MCAT General Chemistry Review - Steven A. Leduc 2015
MCAT Math for General Chemistry
Proportions
The concept of proportionality is fundamental to analyzing the behavior of many physical phenomena and is a common topic for MCAT questions.
14.1 DIRECT PROPORTIONS
If one quantity is always equal to a constant times another quantity, we say that the two quantities are proportional (or directly proportional, if emphasis is desired). For example, if k is some nonzero constant and the equation A = kB is always true, then A and B are proportional, and k is called the proportionality constant. We express this fact mathematically by using this symbol: ∝, which means is proportional to. So, if A = kB, we’d write A ∝ B. Of course, if A = kB, then B = (1/k)A, so we could also say that B ∝ A.
Here are a few examples:
Example 14-1: Energy is equal to Planck’s constant times frequency, E = hf. Therefore E ∝ f.
Example 14-2: The ideal gas law states that PV = nRT. If n, V, and R are constant, P ∝ T.
Example 14-3: The rate law for a chemical reaction that is first order with respect to reactant A is rate = k[A]. Assuming k is constant, rate ∝ [A].
The most important fact about direct proportions is this:
If A ∝ B, and B is multiplied by a factor of b, then A will also be multiplied by a factor of b.
After all, if A = kB, then bA = k(bB).
Example 14-4: Since the energy of a photon is proportional to its frequency, E ∝ f, then, if the frequency is doubled, so is the energy. If the frequency is reduced by half, so is the energy. If the frequency is tripled, so is the energy.
Example 14-5: Since the pressure inside a system is proportional to its temperature when volume and the number of moles present are constant, P ∝ T, when the temperature is quadrupled, the pressure is quadrupled. When the pressure is decreased by a factor of 3, the temperature is also decreased by a factor of 3.
Example 14-6: Since the rate of a first order chemical reaction is proportional to the concentration of reactant A, [rate] ∝ [A], if [A] is increased by a factor of 2, the rate also increases by a factor of 2. If [A] is decreased by a factor of 4 (same as multiplying by ¼), the rate of reaction is ¼ of what it was originally.
It’s important to notice that the actual numerical value of the proportionality constant was irrelevant in the statements made above. For example, the fact that h is the proportionality constant in the equation E = hf did not affect the conclusions made above. If E and f were some other quantities and E happened to always be equal to (17,000)f, we’d still say E ∝ f, and all the conclusions made in Example 14-4 above would still be correct.
Graphically, proportions are easy to spot. If the horizontal and vertical axes are labeled linearly (as they usually are), then the graph of a proportion is a straight line through the origin. Be careful not to make the common mistake of thinking that any straight line is the graph of a proportion. If the line doesn’t go through the origin, then it’s not the graph of a proportion.
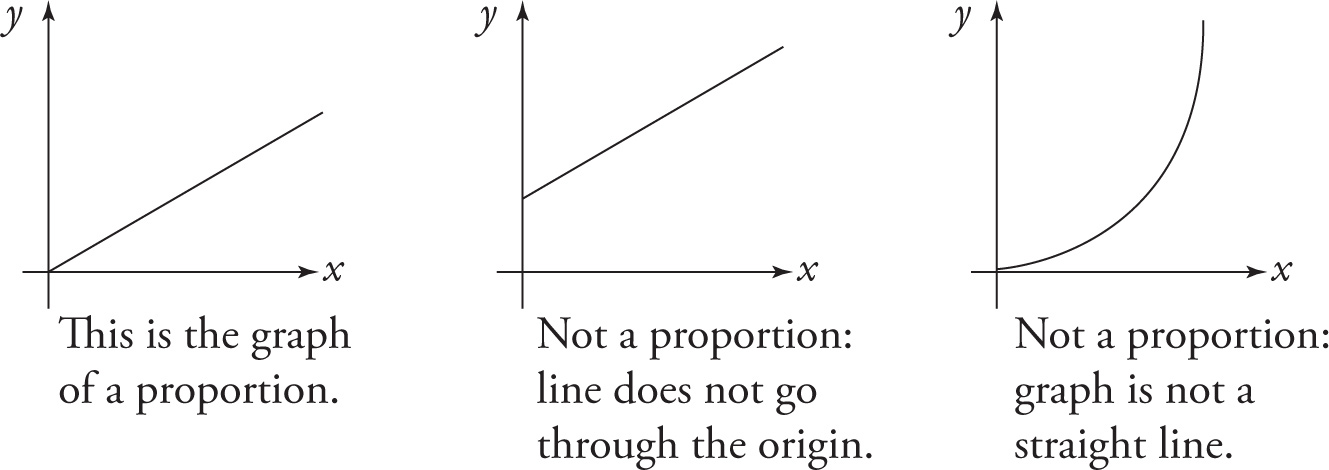
