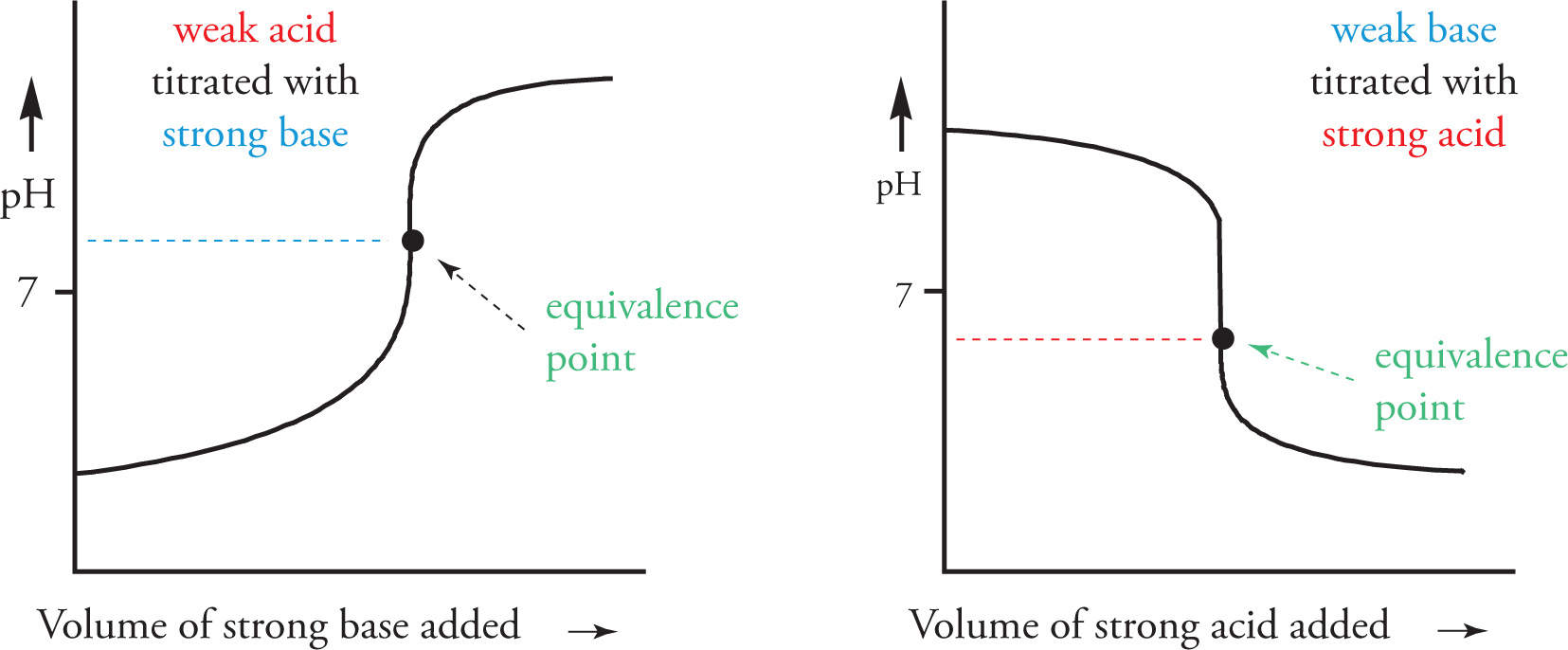
As mentioned above, the titration curve for a strong acid-strong base titration would have the equivalence point at a neutral pH of 7, as shown below.
