MCAT General Chemistry Review - Steven A. Leduc 2015
Chemistry Fundamentals
3.1 METRIC UNITS
Before we begin our study of chemistry, we will briefly go over metric units. Scientists use the Système International d’Unitès (the International System of Units), abbreviated SI, to express measurements of physical quantities. Six of the seven base units of SI are given below:
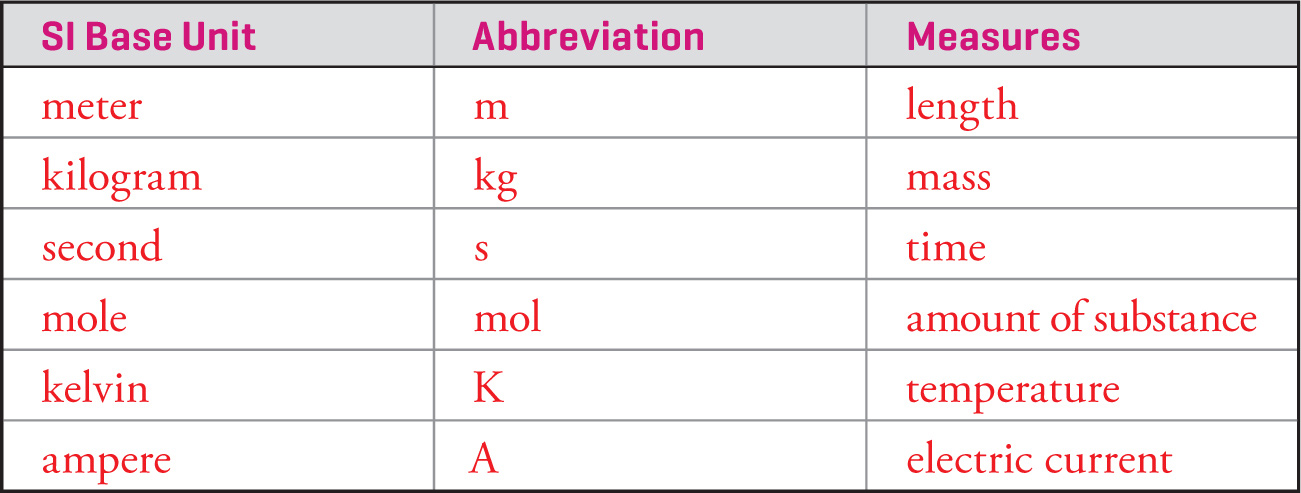
(The seventh SI base unit, the candela [cd], measures luminous intensity, but we will not need to worry about this one.) The units of any physical quantity can be written in terms of the SI base units. For example, the SI unit of speed is meters per second (m/s), the SI unit of energy (the joule) is kilograms times meters2 per second2 (kg • m2/s2), and so forth.
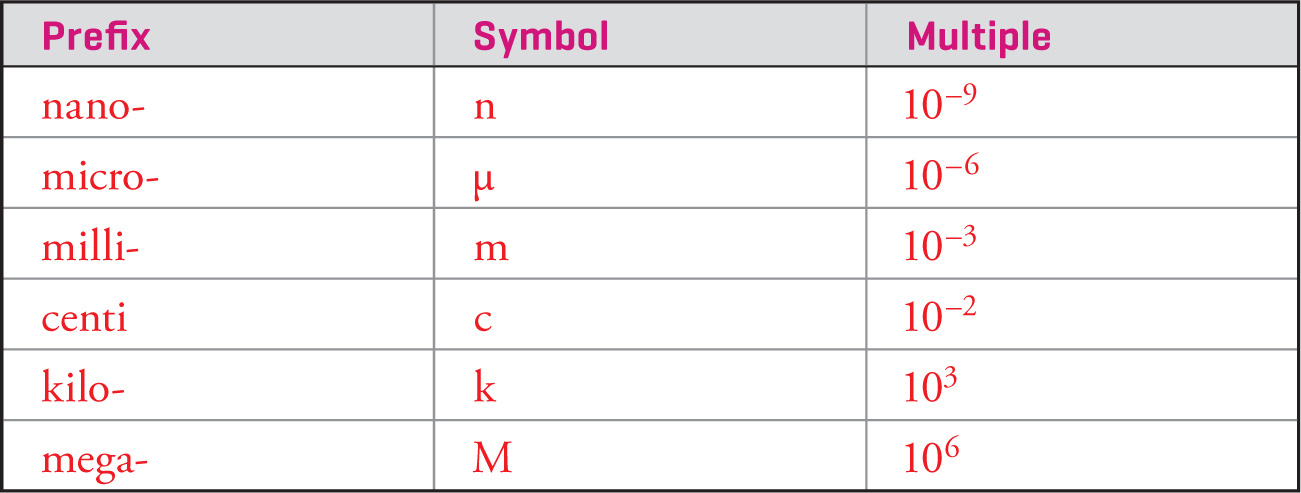
Multiples of the base units that are powers of ten are often abbreviated and precede the symbol for the unit. For example, m is the symbol for milli-, which means 10−3 (one thousandth). So, one thousandth of a second, 1 millisecond, would be written as 1 ms. The letter M is the symbol for mega-, which means 106 (one million); a distance of one million meters, 1 megameter, would be abbreviated as 1 Mm. Some of the most common power-of-ten prefixes are given in the list below:
Two other units, ones that are common in chemistry, are the liter and the angstrom. The liter (abbreviated L) is a unit of volume equal to 1/1000 of a cubic meter:
1000 L = 1 m3
1 L = 1000 cm3
The standard SI unit of volume, the cubic meter, is inconveniently large for most laboratory work. The liter is a smaller unit. Furthermore, the most common way of expressing solution concentrations, molarity (M), uses the liter in its definition: M = moles of solute per liter of solution.
In addition, you will see the milliliter (mL) as often as you will see the liter. A simple consequence of the definition of a liter is the fact that one milliliter is the same volume as one cubic centimeter:
1 mL = 1 cm3 = 1 cc
While the volume of any substance can, strictly speaking, be expressed in liters, you rarely hear of a milliliter of gold, for example. Ordinarily, the liter is used to express the volumes of liquids and gases, but not solids.
