MCAT General Chemistry Review - Steven A. Leduc 2015
MCAT G-Chem Formula Sheet
Stoichiometry
Avogadro’s number: NA = 6.02 × 1023
# moles = 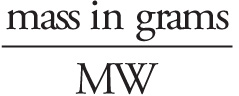
% composition by mass of X = 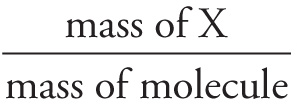 × 100%
× 100%
Mole fraction: Xs = 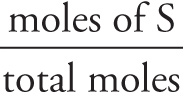
Molarity : M = 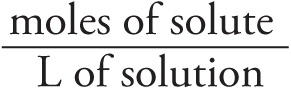
Nuclear and Atomic Chemistry
NA amu (u)= 1 gram
Ephoton = hf = hc/λ
electron energy: En = 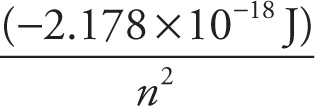 for any 1-electron (Bohr) atom
for any 1-electron (Bohr) atom
Z = # of protons = atomic number, N = # of neutrons
A = Z + N = mass number
Bonding and Intermolecular Forces
formal charge: |
FC = V − ( |
V = (# of valence e− s) |
|
B = (# of bonding e− s) |
|
L = (# of lone-pair e− s) |
