MCAT General Chemistry Review - Steven A. Leduc 2015
MCAT Math for General Chemistry
Arithmetic, Algebra, and Graphs
PREFACE
The MCAT is primarily a conceptual exam, with little actual mathematical computation. Any math that is on the MCAT is fundamental: just arithmetic, algebra, and trigonometry (and there is virtually no trigonometry in General Chemistry). There is absolutely no calculus. The purpose of this section of the book is to go over some math topics (as they pertain to General Chemistry) with which you may feel a little rusty1.
This text is intended for reference and self-study. Therefore, there are lots of examples, all completely solved. Practice working through these examples and master the fundamentals!
1 For a complete discussion of all the math found on the MCAT, see our book MCAT Physics and Math Review.
Arithmetic, Algebra, and Graphs
13.1 THE IMPORTANCE OF APPROXIMATION
Since you aren’t allowed to use a calculator on the MCAT, you need to practice doing arithmetic calculations by hand again. Fortunately, the amount of calculation you’ll have to do is small, and you’ll also be able to approximate. For example, let’s say you were faced with performing the following calculation:
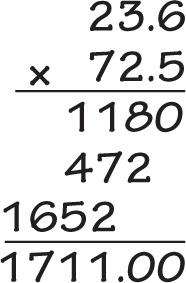
Your first inclination would be to reach for your calculator, but … you don’t have one available. Now what? Realize that on the Chemical and Physical Foundations of Biological Systems section of the MCAT, you have roughly a minute and twenty-five seconds per question, so there simply cannot be questions requiring lengthy, complicated computation. Instead, we’ll figure out a reasonably accurate (and fast) approximation of the value of the expression above:
So, if the answer to an MCAT question was the value of the expression above, and the four answer choices were, say, 1324, 1617, 1711, and 1856, we’d know right away that the answer is 1711. The choices are far enough apart that even with our approximations, we were still able to tell which choice was the correct one. Just as importantly, we didn’t waste time trying to be more precise; it was unnecessary, and it would have decreased the amount of time we had to spend on other questions.
If you find yourself writing out lengthy calculations on your scratch paper when you’re working through MCAT questions that contain some mathematical calculation, it’s important that you recognize that you’re not using your time efficiently. Say to yourself, “I’m wasting valuable time trying to get a precise answer, when I don’t need to be precise.”
Try this one: What’s 1583 divided by 32.1? (You have five seconds. Go.)
For the previous practice exercise, you should have written (or done in your head): 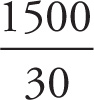 = 50
= 50
13.2 SCIENTIFIC NOTATION, EXPONENTS, AND RADICALS
It’s well known that very large or very small numbers can be handled more easily when they’re written in scientific notation, that is, in the form ± m × 10n, where 1 ≤ m < 10 and n is an integer. For example:
602,000,000,000,000,000,000,000 = 6.02 × 1023
−35,000,000,000 = −3.5 × 1010
0.000000004 = 4 × 10−9
Quantities like these come up all the time in physical problems, so you must be able to work with them confidently. Since a power of ten (the term 10n) is part of every number written in scientific notation, the most important rules for dealing with such expressions are the Laws of Exponents:

Example 13-1: Simplify each of the following expressions, writing your answer in scientific notation:
a) (4 × 10−3)(5 × 109)
b) (4 × 10−3)/(5 × 109)
c) (3 × 10−4)3
d) [(1 × 10−2)/(5 × 10−7)]2
Solution:
a) (4 × 10−3)(5 × 109) = (4)(5) × 10−3+9 = 20 × 106 = 2 × 107
b) (4 × 10−3)/(5 × 109) = (4/5) × 10−3—9 = 0.8 × 10−12 = 8 × 10−13
c) (3 × 10−4)3 = 33 × (10−4)3 = 27 × 10−12 = 2.7 × 10−11
d) [(1 × 10−2)/(5 × 10−7)]2 = (1 × 10−2)2/(5 × 10−7)2 = (1 × 10−4)/(25 × 10−14) = (1/25) × 10−4—(—14) = (4/100) × 1010 = 4 × 108
Another important skill involving numbers written in scientific notation involves changing the power of 10 (and compensating for this change so as not to affect the original number). The approximation carried out in the very first example in this chapter is a good example of this. To find the square root of 5 × 10−7, it is much easier to first rewrite this number as 50 × 10−8, because then the square root is easy:
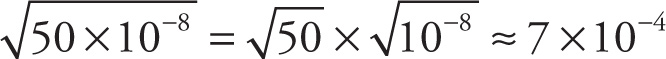
Other examples of this procedure are found in Example 13-1 above; for instance,
20 × 106 = 2 × 107
0.8 × 10−12 = 8 × 10−13
27 × 10−12 = 2.7 × 10−11
In writing 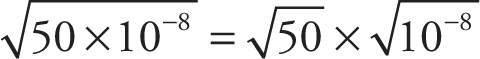 ≈ 7 × 10−4, I used a familiar law of square roots, that the square root of a product is equal to the product of the square roots. Here’s a short list of rules for dealing with radicals:
≈ 7 × 10−4, I used a familiar law of square roots, that the square root of a product is equal to the product of the square roots. Here’s a short list of rules for dealing with radicals:

A couple of remarks about this list: First, Laws 1 and 2 illustrate how to handle square roots, which are the most common. However, the same laws are true even if the index of the root is not 2. [The index of a root (or radical) is the number that indicates the root that’s to be taken; it’s indicated by the little q in front of the radical sign in Law 3. Cube roots are index 3 and written 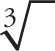 ; fourth roots are index 4 and written
; fourth roots are index 4 and written 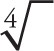 ; and square roots are index 2 and written
; and square roots are index 2 and written 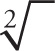 , although we hardly ever write the little 2.] Second, Law 3 provides the link between exponents and radicals.
