MCAT General Chemistry Review - Steven A. Leduc 2015
Bonding and Intermolecular Forces
The physical properties of a substance are determined at the molecular level, and the chemistry of molecules is dominated by the reactivity of covalent and ionic bonds. An understanding of the fundamentals of bonding can provide the intuitive grasp necessary to answer a wide range of questions in both general and organic chemistry. This chapter will briefly outline some basic principles, that when mastered, will help lay a strong foundation for many chemistry concepts you will encounter on the MCAT.
5.1 LEWIS DOT STRUCTURES
Each dot in the picture below represents one of fluorine’s valence electrons. Fluorine is a halogen, with a general valence-shell configuration of ns2np5, so there are 2 + 5 = 7 electrons in its valence shell. We simply place the dots around the symbol for the element, one on each side, and, if there are more than 4 valence electrons, we just start pairing them up. So, for fluorine, we’d have:
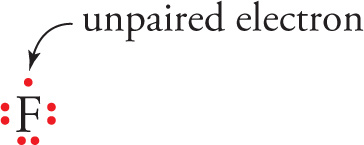
This is known as a Lewis dot symbol. Here are some others:

(Note: Electrons in d subshells are not considered valence electrons for transition metals since valence electrons are in the highest n level.)
Example 5-1: Consider this Lewis dot symbol:

Among the following, X could represent:
A) carbon.
B) nitrogen.
C) sulfur.
D) argon.
Solution: Since there are four dots in the Lewis symbol, X will be an element in Group 4 of the periodic table. Of the choices given, only carbon (choice A) is in Group 4.
Lewis dot structures are one type of model we use to represent what compounds look like at the molecular level. Since it’s the valence electrons that are responsible for creating bonds in molecules, a Lewis dot structure that accounts for the number and location of all valence electrons gives us a sense of how molecules are held together and helps us understand their reactivity.
To create a Lewis dot structure for a molecule, we begin to pair up electrons from two separate atoms since two electrons are required to form a single bond. By sharing a pair of electrons to form a bond, each atom may acquire an octet configuration, thereby stabilizing both atoms. For example, each of the fluorine atoms below can donate its unpaired valence electron to form a bond and give the molecule F2. The shared electrons are attracted by the nuclei of both atoms in the bond, which hold the atoms together.
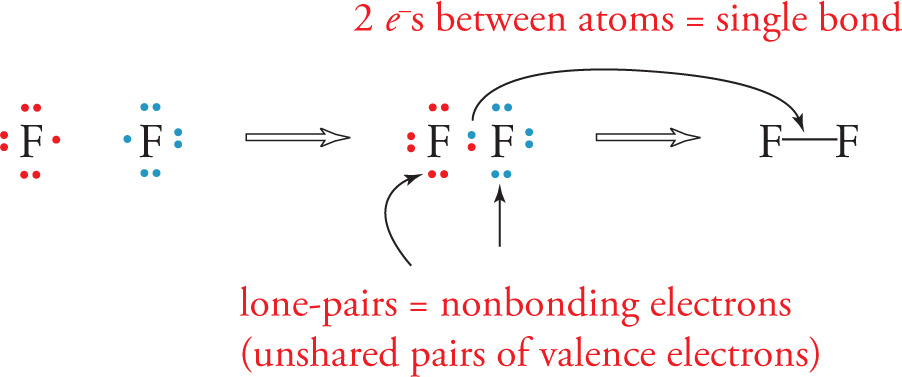
Note that in addition to the single bond (a bond formed from two electrons) between the fluorine atoms, each fluorine atom has three pairs of electrons that are not part of a bond. They help satisfy the octets of the F atoms and are known as “lone pairs” of electrons. We’ll see in a bit how these lone pairs are important for determining physical properties of compounds, so don’t forget to write these out too.
We can also use Lewis dot structures to show atoms that form multiple bonds—double bonds use four electrons while triple bonds require six. Here are a couple of examples:
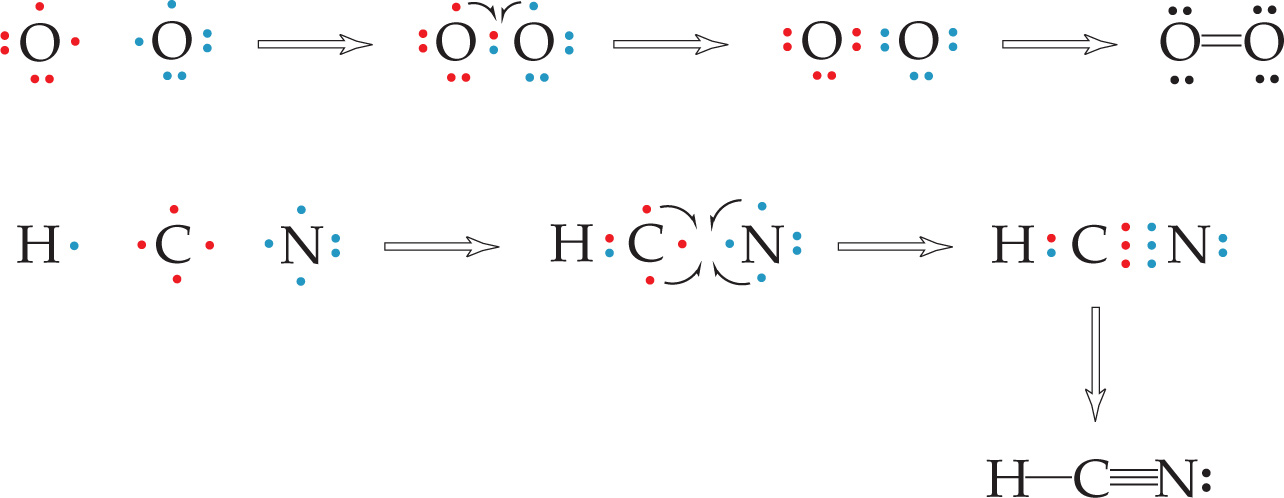
Formal Charge
The last Lewis dot structure shown above for the molecule consisting of 1 atom each of hydrogen, carbon, and nitrogen was drawn with C as the central atom. However, it could have been drawn with N as the central atom, and we could have still achieved closed-shell configurations for all the atoms:

The problem is this doesn’t give the correct structure for this molecule. The nitrogen atom is not actually bonded to the hydrogen. A helpful way to evaluate a proposed Lewis structure is to calculate the formal charge of each atom in the structure. These formal charges won’t give the actual charges on the atoms; they’ll simply tell us if the atoms are sharing their valence electrons in the “best” way possible, which will happen when the formal charges are all zero (or at least as small as possible). The formula for calculating the formal charge of an atom in a covalent compound is:
Formal charge (FC) = V − ½B − L
where V is the number of valence electrons, B is the number of bonding electrons, and L is the number of lone-paired (non-bonding) electrons. We’ll show the calculations of the formal charges for each atom in both Lewis structures:

The best Lewis structures have an octet of electrons and a formal charge of zero on all the atoms. (Sometimes, this simply isn’t possible, and then the best structure is the one that minimizes the magnitudes of the formal charges.) The fact that the HCN structure has formal charges of zero for all the atoms, but the HNC structure does not, tells us right away that the HCN structure is the better one. For dot structures that must contain formal charges on one or more atoms, the best structures have negative formal charges on the more electronegative element.
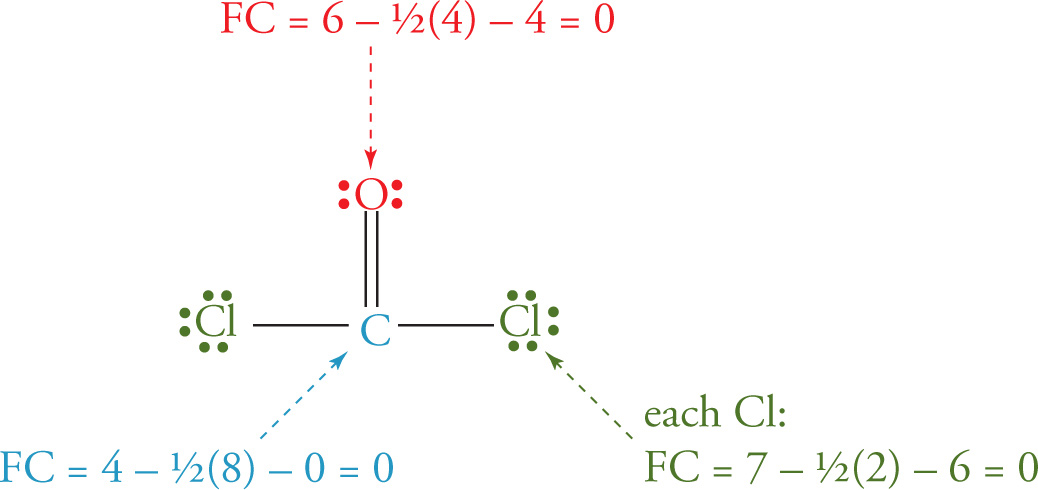
Example 5-2: What’s the formal charge on each atom in phosgene, COCl2?
Solution:
Example 5-3: Which of the following is the best Lewis structure for CH2O?
A. 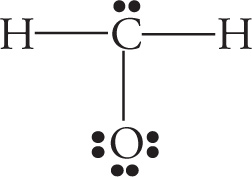
B. 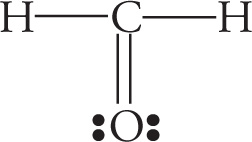
C. 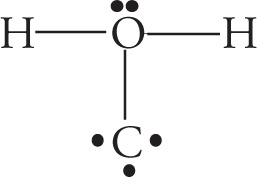
D. 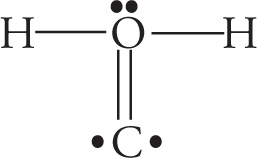
Solution: When faced with a question like this on the MCAT (and they’re rather common), the first thing you should do is simply count the electrons. The correct structure for the molecule CH2O must account for 4 + 2(1) + 6 = 12 valence electrons. The structure in choice A has 14 and the structure in choice C has 11. Answer choices B and D both have 12 valence electrons. However, in choice D, oxygen is surrounded by 10 total electrons. This is not possible because oxygen, like all elements in the second row of the periodic table, cannot violate the octet rule and exceed 8 valence electrons. Choice B, then, with 12 valence electrons and the least electronegative atom as the central atom, is the best choice.
Resonance
Recall that Lewis dot structures are a model that we use to help us understand where the valence electrons are in a molecule. All models, being simplifications of reality, have limitations, and Lewis dot structures are no exception. Sometimes it is impossible for one structure to accurately represent the reality of a molecule’s electron distribution. To account for this complexity, we need two or more structures, called resonance structures, to accurately depict the bonding in a molecule. These structures are often needed when there are double or triple bonds in molecules along with one or more lone pairs of electrons.
Let’s draw the Lewis structure for sulfur dioxide.