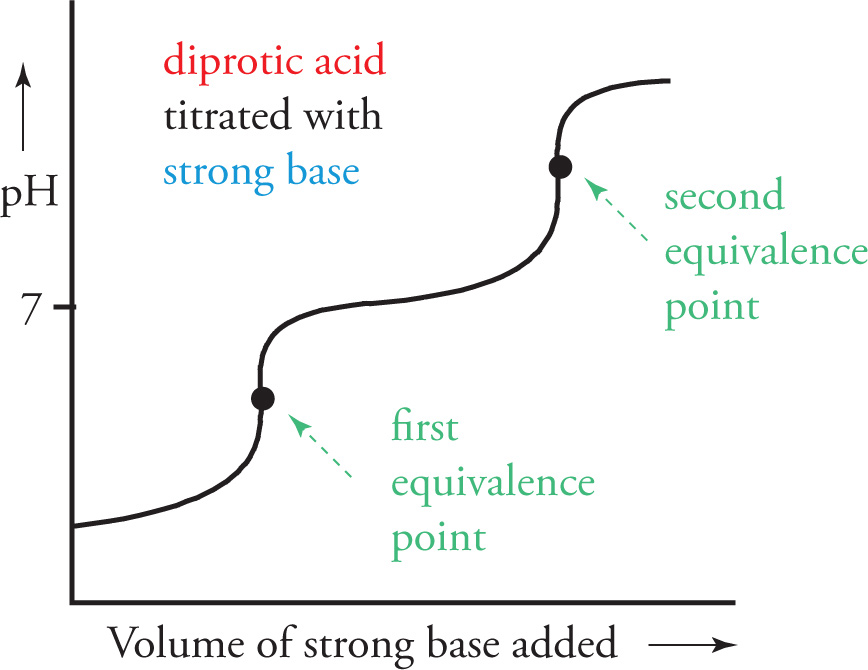
The titration curve for the titration of a polyprotic acid (like H2SO4 or H3PO4) will have more than one equivalence point. The number of equivalence points is equal to the number of ionizable hydrogens the acid can donate.
Example 11-20: A fifty mL solution of HCOOH (formic acid) is titrated with 0.2 M NaOH. The equivalence point is reached when 40 mL of the NaOH solutionhas been added. What was the original concentration of the formic acid solution?
Solution: Using our formula a × [A] × VA = b × [B] × VB, we find that
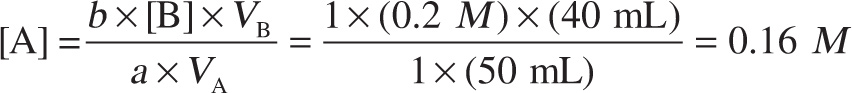
Example 11-21: Methyl red is an indicator that changes from red to yellow in the pH range 4.4—6.2. For which of the following titrations would methyl red be useful for indicating the equivalence point?
A) HCN titrated with KOH
B) NaOH titrated with HI
C) C6H5COOH (benzoic acid) titrated with LiOH
D) C6H5NH2 (aniline) titrated with HNO3
Solution: Since methyl red changes color in a range of acidic pH values, it would be an appropriate indicator for a titration whose equivalence point occurs at a pH less than 7. For a weak base titrated with a strong acid, the equivalence point occurs at a pH less than 7. Only choice D describes such a titration.
Chapter 11 Summary
• Acids are proton donors and electron acceptors; bases are proton acceptors and electron donors.
• Strong acids completely dissociate in water (Ka > 1). You should memorize the list of strong acids and bases.
• The higher the Ka (lower the pKa), the stronger the acid. The higher the Kb (lower the pKb), the stronger the base.
• For any conjugate acid and base pair, KaKb = Kw. Therefore, it follows that the stronger the acid, the weaker its conjugate base. Conjugates of strong acids and bases have no acid/base properties in water.
• Amphoteric substances may act as either acids or bases.
• Water is amphoteric, and autoionizes into OH− and H3O+. The equilibrium constant for the autoionization of water is Kw = [OH−][H3O+]. At 25°C, Kw = 1 × 10−14.
• pH = −log[H3O+]. For a concentration of H3O+ given in a 10−x M notation, simply take the negative exponent to find the pH. The same is true for the relationship between [OH−] and pOH, Ka and pKa, and Kb and pKb.
• At 25°C, pKa + pKb = 14.
