MCAT General Chemistry Review - Steven A. Leduc 2015
Atomic Structure and Periodic Trends
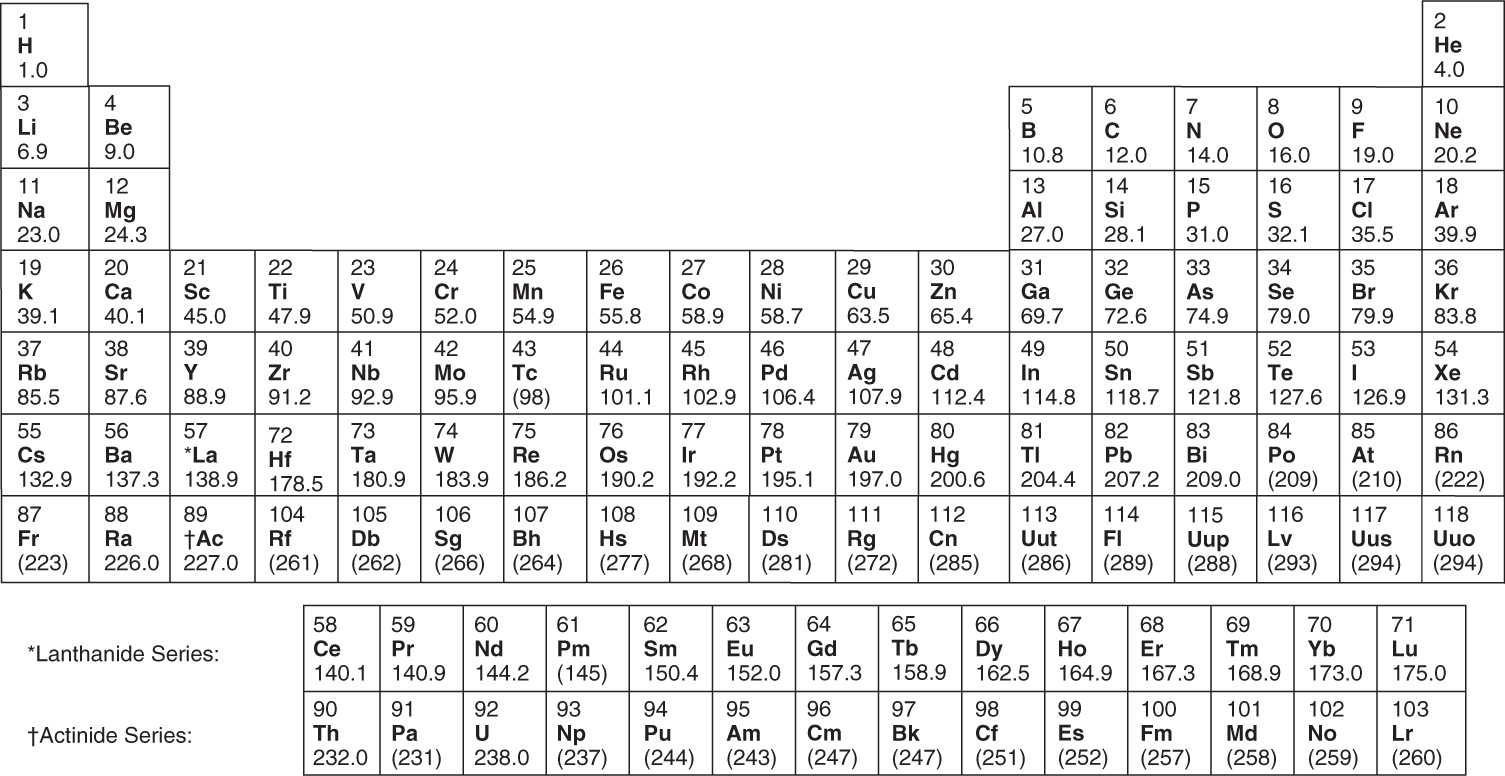
Periodic Table of the Elements
4.1 ATOMS
The smallest unit of any element is one atom of the element. All atoms have a central nucleus, which contains protons and neutrons, known collectively as nucleons. Each proton has an electric charge of +1 elementary unit; neutrons have no charge. Outside the nucleus, an atom contains electrons, and each electron has a charge of —1 elementary unit.
In every neutral atom, the number of electrons outside the nucleus is equal to the number of protons inside the nucleus. The electrons are held in the atom by the electrical attraction of the positively charged nucleus.
The number of protons in the nucleus of an atom is called its atomic number, Z. The atomic number of an atom uniquely determines what element the atom is, and Z may be shown explicitly by a subscript before the symbol of the element. For example, every beryllium atom contains exactly four protons, and we can write this as 4Be.
A proton and a neutron each have a mass slightly more than one atomic mass unit (1 amu = 1.66 × 10−27 kg), and an electron has a mass that’s only about 0.05 percent the mass of either a proton or a neutron. So, virtually all the mass of an atom is due to the mass of the nucleus.
The number of protons plus the number of neutrons in the nucleus of an atom gives the atom’s mass number, A. If we let N stand for the number of neutrons, then A = Z + N.
In designating a particular atom of an element, we refer to its mass number. One way to do this is to write A as a superscript. For example, if a beryllium atom contains 5 neutrons, then its mass number is 4 + 5 = 9, and we would write this as ![]() or simply as 9Be. Another way is simply to write the mass number after the name of the elements, with a hyphen; 9Be is beryllium-9.
or simply as 9Be. Another way is simply to write the mass number after the name of the elements, with a hyphen; 9Be is beryllium-9.
4.2 ISOTOPES
If two atoms of the same element differ in their numbers of neutrons, then they are called isotopes. The atoms shown below are two different isotopes of the element beryllium. The atom on the left has 4 protons and 3 neutrons, so its mass number is 7; it’s 7Be (or beryllium-7). The atom on the right has 4 protons and 5 neutrons, so it’s 9Be (beryllium-9).
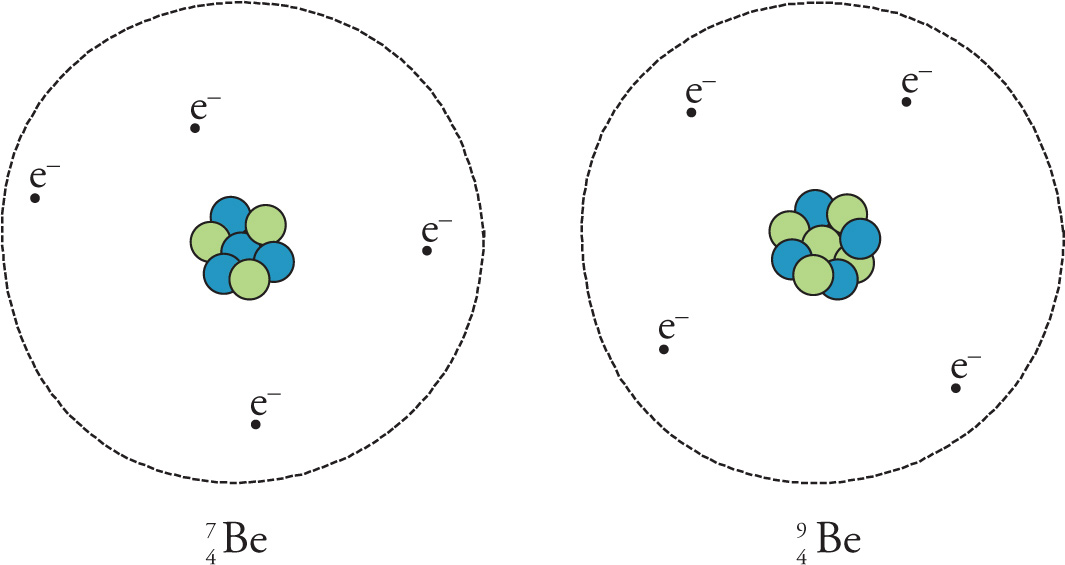
(These figures are definitely not to scale. If they were, each dashed circle showing the “outer edge” of the atom would literally be about 1500 m—almost a mile across! The nucleus occupies only the tiniest fraction of an atom’s volume, which is mostly empty space.) Notice that these atoms—like all isotopes of a given element—have the same atomic number but different mass numbers.
Example 4-1: An atom with 7 neutrons and a mass number of 12 is an isotope of what element?
A) Boron
B) Nitrogen
C) Magnesium
D) Potassium
Solution: If A = 12 and N = 7, then Z = A — N = 12 — 7 = 5. The element with an atomic number of 5 is boron. Therefore, choice A is the answer.
Atomic Weight
Elements exist naturally as a collection of their isotopes. The atomic weight of an element is a weighted average of the masses of its naturally occurring isotopes. For example, boron has two naturally occurring isotopes: boron-10, with an atomic mass of 10.013 amu, and boron-11, with an atomic mass of 11.009 amu. Since boron-10 accounts for 20 percent of all naturally occurring boron, and boron-11 accounts for the other 80 percent, the atomic weight of boron is
(20%)(10.013 amu) + (80%)(11.009 amu) = 10.810 amu
and this is the value listed in the periodic table. (Recall that the atomic mass unit is defined so that the most abundant isotope of carbon, carbon-12, has a mass of precisely 12 amu.)
4.3 IONS
When a neutral atom gains or loses electrons, it becomes charged, and the resulting atom is called an ion. For each electron it gains, an atom acquires a charge of —1 unit, and for each electron it loses, an atom acquires a charge of +1 unit. A negatively charged ion is called an anion, while a positively charged ion is called a cation.
We designate how many electrons an atom has gained or lost by placing this number as a superscript after the chemical symbol for the element. For example, if a lithium atom loses 1 electron, it becomes the lithium cation Li1+, or simply Li+. If a phosphorus atom gains 3 electrons, it becomes the phosphorus anion P3—, or phosphide.
Example 4-2: An atom contains 16 protons, 17 neutrons, and 18 electrons. Which of the following best indicates this ion?
A) 33Cl−
B) 34Cl−
C) 33S2—
D) 34S2—
Solution: Any nucleus that contains 16 protons is sulfur, so we can eliminate choices A and B immediately. Now, because Z = 16 and N = 17, the mass number, A, is Z + N = 16 + 17 = 33. Therefore, the answer is C.
Example 4-3: Of the following atoms/ions, which one contains the greatest number of neutrons?
A) 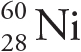
B) 
C) 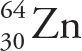
D) 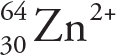
Solution: To find N, we just subtract Z (the subscript) from A (the superscript). The atom in choice A has N = 60 — 28 = 32; the ion in choice B has N = 64 — 29 = 35, and the atom or ion in both choices C and D have N = 64 — 30 = 34. Therefore, of the choices given, the ion in choice B contains the greatest number of neutrons.
4.4 NUCLEAR STABILITY AND RADIOACTIVITY
The protons and neutrons in a nucleus are held together by a force called the strong nuclear force. It’s stronger than the electrical force between charged particles, since for all atoms besides hydrogen, the strong nuclear force must overcome the electrical repulsion between the protons. In fact, of the four fundamental forces of nature, the strong nuclear force is the most powerful even though it only works over extremely short distances, as seen in the nucleus.

Unstable nuclei are said to be radioactive, and they undergo a transformation to make them more stable, altering the number and ratio of protons and neutrons or just lowering their energy. Such a process is called radioactive decay, and we’ll look at three types: alpha, beta and gamma. The nucleus that undergoes radioactive decay is known as the parent, and the resulting more stable nucleus is known as the daughter.
Alpha Decay
When a large nucleus wants to become more stable by reducing the number of protons and neutrons, it emits an alpha particle. An alpha particle, denoted by 42α, consists of 2 protons and 2 neutrons:

This is equivalent to a helium-4 nucleus, so an alpha particle can also be denoted by 42He. Alpha decay reduces the parent’s atomic number by 2 and the mass number by 4. For example, polonium-210 is an α-emitter. It undergoes alpha decay to form the stable nucleus lead-206:
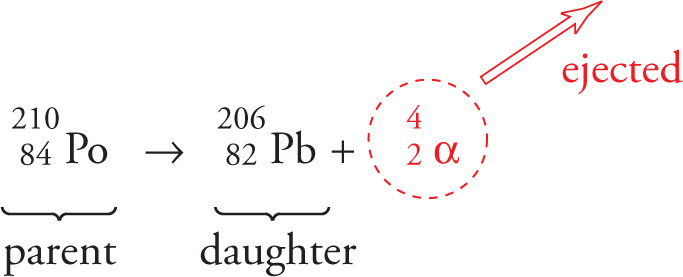
Although alpha particles are emitted with high energy from the parent nucleus, this energy is quickly lost as the particle travels through matter or air. As a result, the particles do not typically travel far, and can be stopped by the outer layers of human skin or a piece of paper.
Beta Decay
There are actually three types of beta decay: β−, β+, and electron capture. Each type of beta decay involves the conversion of a neutron into a proton (along with some other particles that are beyond the scope of the MCAT), or vice versa, through the action of the weak nuclear force.