The examples we’ve seen so far have been the equations E = hf, PV = nRT, and rate = k[A]. Notice that in all of these equations, all the variables are present to the first power. But what about an equation like this: KE = ![]() mv2? This equation gives the kinetic energy of an object of mass m moving with speed v. So, if m is constant, KE is proportional to v2. Now, what if v were multiplied by, say, a factor of 3, what would happen to KE? Because KE ∝ v2, if v increases by a factor of 3, then KE will increase by a factor of 32, which is 9. (By the way, this does not mean that if we graph KE versus v, we’ll get a straight line through the origin. KE is not proportional to v; it’s proportional to v2. If we were to graph KE vs. v2, then we’d get a straight line through the origin.) Here’s another example using the same proportion, KE ∝ v2: If v were decreased by a factor of 2, then KE would decrease by a factor of 22 = 4.
mv2? This equation gives the kinetic energy of an object of mass m moving with speed v. So, if m is constant, KE is proportional to v2. Now, what if v were multiplied by, say, a factor of 3, what would happen to KE? Because KE ∝ v2, if v increases by a factor of 3, then KE will increase by a factor of 32, which is 9. (By the way, this does not mean that if we graph KE versus v, we’ll get a straight line through the origin. KE is not proportional to v; it’s proportional to v2. If we were to graph KE vs. v2, then we’d get a straight line through the origin.) Here’s another example using the same proportion, KE ∝ v2: If v were decreased by a factor of 2, then KE would decrease by a factor of 22 = 4.
Here is one more example:
Example 14-7: The reaction quotient Q for a reaction is described by Q = [A][B]3. Therefore, Q is proportional to the concentration of [B]3: Q ∝ [B]3. So, for example, if [B] were doubled, Q would increase by a factor 23 = 8.
14.2 INVERSE PROPORTIONS
If one quantity is always equal to a nonzero constant divided by another quantity (that is, if A = k/B, where k is some constant), we say that the two quantities are inversely proportional. Here are two equivalent ways of saying this:
(i) If the product of two quantities is a constant (AB = k), then the quantities are inversely proportional.
(ii) If A is proportional to 1/B [that is, if A = k(1/B)], then A and B are inversely proportional.
In fact, we’ll use this final description to symbolize an inverse proportion. That is, if A is inversely proportional to B, then we’ll write A ∝ 1/B. (There’s no commonly accepted single symbol for inversely proportional to.) Of course, if A = k/B, then B = k/A, so we could also say that B ∝ 1/A.
Here are a couple of examples:
Example 14-8: The pressure P and volume V of a sample containing n moles of an ideal gas at a fixed temperature T is given by the equation PV = nRT, where R is a constant. Therefore, the pressure is inversely proportional to the volume: P ∝ 1/V.
Example 14-9: For electromagnetic waves traveling through space, the wavelength λ and frequency f are related by the equation λ f = c, where c is the speed of light (a universal constant). Therefore, wavelength is inversely proportional to frequency: λ ∝ 1/f.