• If a salt is dissolved in water and the cation is a stronger acid than water, the resulting solution will have a pH < 7. If the anion is a base stronger than water, the resulting solution will have a pH > 7.
• Buffers resist pH change upon the addition of a small amount of strong acid or base. A higher concentration of buffer resists pH change better than a lower concentration of buffer (that is, the solution has a higher buffering capacity).
• A buffer consists of approximately equal molar amounts of a weak acid and its conjugate base, and maintains a pH close to its pKa.
• The Henderson-Hasselbalch equation can be used to determine the pH of a buffer solution.
• Indicators are weak acids that change color upon conversion to their conjugate base. An indicator changes color in the range +/− 1 pH unit from its pKa.
• In a titration, the equivalence point is the point at which all of the original acid or base has been neutralized.
• When a strong acid is titrated against a weak base, the pH at the equivalence point is < 7. When a strong base is titrated against a weak acid, the pH at the equivalence point is > 7. When a strong base is titrated against a strong acid, the pH at the equivalence point is = 7.
• At the half equivalence point of a titration of a weak plus a strong acid or base, the solution has equal concentrations of acid and conjugate base, and pH = pKa.
CHAPTER 11 FREESTANDING PRACTICE QUESTIONS
1. The pH of a CH3COOH solution is < 7 because when this compound is added to water:
A) CH3COOH donates H+, making [H+] > [OH−].
B) CH3COOH loses OH−, making [H+] < [OH−].
C) CH3COO− deprotonates H2O, increasing [OH−].
D) CH3COOH dissociation increases [H+], thereby increasing Kw.
2. All of the following are amphoteric EXCEPT:
A) HCO3−
B) H2PO4−
C) SO42−
D) HOOCCOO−
3. A graph depicting a titration of a weak acid with a strong base will start at a:
A) high pH and slope downwards with an equivalence pH equal to 7.
B) high pH and slope downwards with an equivalence pH below 7.
C) low pH and slope upwards with an equivalence pH equal to 7.
D) low pH and slope upwards with an equivalence pH above 7.
4. List the following compounds by increasing pKa:
I. H2SO4
II. NH3
III. CH3CH2COOH
IV. HF
A) I < III < II < IV
B) I < IV < III < II
C) III < I < IV < II
D) II < III < IV < I
5. The amino and carboxyl terminals of alanine lose protons according to the following equilibrium:
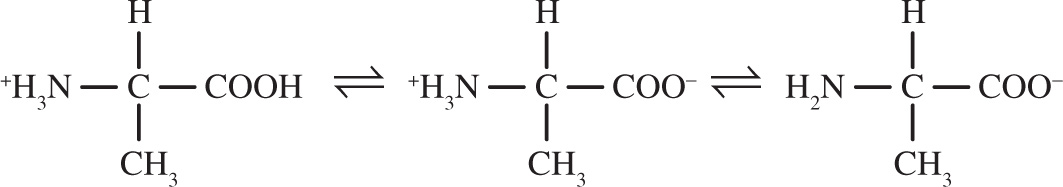
Which of the following indicators would be best used to determine the second equivalence point when alanine is titrated with sodium hydroxide?
A) Methyl violet (pKb = 13.0)
B) Methyl yellow (pKb = 10.5)
C) Thymol blue (pKb = 12.0)
D) Phenolphthalein (pKb = 4.9)
6. The Ka of HSCN is equal to 1 × 10−4. The pH of a HSCN solution:
A) will be approximately 4.
B) will be approximately 10.
C) will increase as [HSCN] increases.
D) cannot be determined from the information given.
7. A 25.0 mL solution of 0.2 M acetic acid (pKa = 4.76) is mixed with 50 mL of 1.0 M sodium acetate (pKb = 9.24). What is the final pH?
A) 4.8
B) 5.8
C) 9.2
D) 10.2
CHAPTER 11 PRACTICE PASSAGE
Blood pH homeostasis is the result of several systems operating within the bloodstream. They collectively maintain blood plasma pH at 7.4, since a drop in pH below 6.8 or rise above 7.8 may result in death.