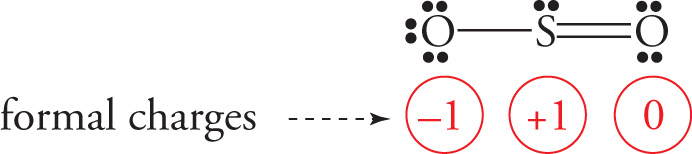
We could also draw the structure like this:
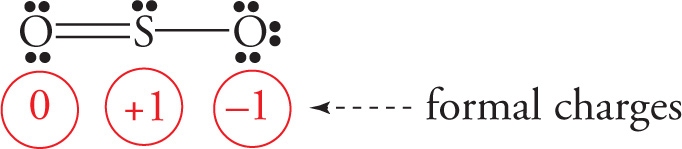
In either case, there’s one S—O single bond and one S=O double bond. This would imply that the double-bonded O would be closer to the S atom than the single-bonded O (see Section 5.2, Bond Length and Bond Dissociation Energy). Experiment, however, reveals that the bond lengths are the same. Therefore, to describe this molecule, we say that it’s an “average” (or, technically, a resonance hybrid) of the equivalent Lewis structures shown:
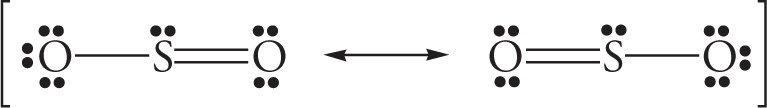
We can also symbolize the resonance hybrid with a single picture, like this:
![]()
The dotted lines in the structure above indicate some double bond character for both S—O bonds, more of a “bond and a half.” A molecule may be a resonance hybrid of more than two equivalent Lewis structures; for example, consider the carbonate ion, CO32—:
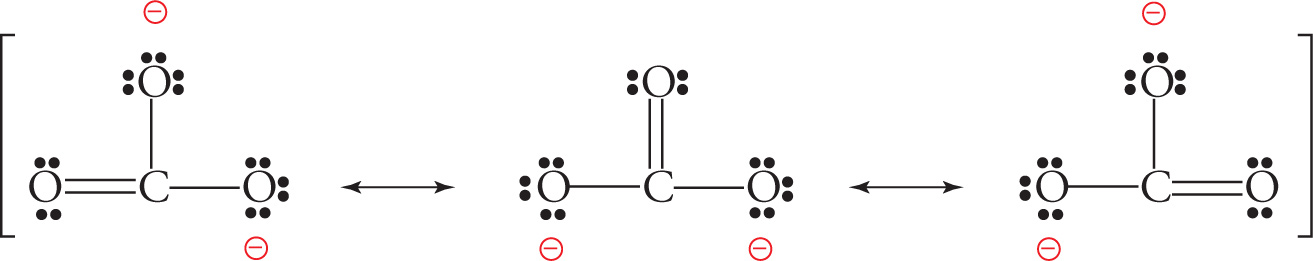
or, more simply,
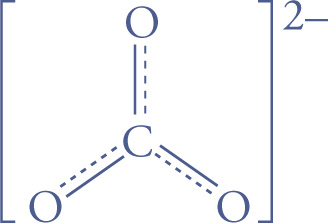
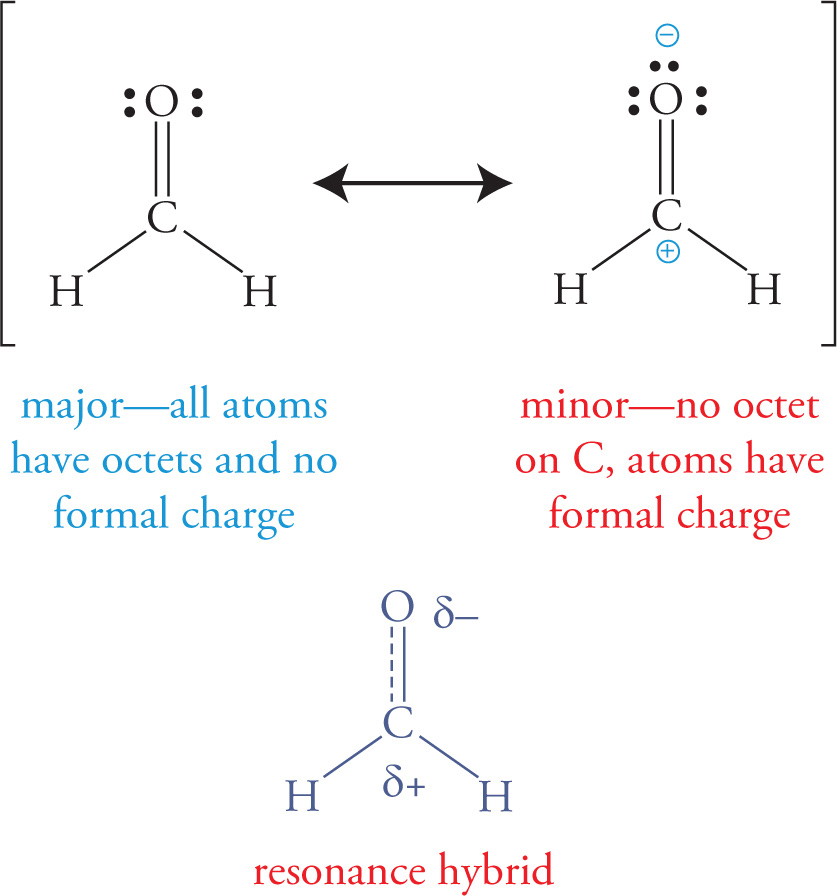
In addition, a molecule may have two or more non-equivalent resonance structures, and the resonance hybrid is then a weighted average of them, as shown with formaldehyde below:
Example 5-4: Resonance structures are two or more structures where:
A) only atoms may move around.
B) only bonding electrons may move around.
C) only nonbonding electrons may move around.
D) only nonbonding electrons, and double and triple bonds may move around.
Solution: Choice D is the correct answer. (This definition is particularly important in organic chemistry.)
5.2 BOND LENGTH AND BOND DISSOCIATION ENERGY
While the term bond length makes good intuitive sense (the distance between two nuclei that are bonded to one another), bond dissociation energy (BDE) is not quite as intuitive. Bond dissociation energy is the energy required to break a bond homolytically. In homolytic bond cleavage, one electron of the bond being broken goes to each fragment of the molecule. In this process two radicals form. This is not the same thing as heterolytic bond cleavage (also known as dissociation). In heterolytic bond cleavage, both electrons of the electron pair that make up the bond end up on the same atom; this forms both a cation and an anion.


These two processes are very different and hence have very different energies associated with them. Here, we will only consider homolytic bond dissociation energies.
When one examines the relationship between bond length and bond dissociation energy for a series of similar bonds, an important trend emerges: For similar bonds, the higher the bond order, the shorter and stronger the bond. Bond order is defined as the number of bonds between adjacent atoms, so a single bond has a bond order of 1 while a triple bond has a bond order of 3. The following table, which lists the bond dissociation energies (BDE, in kcal/mol) and the bond lengths (r, in angstroms, where 1 Å = 10−10 m) for carbon-carbon and carbon-oxygen bonds, illustrates this trend:
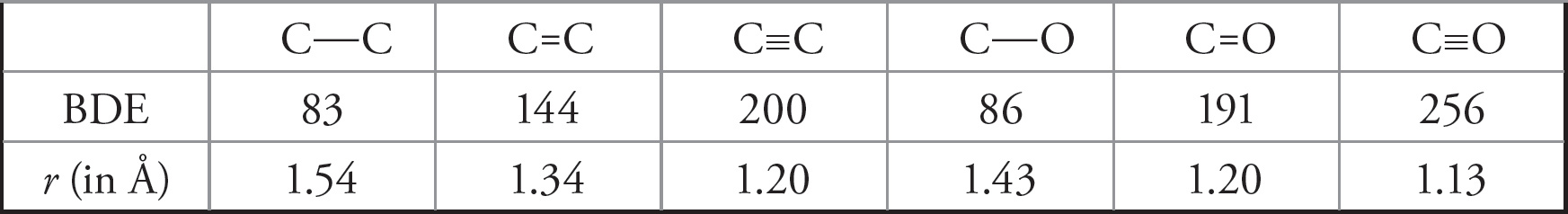
An important caveat arises because of the varying atomic radii: bond length/BDE comparisons should only be made for similar bonds. Thus, carbon-carbon bonds should be compared only to other carbon-carbon bonds; carbon-oxygen bonds should be compared only to other carbon-oxygen bonds, and so on.
Recall the shapes of atomic orbitals: s orbitals are spherical about the atomic nucleus, while p orbitals are elongated “dumbbell”-shaped about the atomic nucleus
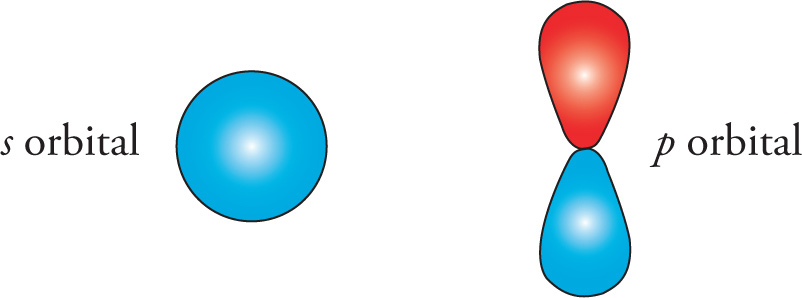
When comparing the same type of bonds, the greater the s character in the hybrid orbitals, the shorter the bond (because s-orbitals are closer to the nucleus than p-orbitals). A greater percentage of p character in the hybrid orbital also leads to a more directional hybrid orbital that is farther from the nucleus and thus a longer bond (see section 5.5 for all the details on hybridization). In addition, when comparing the same types of bonds, the longer the bond, the weaker it is; the shorter the bond, the stronger it is. In the following diagram, compare all the C—C bonds and all the C—H bonds:
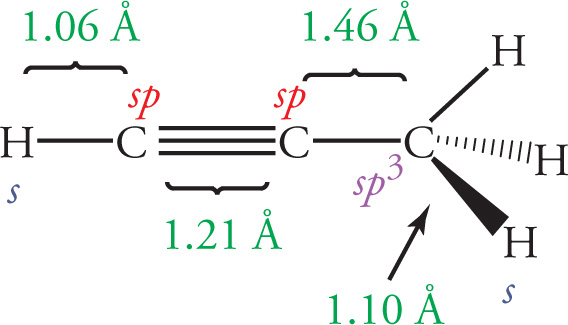
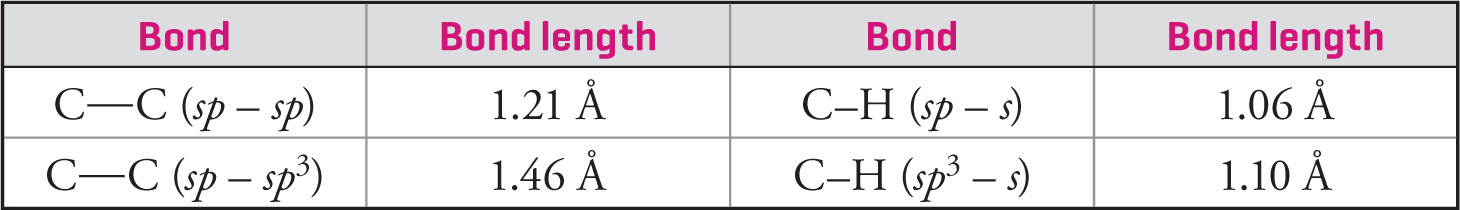
5.3 TYPES OF BONDS
Covalent Bonds
A covalent bond is formed between atoms when each contributes one or more of its unpaired valence electrons. The electrons are shared by both atoms to help complete both octets. There are minor variations in how the electrons are shared, however, so there are several classes of covalent bonds.
Polarity of Covalent Bonds
Recall that electronegativity refers to an atom’s ability to attract another atom’s valence electrons when it forms a bond. Electronegativity, in other words, is a measure of how much an atom will “hog” the electrons that it’s sharing with another atom.
Consider the Lewis dot structures of hydrogen fluoride and fluorine:
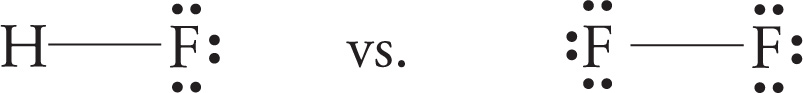
Fluorine is more electronegative than hydrogen (remember the order of electronegativity?), so the electron density will be greater near the fluorine than near the hydrogen in HF. That means that the H—F molecule is partially negative (denoted by δ−) on the fluorine side and partially positive (denoted by δ+) on the hydrogen side. We refer to this as polarity and say that the molecule has a dipole moment. A bond is polar if the electron density between the two nuclei is uneven. This occurs if there is a difference in electronegativity of the bonding atoms, and the greater the difference, the more uneven the electron density and the greater the dipole moment.