Beta particles are more dangerous than alpha particles since they are significantly less massive. They therefore have more energy and a greater penetrating ability. However, they can be stopped by aluminum foil or a centimeter of plastic or glass.
β− Decay
When an unstable nucleus contains too many neutrons, it may convert a neutron into a proton and an electron (also known as a β− particle), which is ejected. The atomic number of the resulting daughter nucleus is 1 greater than the radioactive parent nucleus, but the mass number remains the same. The isotope carbon-14, the decay of which is the basis of radiocarbon dating of archaeological artifacts, is an example of a radioactive nucleus that undergoes β− decay:
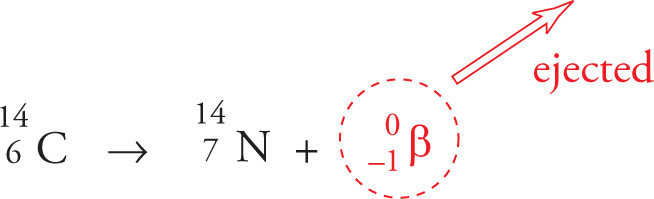
β− decay is the most common type of beta decay, and when the MCAT mentions “beta decay” without any further qualification, it means β− decay.
β+ Decay (or Positron Emission)
When an unstable nucleus contains too few neutrons, it converts a proton into a neutron and a positron, which is ejected. This is known as β+ decay. The positron is the electron’s antiparticle; it’s identical to an electron except its charge is positive. The atomic number of the resulting daughter nucleus is 1 less than the radioactive parent nucleus, but the mass number remains the same. The isotope fluorine-18, which can be used in medical diagnostic bone scans in the form Na18F, is an example of a positron emitter:
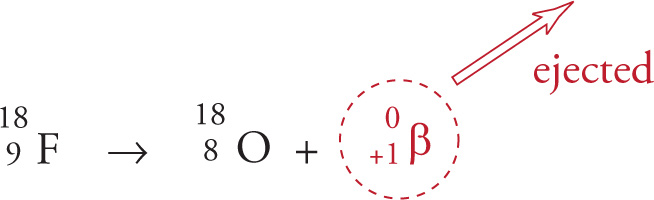
Electron Capture
Another way for an unstable nucleus to increase its number of neutrons is to capture an electron from the closest electron shell (the n = 1 shell) and use it in the conversion of a proton into a neutron. Just like positron emission, electron capture causes the atomic number to be reduced by 1 while the mass number remains the same. The nucleus chromium-51 is an example of a radioactive nucleus that undergoes electron capture, becoming the stable nucleus vanadium-51:
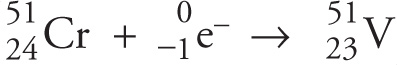
Gamma Decay
A nucleus in an excited energy state—which is usually the case after a nucleus has undergone alpha or any type of beta decay—can “relax” to its ground state by emitting energy in the form of one or more photons of electromagnetic radiation. These photons are called gamma photons (symbolized by γ) and have a very high frequency and energy. Gamma photons (or gamma rays) have neither mass nor charge, and can therefore penetrate matter most effectively. A few inches of lead or about a meter of concrete will stop most gamma rays. Their ejection from a radioactive atom changes neither the atomic number nor the mass number of the nucleus. For example, after silicon-31 undergoes β− decay, the resulting daughter nucleus then undergoes gamma decay:
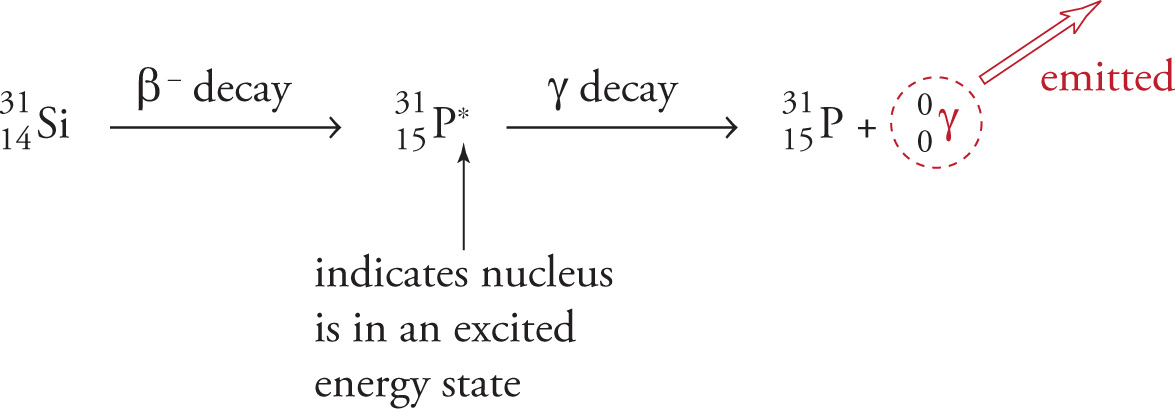
Notice that alpha and beta decay change the identity of the nucleus, but gamma decay does not. Gamma decay is simply an expulsion of energy.
Summary of Radioactive Decay


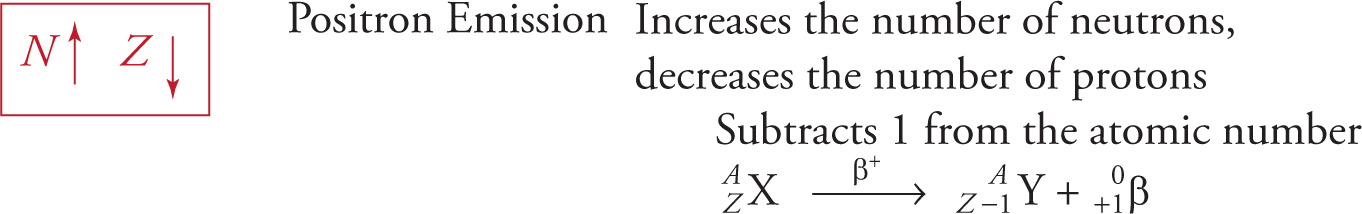
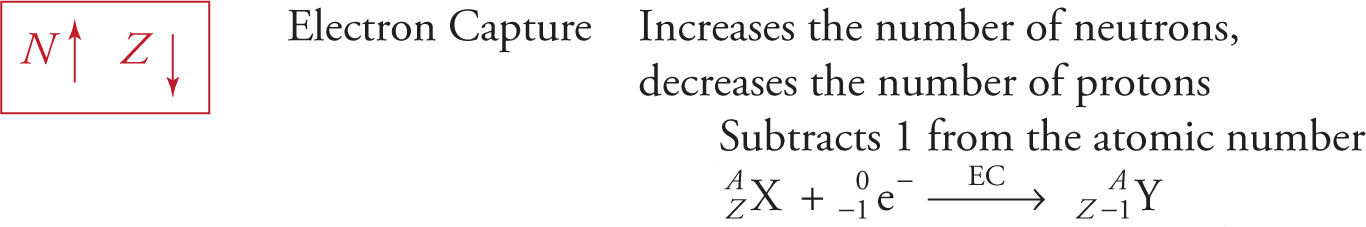
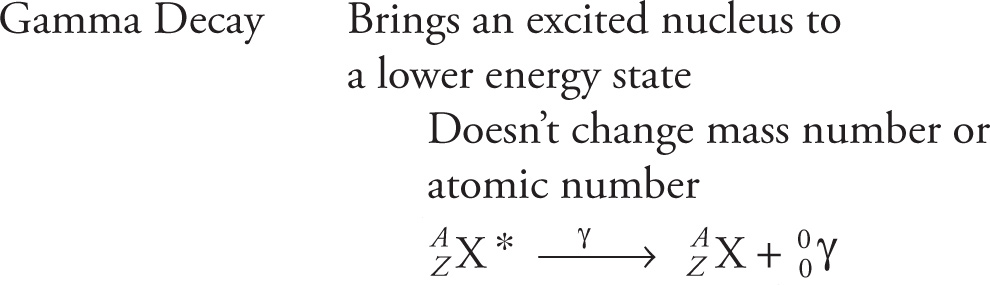
Example 4-4: Radioactive calcium-47, a known β− emitter, is administered in the form of 47CaCl2 by I.V. as a diagnostic tool to study calcium metabolism. What is the daughter nucleus of 47Ca?
A) 46K
B) 47K
C) 47Ca+
D) 47Sc
Solution: Since β− decay will always change the identity of an element, eliminate choice C. The β− decay of 47Ca is described by this nuclear reaction:
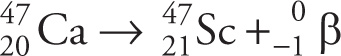
Therefore, the daughter nucleus is scandium-47, choice D.
Example 4-5: Americium-241 is used to provide intracavitary radiation for the treatment of malignancies. This radioisotope is known to undergo alpha decay. What is the daughter nucleus?
A) 237Np
B) 241Pu
C) 237Bk
D) 243Bk
Solution: Alpha decay will reduce the mass by 4, to 237, so eliminate choices B and D. It will reduce the nuclear charge by 2 from 95 to 93, so choose A. The α decay of 241Am is described by this nuclear reaction:
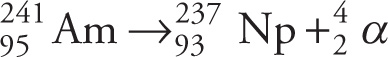
Example 4-6: Vitamin B12 can be prepared with radioactive cobalt (58Co), a known β+ emitter, and administered orally as a diagnostic tool to test for defects in intestinal vitamin B12 absorption. What is the daughter nucleus of 58Co?
A) 57Fe
B) 58Fe
C) 59Co
D) 59Ni
Solution: All types of β+ decay leave the mass of the daughter and parent elements the same, thus the mass must be 58, making choice B the only option. The β+ decay of 58Co is described by this nuclear reaction:
Example 4-7: A certain radioactive isotope is administered orally as a diagnostic tool to study pancreatic function and intestinal fat absorption. This radioisotope is known to undergo β− decay, and the daughter nucleus is xenon-131. What is the parent radioisotope?
A) 131Cs
B) 131I
C) 132I
D) 132Xe
Solution: Eliminate choices C and D since the mass number should remain the same for all forms of β− decay. The β− decay that results in 131Xe is described by this nuclear reaction:
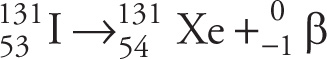
Therefore, the parent nucleus is iodine-131, choice B.
Example 4-8: Which of these modes of radioactive decay causes a change in the mass number of the parent nucleus?
A) α
B) β−
C) β+
D) γ
Solution: Gamma decay causes no changes in the number of protons or neutrons, so we can eliminate choice D. Beta decay (β−, β+, and EC) changes both N and Z by 1, but always such that the change in the sum N + Z (which is the mass number, A) is zero. Therefore, we can eliminate choices B and C. The answer is A.
Example 4-9: One of the naturally occurring radioactive series begins with radioactive 238U. It undergoes a series of decays, one of which is: alpha, beta, beta, alpha, alpha, alpha, alpha, alpha, beta, beta, alpha, beta, alpha, beta. What is the final resulting nuclide of this series of decays?
A) 204Pb
B) 204Pt
C) 206Pb
D) 206Pt
Solution: Since there are so many individual decays, let’s find the final daughter nucleus using a simple shortcut: For every alpha decay, we’ll subtract 4 from the mass number (the superscript) and subtract 2 from the atomic number (the subscript); for every beta decay, we’ll add 0 to the mass number and 1 to the atomic number. Since there are a total of 8 alpha-decays and 6 beta-decays, we get
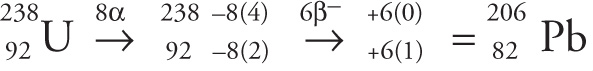
Therefore, the final daughter nucleus is lead-206, choice C.
Half Life
Different radioactive nuclei decay at different rates. The half-life, which is denoted by t1/2, of a radioactive substance is the time it takes for one-half of some sample of the substance to decay. Thus, the shorter the half-life, the faster the decay. The amount of a radioactive substance decreases exponentially with time, as illustrated in the following graph.