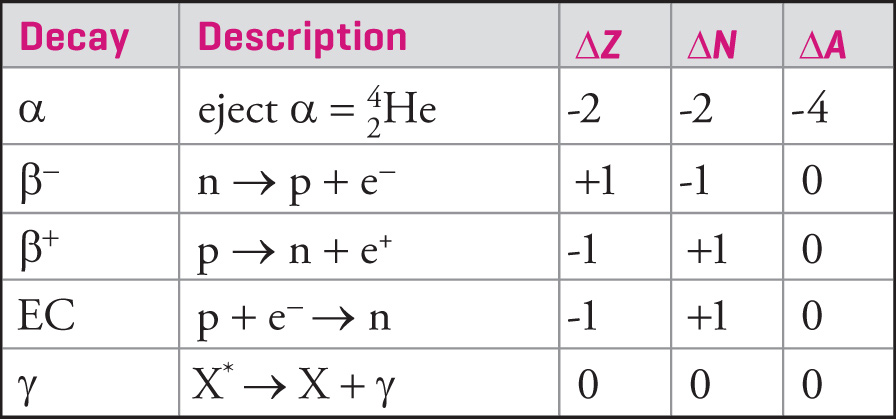
VSEPR Theory
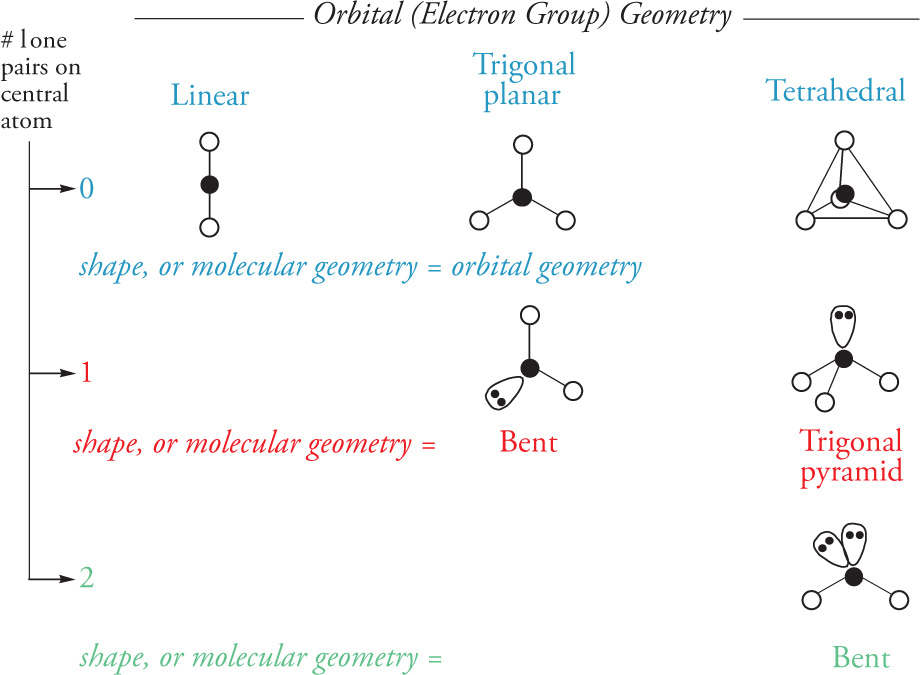
intermolecular forces (D=dipole, I=induced, i=instantaneous):
ion—ion > ion—D > H-bonds > D—D > D—ID > iD—ID (London dispersion)
Periodic Trends
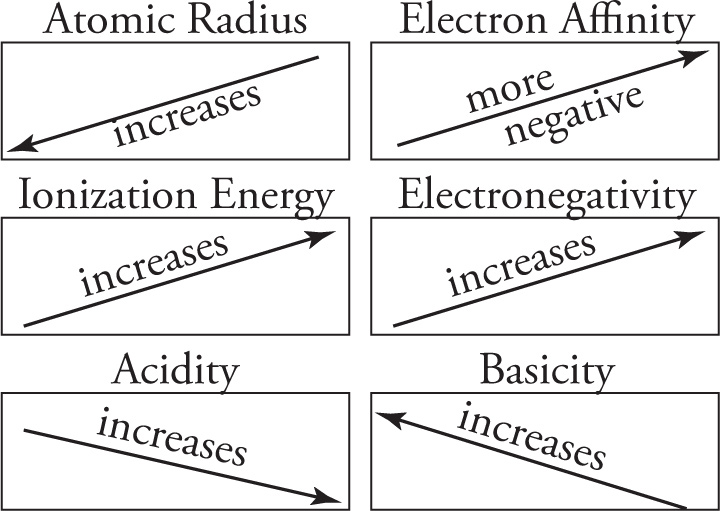
electronegativity of some common atoms:
F > O > (N ≈ Cl) > Br > (I ≈ S ≈ C) > H
Thermodynamics
T (in K) = T°C + 273, 1 cal ≈ 4.2 J, q= heat
q = mc∆T = C∆T (if no phase changes)
q = n∆Hphase change (∆T = 0 during phase change)
enthalpy change: ∆H = heat of rxn at const P
∆H < 0 ![]() exothermic, ∆H > 0
exothermic, ∆H > 0 ![]() endothermic
endothermic
standard state: 1 M, 25°C, 1 atm
∆H°rxn = Σn∆H°f,products − Σn∆H°f,reactants
Laws of Thermodynamics (E = energy, S = entropy):
1) Euniverse is constant. ∆Esystem = Q − W.