, although we hardly ever write the little 2.] Second, Law 3 provides the link between exponents and radicals.
Example 13-2: Approximate each of the following expressions, writing your answer in scientific notation:
a) 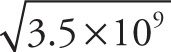
b) 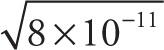
c) 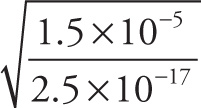
Solution:
a)
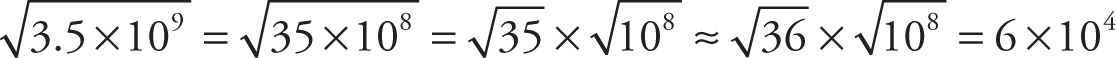
b)
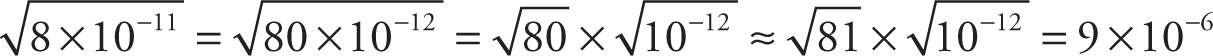
c)
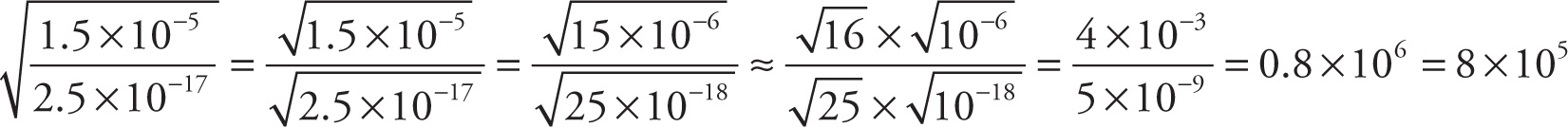
Example 13-3: Approximate each of the following expressions, writing your answer in scientific notation:
a) The mass (in grams) of 4.7 × 1024 molecules of CCl4:
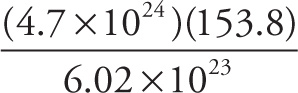
b) The electrostatic force (in newtons) between the proton and electron in the ground state of hydrogen:
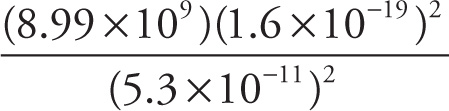
Solution:
a)
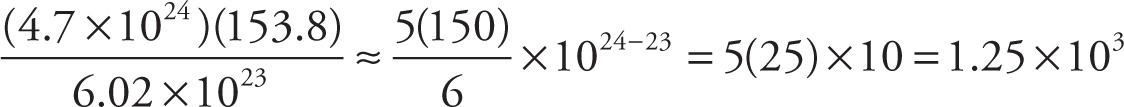
b)
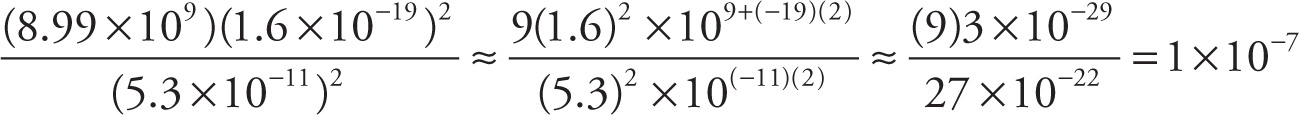
13.3 FRACTIONS, RATIOS, AND PERCENTS
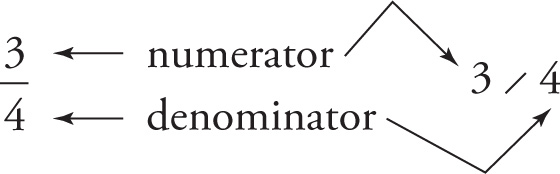
A fraction indicates a division; for example, 3/4 means 3 divided by 4. The number above (or to the left of) the fraction bar is the numerator, and the number below (or to the right) of the fraction bar is called the denominator.
Our quick review of the basic arithmetic operations on fractions begins with the simplest rule: the one for multiplication:
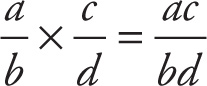
In words, just multiply the numerators and then, separately, multiply the denominators.
Example 13-4: What is 4/9 times 2/5?
Solution:
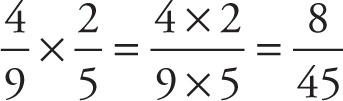
The rule for dividing fractions is based on the reciprocal. If a ≠ 0, then the reciprocal of a/b is simply b/a; that is, to form the reciprocal of a fraction, just flip it over. For example, the reciprocal of 3/4 is 4/3; the reciprocal of —2/5 is —5/2; the reciprocal of 3 is 1/3; and the reciprocal of —1/4 is —4. (The number 0 has no reciprocal.) As a result of this definition, we have the following basic fact: The product of any number and its reciprocal is 1.
Example 13-5: Find the reciprocal of each of these numbers:
a) 2.25
b) 5 × 10−4
c) 4 × 105
Solution:
a) 2.25 is equal to 2 + (1/4), which is 9/4. The reciprocal of 9/4 is 4/9.
b) 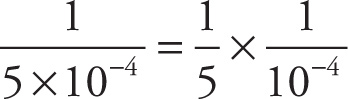 = 0.2 × 104 = 2 × 103
= 0.2 × 104 = 2 × 103
c) 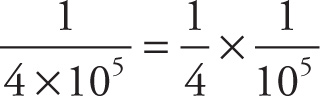 = 0.25 × 10−5 = 2.5 × 10−6
= 0.25 × 10−5 = 2.5 × 10−6
Now, in words, the rule for dividing fractions reads: multiply by the reciprocal of the divisor. That is, flip over whatever you’re dividing by, and then multiply:
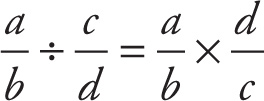
Example 13-6: What is 4/9 divided by 2/5?