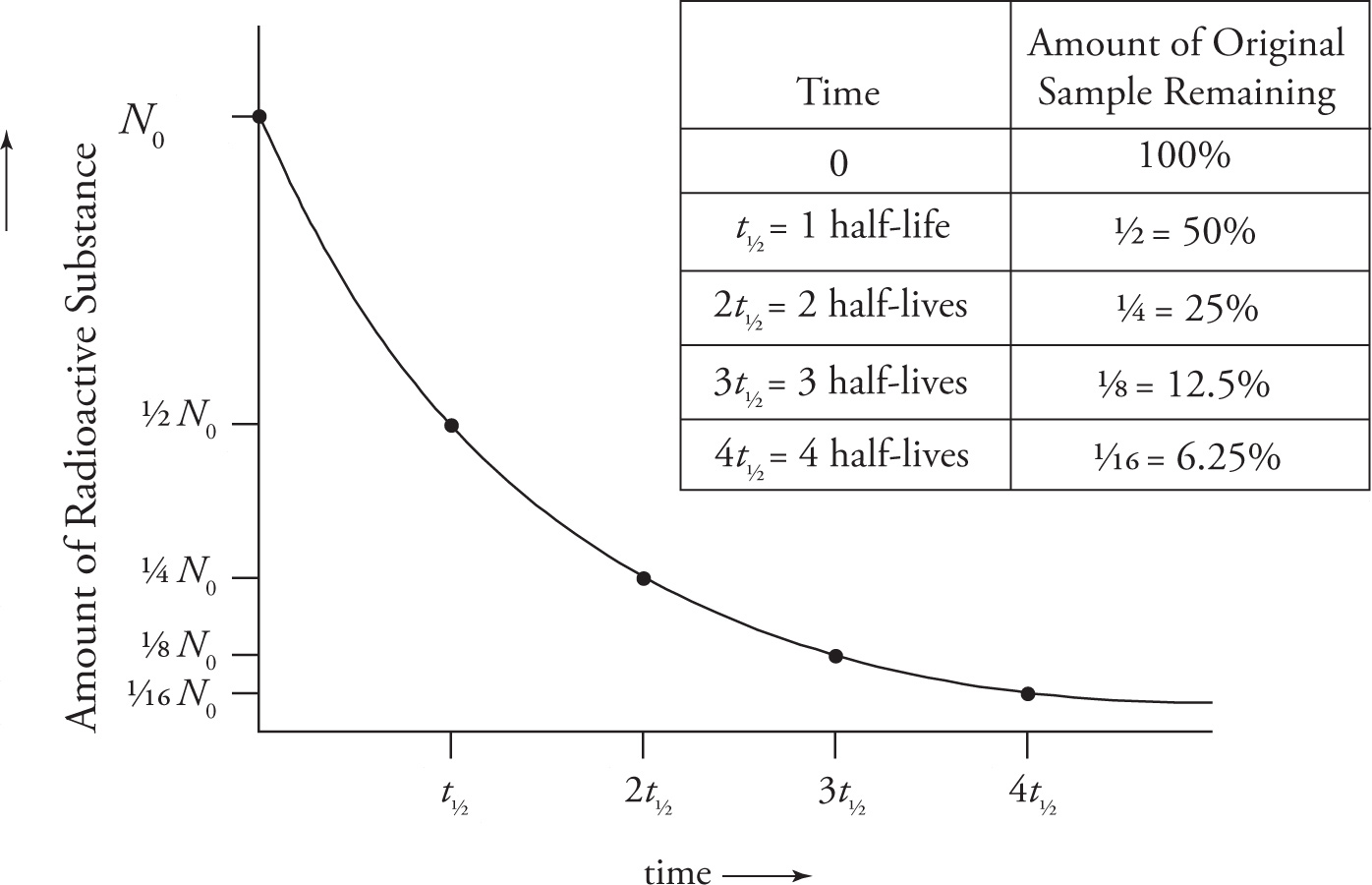
For example, a radioactive sample with an initial mass of 80 grams and a half-life of 6 years will decay as follows:
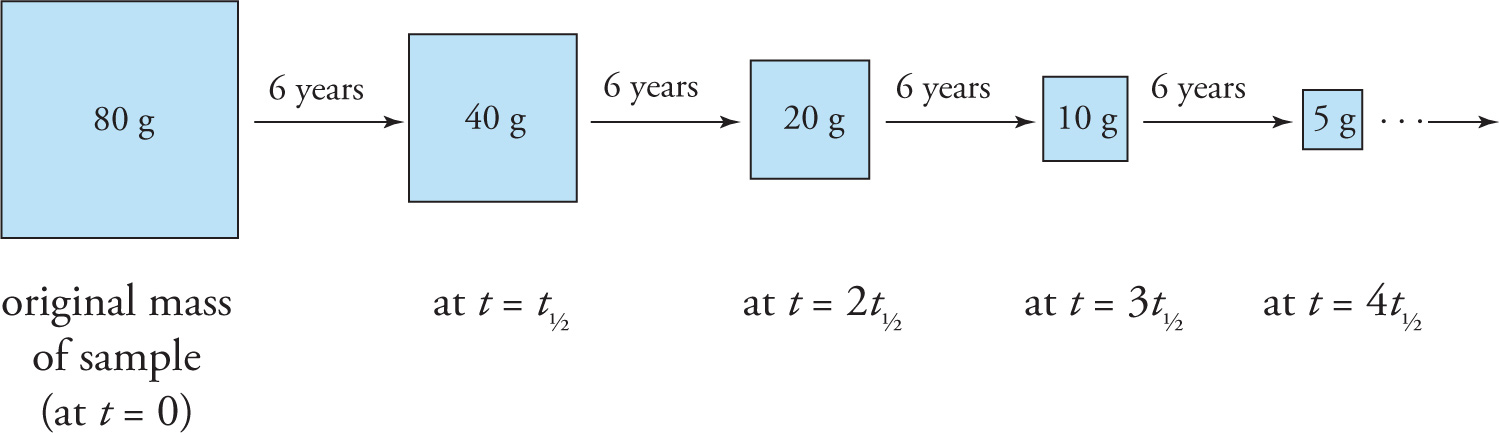
The equation for the exponential decay curve shown above is often written as N = N0e−kt, but a simpler—and much more intuitive way—is
N = N0(1/2)t/t1/2
where t1/2 is the half-life. For example, when t = 3t1/2, the number of radioactive nuclei remaining, N, is N0(1/2)3= 1/8 N0, just what we expect. If the form N0e−kt is used, the value of k (known as the decay constant) is inversely proportional to the half-life: k = (ln 2)/t1/2. The shorter the half-life, the greater the decay constant, and the more rapidly the sample decays.
Example 4-10: Cesium-137 has a half-life of 30 years. How long will it take for only 0.3 g to remain from a sample that had an original mass of 2.4 g?
A) 60 years
B) 90 years
C) 120 years
D) 240 years
Solution: Since 0.3 grams is 1/8 of 2.4 grams, the question is asking how long it will take for the radioisotope to decrease to 1/8 its original amount. We know that this requires 3 half-lives, since 1/2 × 1/2 × 1/2 = 1/8. So, if each half-life is 30 years, then 3 half-lives will be 3(30) = 90 years, choice B.
Example 4-11: Radiolabeled vitamin B12 containing radioactive cobalt-58 is administered to diagnose a defect in a patient’s vitamin-B12 absorption. If 58Co has a half-life of 72 days, approximately what percentage of the radioisotope will still remain in the patient a year later?
A) 3%
B) 5%
C) 8%
D) 10%
Solution: One year is approximately equal to 5 half-lives of this radioisotope, since 5 × 72 = 360 days = 1 year. After 5 half-lives, the amount of the radioisotope will drop to (1/2)5 = 1/32 of the original amount administered. Because 1/32 = 3/100 = 3%, the best answer is choice A.
Example: 4-12 Iodinated oleic acid, containing radioactive iodine-131, is administered orally to study a patient’s pancreatic function. If 131I has a half-life of 8 days, how long after the procedure will the amount of 131I remaining in the patient’s body be reduced to 1/5 its initial value?
A) 19 days
B) 32 days
C) 40 days
D) 256 days
Solution: Although the fraction 1/5 is not a whole-number power of 1/2, we do know that it’s between 1/4 and 1/8. If 1/4 of the sample were left, we’d know that 2 half-lives had elapsed, and if 1/8 of the sample were left, we’d know that 3 half-lives had elapsed. Therefore, because 1/5 is between 1/4 and 1/8, we know that the amount of time will be between 2 and 3 half-lives. Since each half-life is 8 days, this amount of time will be between 2(8) = 16 days and 3(8) = 24 days. Of the choices given, only choice A is in this range.
4.5 ATOMIC STRUCTURE
Emission Spectra
Imagine a glass tube filled with a small sample of an element in gaseous form. When electric current is passed through the tube, the gas begins to glow with a color characteristic of that particular element. If this light emitted by the gas is then passed through a prism—which will separate the light into its component wavelengths—the result is the element’s emission spectrum.

An atom’s emission spectrum gives an energetic “fingerprint” of that element because it consists of a unique sequence of bright lines that correspond to specific wavelengths and energies. The energies of the photons, or particles of light that are emitted, are related to their frequencies, f, and wavelengths, λ, by the equation
Ephoton = hf = h![]()
where h is a universal constant called Planck’s constant (6.63 × 10−34 J⋅s) and c is the speed of light. For the following discussion, a general understanding of the electromagnetic spectrum will be useful. More detail on this topic can be found in Section 13.1 of the MCAT Physics Review.
The Bohr Model of the Atom
In 1913 the Danish physicist Niels Bohr realized that the model of atomic structure of his time was inconsistent with emission spectral data. In order to account for the limited numbers of lines that are observed in the emission spectra of elements, Bohr described a new model of the atom. In this model that would later take his name, he proposed that the electrons in an atom orbited the nucleus in circular paths, much as the planets orbit the sun in the solar system. Distance from the nucleus was related to the energy of the electrons; electrons with greater amounts of energy orbited the nucleus at greater distances. However, the electrons in the atom cannot assume any arbitrary energy, but have quantized energy states, and thereby only orbit at certain allowed distances from the nucleus.
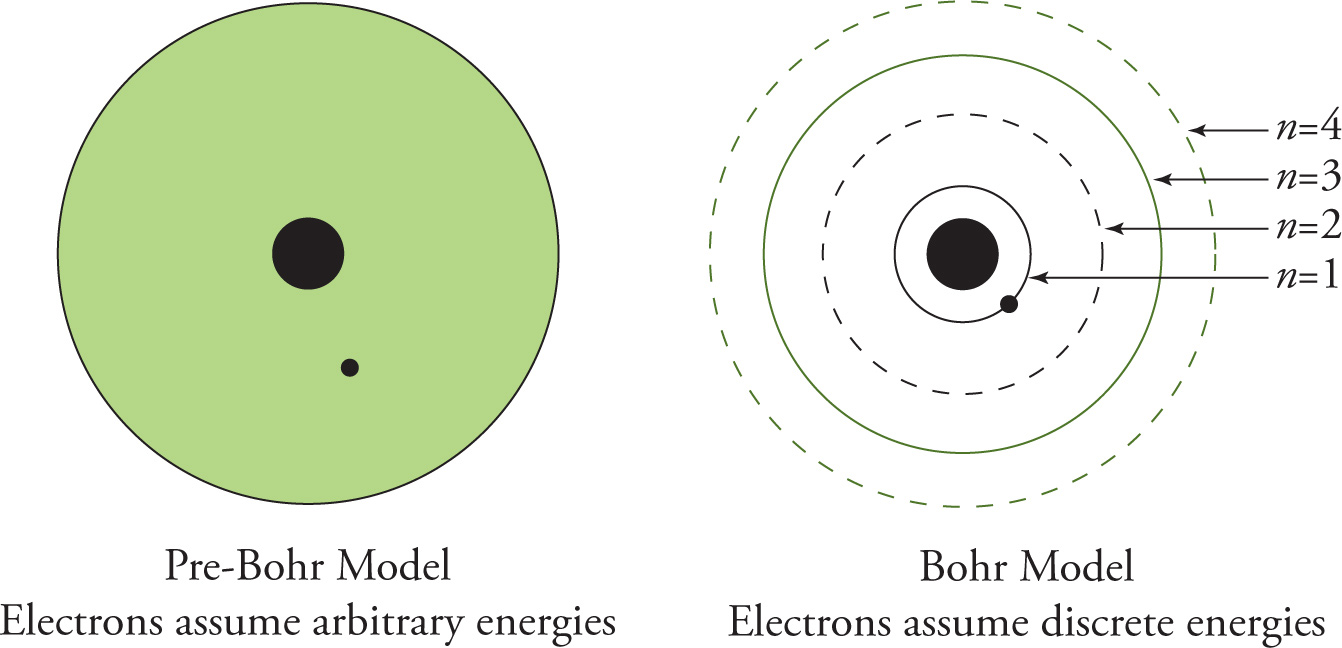
If an electron absorbs energy that’s exactly equal to the difference in energy between its current level and that of an available higher lever, it “jumps” to that higher level. The electron can then “drop” to a lower energy level, emitting a photon with an energy exactly equal to the difference between the levels. This model predicted that elements would have line spectra instead of continuous spectra, as would be the case if transitions between all possible energies could be expected. An electron could only gain or lose very specific amounts of energy due to the quantized nature of the energy levels. Therefore, only photons with certain energies are observed. These specific energies corresponded to very specific wavelengths, as seen in the emission line spectra.