A bond is nonpolar if the electron density between the two nuclei is even. This occurs when there is little to no difference in electronegativity between the bonded atoms, generally when two atoms of the same element are bonded to each other, as we see in F2.
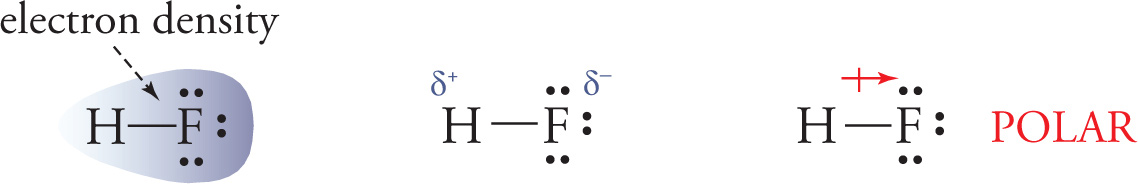
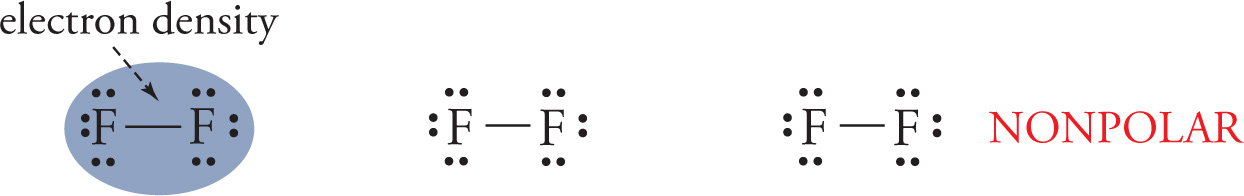
Coordinate Covalent Bonds
Sometimes, one atom will donate both of the shared electrons in a bond. That is called a coordinate covalent bond. For example, the nitrogen atom in NH3 donates both electrons in its lone pair to form a bond to the boron atom in the molecule BF3 to give the coordinate covalent compound F3BNH3:
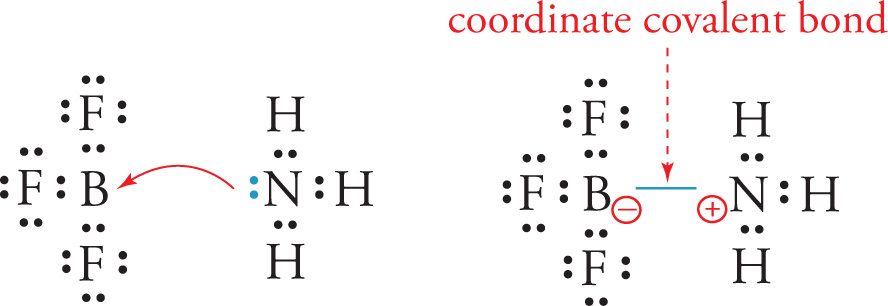
Since the NH3 molecule donates a pair of electrons, it is known as a Lewis base. A Lewis Base can act as a ligand, or a nucleophile (nucleus loving), and so all three terms are synonymous. Since the BF3 molecule accepts a pair of electrons, it’s known as a Lewis acid or electrophile (electron loving). When a coordinate covalent bond breaks, the electrons that come from the ligand will leave with that ligand.
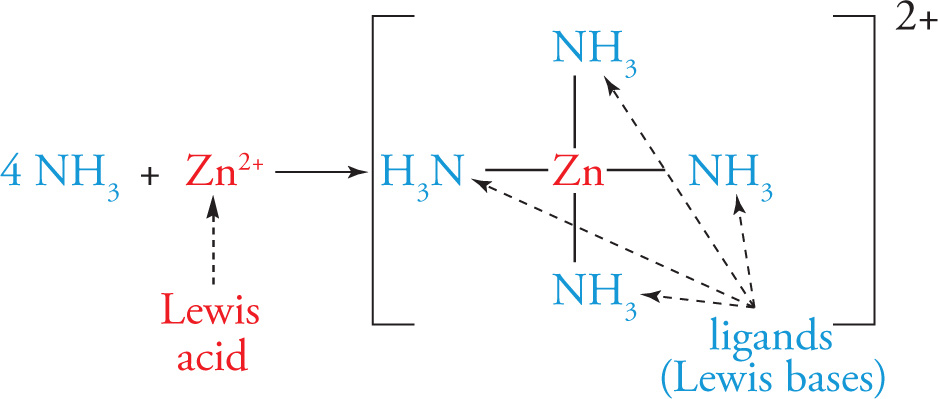
Example 5-5: Identify the Lewis acid and the Lewis base in the following reaction, which forms a coordination complex:
4 NH3 + Zn2+ → Zn(NH3)42+
Solution: Each of the NH3 molecules donates its lone pair to the zinc atom, thus forming four coordinate covalent bonds. Since the zinc ion accepts these electron pairs, it’s the Lewis acid; since each ammonia molecule donates an electron pair, they are Lewis bases (or ligands):
Example 5-6: Which one of the following anions cannot behave as a Lewis base/ligand?
A) F−
B) OH−
C) NO3−
D) BH4−
Solution: A Lewis base/ligand is a molecule or ion that donates a pair of nonbonding electrons. So, in order to even be a candidate Lewis base/ligand, a molecule must have a pair of nonbonding electrons in the first place. The ion in choice D does not have any nonbonding electrons.
Example 5-7: Carbon atoms with nonbonding electrons are excellent Lewis bases/ligands. Therefore, which of the following molecules is not a potential Lewis base/ligand?
A) CO2
B) CO
C) CN−
D) CH3−
Solution: The Lewis structures for the given molecules/ions are as follows:

Therefore, choice A (carbon dioxide) is not a good ligand and is the correct answer here.
Ionic Bonds
While sharing valence electrons is one way atoms can achieve the stable octet configuration, the octet may also be obtained by gaining or losing electrons. For example, a sodium atom will give its valence electron to an atom of chlorine. This results in a sodium cation (Na+) and a chloride anion (Cl−), which form sodium chloride. They’re held together by the electrostatic attraction between a cation and anion; this is an ionic bond.
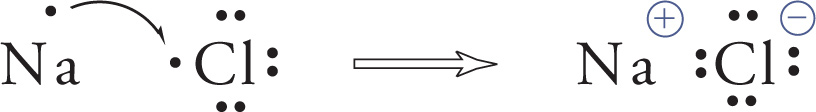
For an ionic bond to form between a metal and a non-metal, there has to be a big difference in electronegativity between the two elements. Generally speaking, the strength of the bond is proportional to the charges on the ions, and it decreases as the ions get farther apart, or as the ionic radii increase. We can use this to estimate the relative strength of ionic systems. For example, consider MgS and NaCl. For MgS, the magnesium ion has a +2 charge and sulfide ion has a —2 charge, while for NaCl, the charges are +1 for sodium and —1 for chloride. Therefore, the MgS “bond” is expected to be about four times stronger than the NaCl “bond,” assuming the sizes of the ions are very nearly the same.
Example 5-8: Which of the following is most likely an ionic compound?
A) NO
B) HI
C) ClF
D) KBr
Solution: A diatomic compound is ionic if the electronegativities of the atoms are very different. Of the atoms listed in the choices, those in choice D have the greatest electronegativity difference (K is an alkali metal, and Br is a halogen); K will give up its lone valence electron to Br, forming an ionic bond.
5.4 VSEPR THEORY
The shapes of simple molecules are predicted by valence shell electron-pair repulsion (VSEPR) theory. There’s one rule: Since electrons repel one another, electron pairs, whether bonding or nonbonding, attempt to move as far apart as possible.
For example, the bonding electrons in beryllium hydride, BeH2, repel one another and attempt to move as far apart as possible. In this molecule, two pairs of electrons point in opposite directions:
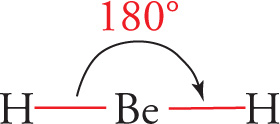
The angle between the bonds is 180°. A molecule with this shape is said to be linear.