Solution:
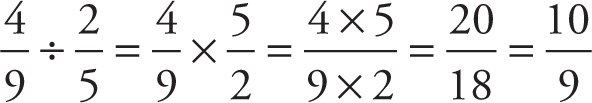
Finally, we turn to addition and subtraction. In elementary and junior high school, you were probably taught to find a common denominator (preferably, the least common denominator, known as the LCD), rewrite each fraction in terms of this common denominator, then add or subtract the numerators. If a common denominator is easy to spot, this may well be the fastest way to add or subtract fractions:
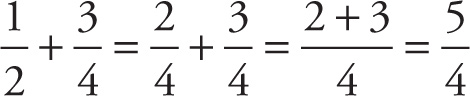
However, it’s now time to learn the grown-up way to add or subtract fractions:
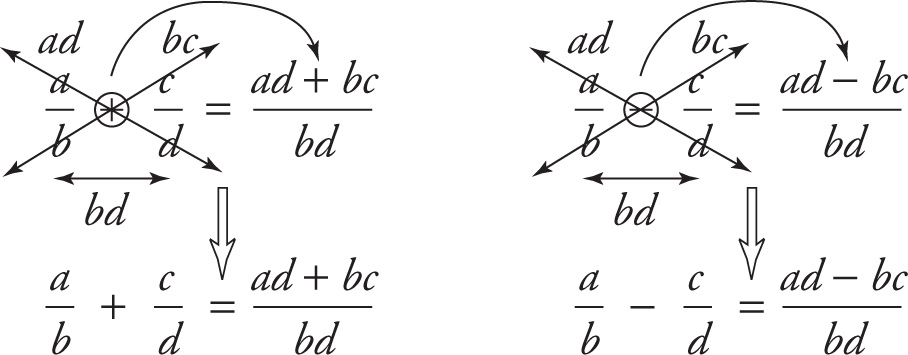
Here’s what the arrows in the top line represent: “Multiply up (d times a gives ad), multiply up again (b times c gives bc), do the adding or subtracting of these products, and place the result over the product of the denominators (bd).” The length of this last sentence hides the simplicity of the rule, but it describes the recipe to follow. For example,

Example 13-7:
a) Approximate the sum 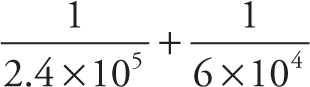
b) What is the reciprocal of this sum?
c) Simplify: 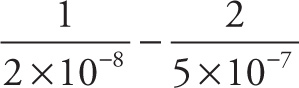
Solution:
a) Using the rule illustrated above, we find that
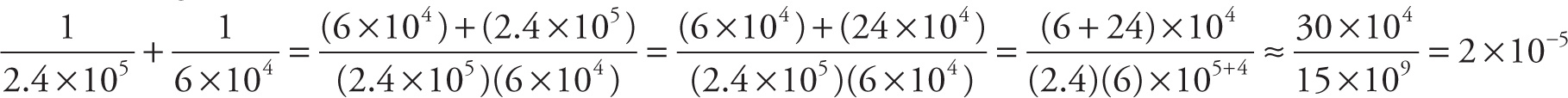
b) The reciprocal of this result is 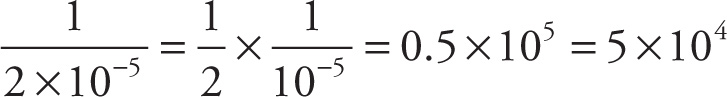 .
.
c)
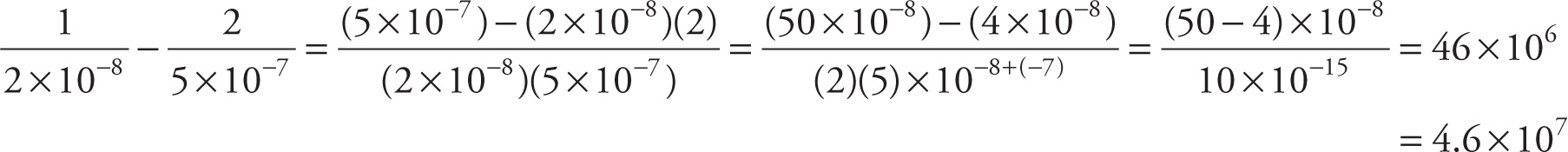
Let’s now move on to ratios. A ratio is simply another way of saying fraction. For example, the ratio of 3 to 4, written 3:4, is equal to the fraction 3/4. Here’s an illustration using isotopes of chlorine: The statement the ratio of 35Cl to 37Cl is 3:1 means that there are 3/1 = 3 times as many 35Cl atoms as there are 37Cl atoms.
A particularly useful way to interpret a ratio is in terms of parts of a total. A ratio of a:b means that there are a + b total parts, with a of them being of the first type and b of the second type. Therefore, the ratio of 35Cl to 37Cl is 3:1 means that if we could take all 35Cl and 37Cl atoms, we could partition all them into 3 + 1 = 4 equal parts such that 3 of these parts will all be 35Cl atoms, and the remaining 1 part will all be 37Cl atoms. We can now restate the original ratio as a ratio of these parts to the total. Since 35Cl atoms account for 3 parts out of the 4 total, the ratio of 35Cl atoms to all Cl atoms is 3:4; that is, 3/4 of all Cl atoms are 35Cl atoms. Similarly, the ratio of 37Cl atoms to all Cl atoms is 1:4, which means that 1/4 of all Cl atoms are 37Cl atoms.