The most important fact about inverse proportions is this:
If A ∝ 1/B, and B is multiplied by a factor of b, then A will be multiplied by a factor of 1/b.
After all, if A = k/B, then (1/b)A = k/(bB). Intuitively, if one quantity is increased by a factor of b, the other quantity will decrease by the same factor, and vice versa.
Example 14-10: Since the pressure of an ideal gas at constant temperature is inversely proportional to the volume, P ∝ 1/V, then if the volume is doubled, the pressure is reduced by a factor of 2. If the volume is quadrupled, the pressure is reduced by a factor of 4. If the volume is divided by 3 (which is the same as saying it’s multiplied by 1/3), then the pressure will increase by a factor of 3.
Example 14-11: Because for electromagnetic waves traveling through space, the wavelength is inversely proportional to frequency, λ ∝ 1/f, if f is increased by a factor of 10, λ will decrease by a factor of 10. If the frequency is decreased by a factor of 2, the wavelength will increase by a factor of 2.
The graph of an inverse proportion is a hyperbola. In the graph below, xy = k, so x and y are inversely proportional to each other.
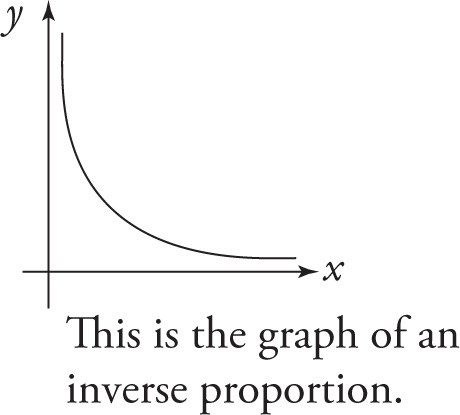
The examples we’ve seen so far have been where one quantity is inversely proportional to the first power of another quantity. But what about an equation like this:
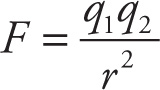
This equation gives the electrostatic force between two point charges of magnitude q1 and q2 separated by a distance r. So, if q1 and q2 are constant, F is inversely proportional to r2. Now, if r were increased by, say, a factor of 3, what would happen to F ? Because F ∝ 1/r2, if r increases by a factor of 3, then F will decrease by a factor of 32, which is 9. Here’s another example using the same proportion, F ∝ 1/r 2: If r were decreased by a factor of 2, then F would increase by a factor of 22 = 4.
Example 14-12: Graham’s law of effusion states that
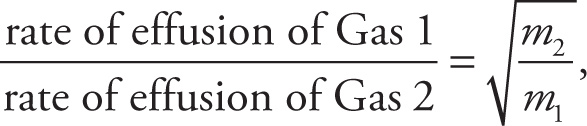
where m2 is the molecular mass of Gas 2 and m1 is the molecular mass of Gas 1. Therefore, the rate of effusion of Gas 1 is inversely proportional to the square root of its molecular mass, rate of effusion of Gas 1 ∝ 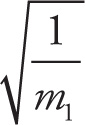 . So, if Gas 1 were changed to a molecule whose mass was 4 times greater, the rate of effusion of Gas 1 would decrease by a factor of 2.
. So, if Gas 1 were changed to a molecule whose mass was 4 times greater, the rate of effusion of Gas 1 would decrease by a factor of 2.
Example 14-13: The kinetic energy of an object of mass m traveling with speed v is given by the formula KE = mv2/2.
a) If v is increased by a factor of 6, what happens to KE?
b) In order to increase KE by a factor of 6, what must happen to v?
Solution:
a) Since KE ∝ v2, if v increases by a factor of 6, then KE increases by a factor of 62 = 36.
b) Since KE ∝ v2, it follows that ![]() ∝ v. So, if KE is to increase by a factor of 6, then v must be increased by a factor of
∝ v. So, if KE is to increase by a factor of 6, then v must be increased by a factor of ![]() .
.