One component of this system is the enzyme carbonic anhydrase, which catalyzes the conversion of CO2 in the blood to carbonic acid. Carbonic acid, in turn, ionizes to form the carbonic acid-bicarbonate buffer. The interdependence of these reactions is shown below in Equation 1.
CO2(g) + H2O(l) ![]() H2CO3(aq)
H2CO3(aq) ![]() H+(aq) + HCO−3(aq)
H+(aq) + HCO−3(aq)
Equation 1
Uncatalyzed blood CO2 and H+ can be found binding to hemoglobin after oxygen liberation in peripheral tissues. As the blood reaches the lungs these actions reverse themselves; hemoglobin binds with oxygen, releasing the CO2 and H+ ions. The exchange of gases between the lungs and the blood and other tissues in the body is a physiologic process known as respiration.
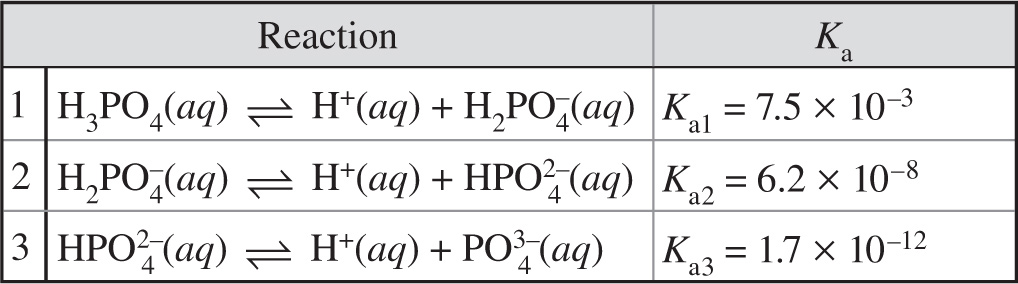
A second system, the phosphoric acid buffer, plays a minor role compared to the carbonic acid-bicarbonate buffer. Phosphoric acid (H3PO4), the primary reactant of this system, is a triprotic acid, which can ionize three protons. This three-step process is illustrated below:
1. Carbonic acid is best described as:
A) amphoteric.
B) polyprotic.
C) a strong acid.
D) the conjugate acid for CO2.
2. If CO2 gas is bubbled continuously in a beaker of water to form carbonic acid, which of the following would be true?
I. Addition of carbonic anhydrase will increase the Keq of the reaction.
II. Carbonic acid will increase in concentration until Keq is reached.
III. Addition of bicarbonate will increase the pH of the system.
A) I only
B) II only
C) III only
D) II and III
3. All of the following statements are true regarding human respiration EXCEPT:
A) when a person’s breathing is hampered by conditions such as asthma or emphysema, the blood [H+] increases.
B) exercise stimulates deeper and more rapid breathing, which increases blood plasma pH.
C) slow, shallow breathing allows CO2 to accumulate in the blood.
D) hyperventilation can result in the loss of too much CO2, causing the accumulation of bicarbonate ions.
4. In the dissociation of phosphoric acid, the trend Ka1 > Ka2 > Ka3 is predominantly due to:
A) an equilibrium shift towards the reactants side in Reactions 2 and 3 due to the release of H+ in Reaction 1.
B) a smaller radius in the H+ liberated in Reaction 1 compared to that in Reactions 2 and 3.
C) a slower rate of reaction after subsequent ionizations.
D) an increasing influence of the anion after subsequent ionizations.
5. What is the relationship between the Ka1 value for phosphoric acid and the Kb1 value for dihydrogen phosphate?
A) Ka1 and Kb1 are inversely related through the dissociation constant for water, Kw.
B) Ka1 and Kb1 are directly related through the dissociation constant for water, Kw.
C) The Ka1 is less than the Kb1.
D) There is no relationship between Ka1 and Kb1.
6. What would be the pH of a solution made from combining 50 mL of 0.030 M acetic acid (Ka = 1.8 × 10−5) and 10 mL of 0.15 M sodium acetate?