Example 13-8: The formula for the compound TNT (trinitrotoluene) is C7H5N3O6.
a) What fraction of the atoms in this compound are nitrogen atoms?
b) If the molar masses of C, H, N, and O are 12 g, 1 g, 14 g, and 16 g, respectively, what is the ratio of the mass of all the nitrogens to the total mass?
Solution:
a) There are a total of 7 + 5 + 3 + 6 = 21 atoms per molecule. The ratio of N atoms to the total is 3:21, or, more simply, 1:7. Therefore, 1/7 of the atoms in this compound are nitrogen atoms.
b) The desired ratio of masses is calculated like this:
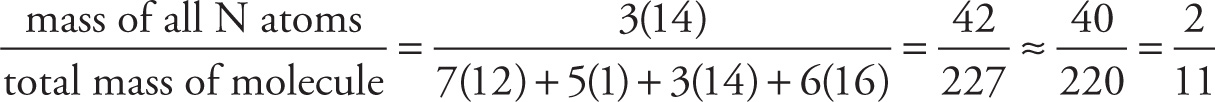
Example 13-9: In a simple hydrocarbon (molecular formula CxHy), the ratio of C atoms to H atoms is 5:4, and the total number of atoms in the molecule is 18. Find x and y.
Solution: Since the ratio of C atoms to H atoms is 5:4, there are 5 parts C atoms and 4 parts H atoms, for a total of 9 equal parts. These 9 equal parts account for 18 total atoms, so each part must contain 2 atoms. Thus, C (which has 5 parts) has 5 × 2 = 10 atoms, and H (which has 4 parts) has 4 × 2 = 8 atoms. Therefore, x = 10 and y = 8.
Example 13-10: The ratio of O atoms to C atoms in each molecule of triethylene glycol is 2:3, and the ratio of O atoms to the total number of C atoms and H atoms is 1:5. If there are 24 atoms (C, H, and O only) per molecule, find the formula for this compound.
Solution: The ratio of O to C atoms is 2:3, which tells us there are 2 parts O atoms and 3 parts C atoms, for a total of 5 parts C and O. Since the ratio of O to (C and H) atoms is 1:5, there are 5 times as many C and H atoms as there are O atoms. But, we have found that there are 2 parts O atoms, so C and H must account for 5 times as many: 10 parts. And, because there are 3 parts C atoms, there must be 10 — 3 = 7 parts H atoms. We therefore have 2 + 3 + 7 = 12 parts total, accounting for 24 atoms, which means 2 atoms per part. So, there must be 2 × 2 = 4 O atoms, 3 × 2 = 6 C atoms, and 7 × 2 = 14 H atoms. The formula is C6H14O4.
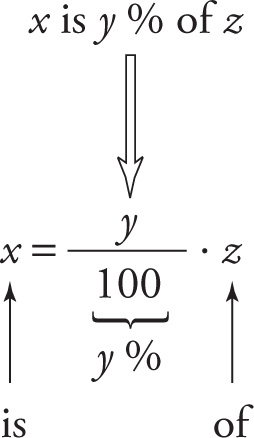
The word percent, symbolized by %, is simply an abbreviation for the phrase “out of 100”. Therefore, a percentage is represented by a fraction whose denominator is 100. For example, 60% means 60/100, or 60 out of 100. The three main question types involving percents are as follows:
1) What is y % of z?
2) x is what percent of z?
3) x is y % of what?
Fortunately, all three question types fit into a single form and can all be answered by one equation. Translating the statement x is y % of z into an algebraic equation, we get
So, if you know any two of the three quantities x, y, and z, you can use the equation above to figure out the third.
Example 13-11:
a) What is 25% of 200?
b) 30 is what percent of 150?
c) 400 is 80% of what?
Solution:
a) Solving the equation x = (25/100) × 200, we get x = 25 × 2 = 50.
b) Solving the equation 30 = (y/100) × 150, we get y = (30/150) × 100 = (1/5) × 100 = 20.
c) Solving the equation 400 = (80/100) × z, we get z = (100/80) × 400 = 100 × 5 = 500.
It’s also helpful to think of a simple fraction that equals a given percent, which can be used in place of y/100 in the equation above. For example, 25% = 1/4, 50% = 1/2, and 75% = 3/4. Other common fractional equivalents are: 20% = 1/5, 40% = 2/5, 60% = 3/5, and 80% = 4/5; 33 1/3% = 1/3 and 66 2/3% = 2/3; and 10n% = n/10 (for example, 10% = 1/10, 30% = 3/10, 70% = 7/10, and 90% = 9/10).
Example 13-12:
a) What is 60% of 35?
b) 12 is 75% of what?
c) What is 70% of 400?
Solution:
a) Since 60% = 3/5, we find that x = (3/5) × 35 = 3 × 7 = 21.
b) Because 75% = 3/4, we solve the equation 12 = (3/4) × z, and find z = 12 × (4/3) = 16.
c) Since 70% = 7/10, we find that x = (7/10) × 400 = 7 × 40 = 280.
Example 13-13:
a) What is the result when 50 is increased by 50%?
b) What is the result when 80 is decreased by 40%?
Solution:
a) “Increasing 50 by 50%” means adding (50% of 50) to 50. Since 50% of 50 is 25, increasing 50 by 50% gives us 50 + 25 = 75.
b) “Decreasing 80 by 40%” means subtracting (40% of 80) from 80. Since 40% of 80 is 32, decreasing 80 by 40% gives us 80 — 32 = 48.
Example 13-14:
a) What is 250% of 60?
b) 2400 is what percent of 500?