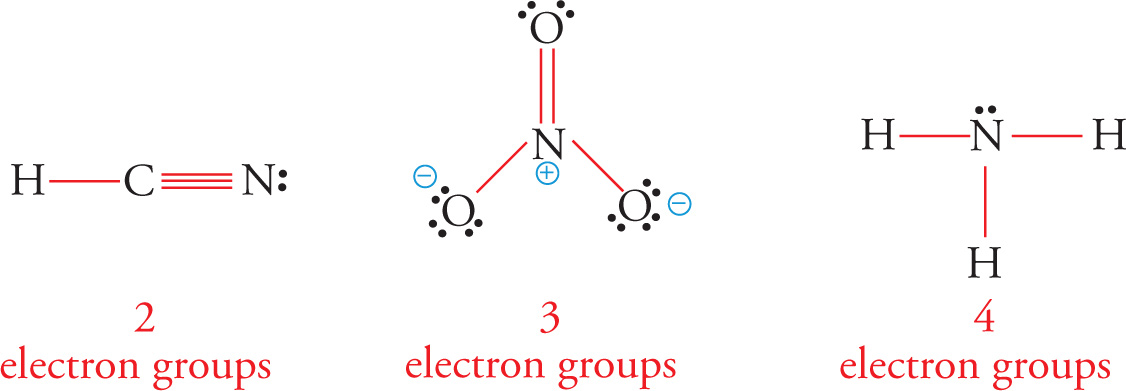
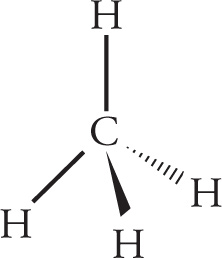
As the BeH2 example shows, the total number of electron groups on the central atom of a molecule determines its bond angles and orbital geometry. Electron groups are defined as any type of bond (single, double, triple) and lone pairs of electrons. Double and triple bonds count only as one electron group, even though they involve two and three pairs of electrons, respectively. To illustrate, the number of electron groups and orbital geometries of the central atom are shown for some example molecules:
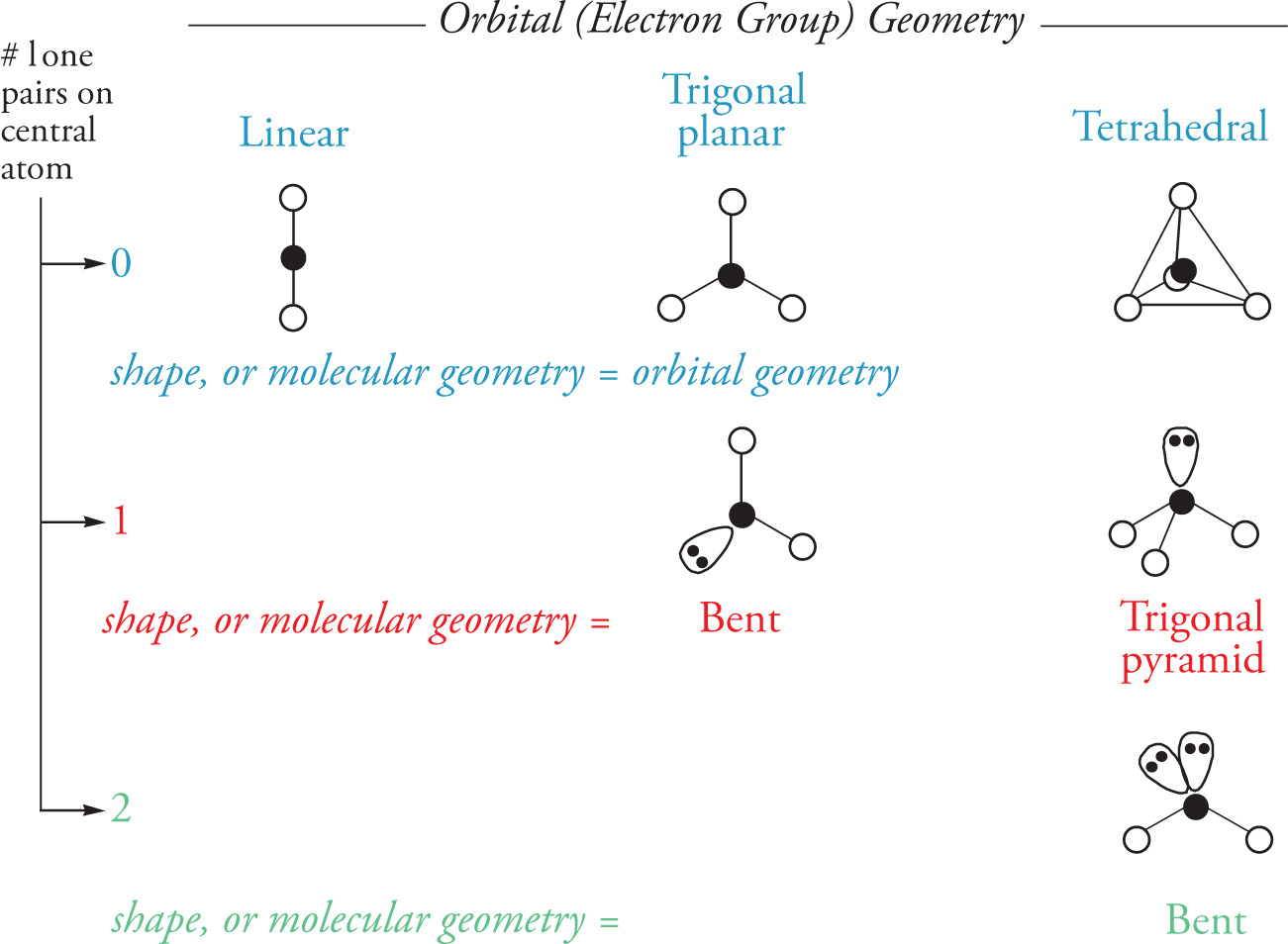
The shape of a molecule (also referred to as the molecular geometry) is also a function of the location of the nuclei of its constituent atoms. Therefore, when lone electron pairs are present on the central atom of a molecule, as in NH3 above, the shape is not the same as the orbital geometry. The table below shows how the presence of lone pairs determines the shape of a molecule:
Example 5-9: Determine the orbital geometry and predict the shape of each of the following molecules or ions:
a) H2O
b) SO2
c) NH4+
d) PCl3
e) CO32—
Solution:
a) 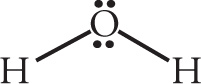
orbital geometry: tetrahedral shape: bent
b) 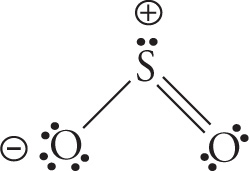
orbital geometry: trigonal planar shape: bent
c) 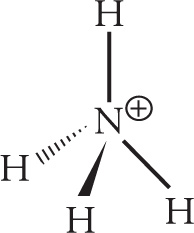
orbital geometry: tetrahedral shape: tetrahedral
d) 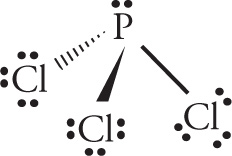
orbital geometry: tetrahedral shape: trigonal pyramid
e) 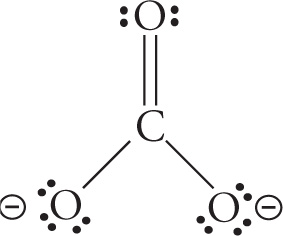
orbital geometry: trigonal planar shape: trigonal planar
5.5 HYBRIDIZATION
In order to rationalize observed chemical and structural trends, chemists developed the concept of orbital hybridization. In this model, one imagines a mathematical combination of atomic orbitals centered on the same atom to produce a set of composite, hybrid orbitals. For example, consider an s and a p orbital on an atom.
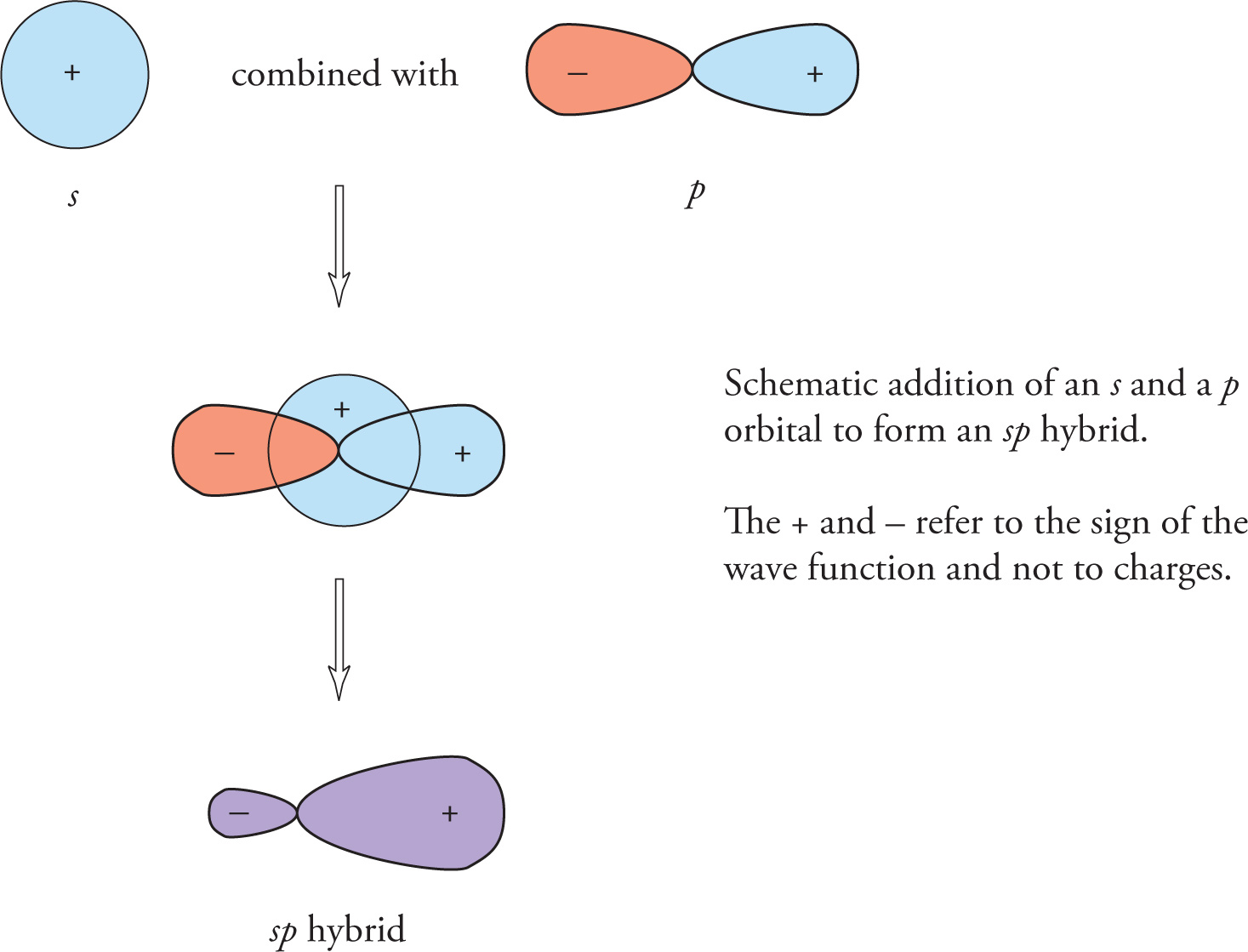
Notice that the new orbital is highly directional; this allows for better overlap when bonding.
There will be two such sp hybrid orbitals formed because two orbitals (the s and the p) were originally combined; that is, the total number of orbitals is conserved in the formation of hybrid orbitals. For this reason, the number of hybrid orbitals on a given atom of hybridization spx is 1 + x (1 for the s, x for the p’s), where x may be either 1, 2, or 3.
The percentages of the s character and p character in a given spx hybrid orbital are listed below:

To determine the hybridization for most atoms in simple molecules, add the number of attached atoms to the number of non-bonding electron pairs (localized) and use the brief table below (which also gives the ideal bond angles and orbital geometry). The number of attached atoms plus the number of lone pairs is equal to the number of orbitals combined to make the new hybridized orbitals.