The angstrom, abbreviated Å, is a unit of length equal to 10−10 m. The angstrom is convenient because atomic radii and bond lengths are typically around 1 to 3 Å.
Example 3-1: By how many orders of magnitude is a centimeter longer than an angstrom?
Solution: An order of magnitude is a factor of ten. Since 1 cm = 10−2 m and 1 Å = 10−10 m, a centimeter is 8 factors of ten, or 8 orders of magnitude, greater than an angstrom.
3.2 DENSITY
The density of a substance is its mass per volume:
Density: ρ = 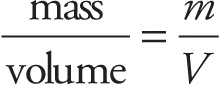
In SI units, density is expressed in kilograms per cubic meter (kg/m3). However, in chemistry, densities are more often expressed in grams per cubic centimeter (g/cm3). This unit of density is convenient because most liquids and solids have a density of around 1 to 20 g/cm3. Here is the conversion between these two sets of density units:
g/cm3 → multiply by 1000 → kg/m3
g/cm3 ← divide by 1000 ← kg/m3
For example, water has a density of 1 g/cm3 (it varies slightly with temperature, but this is the value the MCAT will expect you to use). To write this density in kg/m3, we would multiply by 1000. The density of water is 1000 kg/m3. As another example, the density of copper is about 9000 kg/m3, so to express this density in g/cm3, we would divide by 1000: The density of copper is 9 g/cm3.
Example 3-2: Diamond has a density of 3500 kg/m3. What is the volume, in cm3, of a 1 3/4-carat diamond (where, by definition, 1 carat = 0.2 g)?
Solution: If we divide mass by density, we get volume, so, converting 3500 kg/m3 into 3.5 g/cm3, we find that
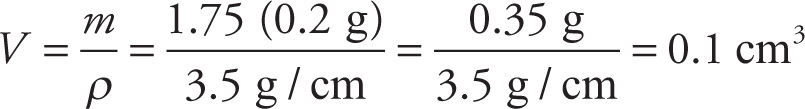
3.3 MOLECULAR FORMULAS
When two or more atoms form a covalent bond they create a molecule. For example, when two atoms of hydrogen (H) bond with one atom of oxygen (O), the resulting molecule is H2O, water. A compound’s molecular formula gives the identities and numbers of the atoms in the molecule. For example, the formula C4H4N2 tells us that this molecule contains 4 carbon atoms, 4 hydrogen atoms, and 2 nitrogen atoms.
Example 3-3: What is the molecular formula of para-nitrotoluene?