Solution:
a) Solving the equation x = (250/100) × 60, we get x = 25 × 6 = 150.
b) Solving the equation 2400 = (y/100) × 500, we get 2400 = 5y, so y = 2400/5 = 480.
Example 13-15: There are three stable isotopes of magnesium: 24Mg, 25Mg, and 26Mg. The relative abundance of 24Mg is 79%. Consider a sample of natural magnesium containing a total of 8 × 1024 atoms.
a) About how many atoms in the sample are 24Mg atoms?
b) If the number of 25Mg atoms in the sample is 8 × 1023, what is the relative abundance (as a percentage) of 25Mg?
c) What’s the relative abundance of 26Mg?
Solution:
a) Since the question is asking, What is 79% of 8 × 1024?, we have
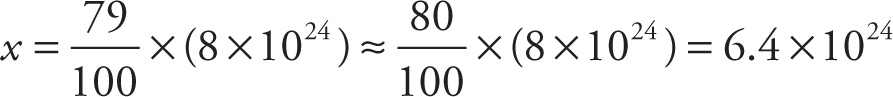
b) The question is asking, 8 × 1023 is what percent of 8 × 1024?, so we write
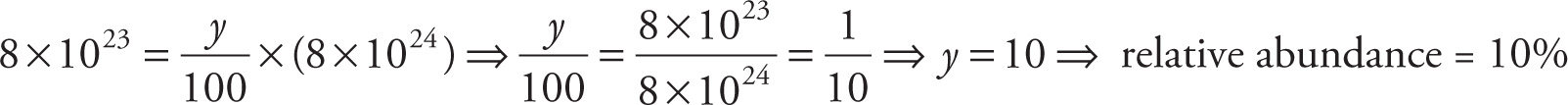
c) Assuming that these three isotopes account for all naturally occurring magnesium, the sum of the relative abundance percentages should be 100%. Therefore, we need only solve the equation 79% + 10% + Y% = 100%, from which we find that Y = 11.
Example 13-16: What is the percentage by mass, of carbon in C7H5N3O6? (Given: Molar mass of compound = 227 g.)
A) 26%
B) 37%
C) 49%
D) 62%
Solution: Once the fraction of the total molar mass of the compound that’s contributed by carbon is calculated, we obtain a percentage by multiplying this fraction by 100%. Since the molar mass of carbon is 12 g, and the molecule contains 7 C atoms, we have
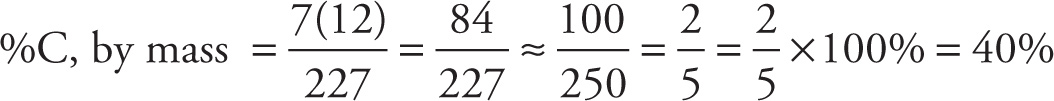
Therefore, choice B is best.
13.4 EQUATIONS AND INEQUALITIES
You may have several questions on the MCAT that require you to solve—or manipulate—an algebraic equation or inequality. Fortunately, these equations and inequalities won’t be very complicated.
When manipulating an algebraic equation, there’s basically only one rule to remember: Whatever you do to one side of the equation, you must do to the other side. (Otherwise, it won’t be a valid equation anymore.) For example, if you add 5 to the left-hand side, then add 5 to the right-hand side; if you multiply the left-hand side by 2, then multiply the right-hand side by 2, and so forth.
Inequalities are a little more involved. While it’s still true that whatever you do to one side of an inequality you must also do the other side, there are a couple of additional rules, both of which involve flipping the inequality sign—that is, changing > to < (or vice versa) or changing ≥ to ≤ (or vice versa).
1) If you multiply both sides of an inequality by a negative number, then you must flip the inequality sign.
For example, let’s say you’re given the inequality —2x > 6. To solve for x, you’d multiply both sides by —1/2. Since this is a negative number, the inequality sign must be flipped: x < —3.
2) If both sides of an inequality are positive quantities, and you take the reciprocal of both sides, then you must flip the inequality sign.
For example, let’s say you’re given the inequality 2/x ≤ 6, where it’s known that x must be positive. To solve for x, you can take the reciprocal of both sides. Upon doing so, the inequality sign must be flipped: x/2 ≥ 1/6, so x ≥ 1/3.
Example 13-17:
a) Solve for T: PV = nRT
b) Solve for v: KE = (1/2)mv2
c) Solve for x (given that x is positive): 4x2 = 2.4 × 10−11
d) Solve for B: h = k + log(B/A)
e) If F = q1q2/r2 and r is positive, solve for r in terms of F, q1, and q2.
f) Solve for x: 3(2 — x) < 18
g) Find all positive values of λ that satisfy
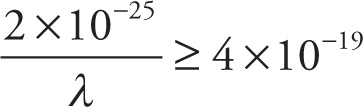
Solution:
a) Dividing both sides by nR, we get T = PV/(nR).
b) Multiply both sides 2/m, then take the square root: v = 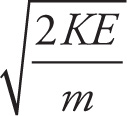 .