2) Spontaneous rxn ![]() ∆Suniverse > 0
∆Suniverse > 0
3) S = 0 for pure crystal at T = 0 K
Gibbs Free Energy: ∆G = ∆H − T∆S [const.T]
∆G < 0 ![]() forward reaction is spontaneous
forward reaction is spontaneous
∆G = 0 ![]() at equilibrium
at equilibrium
∆G > 0 ![]() reverse reaction is spontaneous
reverse reaction is spontaneous
∆G° = −RT ln K ≈ −2.3RT log K
Gases
STP:
T =0 °C = 273 K, P = 1 atm = 760 torr = 760 mmHg
Avogadro’s Law: V ∝ n
Vat STP = n(22.4 L)
Boyle’s Law: V ∝ 1/P (at constant T)
Charles’s Law: V ∝ T (at constant P)
Combined: P1V1 / T1 = P2V2 / T2
Ideal-Gas Law: PV = nRT
van der Waals: 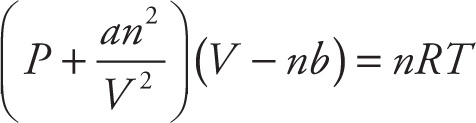
Dalton’s law of partial pressures: Ppot = Σ Pi
Graham’s law of effusion:
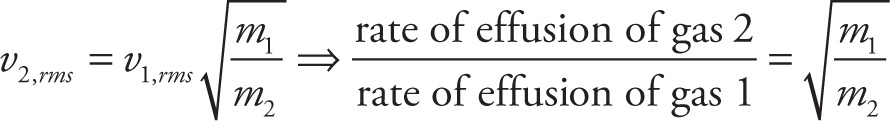
Kinetics
Concentration rate =
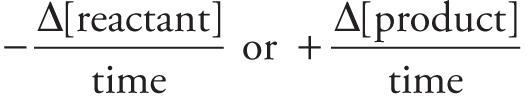
Reaction rate =
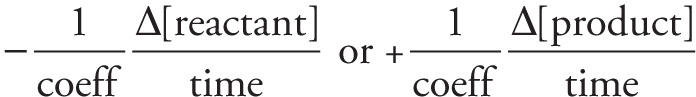
Rate law for rate-determining step:
rate = k[reactant1]coeff1…
Arrhenius equation: k = Ae−Ea/RT
Equilibrium
For generic balanced reaction aA + bB ![]() cC + dD,
cC + dD,
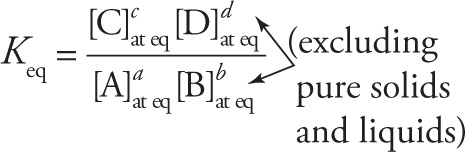
(gas rxns use partial pressures in Kp expression)
Keq is a constant at a given temperature
Keq < 1![]() equilibrium favors reactants
equilibrium favors reactants
Keq > 1 ![]() equilibrium favors products
equilibrium favors products
Reaction quotient: Q = 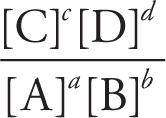
Le Châtelier’s Principle
Q < Keq ![]() rxn proceeds forward
rxn proceeds forward
Q = Keq ![]() rxn at equilibrium
rxn at equilibrium
Q > Keq ![]() rxn proceeds in reverse
rxn proceeds in reverse
Acids and Bases
pH = −log[H+] = −log[H3O+]
pOH = −log[OH−]
Kw = [H+][OH−] = 1 × 10−14 at 25 °C
pH + pOH = 14 at 25 °C
Ka = 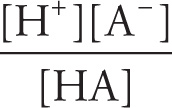
pKa = − log Ka
Kb = 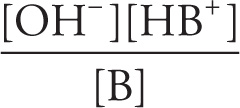
pKb = − log Kb
KaKb = Kw = ion-product constant for water
Henderson-Hasselbalch equations:

acid-base neutralization:
a × [A] × VA = b × [B] × VB
Redox and Electrochemistry
Rules for determining oxidation state (OS):*
1) OS of pure element = 0
2) sum of OS’s = 0 in neutral molecule
sum of OS’s = charge on ion
3) Group 1 metals: OS = +1
Group 2 metals: OS = +2
4) OS of F = −1
5) OS of H = +1
6) OS of O = −2
7) OS of halogens = −1 of O family = −2
If one rule contradicts another, rule higher in list takes precedence.
F = faraday = 96,500 C/mol e−
∆G = −nFEcell
Ecell > 0 ![]() spontaneous
spontaneous
Ecell < 0 ![]() reverse rxn is spontaneous
reverse rxn is spontaneous
Nernst equation: E = E° −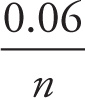 log Q
log Q
Faraday’s Law of Electrolysis:
The amount of chemical charge is proportional to the amount of electricity that flows through the cell.
* These rules work 99 percent of the time.