MCAT General Chemistry Review - Alexander Stone Macnow, MD 2019-2020
The Gas Phase
Explanations to Discrete Practice Questions
1. A
Gases deviate from ideal behavior at higher pressures and lower volumes and temperatures, all of which force molecules closer together. The closer they are, the more they can participate in intermolecular forces, which violates the definition of an ideal gas. At low temperatures, the kinetic energy of the particles is reduced, so collisions with other particles or the walls of the container are more likely to result in significant changes in kinetic energy.
2. D
Density equals mass divided by volume. The mass of 1 mole of neon gas equals 20.2 grams. At STP, 1 mole of neon occupies 22.4 L.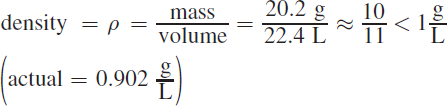
3. D
Graham’s law of effusion states that the relative rates of effusion of two gases at the same temperature and pressure are given by the inverse ratio of the square roots of the masses of the gas particles. In other words, a gas with a higher molar mass will leak more slowly than a gas with a lower molar mass. Both neon and oxygen gases will leak at slower rates than helium because they both have more mass than helium.
4. B
The pressure of the gas is calculated by subtracting the vapor pressure of water from the measured pressure during the experiment: 784 mmHg — 24 mmHg = 760 mmHg, or 1 atm. This is because the reaction is carried out in an aqueous environment; the water present will contribute to the partial pressures of the gas over the liquid. The ideal gas law can be used to calculate the moles of hydrogen gas. The volume of the gas is 0.100 L, the temperature is 298 K, and ![]() Plugging in gives:
Plugging in gives:
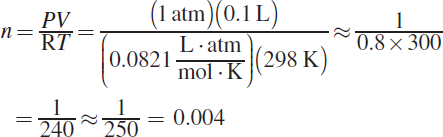
(A) incorrectly substitutes 8.314 into the gas law, rather than 0.0821. Remember that the value of R depends on the other variables in the equation; using 1 atm in the numerator necessitates using 0.0821. (C) incorrectly substitutes the wrong R and keeps the pressure in mmHg. (D) also keeps the pressure in mmHg.
5. B
Ideal gases are said to have no attractive forces between molecules. While each particle within the gas is considered to have negligible volume, ideal gases as a whole certainly do have a measurable volume, thus option I is eliminated. Gases have molar masses, thus option III is eliminated.
6. D
The first thing to do is balance the given chemical equation: NH4NO3 (s) → N2O (g) + 2 H2O (g). The mass given is 8.00 g, which represents 0.1 mol NH4NO3 ( ![]() ). When 0.1 mol of the solid decomposes, it will form 0.1 mol N2O and 0.2 mol water. This gives approximately 0.3 moles of gas product. The ideal gas equation can be used to obtain the pressure in the flask:
). When 0.1 mol of the solid decomposes, it will form 0.1 mol N2O and 0.2 mol water. This gives approximately 0.3 moles of gas product. The ideal gas equation can be used to obtain the pressure in the flask:
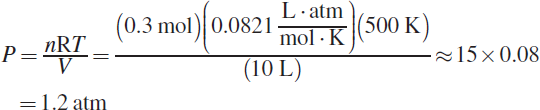
(C) is the result if one assumes the equation is balanced, obtaining 0.2 mol gas as the product.
7. A
The average kinetic energy is directly proportional to the temperature of a gas in kelvins. The kinetic molecular theory states that collisions between molecules are elastic and thus do not result in a loss of energy, eliminating (B). Gas particles are assumed to take up negligible space in kinetic molecular theory, eliminating (C). While the average kinetic energy of any gas as a whole is the same at a given temperature, the particles themselves have a distribution of speeds (as seen in the Maxwell—Boltzmann distribution curve), eliminating (D).
8. C
At STP, the difference between the distribution of speeds for helium and bromine gas is due to the difference in molar mass. Helium has a smaller molar mass than bromine. Particles with small masses travel faster than those with large masses, so the helium gas corresponds to curve B, which has a higher average speed. Because the gases are at the same temperature (273 K), they have the same average kinetic energy, eliminating (B) and (D).
9. A
The solubility of gases in liquids is directly proportional to the atmospheric pressure, as shown by Henry’s law.
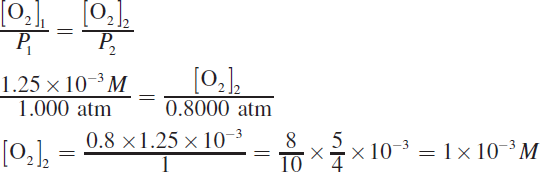
Note that the use of fractions allows this problem to be simplified more readily than it would be with decimals.
10. C
The ideal gas law can be modified to include density (ρ) because the number of moles of gas, n, is equal to the mass divided by the molar mass. Thus, ![]()
![]() Isolating for temperature gives:
Isolating for temperature gives:
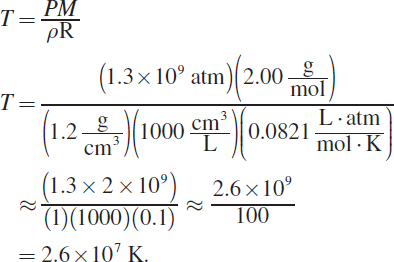
11. B
Gases are easily compressible because they travel freely with large amounts of space between molecules. Because gas particles are far apart from each other and in rapid motion, they tend to take up the volume of their container. Many gases exist as diatomic molecules, but this is not a property that characterizes all gases, eliminating option III.
12. B
We will use Charles’s law. First, we must convert the temperature to kelvins by adding 273 to get 300 K as the initial temperature. Think of this as a proportionality: If the volume is multiplied by ![]() the temperature will also have to be multiplied by
the temperature will also have to be multiplied by ![]() Thus the final temperature is 450 K, which represents a 150 K increase (which is equivalent to an increase of 150°C).
Thus the final temperature is 450 K, which represents a 150 K increase (which is equivalent to an increase of 150°C).
13. B
The partial pressure of each gas is found by multiplying the total pressure by the mole fraction of the gas. Because 80 percent of the molecules are nitrogen, the mole fraction of nitrogen gas is equal to 0.80. Similarly, for helium, the mole fraction is 0.20. To find the pressure exerted by nitrogen, multiply the total pressure (150 torr) by 0.80 to obtain 120 torr of nitrogen. The remainder, 30 torr, is attributable to helium.
14. C
Both a change in temperature and a change in volume can affect a gas’s pressure. So if one of those two variables is kept constant, as in (A) and (B), we’ll definitely be able to predict which way the pressure will change. At a constant volume, heating the gas will increase its pressure, and cooling the gas will decrease it. What about when both temperature and volume are changing? If both changes have the same effect on pressure, then we can still predict which way it will change. This is the case in (D). Cooling the gas and increasing its volume both decrease pressure. (C), on the other hand, presents too vague a scenario for us to predict definitively the change in pressure. Heating the gas would amplify the pressure, while increasing the volume would decrease it. Without knowing the magnitude of each influence, it’s impossible to say whether the pressure would increase, decrease, or stay the same.
15. C
Initially the concentration of the gas is decreased to one-half its original value. Recall that concentration (solubility) and partial pressure are directly related—as one increases, the other increases. If the experimenters then quadruple the partial pressure of oxygen in the vessel, the solubility is also increased by a factor of four. One-half times four gives twice the original concentration value. Misreading the answer choices as being related to the concentration before the experimenters increased the partial pressure leads to (D).