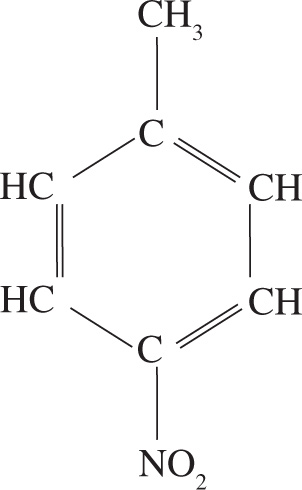
A) C6H5NO2
B) C7H7NO2
C) C7H8NO2
D) C7H9NO2
Solution: There are a total of seven C’s, seven H’s, one N, and two O’s, so choice B is the correct answer.
3.4 EMPIRICAL FORMULAS
Let’s look again at the molecule C4H4N2. There are four atoms each of carbon and hydrogen, and half as many (two) nitrogen atoms. Therefore, the smallest whole numbers that give the same ratio of atoms (carbon to hydrogen to nitrogen) in this molecule are 2:2:1. If we use these numbers for the atoms, we get the molecule’s empirical formula: C2H2N. In general, to reduce a molecular formula to the empirical formula, divide all the subscripts by their greatest common factor. Here are a few more examples:
|
Molecular Formula |
Empirical Formula |
C6H12O6 |
CH2O |
K2S2O8 |
KSO4 |
Fe4Na8O35P10 |
Fe4Na8O35P10 |
C30H27N3O15 |
C10H9NO5 |
Example 3-4: What is the empirical formula for ethylene glycol, C2H6O2?
A) CH3O
B) CH4O
C) CH6O
D) C2H6O2
Solution: Dividing each of the subscripts of C2H6O2 by 2, we get CH3O, choice A.
3.5 FORMULA AND MOLECULAR WEIGHT
If we know the chemical formula, we can figure out the formula weight, which is the sum of the atomic weights of all the atoms in the molecule. The unit for atomic weight is the atomic mass unit, abbreviated amu. (Note: Although weight is the popular term, it should really be mass.) One atomic mass unit is, by definition, equal to exactly 1/12 the mass of an atom of carbon-12 (12C), the most abundant naturally occurring form of carbon. The periodic table lists the mass of each element; it is actually a weighted average of the atomic masses of all its naturally occurring forms (isotopes) based on their relative abundance. For example, the atomic mass of hydrogen is listed as 1.0 (amu), and that of nitrogen as 14.0 (amu). Therefore, the formula weight for C4H4N2 is
4(12) + 4(1) + 2(14) = 80
(The unit amu is often not explicitly included.) When a compound exists as discrete molecules, the term molecular weight (MW) is usually used instead of formula weight. For example, the molecular weight of water, H2O, is 2(1) + 16 = 18. The term formula weight is usually used for ionic compounds, such as NaCl. The formula weight of NaCl is 23 + 35.5 = 58.5.
Example 3-5: What is the formula weight of calcium phosphate, Ca3(PO4)2?
A) 310 amu
B) 350 amu
C) 405 amu
D) 450 amu
Solution: The masses of the elements are Ca = 40 amu, P = 31 amu, and O = 16 amu. Therefore, the formula weight of calcium phosphate is
3(40 amu) + 2(31 amu) + 8(16 amu) = 310 amu
Choice A is the answer.
3.6 THE MOLE
A mole is simply a particular number of things, like a dozen is any group of 12 things. One mole of anything contains 6.02 × 1023 entities. A mole of atoms is a collection of 6.02 × 1023 atoms; a mole of molecules contains 6.02 × 1023 molecules, and so on. This number, 6.02 × 1023, is called Avogadro’s number, denoted by NA (or N0). What is so special about 6.02 × 1023? The answer is based on the atomic mass unit, which is defined so that the mass of a carbon-12 atom is exactly 12 amu. The number of carbon-12 atoms in a sample of mass of 12 grams is 6.02 × 10 23. Avogadro’s number is the link between atomic mass units and grams. For example, the periodic table lists the mass of sodium (Na, atomic number 11) as 23.0. This means that 1 atom of sodium has a mass of 23 atomic mass units, or that 1 mole of sodium atoms has a mass of 23 grams.