.
c) 4x2 = 2.4 × 10−11 ![]() x2 = 6 × 10−12
x2 = 6 × 10−12 ![]() x =
x = ![]() × 10−6 ≈ 2.5 × 10−6
× 10−6 ≈ 2.5 × 10−6
d) h = k + log ![]()
![]() log
log ![]() = h − k
= h − k ![]() 10h − k =
10h − k = ![]()
![]() B = 10h − k A [see Section 3.2]
B = 10h − k A [see Section 3.2]
e) 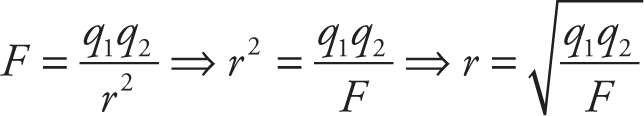
f) 3(2 − x) < 18 ![]() 2 − x < 6
2 − x < 6 ![]() − x < 4
− x < 4 ![]() x > −4
x > −4
g) 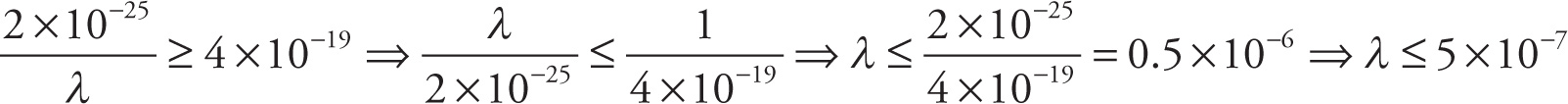
13.5 THE x-y PLANE, LINES, AND OTHER GRAPHS
The figure below shows the familiar x-y plane, which we use to plot data and draw lines and curves showing how one quantity is related to another one:
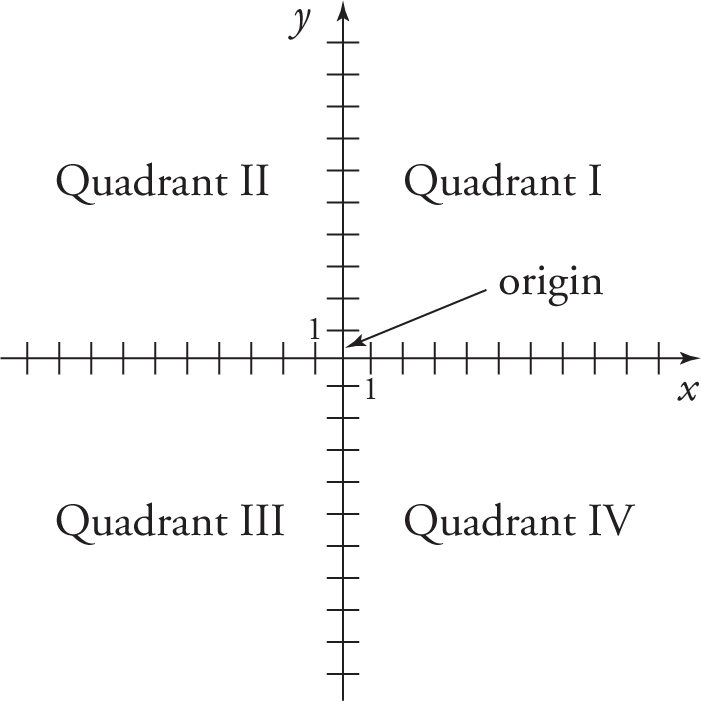
The x-y plane is formed by intersecting two number lines perpendicularly at the origins. The horizontal axis is generically referred to as the x-axis (although the quantity measured along this axis might be named by some other letter, such as time, t), and the vertical axis is generically known as the y-axis. The axes split the plane into four quadrants, which are numbered consecutively in a counterclockwise fashion. Quadrant I is in the upper right and represents all points (x, y) where x and y are both positive; in Quadrant II, x is negative and y is positive; in Quadrant III, x and y are both negative; and in Quadrant IV, x is positive and y is negative.
Suppose that two quantities, x and y, were related by the equation y = 2x2. We would consider x as the independent variable, and y as the dependent variable, since for each value of x we get a unique value of y (that is, y depends uniquely on x). The independent variable is plotted along the horizontal axis, while the dependent variable is plotted long the vertical axis. Constructing a graph of an equation usually consists of plotting specific points (x, y) that satisfy the equation—in this case, examples include (0, 0), (1, 2), (2, 8), (—1, 2), (—2, 8), etc.—and then connecting these points with a line or other smooth curve. The first coordinate of each point—the x coordinate—is known as the abscissa, and the second coordinate of each point—the y coordinate—is known as the ordinate.
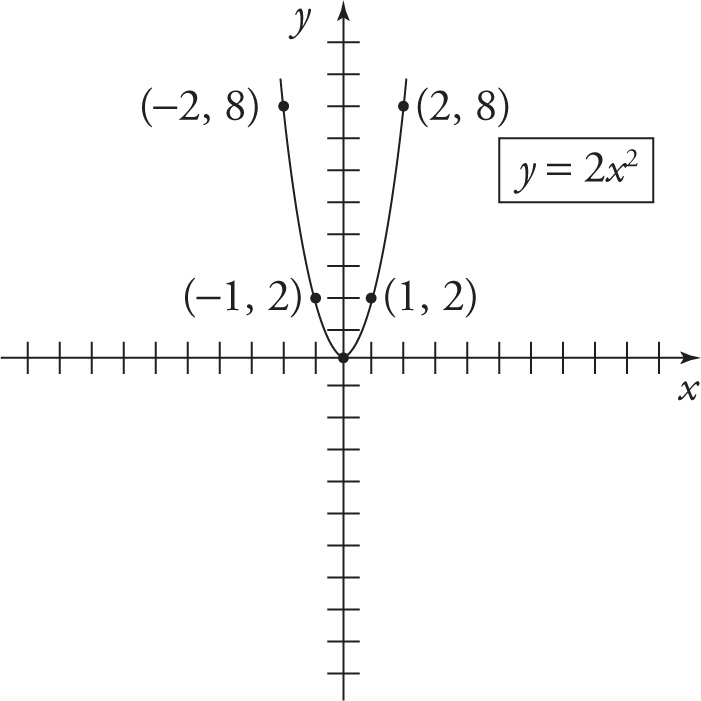
Lines
One of the simplest and most important graphs is the (straight) line. A line is determined by its slope—its steepness—and one specific point on the line, such as its intersection with either the x- or y-axis. The slope of a line is defined to be a change in y divided by the corresponding change in x (“rise over run”). Lines with positive slope rise to the right; those with negative slope fall to the right. And the greater the magnitude (absolute value) of the slope, the steeper the line.