In the transition depicted below, an electron is initially in its ground state (n = 1), or its lowest possible energy level. When this electron absorbs a photon it jumps to a higher energy level, known as an excited state (in this case n = 3). Electrons excited to high energy don’t always relax to the ground state in large jumps, rather they can relax in a series of smaller jumps, gradually coming back to the ground state. From this excited state the electron can relax in one of two ways, either dropping into the n = 2 level, or directly back to the n = 1 ground state. In the first scenario, we can expect to detect a photon with energy corresponding to the difference between n = 3 and n = 2. In the latter case we’d detect a more energetic photon of energy corresponding to the difference between n = 3 and n = 1.
Note: Distances between energy levels are not drawn to scale.
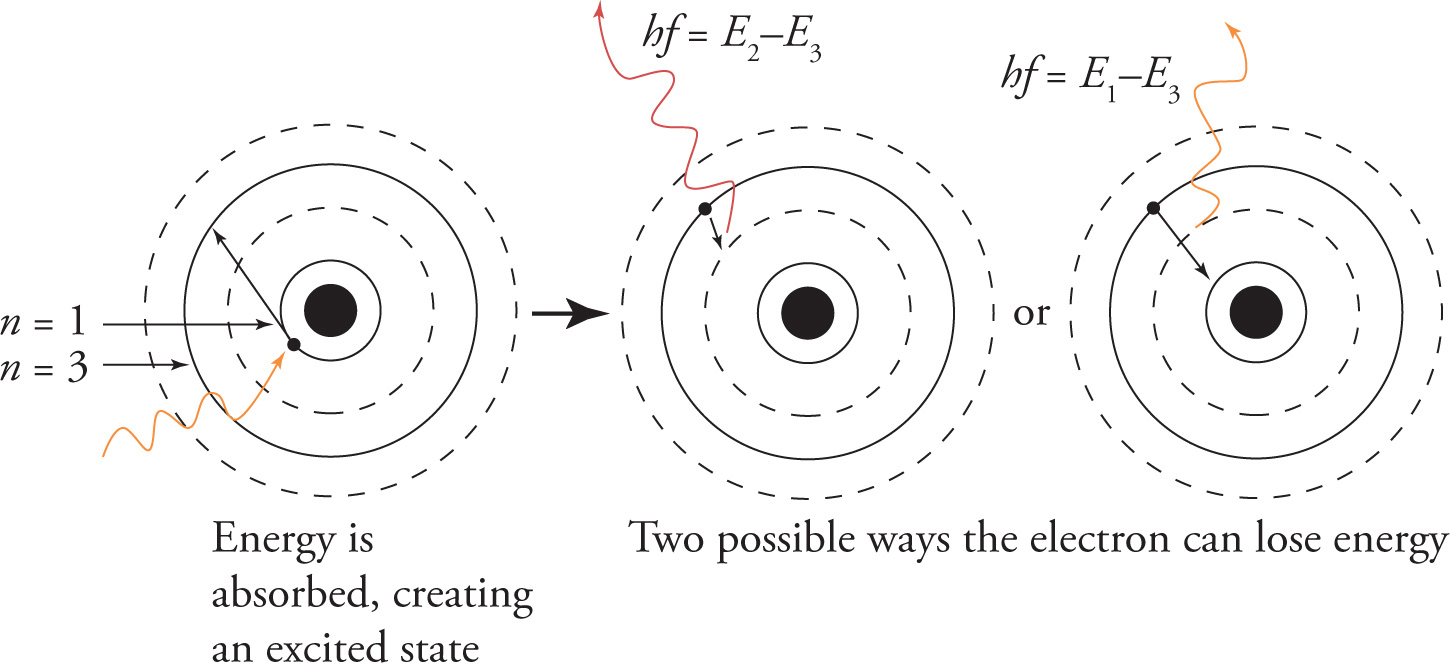
The energies of these discrete energy levels were given by Bohr in the following equation, which only accurately predicted the behavior of atoms or ions containing one electron, now known as Bohr atoms. The value n in this case represents the energy level of the electron.
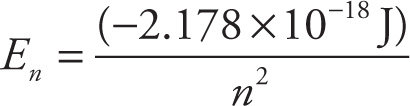
Since we can calculate the energies of the levels of a Bohr atom, we can predict the wavelengths of photons emitted or absorbed when electrons transition between any two energy levels. To do this we calculate the energy differences between discrete levels by subtracting the initial energy of the electron from the final energy of the electron. We can find the energies of the two possible emitted photons shown above as follows:
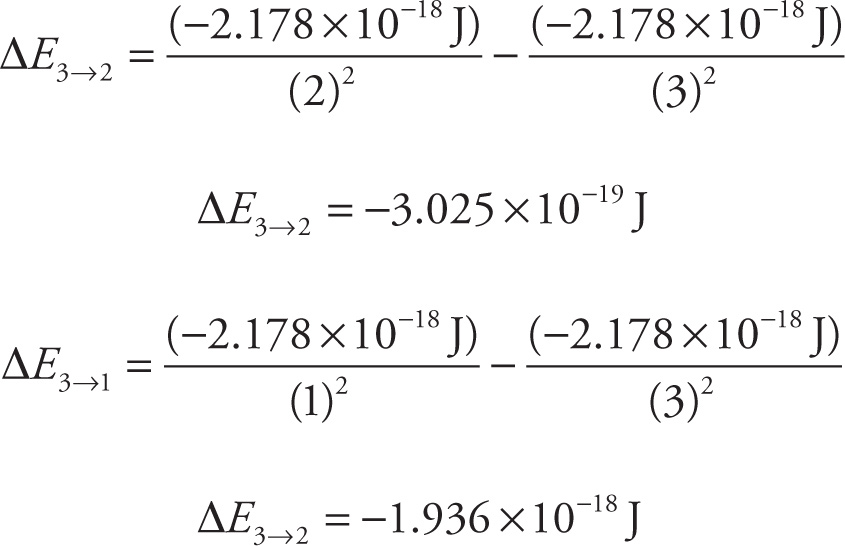
Note that both energies calculated above are negative, indicating that energy is being released by the electron as it falls from its excited state to a lower energy level. For electron transitions from the ground state to an excited state, the ∆E values will be positive, indicating energy is absorbed by the electron.
Once the energy is calculated, the wavelength of the photon can be found by employing the relation ∆E = h![]() . Not all electron transitions produce photons we can see with the naked eye, but all transitions in an atom will produce photons either in the ultraviolet, visible, or infrared region of the electromagnetic spectrum.
. Not all electron transitions produce photons we can see with the naked eye, but all transitions in an atom will produce photons either in the ultraviolet, visible, or infrared region of the electromagnetic spectrum.
Example 4-13: Which of the following is NOT an example of a Bohr atom?
A) H
B) He+
C) Li2+
D) H+
Solution: A Bohr atom is one that contains only one electron. Since H+ has a positive charge from losing the one electron in the neutral atom thereby having no electrons at all, choice D is the answer.
Example 4-14: The first four electron energy levels of an atom are shown below, given in terms of electron volts. Which of the following gives the energy of a photon that could NOT be emitted by this atom?
________ E4 = −18 eV
________ E3 = −32 eV
________ E2 = −72 eV
________ E1 = −288 eV
A) 14 eV
B) 40 eV
C) 44 eV
D) 54 eV
Solution: The difference between E4 and E3 is 14 eV, so a photon of 14 eV would be emitted if an electron were to drop from level 4 to level 3; this eliminates choice A. Similarly, the difference between E3 and E2 is 40 eV, so choice B is eliminated, and the difference between E4 and E2 is 54 eV, so choice D is eliminated. The answer must be C; no two energy levels in this atom are separated by 44 eV.
Example 4-15: Consider two electron transitions. In the first case, an electron falls from n = 4 to n = 2, giving off a photon of light with a wavelength equal to 488 nm. In the second transition, an electron moves from n = 3 to n = 4. For this transition, we would expect that:
A) energy is emitted, and the wavelength of the corresponding photon will be shorter than the first transition.
B) energy is emitted, and the wavelength of the corresponding photon will be longer than the first transition.
C) energy is absorbed, and the wavelength of the corresponding photon will be shorter than the first transition.
D) energy is absorbed, and the wavelength of the corresponding photon will be longer than the first transition.
Solution: Since the electron is moving from a lower to higher energy level, we would expect that the atom absorbs energy (eliminating choices A and B). Since the electron transitions between energy levels that are closer together, the ∆E between levels is smaller. By the ∆E = h![]() relationship, we know that energy and wavelength are inversely related. Therefore with a smaller energy change, the wavelength of the associated light will be longer. D is the correct answer.
relationship, we know that energy and wavelength are inversely related. Therefore with a smaller energy change, the wavelength of the associated light will be longer. D is the correct answer.
The Quantum Model of the Atom
While one-electron atoms produce easily predicted atomic spectra, the Bohr model does not do a good job of predicting the atomic spectra of many-electron atoms. This shows that the Bohr model cannot describe the electron-electron interactions that exist in many-electron atoms. The quantum model of the atom was developed to account for these differences. Bohr’s model suggested, and we still hold to be true, that electrons held by an atom can exist only at discrete energy levels—that is, electron energy levels are quantized. This quantization is described by a unique “address” for each electron, consisting of four quantum numbers designating the shell, subshell, orbital, and spin. While the details of quantum numbers are beyond the scope of the MCAT, it is still useful to understand the conceptual basis of the quantum model.
The Energy Shell
The energy shell (n) of an electron in the quantum model of the atom is analogous to the circular orbits in the Bohr model of the atom. An electron in a higher shell has a greater amount of energy and a greater average distance from the nucleus. For example, an electron in the 3rd shell (n = 3) has a higher energy than an electron in the 2nd shell (where n = 2), which has more energy than an electron in the 1st shell (n = 1).
The Energy Subshell
In the quantum model of the atom, however, we no longer describe the path of electrons around the nucleus as circular orbits, but focus on the probability of finding an electron somewhere in the atom. Loosely speaking, an orbital describes a three-dimensional region around the nucleus in which the electron is most likely to be found.
A subshell in an atom is comprised of one or more orbitals, and is denoted by a letter (s, p, d, or f) that describes the shape and energy of the orbital(s). The orbitals in the subshells get progressively more complex and higher in energy in the order listed above. Each energy shell has one or more subshells, and each higher energy shell contains one additional subshell. For example, the first energy shell contains the s subshell, while the second energy shell contains both the s and p subshell, etc.
The Orbital Orientation
Each subshell contains one or more orbitals of the same energy (also called degenerate orbitals), and these orbitals have different three-dimensional orientations in space. The number of orientations increases by two in each successive subshell. For example, the s subshell contains one orientation and the p subshell contains three orientations.
You should be able to recognize the shapes of the orbitals in the s and p subshells. Each s subshell has just one spherically symmetrical orbital.
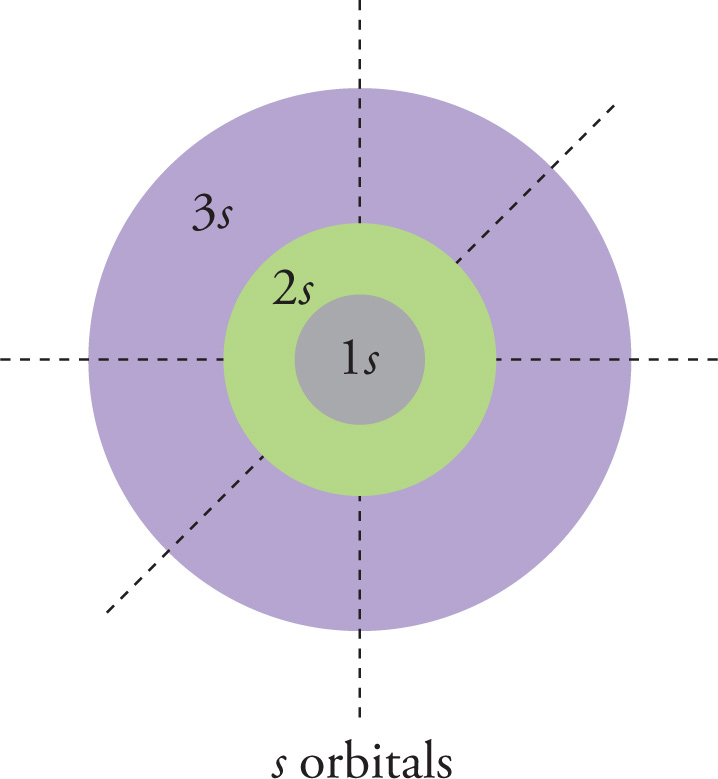
Each p subshell has three orbitals, each depicted as a dumbbell, with different spatial orientations.