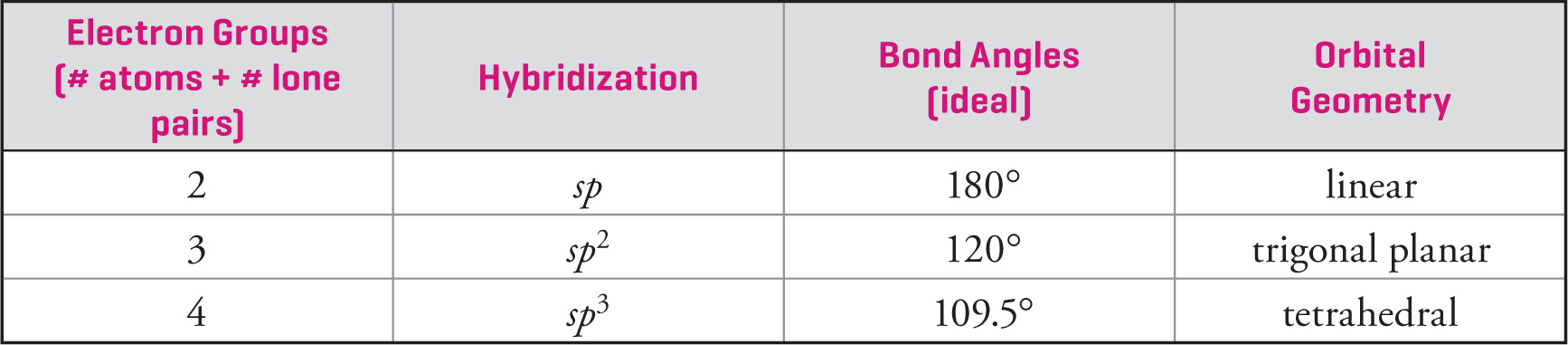
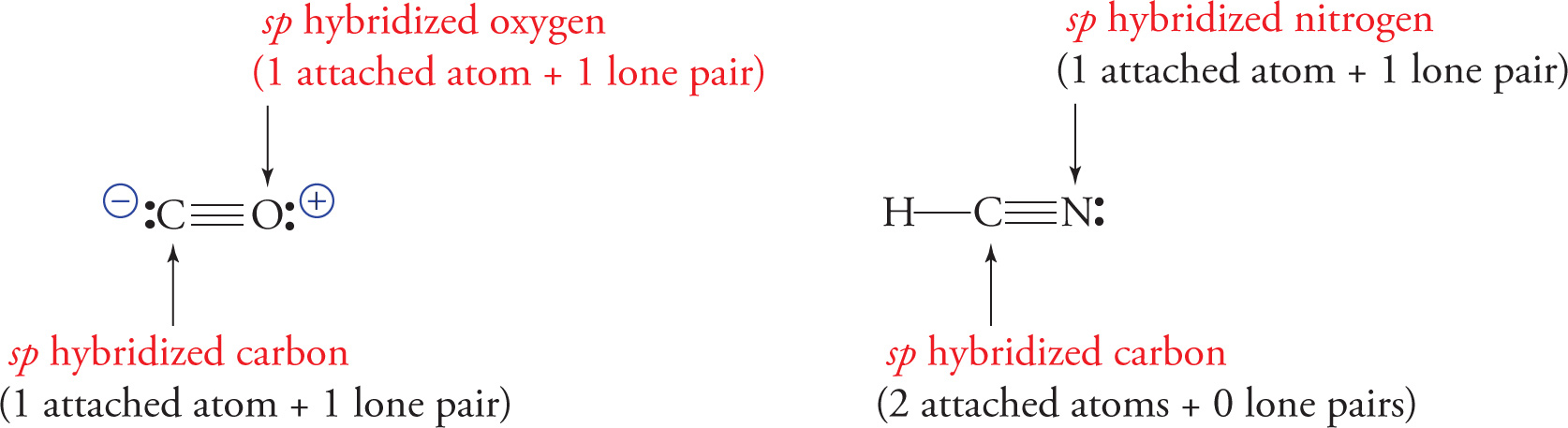
sp hybridization:
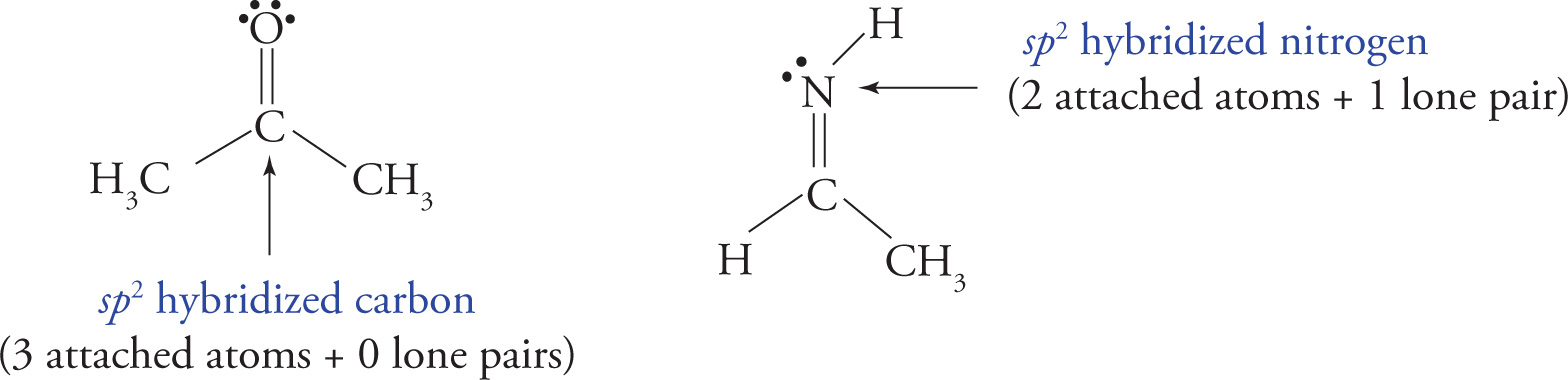
sp2 hybridization:
sp3 hybridization:
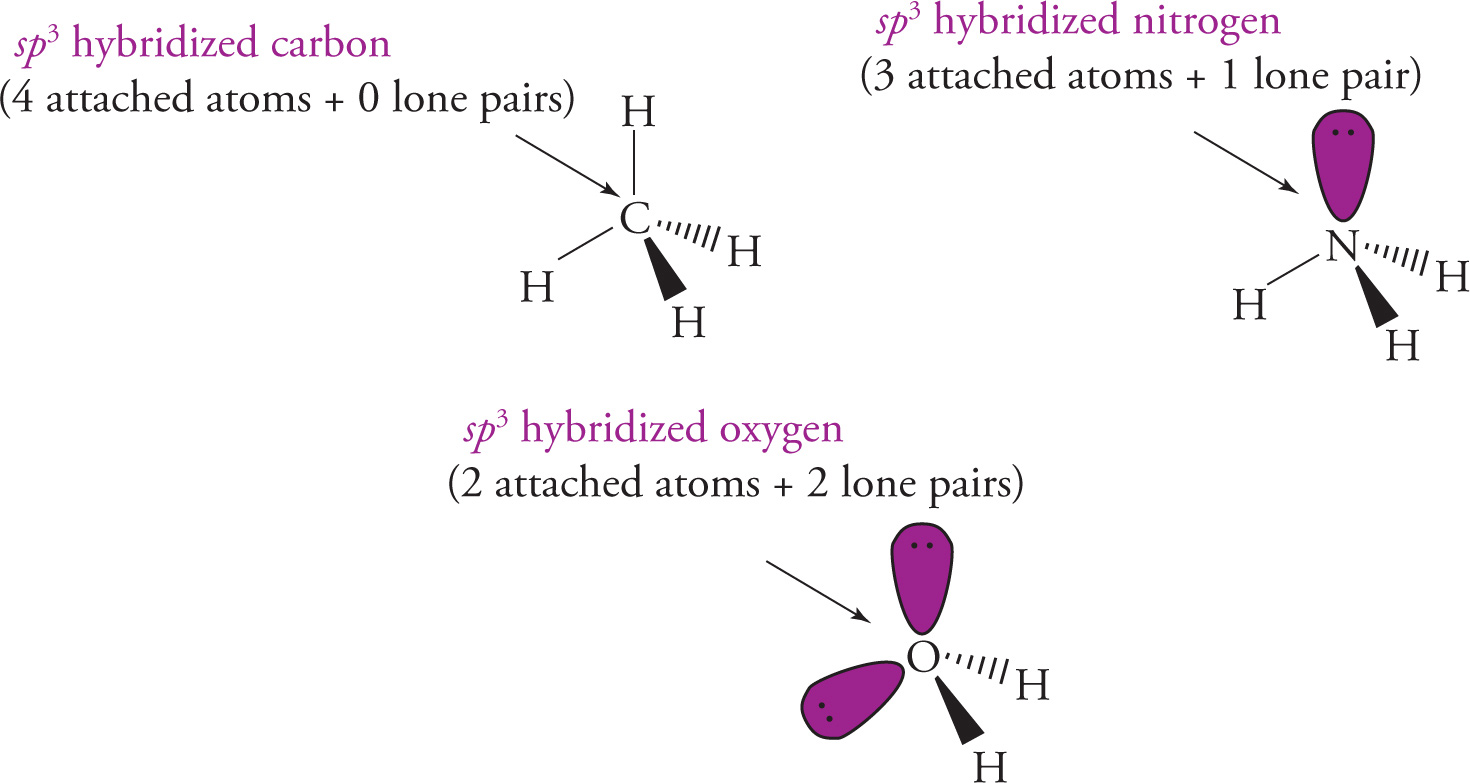
Example 5-10: Determine the hybridization of the central atom in each of the following molecules or ions from the previous example:
a) H2O
b) SO2
c) NH4+
d) PCl3
e) CO32—
Solution:
a) Hybridization of O is sp3.
b) Hybridization of S is sp2.
c) Hybridization of N is sp3.
d) Hybridization of P is sp3.
e) Hybridization of C is sp2.
Sigma (σ) Bonds
A σ bond consists of two electrons that are localized between two nuclei. It is formed by the end-to-end overlap of one hybridized orbital (or an s orbital in the case of hydrogen) from each of the two atoms participating in the bond. Below, we show the σ bonds in ethane, C2H6:
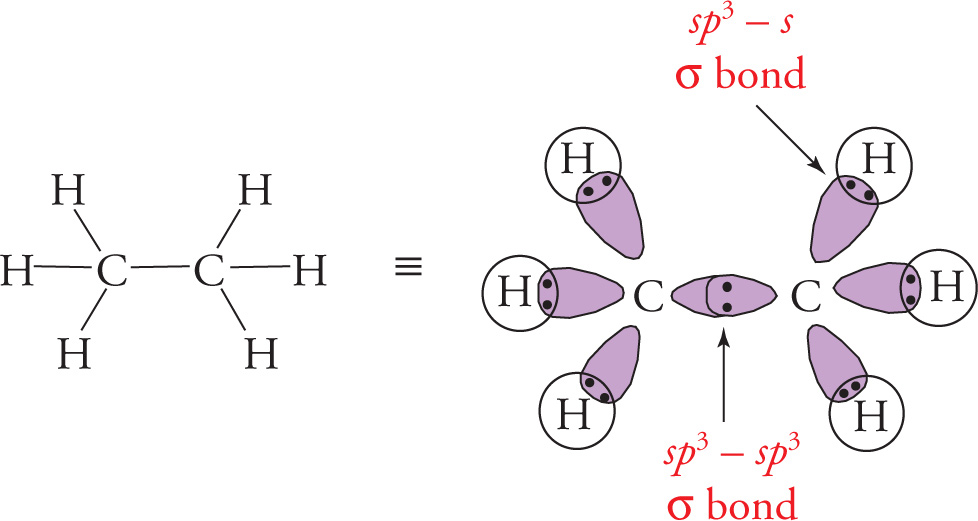
Remember that an sp3 carbon atom has 4 sp3 hybrid orbitals, which are derived from one s orbital and three p orbitals.
Example 5-11: Label the hybridization of the orbitals comprising the σ bonds in the molecules shown below:
a) 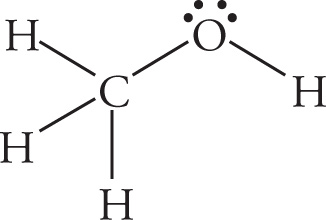
b) 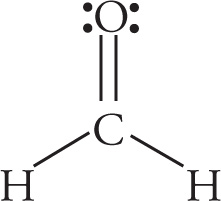
c) 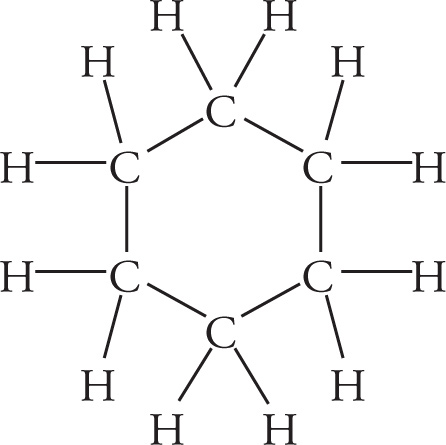
d) 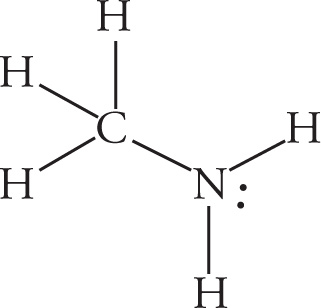
Solution:
a) Bonds to H are sp3−s σ bonds. The C—O bond is an sp3−sp3 σ bond.
b) The bonds to H are sp2−s σ bonds. The C=O bond contains an sp2−sp2 σ bond. (It’s also composed of a π bond, which we’ll discuss in the next section.)
c) All C—C bonds are sp3−sp3 σ bonds, while all C—H bonds are sp3−s σ bonds.
d) All bonds to H are sp3−s σ bonds. The C—N bond is an sp3−sp3 σ bond.