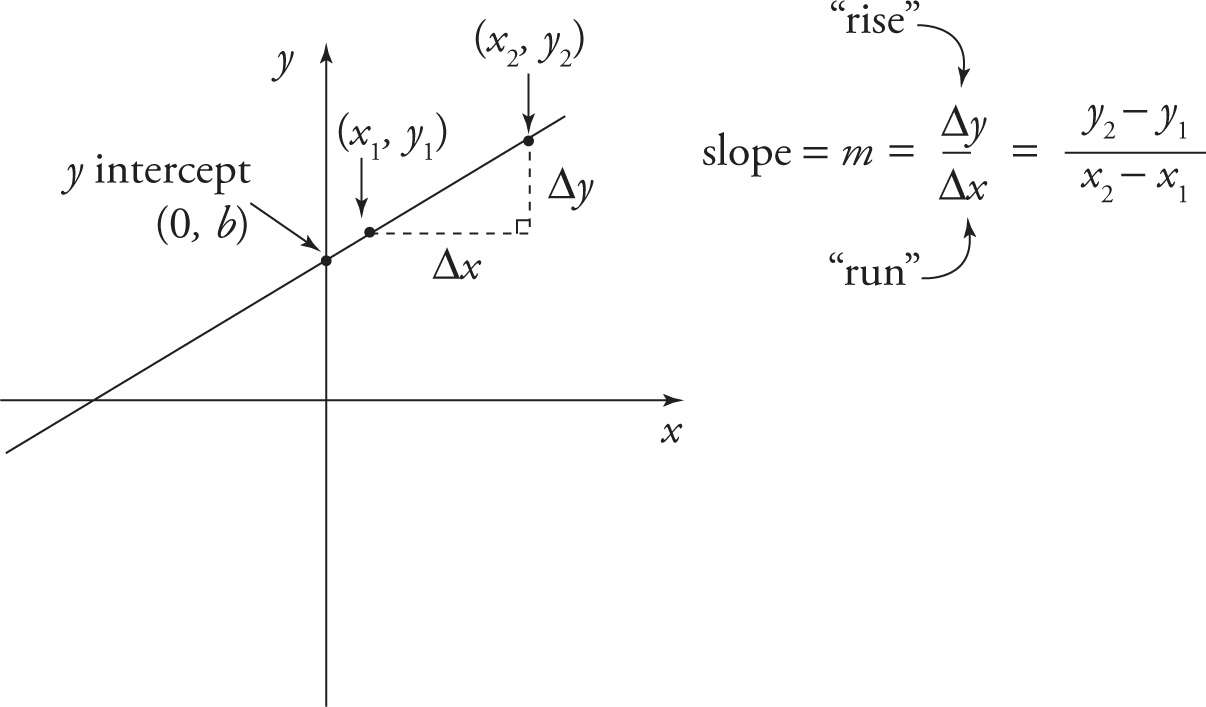
Perhaps the simplest way to write the equation of a line is in terms of its slope and the y-coordinate of the point where it crosses the y-axis. If the slope is m and the y-intercept is b, the equation of the line can be written in the form
y = mx + b
The only time this form doesn’t work is when the line is vertical, since vertical lines have an undefined slope and such a line either never crosses the y-axis (no b) or else coincides with the y-axis. The equation of every vertical line is simply x = a, where a is the x-intercept.
Example 13-18:
a) Where does the line y = 3x — 4 cross the y-axis? the x-axis? What is its slope?
b) Find the equation of the line that has slope —2 and crosses the y-axis at the point (0, 3).
c) Find the equation of the line that has slope 4 and crosses the y-axis at the origin.
d) A linear function is a function whose graph is a line. Let’s say it’s known that some quantity p is a linear function of x. If p = 50 when x = 0 and p = 250 when x = 20, find an equation for p in terms of x. Then use the equation to find the value of p when x = 40.
Solution:
a) The equation y = 3x — 4 matches the form y = mx + b with m = 3 and b = —4. Therefore, this line has slope 3 and crosses the y-axis at the point (0, —4). To find the x-intercept, we set y equal to 0 and solve for x: 0 = 3x — 4 implies that x = 4/3. Therefore, this line crosses the x-axis at the point (4/3, 0).
b) We’re given m = —2 and b = 3, so the equation of the line is y = —2x + 3.
c) We’re given m = 4 and b = 0, so the equation of the line is y = 4x.
d) Since p is a linear function of x, it must have the form p = mx + b for some values of m and b. Because p = 50 when x = 0, we know that b = 50, so p = mx + 50. Now, since p = 250 when x = 20, we have 250 = 20m + 50, so m = 10. Thus, p = 10x + 50. Finally, plugging in x = 40 into this formula, we find that the value of p when x = 40 is (10)(40) + 50 = 450.
Example 13-19: An insulated 50 cm3 sample of water has an initial temperature of Ti = 10°C. If Q calories of heat are added to the sample, the temperature of the water will rise to T, where T = kQ + Ti. When the graph of T vs. Q is sketched (with Q measured along the horizontal axis), it’s found that the point (Q, T) = (200, 14) lies on the graph.
a) What is the value of k?
b) How much heat is required to bring the water to 20°C?
c) If Q = 2200 cal, what will be the value of T?