Since 1 mole of a substance has a mass in grams equal to the mass in amus of 1 formula unit of the substance, we have the following formula:
# moles = 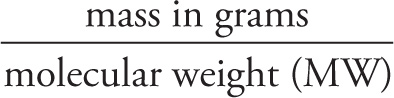
Example 3-6:
a) Which has the greater molecular weight: potassium dichromate (K2Cr2O7) or lead azide Pb(N3)2?
b) Which contains more formula units: a 1-mole sample of potassium dichromate or a 1-mole sample of lead azide?
Solution:
a) The molecular weight of potassium dichromate is
2(39.1) + 2(52) + 7(16) = 294.2
and the molecular weight of lead azide is
207.2 + 6(14) = 291.2
Therefore, potassium dichromate has the greater molecular weight.
b) Trick question. Both samples contain the same number of formula units, namely 1 mole of them. (Which weighs more: a pound of rocks or a pound of feathers?)
Example 3-7: How many molecules of hydrazine, N2H4, are in a sample with a mass of 96 grams?
Solution: The molecular weight of N2H4 is 2(14) + 4(1) = 32. This means that 1 mole of N2H4 has a mass of 32 grams. Therefore, a sample that has a mass of 96 grams contains 3 moles of molecules, because the formula above tells us that
n = 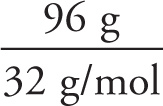 = 3 moles
= 3 moles
3.7 PERCENTAGE COMPOSITION BY MASS
A molecule’s molecular or empirical formula can be used to determine the molecule’s percent mass composition. For example, let’s find the mass composition of carbon, hydrogen, and nitrogen in C4H4N2. Using the compound’s empirical formula, C2H2N, will give us the same answer but the calculations will be easier because we’ll have smaller numbers to work with. The empirical molecular weight is 2(12) + 2(1) + 14 = 40, so each element’s contribution to the total mass is
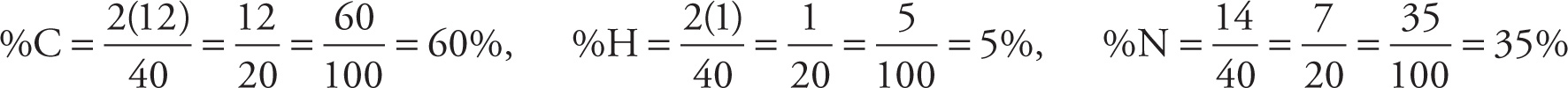
We can also use information about the percentage composition to determine a compound’s empirical formula. Suppose a substance is analyzed and found to consist, by mass, of 70 percent iron and 30 percent oxygen. To find the empirical formula for this compound, the trick is to start with 100 grams of the substance. We choose 100 grams since percentages are based on parts in 100. One hundred grams of this substance would then contain 70 g of Fe and 30 g of O. Now, how many moles of Fe and O are present in this 100-gram substance? Since the atomic weight of Fe is 55.8 and that of O is 16, we can use the formula given above in Section 3.6 and find
# moles of Fe = 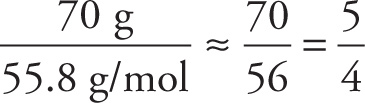
and
# moles of O = 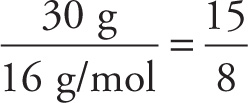
Because the empirical formula involves the ratio of the numbers of atoms, let’s find the ratio of the amount of Fe to the amount of O:
Ratio of Fe to O = 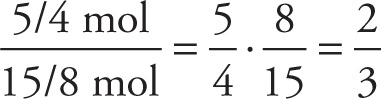
Since the ratio of Fe to O is 2:3, the empirical formula of the substance is Fe2O3.
Example 3-8: What is the percent composition by mass of each element in sodium azide, NaN3?
A) Sodium 25%; nitrogen 75%
B) Sodium 35%; nitrogen 65%
C) Sodium 55%; nitrogen 45%
D) Sodium 65%; nitrogen 35%
Solution: The molecular weight of this compound is 23 + 3(14) = 65. Therefore, sodium’s contribution to the total mass is
%Na = 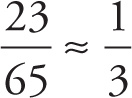 ≈ 33%
≈ 33%
Without even calculating nitrogen’s contribution, we already see that choice B is best.
Example 3-9: What is the percent composition by mass of carbon in glucose, C6H12O6?