A) pH = 1.6
B) pH = 2.5
C) pH = 3.3
D) pH = 4.7
SOLUTIONS TO CHAPTER 11 FREESTANDING PRACTICE QUESTIONS
1. A CH3COOH is acetic acid, a common organic, carboxylic acid. It will dissociate in water to produce H+ and CH3COO−, eliminating choice B. An acidic solution (pH < 7) has more H+ ions in solution than OH− ions, making choice A the best answer. Choice C can be eliminated because if [OH−] were to increase, the pH of the solution would be greater than 7, rather than less than 7. Choice D can be eliminated because the only thing that changes the value of Kw, or any equilibrium constant, is temperature.
2. C An amphoteric substance is one that can act as both an acid and a base. This definition fits choices A, B, and D because they can all donate or accept a proton. Choice C has no protons for donation and cannot be acidic.
3. D A graph showing the titration of a weak acid will start at a low pH and slope upwards as the titrant (in this case a strong base) is added. Therefore, choices A and B cannot be true. As the weak acid and titrant (strong base) react, water and salt are formed as products. The salt will determine the pH at the equivalence point. The conjugate acid of a strong base has no acidic properties and will be neutral in solution. However, the conjugate base of the weak acid will be weakly basic. Because of this, the pH at the equivalence point will be above 7.
4. B A higher pKa means a weaker acid, while a lower pKa means a stronger acid. Since this is a ranking question, start with the extremes. Compound I is a strong acid and will have the lowest pKa, eliminating choices C and D. Compound II is the only base so it will have the largest pKa and choice A can be eliminated.
5. D Alanine is a neutral amino acid with an isoelectric point close to 7. Therefore, the second equivalence point represents when all the ammonium residue of the zwitterion (the middle structure shown in the question) is deprotonated. This must occur at a basic pH. An appropriate indicator will change color if its pKa is ±1 of the pH at this equivalence point. Therefore, the desired indicator should have a pKa > 7, or pKb < 7, making choice D the best answer. Another approach to this question is to recognize that no numerical data are provided and choice D is the only indicator for a basic region. There would be no other reasonable way to choose between choices A, B, and C.
6. D The Ka of an acid is a measure of its ability to dissociate in water, not the pH of a solution (the smaller the Ka the weaker the acid). If we know the [H+] of a solution we can find the pH by finding —log [H+], but we cannot find the pH of a weak acid solution from only the Ka. We must also know the concentration of the acid. Choice A is a trap answer if you confuse pKa with pH. The greater the concentration of an acid, the more H+ ions will be in solution. However, this will decrease the pH of the solution, not increase it (choices B and C can be eliminated). By process of elimination, choice D is the best answer.
7. B The sodium acetate solution will be completely ionized:
NaC2 H3O2 → Na + C2H3 O2−
However, acetic acid will have negligible dissociation in solution:
HC2H3O2 ![]() H+ + C2H3O2− (Ka ≈ 1 × 10−5)
H+ + C2H3O2− (Ka ≈ 1 × 10−5)
Therefore, for the combined solution, it is reasonable to assume that all of the HC2H3O2 is contributed from the acid solution, and all of the C2H3O2− is contributed from the salt solution:
(0.2 M HC2H3O2)(0.025 L) = 5 × 10−3 mol HC2H3O2
(1 M NaC2H3O2)(0.05 L) = 5 × 10−2 mol C2H3O2−
The new volume of 0.075 L cancels out when solving for the pH using the Henderson-Hasselbalch equation:
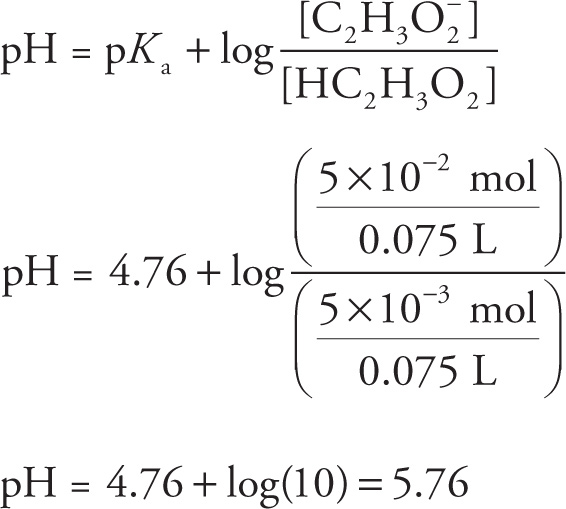
SOLUTIONS TO CHAPTER 11 PRACTICE PASSAGE
1. B Even though the bicarbonate ion is amphoteric and can donate and accept a proton, carbonic acid cannot (eliminate choice A). Choice C is eliminated because carbonic acid is not one of the six strong acids you should know for the MCAT (HI, HBr, HCl, HClO4, H2SO4, HNO3). Choice D is false because carbon dioxide and carbonic acid cannot be a conjugate acid-base pair since they differ by more than one H+. Choice B is correct because carbonic acid has the ability to donate two protons, making it polyprotic.