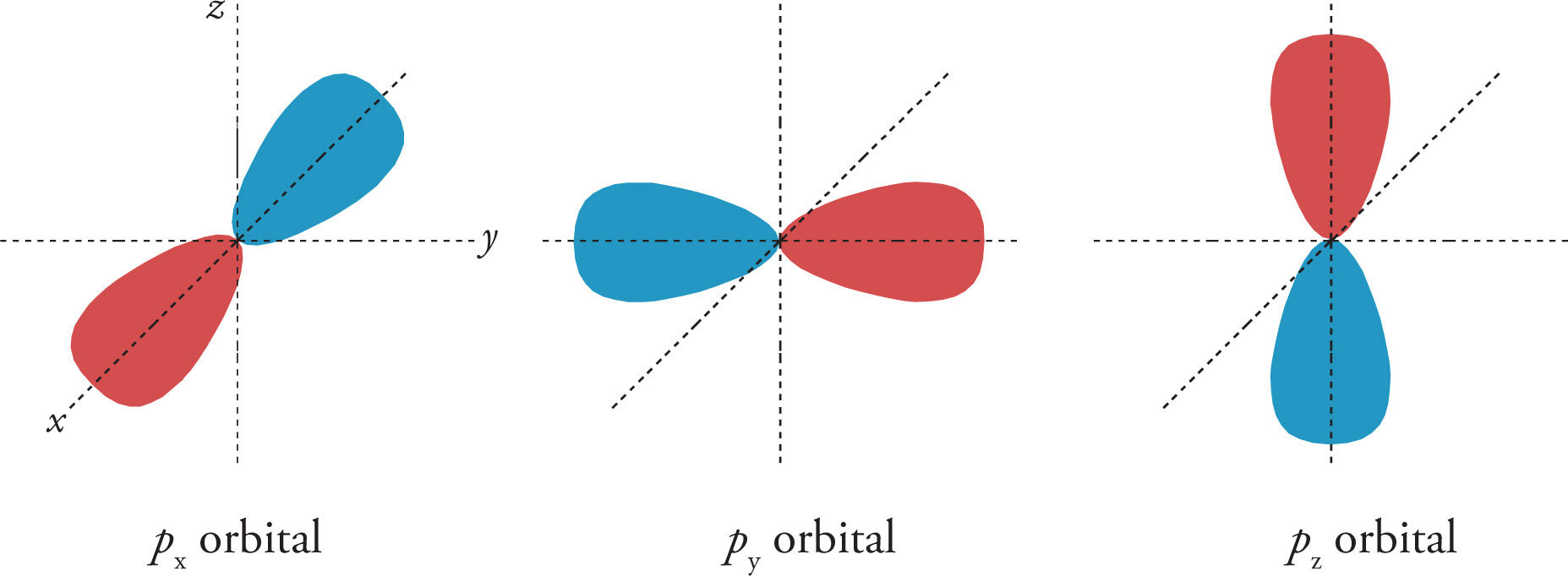
The Electron Spin
Every electron has two possible spin states, which can be considered the electron’s intrinsic magnetism. Because of this every orbital can accommodate a maximum of two electrons, one spin-up and one spin-down. If an orbital is full, we say that the electrons it holds are “spin-paired.”
4.6 ELECTRON CONFIGURATIONS
Now that we’ve described the modern quantum model of the atom, let’s see how this is represented as an electron configuration. There are three basic rules:
1) Electrons occupy the lowest energy orbitals available. (This is the Aufbau principle.) Electron subshells are filled in order of increasing energy. The periodic table is logically constructed to reflect this fact, and therefore one can easily determine shell filling for specific atoms based on where they appear on the table. We will detail this in the next section on “Blocks.”
2) Electrons in the same subshell occupy available orbitals singly, before pairing up. (This is known as Hund’s rule.)
3) There can be no more than two electrons in any given orbital. (This is the Pauli exclusion principle.)
For example, let’s describe the locations for all the electrons in an oxygen atom, which contains eight electrons. Beginning with the first, lowest energy shell, there is only one subshell (s) and only one orientation in that subshell, and there can only be two electrons in that one orbital. Therefore, these two electrons fill the only orbital in the 1s subshell. We write this as 1s2, to indicate that there are two electrons in the 1s subshell.
We still have six electrons left, so let’s move on to the second, next highest, energy shell. There are two subshells (s and p). Since the s subshell is lower in energy than the p subshell, the next two electrons go in the 2s subshell, that is, 2s2.
For the remaining four electrons, there would be three orientations of orbitals in the p subshell. According to Hund’s rule, we place one spin up electron in each of these three orbitals. The eighth electron now pairs up with an electron in one of the 2p orbitals. So, the last four electrons go in the 2p subshell: 2p4 (or more explicitly, 2px22py12pz1).
The complete electron configuration for oxygen can now be written like this:
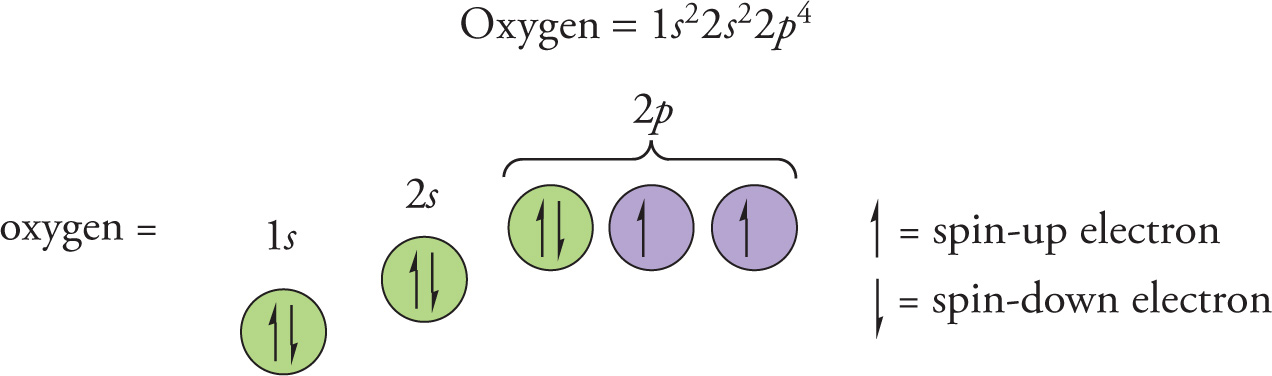
Here are the electron configurations for the first ten elements:

Example 4-16: What’s the maximum number of electrons that can go into any s subshell? Any p subshell? Any d? Any f?
Solution: An s subshell has only one possible orbital orientation. Since only two electrons can fill any given orbital, an s subshell can hold no more than 1 × 2 = 2 electrons.
A p subshell has three possible orbital orientations (two more than an s subshell). Since again only two electrons can fill any given orbital, a p subshell can hold no more than 3 × 2 = 6 electrons.
A d subshell has five possible orbital orientations (two more than a p subshell). Since there are two electrons per orbital, a d subshell can hold no more than 5 × 2 = 10 electrons.
Finally, an f subshell has seven possible orbital orientations (two more than a d subshell). Since there are two electrons per orbital, an f subshell can hold no more than 7 × 2 = 14 electrons.
Example 4-17: Write down—and comment on—the electron configuration of argon (Ar, atomic number 18).
Solution: We have 18 electrons to successively place in the proper subshells, as follows:
1s: 2 electrons
2s: 2 electrons
2p: 6 electrons
3s: 2 electrons
3p: 6 electrons
Therefore,
[Ar] = 1s22s22p63s23p6
Notice that 3s and 3p subshells have their full complement of electrons. In fact, the noble gases (those elements in the last column of the periodic table) all have their outer 8 electrons in filled subshells: 2 in the ns subshell plus 6 in the np. (The lone exception, of course, is helium; but its one and only subshell, the 1s, is filled—with 2 electrons.) Because their 8 valence electrons are in filled subshells, we say that these atoms—Ne, Ar, Kr, Xe, and Rn—have a complete octet, which accounts for their remarkable chemical inactivity.
Diamagnetic and Paramagnetic Atoms
An atom that has all of its electrons spin-paired is referred to as diamagnetic. For example, helium, beryllium, and neon are diamagnetic. A diamagnetic atom must contain an even number of electrons and have all of its occupied subshells filled. Since all the electrons in a diamagnetic atom are spin-paired, the individual magnetic fields that they create cancel, leaving no net magnetic field. Such an atom will be repelled by an externally produced magnetic field.
If an atom’s electrons are not all spin-paired, it is said to be paramagnetic. Paramagnetic atoms are attracted into externally produced magnetic fields.
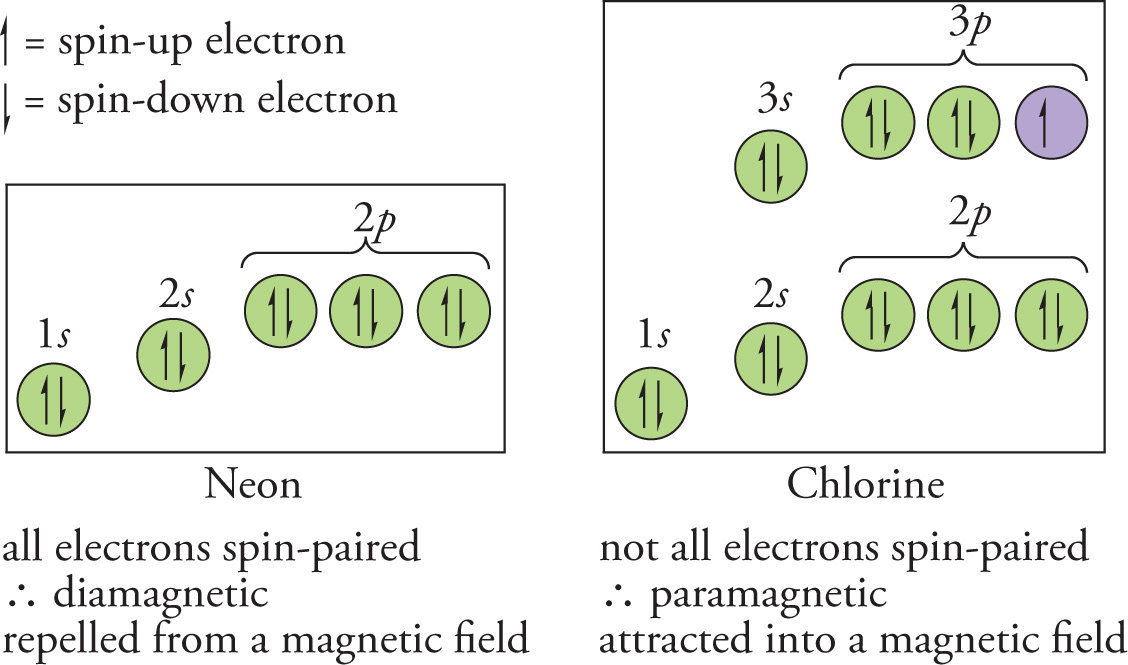
Example 4-18: Which of the following elements is diamagnetic?
A) Sodium
B) Sulfur
C) Potassium
D) Calcium
Solution: First, a diamagnetic atom must contain an even number of electrons, because they all must be spin-paired. So, we can eliminate choices A and C, since sodium and potassium each contain an odd number of electrons (11 and 19, respectively). The electron configuration of sulfur is [Ne] 3s23p4; by Hund’s rule, the 4 electrons in the 3p subshell will look like this:
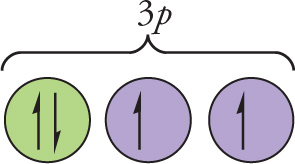
They’re not all spin-paired, so sulfur is not diamagnetic. The answer must be D, calcium, because its configuration is [Ar] 4s2, and all of its electrons are spin-paired.