A) 40%
B) 50%
C) 67%
D) 75%
Solution: The empirical formula for this compound is CH2O, so the empirical molecular weight is 12 + 2(1) + 16 = 30. Therefore, carbon’s contribution to the total mass is
%C = ![]() = 40%
= 40%
So choice A is the answer. We would have found the same answer using the molecular formula, but the numbers would have been messier:
%C = 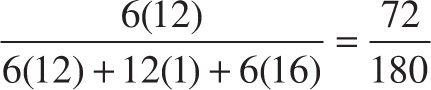 = 40%
= 40%
Example 3-10: What is the empirical formula of a compound that is, by mass, 90 percent carbon and 10 percent hydrogen?
A) CH2
B) C2H3
C) C3H4
D) C4H5
Solution: A 100-gram sample of this compound would contain 90 g of C and 10 g of H. Since the atomic weight of C is 12 and that of H is 1, we have
# moles of C = 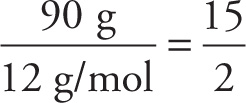
and
# moles of H = 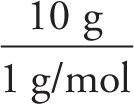 = 10
= 10
Therefore, the ratio of the amount of C to the amount of H is
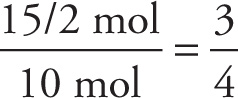
Because the ratio of C to H is 3:4, the empirical formula of the compound is C3H4, and choice C is the answer.
Example 3-11: What is the percent, by mass of water, in the hydrate MgCl2 • 5H2O?
A) 27%
B) 36%
C) 49%
D) 52%
Solution: The formula weight for this hydrate is 24.3 + 2(35.5) + 5[2(1) + 16] = 185.3. Since water’s total molecular weight in this compound is 5[2(1) + 16] = 90, we see that water’s contribution to the total mass is %H2O = 90/185.3, which is a little less than one half (50 percent). Therefore, the answer is C.
Example 3-12: In which of the following compounds is the mass percent of each of the constituent elements nearly identical?
A) NaCl
B) LiBr
C) HCl
D) CaF2
Solution: The question is asking us to identify the compound made up of equal amounts, by mass, of two elements. Looking at the given compounds, we see that
Na (23.0 g/mol) ≠ Cl (35.5 g/mol)
Li (6.9 g/mol) ≠ Br (79.9 g/mol)
H (1.0 g/mol) ≠ Cl (35.5 g/mol)
Ca (40.1 g/mol) ≈ 2 F (2×19) = 38 g/mol)
Therefore, choice D is best.
3.8 CONCENTRATION
Molarity (M) expresses the concentration of a solution in terms of moles of solute per volume (in liters) of solution:
Molarity (M) = 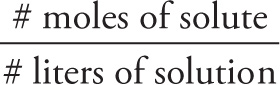
Concentration is denoted by enclosing the solute in brackets. For instance, “[Na+] = 1.0 M” indicates a solution in which the concentration is equivalent to 1 mole of sodium ions per liter of solution.
Mole fraction simply expresses the fraction of moles of a given substance (which we’ll denote here by S) relative to the total moles in a solution:
mole fraction of S = Xs = 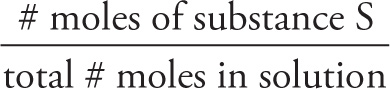
Mole fraction is a useful way to express concentration when more than one solute is present, and is often used when discussing the composition of a mixture of gases.
3.9 CHEMICAL EQUATIONS AND STOICHIOMETRIC COEFFICIENTS
The equation
2 Al + 6 HCl → 2 AlCl3 + 3 H2
describes the reaction of aluminum metal (Al) with hydrochloric acid (HCl) to produce aluminum chloride (AlCl3) and hydrogen gas (H2). The reactants are on the left side of the arrow, and the products are on the right side. A chemical equation is balanced if, for every element represented, the number of atoms on the left side is equal to the number of atoms on the right side. This illustrates the Law of Conservation of Mass (or of Matter), which says that the amount of matter (and thus mass) does not change in a chemical reaction. For a balanced reaction such as the one above, the coefficients (2, 6, 2, and 3) preceding each compound—which are known as stoichiometric coefficients—tell us in what proportion the reactants react and in what proportion the products are formed. For this reaction, 2 atoms of Al react with 6 molecules of HCl to form 2 molecules of AlCl3 and 3 molecules of H2. The equation also means that 2 moles of Al react with 6 moles of HCl to form 2 moles of AlCl3 and 3 moles of H2.
The stoichiometric coefficients give the ratios of the number of molecules (or moles) that apply to the combination of reactants and the formation of products. They do not give the ratios by mass.