Pi (π) Bonds
A π bond is composed of two electrons that are localized to the region that lies on opposite sides of the plane formed by the two bonded nuclei and immediately adjacent atoms, not directly between the two nuclei as with a σ bond. A π bond is formed by the proper, parallel, side-to-side alignment of two unhybridized p orbitals on adjacent atoms. (An sp2 hybridized atom has three sp2 orbitals—which come from one s and two p orbitals—plus one p orbital that remains unhybridized.) Below, we show the π bonds in ethene, C2H4:
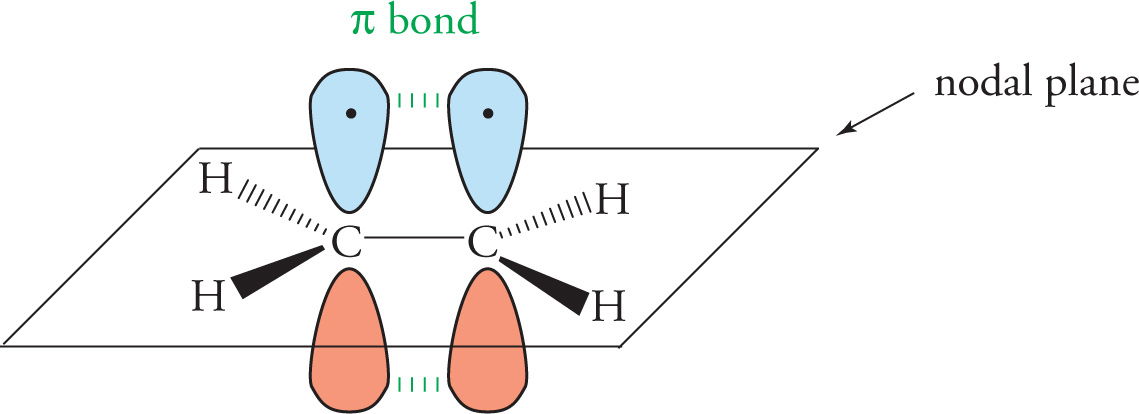
In any multiple bond, there is only one σ bond; the remainder are π bonds. Therefore:
a single bond: |
composed of 1 σ bond |
a double bond: |
composed of 1 σ bond and 1 π bond |
a triple bond: |
composed of 1 σ bond and 2 π bonds |
Example 5-12: Count the number of σ bonds and π bonds in each of the following molecules. (Don’t forget to count all of the C—H σ bonds!)
a) 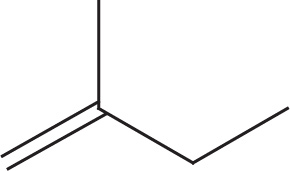
b) 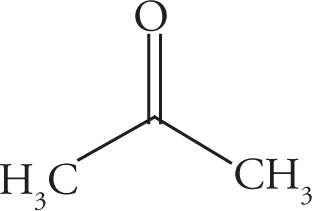
c) 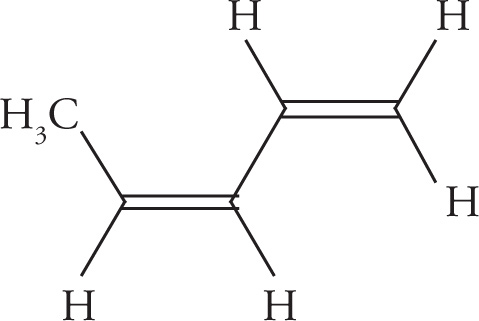
d) ![]()
e) 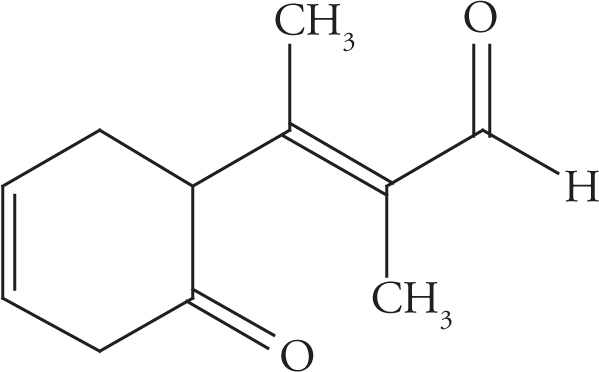
f) 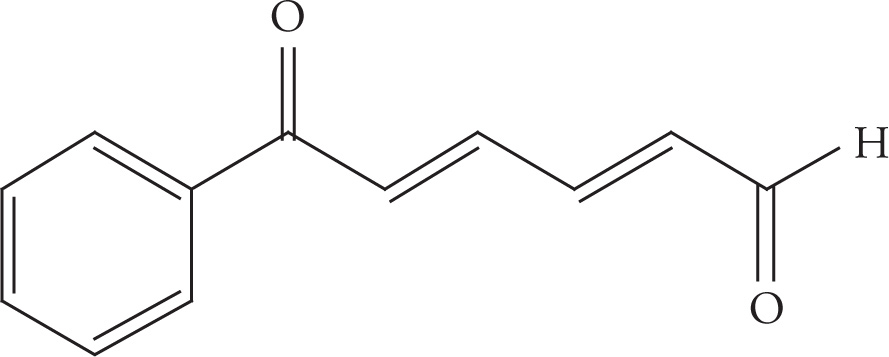
Solution:
a) 14 σ, 1 π
b) 9 σ, 1 π
c) 12 σ, 2 π
d) 2 σ, 2 π
e) 27 σ, 4 π
f) 24 σ, 7 π
5.6 MOLECULAR POLARITY
A molecule as a whole may also be polar or nonpolar. If a molecule contains no polar bonds, it cannot be polar. In addition, if a molecule contains two or more symmetrically oriented polar bonds, the bond dipoles effectively cancel each other out, evenly distributing the electron density over the entire molecule. However, if the polar bonds in a molecule are not symmetrically oriented around the central atom (generally, though not always due to the presence of a lone pair of electrons on the central atom), the individual bond dipoles will not cancel. Therefore, there will be an uneven distribution of electron density over the entire molecule, and this results in a polar molecule.
Example 5-13: For each of the molecules N2, OCS, and CCl4, describe the polarity of each bond and of the molecule as a whole.
Solution:
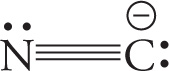
• The N≡N bond is nonpolar (since it’s a bond between two identical atoms), and since this is the molecule, it’s nonpolar, too; no dipole moment.
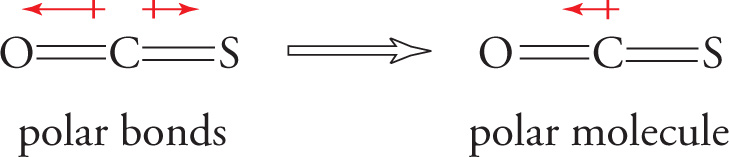
• For the molecule O=C=S, each bond is polar, since it connects two different atoms of unequal electronegativities. Furthermore, the O=C bond is more polar that then C=S bond, because the difference between the electronegativities of O and C is greater than the difference between the electronegativities of C and S. Therefore, the molecule as a whole is polar (that is, it has a dipole moment):
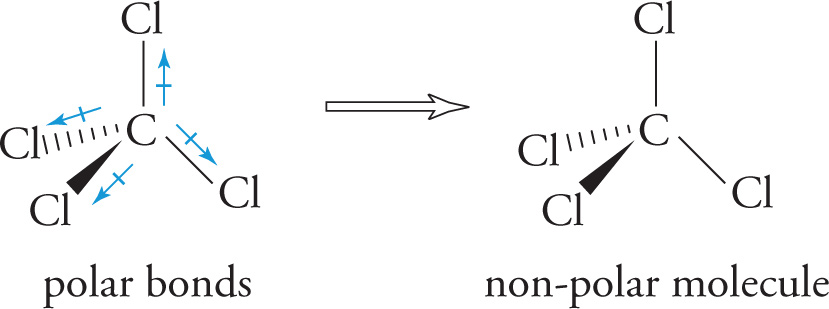
• For the molecule CCl4, each bond is polar, since it connects two different atoms of unequal electronegativities. However, the bonds are symmetrically arranged around the central C atom, leaving the molecule as a whole nonpolar, with no dipole moment:
5.7 INTERMOLECULAR FORCES
Liquids and solids are held together by intermolecular forces, such as dipole-dipole forces and London dispersion forces. Intermolecular forces are the relatively weak interactions that take place between neutral molecules.
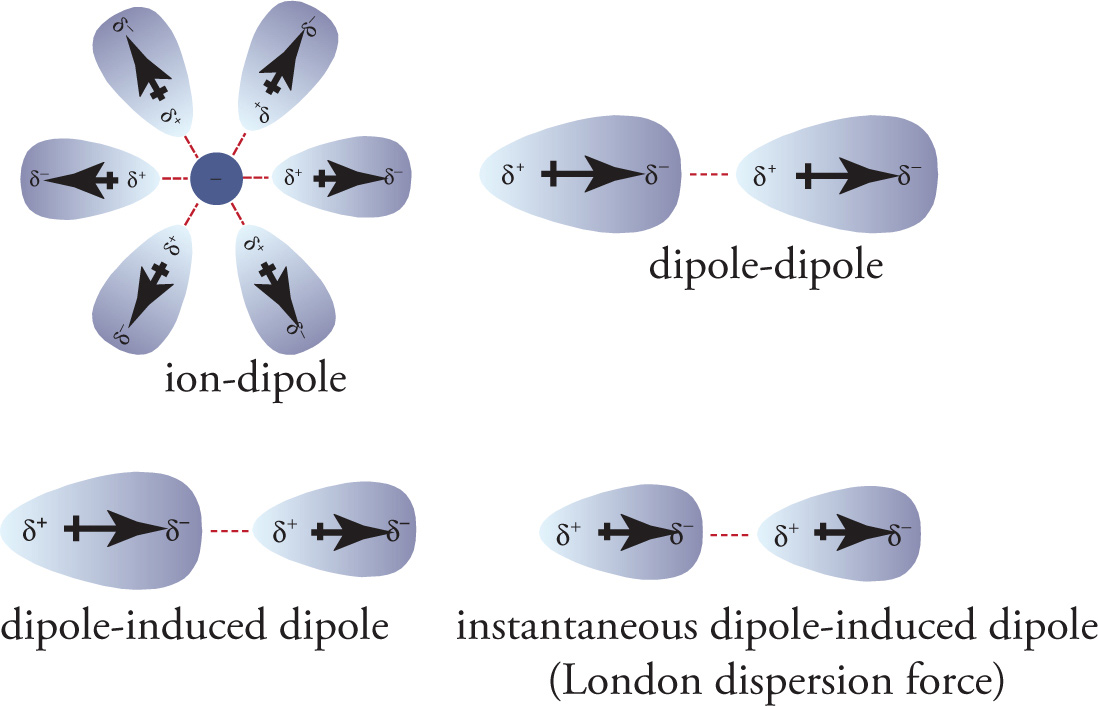
Polar molecules are attracted to ions, producing ion-dipole forces. Dipole-dipole forces are the attractions between the positive end of one polar molecule and the negative end of another polar molecule. (Hydrogen bonding [which we will look at more closely below] is the strongest dipole-dipole force.) A permanent dipole in one molecule may induce a dipole in a neighboring nonpolar molecule, producing a momentary dipole-induced dipole force.
Finally, an instantaneous dipole in a nonpolar molecule may induce a dipole in a neighboring nonpolar molecule. The resulting attractions are known as London dispersion forces, which are very weak and transient interactions between the instantaneous dipoles in nonpolar molecules. They are the weakest of all intermolecular interactions, and they’re the “default” force; all an atom or molecule needs to experience them is electrons. In addition, as the size (molecular weight) of the molecule increases, so does its number of electrons, which increases its polarizability. As a result, the partial charges of the induced dipoles get larger, so the strength of the dispersion forces increases.
Despite being weak, all intermolecular forces, including London dispersion forces, can have a profound impact on the physical properties of a particular molecule. Specifically, substances with stronger intermolecular forces will exhibit greater melting points, greater boiling points, greater viscosities, and lower vapor pressures (more on this below) than similar compounds with weaker intermolecular forces. For example, many substances that experience only dispersion forces, like fluorine (F2) and chlorine (Cl2), exist as gases under standard conditions (1 atm and 25˚C). However, bromine (Br2) is a liquid and iodine (I2) is a solid because the strength of the dispersion forces increase as atomic size increases.