Blocks in the Periodic Table
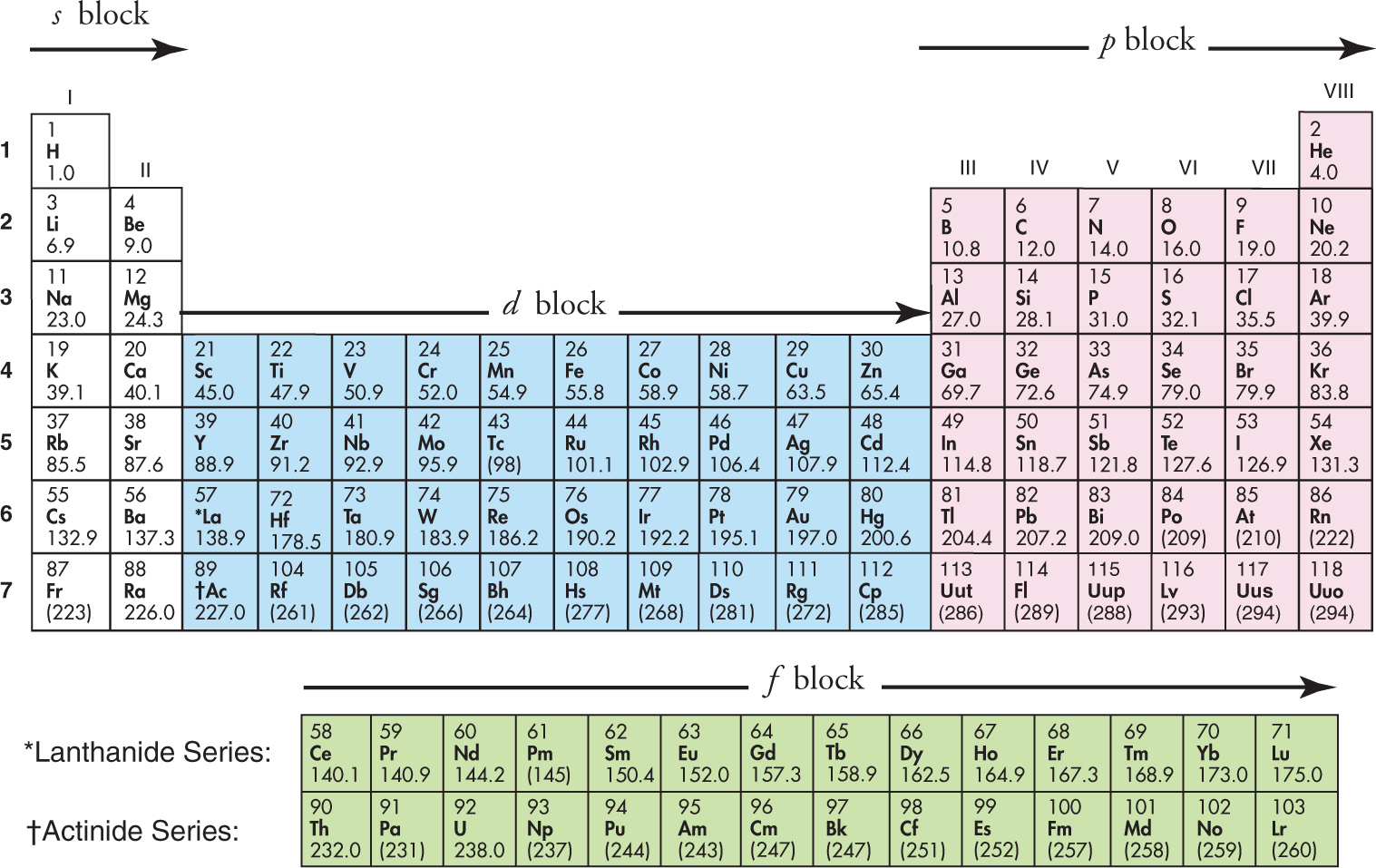
The periodic table can be divided into blocks, as shown above. The name of the block (s, p, d, or f ) indicates the highest-energy subshell containing electrons in the ground-state of an atom within that block. For example, carbon is in the p block, and its electron configuration is 1s22s22p2; the highest-energy subshell that contains electrons (the 2p) is a p subshell. In addition, each horizontal row in the periodic table is called a period, and each vertical column is called a group (or family). The bold numbers next to the rows on the left indicate the period number; for example, potassium (K, atomic number 19) is in Period 4.
How do we use this block diagram to write electron configurations? To illustrate, let’s say we want to write the configuration for chlorine (Z = 17). To get to Z = 17, imagine starting at Z = 1 (hydrogen) and filling up the subshells as we move along through the rows to Z = 17. (Notice that helium has been moved over next to hydrogen for purposes of this block diagram.) We’ll first have 1s2 for the 2 atoms in Period 1, s block (Z = 1 and Z = 2); the 2s2 for the next 2 atoms, which are in Period 2, s block (Z = 3 and Z = 4); then 2p6 for the next 6 atoms, which are in Period 2, p block (Z = 5 through Z = 10); the 3s2 for the next 2 atoms, which are in Period 3, s block (Z = 11 and Z = 12); then, finally, 3p5 for the atoms starting with aluminum, Al, in Period 3, p block and counting through to chlorine, Cl. So, we’ve gone through the rows and blocks from the beginning and stopped once we hit the atom we wanted, and along the way we obtained 1s22s22p63s23p5. This is the electron configuration of chlorine.
The noble gases are often used as starting points, because they are at the end of the rows and represent a shell being completely filled; all that’s left is to count over in the next row until the desired atom is reached. We find the closest noble gas that has an atomic number less than that of the atom for which we want to find an electron configuration. In the case of chlorine (Z = 17), the closest noble gas with a smaller atomic number is neon (Z = 10). Starting with neon, we have 7 additional electrons to take care of. To get to Z= 17, we go through the 2 atoms in the s block of Period 3 (3s2), then notice that Cl is the fifth element in the p block, giving us 3p5. Therefore, the electron configuration of chlorine is the same as that of neon plus 3s23p5, which we can write like this: Cl = [Ne] 3s23p5.
The simple counting through the rows and blocks works as long as you remember this simple rule: Whenever you’re in the d block, subtract 1 from the period number. For example, the first row of the d block (Z = 21 through Z = 30) is in Period 4, but instead of saying that these elements have their outermost (or valence) electrons in the 4d subshell, we subtract 1 from the period number and say that these elements put their valence electrons in the 3d subshell.
In summary: The block in the table tells us in which subshell the outermost (valence) electrons of the atom will be. The period (row) gives the shell, n, as long as we remember the following fact about the atoms in the d block: electrons for an atom in the d block of Period n go into the subshell (n—1)d. For example, the electron configuration for scandium (Sc, atomic number 21) is [Ar]4s23d1. (Note: if you ever need to write the electron configuration for an element in the f block, the rule is: In the f block, subtract 2 from the period number.)
Example 4-19: Which of the following gives the electron configuration of an aluminum atom?
A) 1s22s22p1
B) 1s22s22p2
C) 1s22s22p63s23p1
D) 1s22s22p63s23p2
Solution: Since aluminum (Al) has atomic number 13, a neutral aluminum atom must have 13 electrons. This observation alone eliminates choices A, B, and D (which indicate a total of 5, 6, and 14 electrons, respectively), so the answer must be C.
Example 4-20: What is the maximum number of electrons that can be present in the n = 3 shell?
A) 6
B) 9
C) 12
D) 18
Solution: Every new energy level (n) adds a new subshell. That means that in the first energy level we have only the s subshell, while when n = 2 we have both s and p subshells, and when n = 3, there are s, p, and d subshells. Since there are 1, 3, and 5 s, p, and d orbitals, respectively, for a total of 9 orbitals, and since the maximum number of electrons in an orbital is 2, there can be a maximum of 18 electrons in the n = 3 shell.
Example 4-21: What’s the electron configuration of a zirconium atom (Z = 40)?
A) [Kr] 4d4
B) [Kr] 5s24d2
C) [Kr] 5s25p2
D) [Kr] 5s25d2
Solution: Zirconium (Zr) is in the d block of Period 5. After krypton (Kr, atomic number 36), we’ll have 5s2 for the next 2 atoms in the Period 5, s block (Z = 37 and Z = 38). Then, remembering the rule that electrons for an atom in the d block of Period n go into the subshell (n — 1)d, we know that the last two electrons will go in the 4d (not the 5d) subshell. Therefore, the answer is B.
Some Anomalous Electron Configurations
The process described above (reading across the periodic table, from top to bottom and left to right, using the blocks as a tool for the order of filling of subshells) to determine an atom’s electron configuration works quite well for a large percentage of the elements, but there are a few atoms for which the anticipated electron configuration is not the actual configuration observed.
In a few instances, atoms can achieve a lower energy state (or a higher degree of stability) by having a filled or half-filled, d subshell. For example, consider chromium (Cr, Z = 24). On the basis of the block diagram, we’d expect its electron configuration to be [Ar] 4s23d 4. Recalling that a d subshell can hold a maximum of 10 electrons, it turns out that chromium achieves a more stable state by filling its d subshell with 5 electrons (half-filled) rather than leaving it with 4. This is accomplished by promoting one of its 4s electrons to the 3d subshell, yielding the electron configuration [Ar]4s13d 5. As another example, copper (Cu, Z = 29) has an expected electron configuration of [Ar] 4s23d 9. However, a copper atom obtains a more stable, lower-energy state by promoting one of its 4s electrons into the 3d subshell, yielding [Ar] 4s13d 10 to give a filled d subshell.
Other atoms that display the same type of behavior with regard to their electron configuration as do chromium and copper include molybdenum (Mo, Z = 42, in the same family as chromium), as well as silver and gold (Ag and Au, Z = 47 and Z = 79, respectively, which are in the same family as copper).
Example 4-22: What is the electron configuration of an atom of silver?
Solution: As mentioned above, silver is one of the handful of elements with atoms that actually achieve greater overall stability by promoting one of its electrons into a higher subshell in order to make it filled. We’d expect the electron configuration for silver to be [Kr]5s24d 9. But, by analogy with copper, we’d predict (correctly) that the actual configuration of silver is [Kr] 5s14d 10, where the atom obtains a more stable state by promoting one of its 5s electrons into the 4d subshell, to give a filled d subshell.
Electron Configurations of Ions
Recall that an ion is an atom that has acquired a nonzero electric charge. An atom with more electrons than protons is negatively charged and is called an anion; an atom with fewer electrons than protons is positively charged and is called a cation.
Atoms that gain electrons (anions) accommodate them in the first available orbital, the one with the lowest available energy. For example, fluorine (F, Z = 9) has the electron configuration 1s22s22p5. When a fluorine atom gains an electron to become the fluoride ion, F−, the additional electron goes into the 2p subshell, giving the electron configuration 1s22s22p6, which is the same as the configuration of neon. For this reason, F− and Ne are said to be isoelectronic.
In order to write the electron configuration of an ion for an element in the s or p blocks, we can use the blocks in the periodic table as follows. If an atom becomes an anion—that is, if it acquires one or more additional electrons—then we move to the right within the table by a number of squares equal to the number of electrons added in order to find the atom with the same configuration as the ion.
If an atom becomes a cation—that is, if it loses one or more electrons—then we move to the left within the table by a number of squares equal to the number of electrons lost in order to find the atom with the same configuration as the ion.
Example 4-23: What’s the electron configuration of P3— ? Of Sr+?
Solution: To find the configuration of P3—, we locate phosphorus (P, Z = 15) in the periodic table and move 3 places to the right (because we have an anion with charge of 3−); this lands us on argon (Ar, Z = 18). Therefore, the electron configuration of the anion P3— is the same as that of argon: 1s22s22p63s23p6.
To find the configuration of Sr+, we locate strontium (Sr, Z = 38) in the periodic table and move 1 place to the left (because we have a cation with charge 1+), thus landing on rubidium (Rb, Z = 37). Therefore, the electron configuration of the anion Sr+ is the same as that of rubidium: [Kr] 5s1.
Electrons that are removed (ionized) from an atom always come from the valence shell (the highest n level), and the highest energy orbital within that level. For example, an atom of lithium, Li (1s22s1), becomes Li+ (1s2) when it absorbs enough energy for an electron to escape. However, recall from our discussion above that transition metals (which are the elements in the d block) have both ns and (n — 1)d electrons. To form a cation, atoms will always lose their valence electrons first, and since n > n — 1, transition metals lose s electrons before they lose d electrons. Only after all s electrons are lost do d electrons get ionized. For example, the electron configuration for the transition metal titanium (Ti, Z = 22) is [Ar] 4s23d2. We might expect that the electron configuration of the ion Ti+ to be [Ar] 4s23d1 since the d electrons are slightly higher in energy. However, the actual configuration is [Ar] 4s13d2, and the valence electrons (the ones from the highest n level) are ALWAYS lost first. Similarly, the electron configuration of Ti2+ is not [Ar] 4s2—it’s actually [Ar] 3d2.
Example 4-24: Which one of the following ions has the same electron configuration as the noble gas argon?
A) Na+
B) P2—
C) Al3+
D) Cl−
Solution: Na+ (choice A) has the same electron configuration as the noble gas neon, not argon, since one element to the left of Na is Ne. The ion P2— has the same electron configuration as Cl, which is two elements to the right of P. Al3+, like Na+, has the same configuration as Ne. Of the choices given, only Cl− (choice D) has the same configuration as Ar, since Ar is one element to the right of Cl.
Example 4-25: What’s the electron configuration of Cu+? Of Cu2+? Of Fe3+?
Solution: Copper (Cu, Z = 29) is a transition metal, so it will lose its valence s electrons before losing any d electrons. Recall the anomalous electron configuration of Cu (to give it a filled 3d subshell): [Ar] 4s13d 10. Therefore, the configuration of Cu+ (the cuprous ion, Cu(I)) is [Ar] 3d 10, and that of Cu2+ (the cupric ion, Cu(II)) is [Ar] 3d 9. Since the electron configuration of iron (Fe, Z = 26) is [Ar] 4s23d 6, the configuration of Fe3+ (the ferric ion, Fe(III)) is [Ar] 3d 5, since the transition metal atom Fe first loses both of its valence s electrons, then once they’re ionized, one of its d electrons.
Excited State vs. Ground State
Assigning electron configurations as we’ve just discussed is aimed at constructing the most probable location of electrons, following the Aufbau principle. These configurations are the most probable because they are the lowest in energy, or as they are often termed, the ground state.