Solution:
a) The equation T = kQ + Ti matches the form y = mx + b, so k is the slope of the line. To find the slope, we evaluate the rise-over-run expression—which in this case is ∆T/∆Q—for two points on the line. Using (Q1, T1) = (0, 10) and (Q2, T2) = (200, 14), we find that
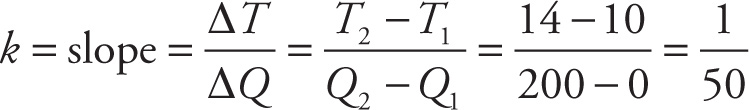
b) We set T equal to 20 and solve for Q:
T = kQ + Ti ![]() T =
T = ![]() Q + 10
Q + 10 ![]() 20 =
20 = ![]() Q + 10
Q + 10 ![]() Q = 500 (cal)
Q = 500 (cal)
c) Here we set Q = 2200 and evaluate T:
T = kQ + Ti ![]() T =
T = ![]() Q + 10
Q + 10 ![]() T =
T = ![]() (2200) + 10 = 44 + 10 = 54 (°C)
(2200) + 10 = 44 + 10 = 54 (°C)
(Technical note: The equation for the temperature of the water, T = kQ + Ti, is valid as long as no phase change occurs.)
Besides lines, there are a few other graphs and features you should be familiar with.
The equation y = kx2, where k ≠ 0, describes the basic parabola, one whose turning point (vertex) is at the origin. It has a U shape, and opens upward if k is positive and downward if k is negative. The graph of the related equation y = k(x — a)2 is obtained from the basic parabola by shifting it horizontally so that its vertex is at the point (a, 0). The graph of the equation y = kx2 + b is obtained from the basic parabola by shifting it vertically so that its vertex is at the point (0, b). Finally, the graph of the equation y = k(x — a)2 + b is obtained from the basic parabola in two shifting steps: First, shift the basic parabola horizontally so that its vertex is at the point (a, 0); next, shift this parabola vertically so that the vertex is at the point (a, b). These parabolas are illustrated below for positive a, b, k, and x:
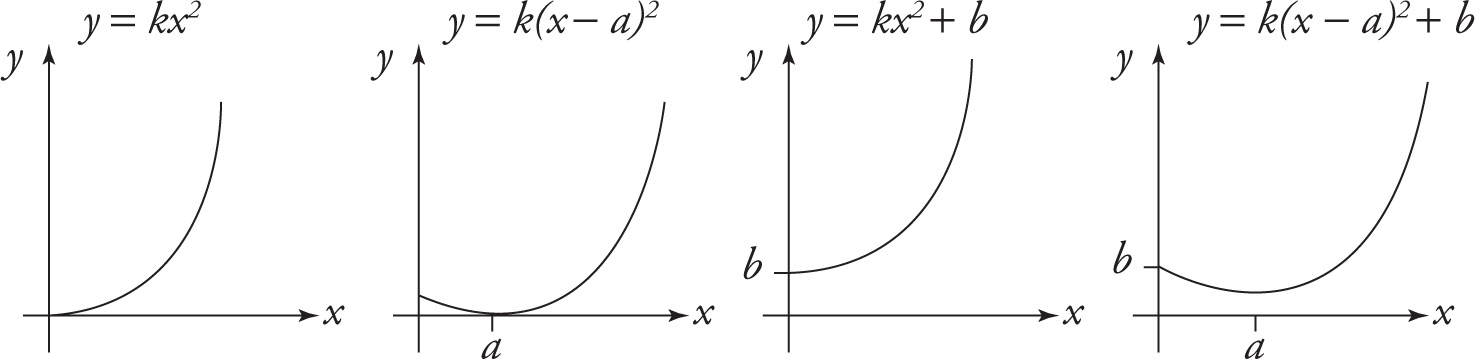
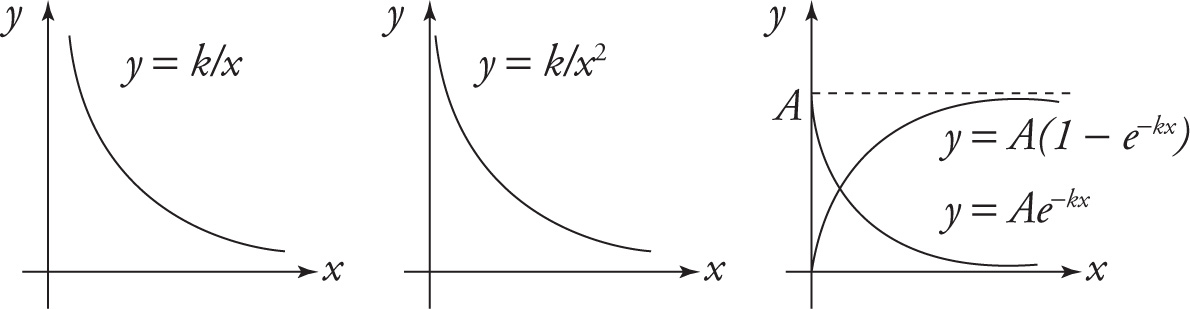
The equation y = k/x, where k ≠ 0, describes a hyperbola. It is the graph of an inverse proportion (see Section 2.2). For small values of x, the values of y are large; and for large values of x, the values of y are small. Notice that the graph of a hyperbola approaches—but never touches—both the x- and y-axes. These lines are therefore called asymptotes.
The equation y = k/x2, where k ≠ 0, has a graph whose shape is similar to a hyperbola but it approaches its asymptotes faster than a hyperbola does (because of the square in the denominator).
The graph of the equation y = Ae−kx (where k is positive) is an exponential decay curve. It intersects the y axis at the point (0, A), and, as x increases, the value of y decreases. Here, the x-axis is an asymptote.
The graph of the equation y = A(1 — e−kx), where k is positive, contains the origin, and as x increases, the graph rises to approach the horizontal line y = A. This line is an asymptote.