A final note: Dipole forces, hydrogen bonding, and London forces are all collectively known as van der forces. However, you may sometimes see the term “van der Waals forces” used to mean only London dispersion forces.
Hydrogen Bonding
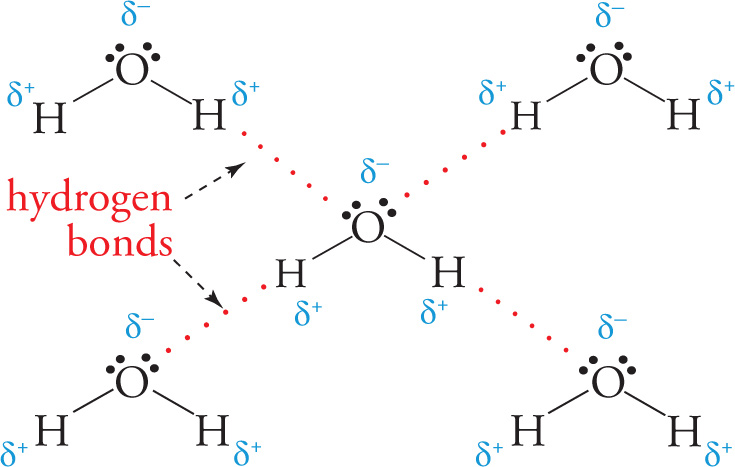
Hydrogen bonding is the strongest type of intermolecular force between neutral molecules. In order for a hydrogen bond to form, two very specific criteria must be fulfilled: 1) a molecule must have a covalent bond between H and either N, O, or F, and 2) another molecule must have a lone pair of electrons on an N, O, or F atom. A very common example of a substance that experiences hydrogen bonding is water:
One of the consequences of hydrogen bonding is the high boiling points of compounds such as NH3, H2O, and HF. The boiling points of these hydrogen-containing compounds are higher than those of the hydrogen-containing compounds of other elements from Groups V, VI, and VII (the groups where N, O, and F reside). For example, the boiling point of H2S is approximately —50°C, while that of H2O is (of course) 100°C.
Example 5-14: Identify the mixture of compounds that cannot experience hydrogen bonding with each other:
A) NH3 / H2O
B) H2O / HF
C) HF / CO2
D) H2S / HCl
Solution: Hydrogen bonding occurs when an H covalently bonded to an F, O, or N electrostatically interacts with another F, O, or N (which doesn’t need to have an H). Therefore, choices A, B, and C can all experience hydrogen bonding. Choice D, however, cannot, and this is the answer.
Vapor Pressure
One of the physical properties determined by the strength of the intermolecular forces of a substance is its vapor pressure. Vapor pressure is the pressure exerted by the gaseous phase of a liquid that evaporated from the exposed surface of the liquid. The weaker a substance’s intermolecular forces, the higher its vapor pressure and the more easily it evaporates. For example, if we compare diethyl ether (H5C2OC2H5) and water, we notice that while water undergoes hydrogen bonding, diethyl ether does not, so despite its greater molecular mass, diethyl ether will vaporize more easily and have a higher vapor pressure than water. Easily vaporized liquids—liquids with high vapor pressures—like diethyl ether are said to be volatile.
While a substance’s vapor pressure is determined in part by its intermolecular forces, vapor pressure is also temperature dependent and increases with the temperature of the substance. Increasing the average kinetic energy of the particles (which is proportional to temperature), allows them to overcome the intermolecular forces holding them together and increases the proportion of particles that can move into the gas phase. As a result, the vapor pressure of a substance is indirectly related to its boiling point, a topic we’ll discuss in more detail in Chapter 7.
Example 5-15: An understanding of intermolecular forces is of critical importance because they govern so many physical properties of a substance. The property least likely to be influenced by intermolecular force strength is:
A) color.
B) melting point.
C) solubility.
D) vapor pressure.
Solution: Any physical property that involves separating molecules from one another will very much depend upon the strength of intermolecular forces. Molecules are spread out during melting (choice B), dissolving (choice C), and evaporation (choice D). Choice A is therefore the best choice here.
5.8 TYPES OF SOLIDS
Ionic Solids
An ionic solid is held together by the electrostatic attraction between cations and anions in a lattice structure. The bonds that hold all the ions together in the crystal lattice are the same as the bonds that hold each pair of ions together. Ionic bonds are strong, and most ionic substances (like NaCl and other salts) are solid at room temperature. As discussed previously, the strength of the bonds is primarily dependent on the magnitudes of the ion charges, and to a lesser extent, the size of the ions. The greater the charge, the stronger the force of attraction between the ions. The smaller the ions, the more they are attracted to each other.
Network Solids
In a network solid, atoms are connected in a lattice of covalent bonds, meaning that all interactions between atoms are covalent bonds. Like in an ionic solid, in a network solid the intermolecular forces are identical to the intramolecular forces. You can think of a network solid as one big molecule; in a network solid there are only intramolecular forces. As a result, network solids are very strong, and tend to be very hard solids at room temperature. Diamond (one of the allotropes of carbon) and quartz (a form of silica, SiO2) are examples of network solids.
Metallic Solids
A sample of metal can be thought of as a covalently bound lattice of nuclei and their inner shell electrons, surrounded by a “sea” or “cloud” of electrons. At least one valence electron per atom is not bound to any one particular atom and is free to move throughout the lattice. These freely roaming valence electrons are called conduction electrons. As a result, metals are excellent conductors of electricity and heat, and are malleable and ductile. Metallic bonds vary widely in strength, but almost all metals are solids at room temperature.
Molecular Solids
The particles at the lattice points of a crystal of a molecular solid are molecules. These molecules are held together by one of three types of intermolecular interactions—hydrogen bonds, dipole-dipole forces, or London dispersion forces. Since these forces are significantly weaker than ionic, network, or metallic bonds, molecular compounds typically have much lower melting and boiling points than the other types of solids above. Molecular solids are often liquids or gases at room temperature, and are more likely to be solids as the strength of their intermolecular forces increase.
Example 5-16: Of the following, which one will have the lowest melting point?
A) MgO
B) CH4
C) Cr
D) HF
Solution: Almost all ionic compounds are solids at room temperature. Therefore, choice A is eliminated. Similarly, all metals except for mercury (Hg) are solids at room temperature, so eliminate choice C. Both answers B and D will be molecular solids. Hydrogen fluoride is able to hydrogen bond and will therefore have stronger intermolecular interactions than the nonpolar methane. Since choice B has the weakest intermolecular forces (London dispersion), it will be easiest to melt.
Chapter 5 Summary
• The best Lewis dot or resonance structures have 1) octets around all atoms, 2) minimized formal charge, and 3) negative charges on more electronegative elements.
• Covalent bonds form between elements with similar electronegativities (two nonmetals).
• Nonpolar bonding means equal electron sharing; polar bonding means unequal electron sharing, and electron density is higher around the more electronegative element.
• Coordinate covalent bonds form between a Lewis base (e− pair donor) and a Lewis acid (e− pair acceptor); electrons are shared.
• Ionic bonds form between elements with large differences in electronegativity (metals + nonmetals), and the strength of that bond depends on the charge and the size of the ions. Larger charges and smaller ions make the strongest ionic bonds.
• VSEPR theory predicts the shape of molecules; angles between electron groups around the central atom are maximized for greatest stability.
• The hybridization of an atom is dependent on the number of electron groups on the atom (two e− groups = sp, three e− groups = sp2, four e− groups = sp3).
• Sigma (σ) bonds generally form through the end-on-end overlap of hybrid orbitals; pi (π) bonds form through the side-to-side overlap of unhybridized p orbitals.
• If bond dipoles are symmetrically oriented in a molecule, the molecule as a whole is nonpolar; if the dipoles are asymmetrical, the molecule will be polar.
• Intermolecular forces are cohesive, and determine the physical properties (melting and boiling points, solubility, vapor pressure, etc.) of a compound based on relative strengths.
• While all molecules have London dispersion forces, they are the predominant intermolecular force that holds nonpolar molecules together. Dipole-dipole forces are the predominant intermolecular force that holds polar molecules together.
• Molecules with an H—F, H—O, or H—N bond and an N, O, or F with a lone electron pair can hydrogen bond.
CHAPTER 5 FREESTANDING PRACTICE QUESTIONS
1. Which of these molecules has the strongest dipole moment?
A) PBr3O
B) PF5
C) CCl4
D) SF6
2. A pure sample of which of the following ions/molecules will participate in intermolecular hydrogen bonding?
I. CH3CO2H
II. CO2
III. H2S
A) I only
B) III only
C) I and II
D) I and III
3. Which of the following best describes the intramolecular bonding present within a cyanide ion (CN−)?
A) Ionic bonding
B) Covalent bonding
C) Van der Waals forces
D) Induced dipole
4. All of the following would be categorized as having tetrahedral orbital geometry EXCEPT:
A) NH3
B) NH4+
C) CO2
D) CH4
5. Rank the following from highest to lowest boiling point:
I. H2SO4
II. NH3
III. CO2
IV. H2O
A) I > IV > II > III
B) II > I > IV > III
C) I > III > IV > II
D) IV > III > I > II
6. Which of the following most specifically accounts for neon’s ability to form a solid at 1 atm and 25 K?
A) Gravitational forces
B) Electrostatic forces
C) London dispersion forces
D) Strong nuclear forces
7. In the following reaction, which of the following most accurately describes the type of bond formed?
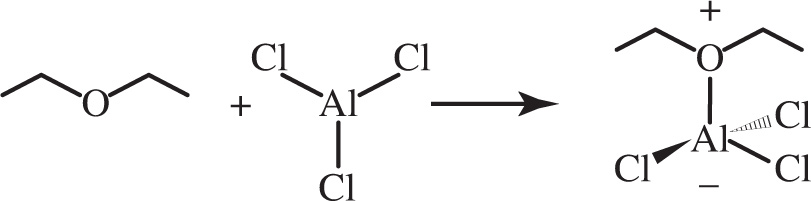
A) Covalent
B) Electrostatic
C) Metallic
D) Coordinate covalent
CHAPTER 5 PRACTICE PASSAGE
Molecules are not rigid, unchanging structures. Their atoms are in constant motion even relative to each other, ceaselessly oscillating around their average bond lengths and bond angles. For instance, in non-linear triatomic molecules there are three possible modes of vibration. There is the symmetric stretch in which both bonds in the molecule lengthen and contract in unison. In the asymmetric stretch, one bond lengthens while the other contracts. Finally, there is the bend in which the bond angle alternately widens and narrows.
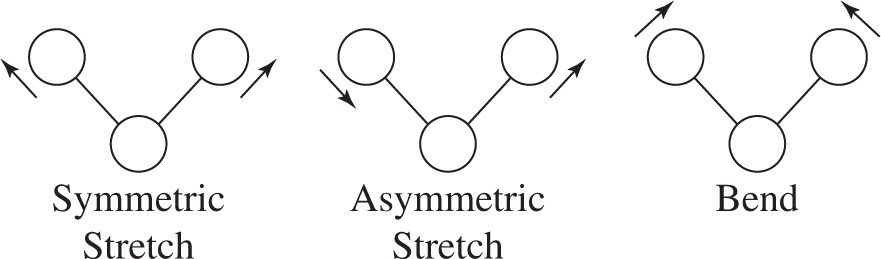
Figure 1 Vibrations of a Triatomic Molecule
More generally, each atom in a molecule is capable of moving in three distinct directions, often represented by x, y and z. In a molecule with N atoms, there will be 3N possible atomic movements. However, if all the atoms in a molecule move in the same direction, translational movement and not vibration will result. Likewise, there are some combinations of atomic motions that result in rotation of the molecule and not vibration. Taking this into account, in a molecule containing N atoms there will be 3N — 6 normal modes of vibration in non-linear molecules and 3N — 5 normal modes of vibration in linear molecules.