Any electron configuration of an atom that is not as we would assign it, provided it doesn’t break any physical rules (no more than 2 e− per orbital, no assigning non existent shells such as 2d, etc.…) is an excited state. The atom has absorbed energy, so the electrons now inhabit states we wouldn’t predict as the most probable ones.
Example 4-26: Which of the following could be the electron configuration of an excited oxygen atom?
A) 1s22s22p4
B) 1s22s22p5
C) 1s22s22p33s1
D) 1s22s22p43s1
Solution: An oxygen atom contains 8 electrons; when excited, one (or more) of these electrons will jump to a higher energy level. Choice A is the configuration of a ground-state oxygen atom, and choices B and D show the placement of 9 electrons, not 8, so both may be eliminated. The answer must be C; one of the 2p electrons has jumped to the 3s subshell. (Note carefully that an excited atom is not an ion; electrons are not lost or gained; they simply jump to higher energy levels within the atom.
4.7 GROUPS OF THE PERIODIC TABLE AND THEIR CHARACTERISTICS
We will use the electron configurations of the atoms to predict their chemical properties, including their reactivity and bonding patterns with other atoms.
Recall that each horizontal row in the periodic table is called a period, and each vertical column is called a group (or family). Within any group in the periodic table, all of the elements have the same number of electrons in their outermost shell. For instance, the elements in Group II all have two electrons in their outermost shell. Electrons in an atom’s outermost shell are called valence electrons, and it’s the valence electrons that are primarily responsible for an atom’s properties and chemical behavior.
Some groups (families) have special names.
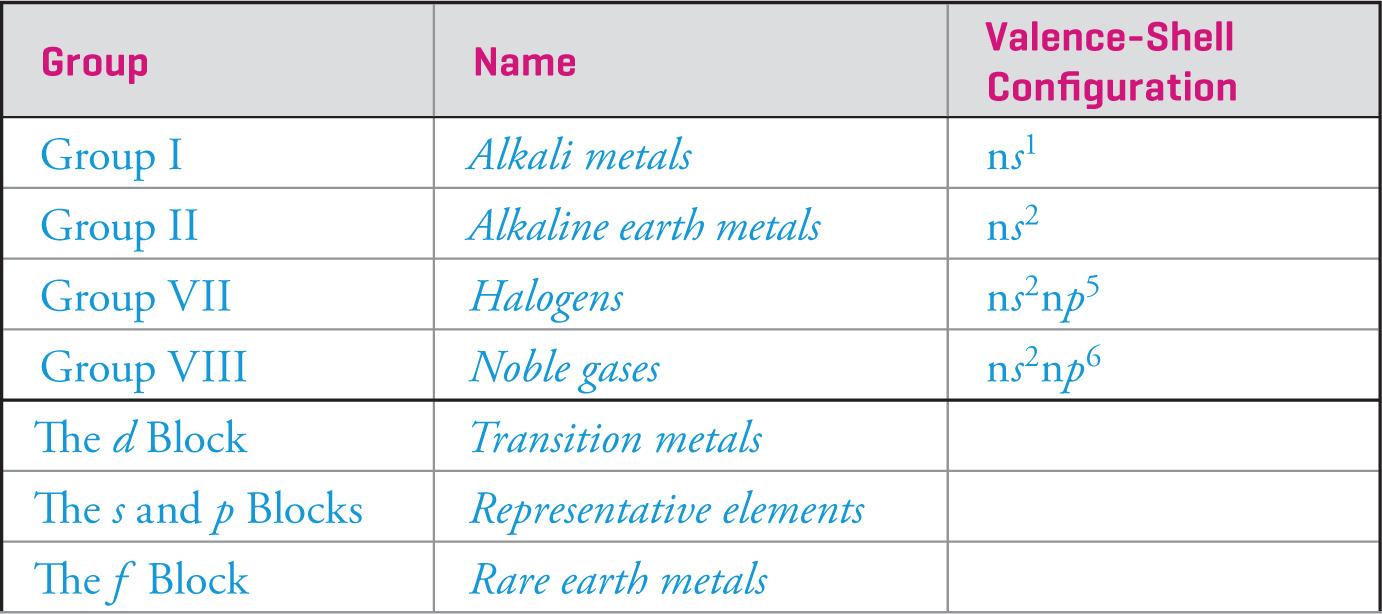
The valence-shell electron configuration determines the chemical reactivity of each group in the table. For example, in the noble gas family each element has eight electrons in its outermost shell (ns2np6). Such a closed-shell (fully-filled valence shell) configuration is called an octet and results in great stability (and therefore low reactivity) for an atom. For this reason, noble gases do not generally undergo chemical reactions, so most group VIII elements are inert. Helium is inert as well, but has a closed shell with a stable duet (1s2) of electrons.
Other elements experience similar increases in stability upon reaching this stable octet electron configuration, and most chemical reactions can be regarded as the quest for atoms to achieve such closed-shell stability. The alkali metals and alkaline earth metals, for instance, possess one (ns1) or two (ns2) electrons in their valence shells, respectively, and behave as reducing agents (i.e., lose valence electrons) in redox reactions in order to obtain a stable octet, generally as an M+ or M2+ cation.
Similarly, the halogens (ns2np5) require only a single electron to achieve a stable octet. To achieve this state in their elemental form, halogens naturally exist as diatomic molecules (e.g., F2) where one electron from each atom is shared in a covalent bond. When combined with other elements, the halogens behave as powerful oxidizing agents (that is, gain electrons); they can become stable either as X− anions or by sharing electrons with other nonmetals (more on bonding in Ch. 5).
Reactions between elements on opposite sides of the periodic table can be quite violent. This occurs due to the great degree of stability gained for both elements when the valence electrons are transferred from the metal to the nonmetal. The relative reactivities within these and all other groups can be further explained by the periodic trends detailed in the next section.
Example 4-27: Which of the following elements has a closed valence shell, but not an octet?
A) He
B) Ne
C) Br
D) Rn
Solution: Choice A, He, is the correct choice because He, along with H− and Li+, has a completed n = 1 shell with only 2 electrons, since the n = 1 shell can fit only 2 electrons.
Example 4-28: Which of the following could describe an ion with the same electron configuration as a noble gas?
A) An alkali metal that has gained an electron
B) A halogen that has lost an electron
C) A transition metal that has gained an electron
D) An alkaline earth metal that has lost two electrons
Solution: Choice A is wrong since it says “gained” rather than “lost.” Choice B is incorrect since it says “lost” rather than “gained.” Choice C is also incorrect, because no element in the d block could acquire a noble-gas configuration by gaining a single electron. The answer must be D. If an element in Group II loses two electrons, it can acquire a noble-gas electron configuration. (For example, Mg2+ has the same configuration as Ne, and Ca2+ has the same configuration as Ar.)
Example 4-29: Of the following, the element that possesses properties of both metals and nonmetals is:
A) Si
B) Al
C) Zn
D) Hg
Solution: Elements that possess qualities of both metals and nonmetals are called metalloids. These elements are shown below. Thus, choice A is the correct answer; the other choices are metals.
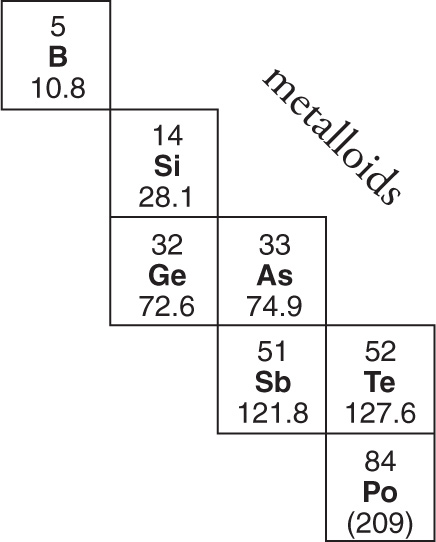
4.8 PERIODIC TRENDS
Shielding
Each filled shell between the nucleus and the valence electrons shields—or “protects”—the valence electrons from the full effect of the positively charged protons in the nucleus. This is called nuclear shielding or the shielding effect. As far as the valence electrons are concerned, the electrical pull by the protons in the nucleus is reduced by the negative charges of the electrons in the filled shells in between; the result is an effective reduction in the positive elementary charge, from Z to a smaller amount denoted by Zeff (for effective nuclear charge).
Example 4-30: The electrons in a solitary He atom are under the influence of two forces, one attractive and one repulsive. What are these forces?
A) Electrostatic attraction between the electrons and the nuclear protons, and electrostatic repulsion between the electrons and nuclear neutrons.
B) Electrostatic attraction between the electrons and the nuclear protons, and electrostatic repulsion between the electrons.
C) Gravitational attraction between the electrons and the nuclear protons, and frictional repulsion between the electrons.
D) Gravitational attraction between the electrons and the entire nucleus, and frictional repulsion between the electrons.
Solution: Compared to the magnitude of electrostatic forces in an atom, gravitational forces between the electrons and nucleons of an atom are negligible, so choices C and D are eliminated. Furthermore, neutrons have no charge and thus do not participate in electrostatic forces, so choice A is eliminated. Remember that opposite charges attract and like charges repel. The best choice is B.
Atomic and Ionic Radius
With progression across any period in the table, the number of protons increases, and hence their total pull on the outermost electrons increases, too. New shells are initiated only at the beginning of a period. So, as we go across a period, electrons are being added, but new shells are not; therefore, the valence electrons are more and more tightly bound to the atom because they feel a greater effective nuclear charge. Therefore, as we move from left to right across a period, atomic radius decreases.
However, with progression down a group, as new shells are added with each period, the valence electrons experience increased shielding. The valence electrons are less tightly bound since they feel a smaller effective nuclear charge. Therefore, as we go down a group, atomic radius increases due to the increased shielding.
If we form an ion, the radius will decrease as electrons are removed (because the ones that are left are drawn in more closely to the nucleus), and the radius will increase as electrons are added. So, in terms of radius, we have X+ < X < X−; that is, cation radius < neutral-atom radius < anion radius.
Ionization Energy
Because the atom’s positively charged nucleus is attracted to the electrons in the atom, it takes energy to remove an electron. The amount of energy necessary to remove the least tightly bound electron from an isolated atom is called the atom’s (first) ionization energy (often abbreviated IE or IE1). As we move from left to right across a period, or up a group, the ionization energy increases since the valence electrons are more tightly bound. The ionization energy of any atom with a noble-gas configuration will always be very large. (For example, the ionization energy of neon is 4 times greater than that of lithium.) The second ionization energy (IE2) of an atom, X, is the energy required to remove the least tightly bound electron from the cation X+. Note that IE2 will always be greater than IE1.
Electron Affinity
The energy associated with the addition of an electron to an isolated atom is known as the atom’s electron affinity (often abbreviated EA). If energy is released when the electron is added, the usual convention is to say that the electron affinity is negative; if energy is required in order to add the electron, the electron affinity is positive. The halogens have large negative electron affinity values, since the addition of an electron would give them the much desired octet configuration. So they readily accept an electron to become an anion; the increase in stability causes energy to be released. On the other hand, the noble gases and alkaline earth metals have positive electron affinities, because the added electron begins to fill a new level or sublevel and destabilizes the electron configuration. Therefore, anions of these atoms are unstable. Electron affinities typically become more negative as we move to the right across a row or up a group (noble gases excepted), but there are anomalies in this trend.
Electronegativity
Electronegativity is a measure of an atom’s ability to pull electrons to itself when it forms a covalent bond; the greater this tendency to attract electrons, the greater the atom’s electronegativity. Electronegativity generally behaves as does ionization energy; that is, as we move from left to right across a period, electronegativity increases. As we go down a group, electronegativity decreases. You should know the order of electronegativity for the nine most electronegative elements:
F > O > N > Cl > Br > I > S > C ≈ H
Acidity
Acidity is a measure of how well a compound donates protons, accepts electrons, or lowers pH in a chemical system. A binary acid has the structure HX, and can dissociate in water in the following manner: HX → H+ + X−. Stronger acids have resulting X− anions that are likely to separate from H+ because they are stable once they do. With respect to the horizontal periodic trend for acidity, the more electronegative the element is (X−) the more stable the anion will be. Therefore acidity increases from left to right across a period. However, the vertical trend for acidity depends on the size of the anion. The larger the anion, the more the negative charge can be delocalized and stabilized. Therefore, acidity increases down a group or family in the periodic table.

Example 4-31: Compared to calcium, beryllium is expected to have:
A) greater electronegativity and ionization energy.
B) smaller electronegativity and ionization energy.
C) greater electronegativity and smaller ionization energy.
D) smaller electronegativity and larger ionization energy
Solution: Beryllium and calcium are in the same group, but beryllium is higher in the column. We therefore expect beryllium to have greater ionization energy and a greater electronegativity than calcium (choice A), since both of these periodic trends tend to increase as we go up within a group.
Example 4-32: Which of the following will have a greater value for phosphorus than for magnesium?