Balancing Equations
Balancing most chemical equations is simply a matter of trial and error. It’s a good idea to start with the most complex species in the reaction. For example, let’s look at the reaction above:
Al + HCl → AlCl3 + H2 (unbalanced)
Start with the most complex molecule, AlCl3. The total number of atoms, or moles of atoms, is calculated by multiplying the coefficient in front of a compound times the subscript within the formula. To get 3 atoms of Cl on the product side, we need to have 3 atoms of Cl on the reactant side; therefore, we put a 3 in front of the HCl:
Al + 3 HCl → AlCl3 + H2 (unbalanced)
We’ve now balanced the Cl’s, but the H’s are still unbalanced. Since we have 3 H’s on the left, we need 3 H’s on the right to accomplish this, so we put a coefficient of 3/2 in front of the H2:
Al + 3 HCl → AlCl3 + 3/2 H2
Notice that we put a 3/2 (not a 3) in front of the H2, because a hydrogen molecule contains 2 hydrogen atoms. All the atoms are now balanced—we see 1 Al, 3 H’s, and 3 Cl’s on each side. Because it’s customary to write stoichiometric coefficients as whole numbers, we simply multiply through by 2 to get rid of the fraction and write
2 Al + 6 HCl → 2 AlCl3 + 3 H2
Example 3-13: Balance each of these equations:
a) NH3 + O2 → NO + H2O
b) CuCl2 + NH3 + H2O → Cu(OH)2 + NH4Cl
c) C3H8 + O2 → CO2 + H2O
d) C8H18 + O2 → CO2 + H2O
Solution:
a) 4 NH3 + 5 O2 → 4 NO + 6 H2O
b) CuCl2 + 2 NH3 + 2 H2O → Cu(OH)2 + 2 NH4Cl
c) C3H8 + 5 O2 → 3 CO2 + 4 H2O
d) 2 C8H18 + 25 O2 → 16 CO2 + 18 H2O
3.10 STOICHIOMETRIC RELATIONSHIPS IN BALANCED REACTIONS
Once the equation for a chemical reaction is balanced, the stoichiometric coefficients tell us the relative amounts of the reactant species that combine and the relative amounts of the product species that are formed. For example, recall that the reaction
2 Al + 6 HCl → 2 AlCl3 + 3 H2
tells us that 2 moles of Al react with 6 moles of HCl to form 2 moles of AlCl3 and 3 moles of H2.
Example 3-14: If 108 grams of aluminum metal are consumed, how many grams of hydrogen gas will be produced?
Solution: Because the stoichiometric coefficients give the ratios of the number of moles that apply to the combination of reactants and the formation of products—not the ratios by mass—we first need to determine how many moles of Al react. Since the molecular weight of Al is 27, we know that 27 grams of Al is equivalent to 1 mole. Therefore, 108 grams of Al is 4 moles. Now we use the stoichiometry of the balanced equation: for every 2 moles of Al that react, 3 moles of H2 are produced. So, if 4 moles of Al react, we’ll get 6 moles of H2. Finally, we convert the number of moles of H2 produced to grams. The molecular weight of H2 is 2(1) = 2. This means that 1 mole of H2 has a mass of 2 grams. Therefore, 6 moles of H2 will have a mass of 6(2 g) = 12 grams.
Example 3-15: How many grams of HCl are required to produce 534 grams of aluminum chloride?
Solution: First, we’ll convert the desired mass of AlCl3 into moles. The molecular weight of AlCl3 is 27 + 3(35.5) = 133.5. This means that 1 mole of AlCl3 has a mass of 133.5 grams. Therefore, 534 grams of AlCl3 is equivalent to 534/133.5 = 4 moles. Next, we use the stoichiometry of the balanced equation. For every 2 moles of AlCl3 that are produced, 6 moles of HCl are consumed. So, if we want to produce 4 moles of AlCl3, we’ll need 12 moles of HCl. Finally, we convert the number of moles of HCl consumed to grams. The molecular weight of HCl is 1 + 35.5 = 36.5. This means that 1 mole of HCl has a mass of 36.5 grams. Therefore, 12 moles of HCl will have a mass of 12(36.5 g) = 438 grams.
Example 3-16: Consider the following reaction:
CS2 + 3 O2 → CO2 + 2 SO2
How much carbon disulfide must be used to produce 64 grams of SO2?