If we make the rough approximation that atoms in a molecule are harmonic oscillators, then the energy of their vibration is given by:
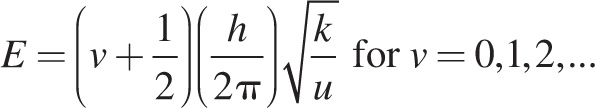
where v is the quantum vibrational number, h is Planck’s constant, k is the force constant of the bond which increases with bond strength, and u is the reduced mass of the molecule. Changes in the vibrational quantum state are associated with energies similar to infrared photons. Thus, IR spectroscopy is the study of the energetics of a molecule’s vibrational quantum states. However, only those normal modes of vibration that induce a change in the dipole moment of a molecule can be excited with IR light.
Table 1 Bond Energies of Select Diatomic Elements
Molecule |
Bond Energy (kJ/mol) |
H2 |
436 |
N2 |
946 |
O2 |
497 |
F2 |
155 |
1. Which of the following molecules has nine normal modes of vibration?
A) NI3
B) CH4
C) PF5
D) SCl6
2. A change in which of the following combinations of molecular movement can never produce a peak in an IR spectrum?
A) Translation and rotation
B) Stretching and bending
C) Vibration and translation
D) Rotation and bending
3. Assuming their reduced masses are the same, which molecule will have the highest energy of vibration in the v = 0 state?
A) N2
B) O2
C) F2
D) Cannot be determined from the information given.
4. All of the following molecules will display absorption peaks in an IR spectrum EXCEPT
A) HClO4
B) SO3
C) CO
D) O2
5. In VSEPR theory, T-shaped is a sub-class of the trigonal bipyramidal geometric family in which the central atom has exactly three atoms bound to it and two lone pairs of electrons. Which of the following molecules is T-shaped?
A) SF4
B) NH3
C) BrCl3
D) FO3−
6. For a diatomic molecule, the reduced mass is given by u = (m1 × m2) / (m1 + m2) where m1 and m2 are the atomic weights of the two bonded atoms. What will be the ratio of the ground state vibration energies of D2 to H2 assuming the force constant k is the same for both?
A) 0.5
B) 0.7
C) 1.4
D) 2.0
7. Nitrate is best described by a resonance average of three structures:
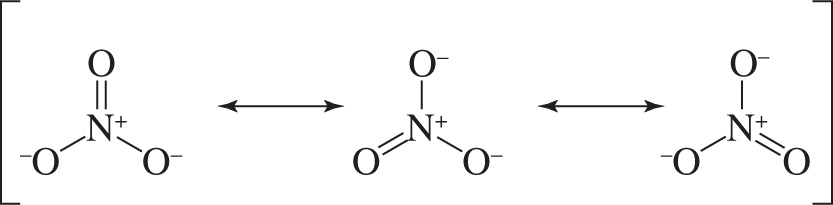
What best describes the peaks in an IR spectrum that result from the three N—O bond stretches?
A) One peak at the double bond N=O stretch frequency and two peaks at the single bond N—O stretch frequency
B) One peak at the double bond N=O stretch frequency and one peak at the single bond N—O stretch frequency
C) One peak at the double bond N=O stretch frequency and one peak between the single N—O and double bond N=O stretch frequencies
D) One peak between the single N—O and double bond N=O stretch frequencies
SOLUTIONS TO CHAPTER 5 FREESTANDING PRACTICE QUESTIONS
1. A A bond has a dipole moment when the two atoms involved in the bond differ in electronegativity. However, an entire molecule can only have a dipole if it contains bond dipoles and is asymmetrical. Choice A is tetrahedral and not all four substituents are the same. Therefore, it is asymmetrical and has a small negative dipole in the direction of the most electronegative substituent, oxygen. The remaining choices are trigonal bipyramidal, tetrahedral, and octahedral respectively. All have identical substituents, are symmetrical, and have no net dipole moment.
2. A In order to participate in intermolecular hydrogen bonding, a molecule must be able to act as both a hydrogen bond donor and acceptor. In order to act as a hydrogen bond donor, a molecule must possess a hydrogen (H) atom covalently bound to a nitrogen (N), oxygen (O), or fluorine (F) atom. In order to act as a hydrogen bond acceptor, a molecule must have an oxygen, nitrogen, or fluorine atom with an unshared pair of electrons. CH3CO2H meets both of these requirements, and is therefore a valid choice. CO2 does not possess any hydrogen atoms and is therefore an invalid option. While H2S may seem like an enticing choice, sulfur is not sufficiently electronegative to produce hydrogen bonding when covalently bound to hydrogen atoms.
3. B Van der Waals forces and induced dipoles are both examples of intermolecular forces, not intramolecular bonding, therefore choices C and D can be eliminated. The disparity in electronegativities between the carbon (C) and nitrogen (N) atoms in cyanide is not sufficient enough to produce ionic bonding, therefore choice B, covalent bonding, is the best answer.
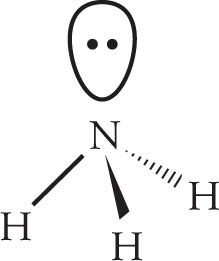
4. C The central atom, N, possesses three bonding electron groups and one lone pair of electrons. NH3 therefore has tetrahedral orbital geometry.
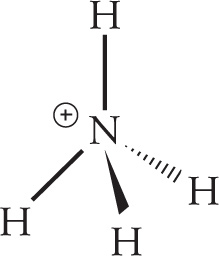
The central atom, N, possesses four bonding electron groups and zero lone pairs. NH4+ therefore has tetrahedral geometry.
The central atom, C, possesses two bonding electron groups and zero lone pairs. Recall that double bonds count as a single electron group. CO2 therefore has linear geometry.
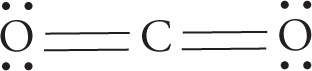
The central atom, C, possesses four bonding electron groups and zero lone pairs. CH4 therefore has tetrahedral geometry.
5. A When answering ranking questions, it is best to determine the extremes and eliminate answer choices. Of the four molecules, only H2O and H2SO4 are liquids at room temperature, and therefore would have higher boiling points than the two gases. Both experience strong hydrogen bonding, but the H2SO4 molecule is substantially larger, and, aside from this, has more sites to accept H-bonds from surrounding molecules. Therefore, H2SO4 should have the highest boiling point, eliminating answer choices B and D. Both NH3 and CO2 are gases at room temperature. However, NH3 experiences hydrogen bonding, and therefore its boiling point would be higher than CO2, eliminating choice C and making choice A the correct answer.
6. C For neon to form a solid, there must be intermolecular forces holding the atoms or molecules in relatively fixed positions. Gravitational force is given by F = G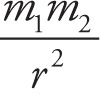 With the constant G on the order of 10−11 and the mass of neon on the order of 10−26, this force is negligible and choice A is eliminated. Electrostatic force is given by F = k
With the constant G on the order of 10−11 and the mass of neon on the order of 10−26, this force is negligible and choice A is eliminated. Electrostatic force is given by F = k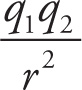 Choice B is incorrect because neon is a neutral atom without any charge, so there are no significant electrostatic forces at play. Choice D is incorrect because strong nuclear forces act over a very small distance essentially limited to the size of the nucleus. Choice C is correct. Neon is a neutral molecule and has induced dipole-dipole interactions, also known as London dispersion forces. Because this is the weakest of the van der Waals forces, neon must be cooled down close to absolute zero before forming a solid.
Choice B is incorrect because neon is a neutral atom without any charge, so there are no significant electrostatic forces at play. Choice D is incorrect because strong nuclear forces act over a very small distance essentially limited to the size of the nucleus. Choice C is correct. Neon is a neutral molecule and has induced dipole-dipole interactions, also known as London dispersion forces. Because this is the weakest of the van der Waals forces, neon must be cooled down close to absolute zero before forming a solid.
7. D This is an example of a Lewis acid-base reaction. In this type of reaction, one species accepts an electron pair from another species and a coordinate covalent bond is formed. One member of the bond donates both electrons in the bond. Whereas a coordinate covalent bond is a type of covalent interaction, the questions asks for the best answer, and coordinate covalent is more specific (eliminate choice A). Therefore, choice D is correct. An electrostatic bond is an ionic bond (eliminate choice B), and a metallic bond involves long-range delocalization of valence electrons, which is not the case in the product molecule (eliminate choice C).
SOLUTIONS TO CHAPTER 5 PRACTICE PASSAGE
1. B None of these molecules are linear. The passage states that the number of normal modes of vibration is 3N — 6 for non-linear molecules. If there are nine normal modes of vibration, 3N — 6 = 9, and N = 5. Only methane, choice B, has 5 atoms.
2. A From the passage, stretching and bending are types of vibration, while translation and rotation are not. All vibrations can potentially produce peaks in IR spectra, so choices B, C, and D are eliminated. Changes in translational and rotational movements alone do not show up in IR.
3. A If v = 0 and h and u are constant, the equation for vibration energy reduces as follows:
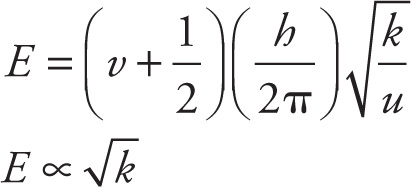
The energy of the ground state is proportional to the square root of the force constant k. The passage states that k is larger for stronger bonds, and the bond in N2 is strongest since it has the highest bond energy (see Table 1). N2 must have the highest vibrational energy in the ground state.
4. D The passage states that only those normal modes of vibration that produce a change in the dipole moment of a molecule will absorb IR light. Since O2 has no dipole, it will not have any peaks in an IR spectrum. Note that while the molecule SO3 as a whole has no dipole, its individual bonds do have detectable dipoles.
5. C As the question states, T-shaped molecules have trigonal bipyramidal orbital geometry because of its three bonded and two lone pairs of electrons around the central atom. SF4 has four atoms bound so it cannot be T-shaped, eliminating choice A. Both FO3− and NH3 have three atoms bound to their central atoms and one lone pair, so they have trigonal pyramidal molecular geometries, eliminating choices B and D. BrCl3 has three atoms bound to it and two lone pairs, making choice C the correct answer.
6. B Deuterium (D) has one proton, one neutron, and an atomic mass of 2. For D2, u = (2 × 2) / (2 + 2) = 1. For H2, u = 1 × 1 / (1 + 1) = 1/2. Since v, h, and k are all constant in this comparison, the vibration energy equation in the passage reduces as follows:
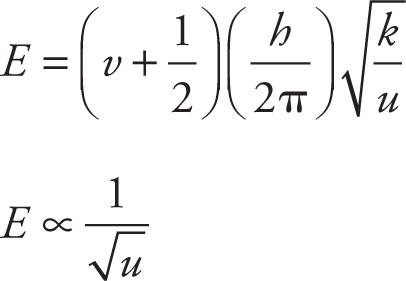
The ratio of this energy for D2 to H2 is:
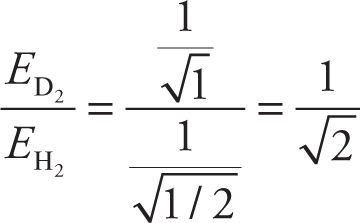
The quantity must be greater than 0.5 but less than 1, so only choice B is possible.
7. D A molecule essentially exists as an average of its resonance structures. No individual resonance structure exists in isolation. In nitrate there are three bonds of equal strength, containing about 33% N=O double bond character and about 67% N—O single bond character. Therefore, there will be only one stretch peak in the IR spectrum intermediate to the N—O and N=O stretches that accounts for all three of these bonds in nitrate.