2. D Addition of a catalyst (such as the enzyme carbonic anhydrase) will simply increase the rate of a reaction. It plays no role in shifting the equilibrium, or changing the equilibrium constant making Item I false (eliminate choice A). As carbon dioxide is bubbled through, carbonic acid will form until its equilibrium concentration is attained, making Item II valid (eliminate choice C). Finally, addition of bicarbonate will shift the carbonic acid equilibrium to the reactant side, consuming H+ in the process. Since the concentration of H+ will decrease, pH will increase, making Item III valid (eliminate choice B).
3. D Choices A and C can be eliminated because they create the same effect. A decrease in breathing rate causes less CO2 to exchange, leading to an increase in CO2 remaining in the blood (i.e., increased CO2 concentration). Consequently, this shifts the equilibria shown in Figure 1 to the right, which results in increased H+ concentration, and decreased pH. Choice B is the opposite effect. Deeper, more rapid breathing expels more CO2, decreasing the CO2 in the blood and increasing pH. Hyperventilation may involve the loss of too much CO2. This loss will shift the equilibria shown in Equation 1 to the left. Loss of bicarbonate ions will result, making choice D the only statement that is NOT true.
4. D Generation of H+ in Reaction 1 is coupled with a release of H2PO4−. Both the product and reactant sides of Reaction 2 are increased proportionally, causing no shift in equilibrium (eliminate choice A). Atomic radius is a function of an atom’s position in the periodic table. Thus, the radius of H+ is the same in all three reactions, eliminating choice B. Equilibrium constants have no relationship to reaction rates, so choice C can be eliminated. The Ka values progressively decrease when removing a proton from a polyprotic acid because it is more difficult to remove a proton from an anion compared to a neutral molecule. In subsequent ionizations, the anion becomes more negative, resulting in greater difficulty liberating a positively charged H+ ion.
5. A The relationship between the Ka value of an acid and the Kb value of its conjugate base is through the dissociation constant of water, where Kw = (Ka)(Kb). Therefore, the relationship between Ka1 and Kb1 is Ka1 = Kw/Kb1; an inverse relationship.
6. D The final solution is composed of (50 mL)(0.03 M) = 1.5 mmol of HC2H3O2 and (10 mL)(0.15 M) = 1.5 mmol of NaC2H3O2 (or 1.5 mmol of C2H3O2−). The total volume will be 60 mL and the starting concentration of acetic acid will be the same as the starting concentration of its conjugate base. Since acetic acid is a weak acid, any subsequent dissociation will be relatively insignificant and the equilibrium concentrations of acid and base will remain approximately the same. When the concentration of the two species in a conjugate pair are equal, the pKa = pH from the Henderson-Hasselbalch equation: pH = pKa + log [conjugate base]/[acid]. The pKa of acetic acid (Ka = 1.8 × 10−5) is approximately 4.7.