A) 38 g
B) 57 g
C) 76 g
D) 114 g
Solution: Since the molecular weight of SO2 is 32.1 + 2(16) = 64, we know that 64 grams of SO2 is equivalent to 1 mole. From the stoichiometry of the balanced equation, we see that for every 1 mole of CS2 that reacts, 2 moles of SO2 are produced. Therefore, to produce just 1 mole of SO2, we need 1/2 mole of CS2. The molecular weight of CS2 is 12 + 2(32.1) ≈ 76, so 1/2 mole of CS2 has a mass of 38 grams. The answer is A.
3.11 THE LIMITING REAGENT
Let’s look again at the reaction of aluminum with hydrochloric acid:
2 Al + 6 HCl → 2 AlCl3 + 3 H2
Suppose that this reaction starts with 4 moles of Al and 18 moles of HCl. We have enough HCl to make 6 moles of AlCl3 and 9 moles of H2. However, there’s only enough Al to make 4 moles of AlCl3 and 6 moles of H2. There isn’t enough aluminum metal (Al) to make use of all the available HCl. As the reaction proceeds, we’ll run out of aluminum. This means that aluminum is the limiting reagent here, because we run out of this reactant first, so it limits how much product the reaction can produce.
Now suppose that the reaction begins with 4 moles of Al and 9 moles of HCl. There’s enough Al metal to produce 4 moles of AlCl3 and 6 moles of H2. But there’s only enough HCl to make 3 moles of AlCl3 and 4.5 moles of H2. There isn’t enough HCl to make use of all the available aluminum metal. As the reaction proceeds, we’ll find that all the HCl is consumed before the Al is consumed. In this situation, HCl is the limiting reagent. Notice that we had more moles of HCl than we had of Al and the initial mass of the HCl was greater than the initial mass of Al. Nevertheless, the limiting reagent in this case was the HCl. The limiting reagent is the reactant that is consumed first, not necessarily the reactant that’s initially present in the smallest amount.
Example 3-17: Consider the following reaction:
2 ZnS + 3 O2 → 2 ZnO + 2 SO2
If 97.5 grams of zinc sulfide undergoes this reaction with 32 grams of oxygen gas, what will be the limiting reagent?
A) ZnS
B) O2
C) ZnO
D) SO2
Solution: Since the molecular weight of ZnS is 65.4 + 32.1 = 97.5 and the molecular weight of O2 is 2(16) = 32, this reaction begins with 1 mole of ZnS and 1 mole of O2. From the stoichiometry of the balanced equation, we see that 1 mole of ZnS would react completely with ![]() = 1.5 moles of O2. Because we have only 1 mole of O2, the O2 will be consumed first; it is the limiting reagent, and the answer is B. Note that choice C and D can be eliminated immediately, because a limiting reagent is always a reactant.
= 1.5 moles of O2. Because we have only 1 mole of O2, the O2 will be consumed first; it is the limiting reagent, and the answer is B. Note that choice C and D can be eliminated immediately, because a limiting reagent is always a reactant.
3.12 SOME NOTATION USED IN CHEMICAL EQUATIONS
In addition to specifying what atoms or molecules are involved in a chemical reaction, an equation may contain additional information. One type of additional information that can be written right into the equation specifies the phases of the atoms or molecules in the reaction; that is, is the substance a solid, liquid, or gas? Another common condition is that a substance may be dissolved in water when the reaction proceeds. In this case, we’d say the substance is in aqueous solution. These four “states” are abbreviated and written in parentheses as follows:
Solid |
(s) |
Liquid |
(l) |
Gas |
(g) |
Aqueous |
(aq) |
These immediately follow the chemical symbol for the reactant or product in the equation. For example, the reaction of sodium metal with water, which produces sodium hydroxide and hydrogen gas, could be written like this:
2 Na(s) + 2 H2O(l) ![]() 2 NaOH(aq) + H2(g)
2 NaOH(aq) + H2(g)
In some cases, the reactants are heated to produce the desired reaction. To indicate this, we write a “∆”—or the word “heat”—above (or below) the reaction arrow. For example, heating potassium nitrate produces potassium nitrite and oxygen gas:
2 KNO3(s) ![]() 2 KNO2(aq) + O2(g)
2 KNO2(aq) + O2(g)
Some reactions proceed more rapidly in the presence of a catalyst, which is a substance that increases the rate of a reaction without being consumed. For example, in the industrial production of sulfuric acid, an intermediate step is the reaction of sulfur dioxide and oxygen to produce sulfur trioxide. Not only are the reactants heated, but they are combined in the presence of vanadium pentoxide, V2O5. We indicate the presence of a catalyst by writing it below the arrow in the equation:
2 SO2 + O2 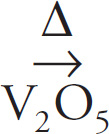 2 SO3
2 SO3
3.13 OXIDATION STATES
An atom’s oxidation state (or oxidation number) is meant to indicate how the atom’s “ownership” of its valence electrons changes when it forms a compound. For example, consider the formula unit NaCl. The sodium atom will transfer its valence electron to the chlorine atom, so the sodium’s “ownership” of its valence electron has certainly changed. To indicate this, we’d say that the oxidation state of sodium is now +1 (or 1 less electron than it started with). On the other hand, chlorine accepts ownership of that 1 electron, so its oxidation state is —1 (that is, 1 more electron than it started with). Giving up ownership results in a more positive oxidation state; accepting ownership results in a more negative oxidation state.