I. Atomic radius
II. Ionization energy
III. Electronegativity
A) I only
B) I and II only
C) II and III only
D) I, II, and III
Solution: Magnesium and phosphorus are in the same period (row), but phosphorus is farther to the right. We therefore expect phosphorus to have a smaller atomic radius, making Roman numeral I false. This allows us to eliminate choices A, B, and D, leaving C as the correct answer. This is also consistent as we expect phosphorus to have a greater ionization energy and a greater electronegativity than magnesium, since both of these periodic trends tend to increase as we move to the right across a row. However, we expect the atomic radius of phosphorus to be smaller than that of magnesium, since atomic radii tend to decrease as we move to the right across a row. Therefore, the answer is C.
Example: 4-33 Of the following, which has the most negative electron affinity?
A) Barium
B) Bromine
C) Phosphorus
D) Chlorine
Solution: Barium is in Group II and therefore has a large positive electron affinity, so we can eliminate choice A immediately. Because electron affinity values tend to become more negative as we go to the right across a row or up within a column, we’d expect chlorine to have a more negative electron affinity than phosphorus or bromine. Therefore, choice D is the answer.
Example 4-34: Of the following, which has the smallest atomic radius?
A) Sodium
B) Oxygen
C) Calcium
D) Silicon
Solution: The atoms with the smallest atomic radius are those in the upper right portion of the periodic table, since atomic radius tends to increase as we move to the left or down a column. We can therefore eliminate choices A and C; these elements are in Groups I and II, respectively, at the far left end of the table. To decide between the remaining choices, we notice that oxygen is farther to the right and in a higher row than silicon, so we’d expect an oxygen atom to have a smaller radius than a silicon atom. Choice B is the best answer.
Example 4-35: Of the following, which is the strongest acid?
A) H2O
B) H2S
C) HCl
D) HBr
Solution: For binary acids, we expect acidity to increase with increasing stability of the conjugate base. When comparing anions in a period, those that are more electronegative are more stable. Since chloride is more electronegative than sulfide, choice B can be eliminated. In addition, when comparing anions in a family, those that are larger are more stable, so choices A and C can also be eliminated, making choice D the best answer.
Chapter 4 Summary
• The nucleus contains protons and neutrons. Their sum corresponds to the mass number (A).
• The number of protons corresponds to the atomic number (Z).
• An overabundance of either protons or neutrons can result in unstable nuclei, which decay via the emission of various particles.
• For nuclear decay reactions, the sum of all mass and atomic numbers in the products must equal the same sum of these numbers in the reactants.
• The rate of nuclear decay is governed by a species’ half-life.
• Electrons exist in discrete energy levels within an atom. Emission spectra are obtained from energy emitted as excited electrons fall from one level to another.
• The periodic table is organized into blocks based on the architecture of electron orbitals. Therefore, valence electron configurations can be determined based on an element’s location in the table.
• In their ground state, electrons occupy the lowest energy orbitals available, and occupy subshell orbitals singly before pairing.
• Atoms and ions are most stable when they have an octet of electrons in their outer shell.
• The d subshell is always backfilled: for an atom in the d block of period n, the d subshell will have a principle quantum number of n − 1.
• A half filled (d5) or filled (d10) d subshell is exceptionally stable.
• Transition metals ionize from their valence s subshell before their d subshell.
• Atomic radius increases to the left and down the periodic table; for charged species, cations < neutral atom < anions for a given element; for isoelectronic ions, the species with more protons will have the smaller radius.
• Ionization energy, electron affinity, and electronegativity increase up and to the right on the periodic table, while acidity increases to the right and down the periodic table.
• The relative electronegativities of common atoms in decreasing order are F O N Cl Br I S C ≈ H.
CHAPTER 4 FREESTANDING PRACTICE QUESTIONS
1. When an atom of plutonium-239 is bombarded with an alpha particle, this element along with one free neutron is created:
A) Californium-240
B) Californium-241
C) Curium-242
D) Curium-243
2. Which of the following represents the correct ground state electronic configuration for ferrous ion, Fe2+?
A) [Ar] 4s23d6
B) [Ar] 4s23d4
C) [Ar] 3d6
D) [Ar] 4s23d2
3. Which atom has three unpaired electrons in its valence energy level?
A) Li
B) Be
C) C
D) N
4. Which of the following elements would be most strongly attracted to a magnetic field?
A) Mg
B) Ca
C) Cr
D) Zn
5. Which of the following colors would appear as a bright band in an emission spectrum of a yellow sodium vapor lamp?
A) Yellow, indicating a lesser wavelength than ultraviolet light
B) Yellow, indicating a greater wavelength than ultraviolet light
C) Blue, indicating a lesser wavelength than ultraviolet light
D) Blue, indicating a greater wavelength than ultraviolet light
6. Which of the following atoms/ions has electrons in the subshell of highest energy?
A) Cl−
B) Ca2+
C) Cr+
D) As
7. Of the following metallic elements, which has the lowest second ionization energy?
A) Na
B) K
C) Mg
D) Ca
8. Which of the following has the smallest atomic or ionic radius?
A) Cl−
B) Ar
C) K+
D) Ca2+
9. Metallic character results from an element’s ability to lose electrons. On the periodic table it is expected that metallic character increases:
A) from left to right, because the decrease in electronegativity would make it easier to lose electrons.
B) from left to right, because the decrease in atomic radius would result in more stable positive ions.
C) from right to left, because the decrease in ionization energy would make it easier to lose electrons.
D) from right to left, because the decrease in electron affinity would result in more stable positive ions.
CHAPTER 4 PRACTICE PASSAGE
Atoms are the building blocks of molecules and they consist of electrons surrounding a nucleus composed of neutrons and protons. The identity of an atom depends on how many protons it contains. The stability and/or reactivity of an atom often depends on how many neutrons and electrons it contains.
Nucleons themselves are composed of elementary particles known as quarks. Quarks are held together by the strong force to generate composite particles referred to as hadrons. This strong force, sometimes called the nuclear force, is also responsible for holding protons and neutrons together in the nucleus and overcomes other forces that may be present. Baryons, which are hadrons containing three quarks, form a charged nucleon when two up quarks and one down quark combine and uncharged nucleons when one up quark combines with two down quarks. Mesons, which are the other family of hadrons, are unstable particles composed of one quark and one antiquark.
1. An atom contains 29 hadrons comprised of two down quarks and one up quark and 28 hadrons comprised of two up quarks and one down quark. Which one of the following is the identity of the atom?
A) Iron-57
B) Nickel-57
C) Copper-57
D) Nickel-58
2. An excited electron drops down to its ground state and in the process a photon of light with a wavelength of 525 nm is emitted. Which of the following types of electromagnetic radiation could the photon be?
A) Infrared
B) Visible light
C) X-ray
D) Cannot be determined from the information given
3. A scientist in a laboratory observes a nucleon composed of three quarks with charges of +2/3 e, −1/3 e, and −1/3 e respectively. Which of the following best describes the nucleon?
A) The particle is a neutron and it contains two down quarks.
B) The particle is a proton and it contains two down quarks.
C) The particle is a neutron and it contains one down quark.
D) The particle is a proton and it contains one down quark.
4. The first, second, and third ionization energies for strontium are 549.5 kJ/mol, 1064.2 kJ/mol, and 4138 kJ/mol respectively. Why is the third ionization energy so much higher than the first two ionization energies?
A) The third electron is being removed from a completely full subshell.
B) The third electron has a larger mass than the first two electrons being removed.
C) The third electron being removed is less attracted to the nucleus than the first two electrons.
D) The third electron is at a higher energy level than the first two electrons.
5. Particles with opposite charges attract one another and particles with like charges repel. How can protons, which are positively charged, coexist in the nucleus?
A) The neutrons in the nucleus prevent the protons from touching one another.
B) The nuclear force is stronger than the repulsive forces between protons.
C) Hadrons do not experience forces with one another.
D) The surrounding cloud of electrons generates an opposing force.
SOLUTIONS TO CHAPTER 4 FREESTANDING PRACTICE QUESTIONS
1. C The process described is transmutation, and the new nucleus can be determined by writing a balanced nuclear equation. The preliminary equation to balance is this:
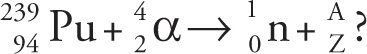
where the question mark stands for the new element formed. Balancing mass number gives 239 + 4 = 1 + A, where A = 242; balancing the atomic number gives 94 + 2 = 0 + Z, where Z = 96. Therefore, element number 96 is curium (eliminate choices A and B), and the appropriate isotope has a mass number of 242.
2. C When answering electron configuration questions, the first step is to eliminate all answer choices that do not display the correct number of electrons. In this case, ferrous ion possesses six electrons beyond those represented by [Ar] (eight for elemental iron minus two to generate the +2 cation). Thus, choices A and D can be eliminated. To choose between B and C, recall that when transition metals ionize, it is the outermost and therefore least tightly held electrons that are removed first. In this case, the 4s electrons are further from the nucleus and are less tightly held (n = 4 represents a greater radial distance from the nucleus than n = 3). Thus they are the first to be removed.
3. D Since Li has only one valence electron and Be has only two, neither choices A nor B can be correct. To choose between C and D, note that the valence configuration of C is 2s22p2. Thus the 2s electrons are paired leaving only two unpaired p electrons. Nitrogen has a valence configuration of 2s22p3, and by Hund’s rule, the three p electrons will singly occupy the px, py, and pz levels rather than pairing up to avoid electron repulsion.
4. C Diamagnetic atoms are repelled by magnetic fields and paramagnetic atoms are attracted to magnetic fields. Paramagnetic atoms have unpaired electrons in their valence orbitals. Mg and Ca are in the same group and have the same valence configuration, so both cannot be the right answer. Zn is at the end of the d block and has a valence shell with all of its electrons paired. Cr only has four electrons in its 3d subshell, resulting in four unpaired electron orbitals. Cr is the only choice that is paramagnetic and would be attracted to a magnetic field.
5. B All visible light has a greater wavelength than ultraviolet, eliminating choices A and C. The sodium lamp glows yellow and would therefore emit a yellow band on a dark background. If the question asked where dark bands would have been in an absorption spectrum, several lines would be seen in regions other than yellow since those colors are absorbed.
6. D Electron energy level is determined by the first two quantum numbers. Given Cl− = [Kr], Ca2+ = [Ar], Cr+ = [Ar] 3d 5, and As = [Ar] 4s23d104p3, arsenic contains electrons in the highest energy subshell, 4p.
7. D After their first ionizations, Na+ and K+ both have octet electron configurations, so a second ionization to remove another electron would require a very high amount of energy. This eliminates choices A and B. Ionization energy decreases down a group, due to increased nuclear shielding, so it is easier to remove electrons from Ca than Mg, making choice D the answer.
8. D All four answer choices have the same number of electrons and the same electron configuration. Ca2+ has the most protons pulling on these electrons, so it will be the smallest.
9. C Choice A is eliminated because electronegativity increases from left to right on the periodic table. Choice B is eliminated because the stability of positive ions increases as you go up and to the left on the periodic table. Finally, choice D is eliminated since electron affinity is the energy released upon gaining an electron and does not relate to the stability of a positive ion. Choice C is the correct answer because ionization energy, or the energy required to remove an electron, decreases from right to left due to a decrease in effective nuclear charge.
SOLUTIONS TO CHAPTER 4 PRACTICE PASSAGE
1. B A hadron with two up quarks and one down quark is a charged nucleon or proton. Because the atom has 28 of these protons, it must be nickel, eliminating choices A and C. Furthermore, a hadron with two down quarks and one up quark is a neutron. The atom has 29 of these, so the atomic mass must be 28 + 29 = 57, eliminating choice D.
2. B Visible light has a wavelength range of 450-700 nm. The photon falls in this range, so visible light must have been emitted.
3. A The charges of the three quarks add up to 0, so the nucleon must be an uncharged neutron, eliminating choices B and D. The passage states that uncharged nucleons have two down quarks, eliminating choice C.
4. A All electrons in an atom have the same mass, eliminating choice B. To remove three electrons from Sr, the first two electrons are removed from the 5s subshell and the third electron is removed from the 4p subshell, eliminating choice D. Since the third electron is at a lower energy level, it experiences less shielding and would be more attracted to the nucleus than the first two electrons removed, eliminating choice C. The third electron comes from the completely full 4p subshell and is much more difficult to remove, making choice A the best answer.
5. B The passage describes the nuclear force as being responsible for holding the nucleons in the nucleus together and that it “overcomes other forces that may be present.” Therefore, choice B is the best answer because it explains how protons are held together in close proximity despite having the same charge. The passage provides us with no indication that neutrons are preventing protons from touching one another, and particles need not physically touch in order to experience repulsion, eliminating choice A. The passage does describe how hadrons are held together by the strong force, eliminating choice C. The electron cloud surrounding the nucleus experiences an attractive force between the positive protons and the negative electrons. It is unlikely that this could generate an opposing force to prevent the repulsion of like-charged nucleons, eliminating choice D.