This example of NaCl is rather special (and easy) since the compound is ionic, and we consider ionic compounds to involve the complete transfer of electrons. But what about a non-ionic (that is, a covalent) compound? The oxidation state of an atom is the “charge” it would have if the compound were ionic. Here’s another way of saying this: the oxidation state of an atom in a molecule is the charge it would have if all the shared electrons were completely transferred to the more electronegative element. Note that for covalent compounds, this is not a real charge, just a bookkeeping trick.
The following list gives the rules for assigning oxidation states to the atoms in a molecule. If following one rule in the list causes the violation of another rule, the rule that is higher in the list takes precedence.
Rules for Assigning Oxidation States
1) The oxidation state of any element in its standard state is 0.
2) The sum of the oxidation states of the atoms in a neutral molecule must always be 0, and the sum of the oxidation states of the atoms in an ion must always equal the ion’s charge.
3) Group 1 metals have a +1 oxidation state, and Group 2 metals have a +2 oxidation state.
4) Fluorine has a —1 oxidation state.
5) Hydrogen has a +1 oxidation state when bonded to something more electronegative than carbon, a —1 oxidation state when bonded to an atom less electronegative than carbon, and a 0 oxidation state when bonded to carbon.
6) Oxygen has a —2 oxidation state.
7) The rest of the halogens have a —1 oxidation state, and the atoms of the oxygen family have a —2 oxidation state.
It’s worth noting a common exception to Rule 6: In peroxides (such as H2O2 or Na2O2), oxygen is in a —1 oxidation state.
As we will discuss later, the order of electronegativities of some elements can be remembered with the mnemonic FONClBrISCH (pronounced “fawn-cull-brish”). This lists the elements in order from the most electronegative (F) to the least electronegative (H). Hence, bonds from H to anything before C in FONClBrISCH will give hydrogen a +1 oxidation state, and bonds from H to anything not found in the list will give H a —1 oxidation state.
Let’s find the oxidation number of manganese in KMnO4. By Rule 3, K is +1, and by Rule 6, O is —2. Therefore, the oxidation state of Mn must be +7 in order for the sum of all the oxidation numbers in this electrically-neutral molecule to be zero (the unbreakable Rule 2).
Like many other elements, transition metals can assume different oxidation states, depending on the compound they’re in. (Note, however, that a metal will never assume a negative oxidation state!) For example, iron has an oxidation number of +2 in FeCl2 but an oxidation number of +3 in FeCl3. The oxidation number of a transition metal is given as a Roman numeral in the name of the compound. Therefore, FeCl2 is iron(II) chloride, and FeCl3 is iron(III) chloride.
Example 3-18: Determine the oxidation state of the atoms in each of the following molecules:
a) NO3−
b) HNO2
c) O2
d) SF4
e) Fe3O4
Solution:
a) By Rule 6, the oxidation state of O is —2; therefore, by Rule 2, the oxidation state of N must be +5
b) By Rule 5, the oxidation state of H is +1, and by Rule 6, O has an oxidation state of —2. Therefore, by Rule 2, N must have an oxidation state of +3 in this molecule.
c) By Rule 1 (which is higher in the list than Rule 5 and thus takes precedence), each O atom in O2 has an oxidation state of 0.
d) By Rule 4, F has an oxidation state of —1. So, by Rule 2, S has an oxidation state of +4.
e) By Rule 6, O has an oxidation state of —2. So, by Rule 2, Fe has an oxidation state of +8/3. (Notice that oxidation states do not have to be whole numbers.)