MCAT General Chemistry Review - Alexander Stone Macnow, MD 2019-2020
Compounds and Stoichiometry
Answers to Concept Checks
· 4.1
1. NaBr: ![]()
SrCl2: ![]()
C6H12O6: ![]()
2. NaBr: ![]()
Note that the denominator is greater than the numerator by approximately 3 percent, and the actual value is less than 1 by approximately 3 percent. This approximation can be used to quickly estimate answers that are close to one.
SrCl2: ![]()
Note that the answer is a fraction based on eighths. As these are commonly used on the MCAT, knowing the values for 1/8 through 7/8 can be useful for many problems.
C6H12O6: ![]()
Note that the final fraction is in ninths. Dividing by nine follows a standard pattern that is useful to know for the MCAT: 1/9 = 0.111, 2/9 = 0.222, 3/9 = 0.333, etc.
3. Both values equal one mole of the given substance. The number of entities in a mole is always the same (Avogadro’s number, 6.022 × 1023 mol—1).
4. Normality is calculated as ![]()
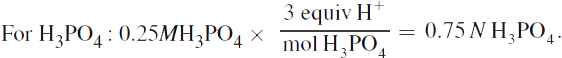
For PO43- the grams must first be converted to moles, then to normality. PO43- has a molecular mass of 95, giving
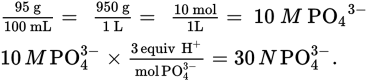
· 4.2
1. Both molecular and empirical formulas contain the same elements in the same ratios. They differ in that molecular formulas give the actual number of atoms of each element in the compound; empirical formulas give only the ratio and therefore may or may not give the actual number of atoms.
2. The molar mass of sodium carbonate is given by (2×23)+(1×12)+(3×16)=106 ![]() The percent compositions are:
The percent compositions are: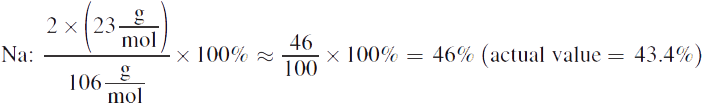
Note that in all three cases, the estimation reduces the value of the denominator, thus making the calculated value larger than the actual value.
3. Start by assuming a 100 g sample, which represents 28.5 g Fe, 24.0 g S, and 49.7 g O. Next, divide each number of grams by the atomic weight to determine the number of moles: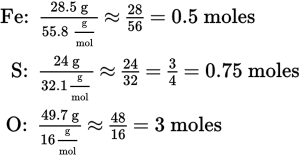
Next, find the multiplier that gives all three compounds integer values of moles. Using sulfur, multiplying 0.75 moles × 4 = 3 moles. Using 4 as a multiplier for all three compounds gives the ratio 2 Fe: 3 S : 12 O. This gives an empirical formula of Fe2S3O12.
· 4.3
1. Ammonium cations swap places with (or displace) zinc cations yielding ammonium nitrate and zinc(II) sulfide. Zinc(II) sulfide then precipitates out of solution as a solid salt.
2.
|
Reactants |
Conditions |
Products |
Reaction Type |
|
2 H2 + O2 |
→ |
2 H2O |
Combination |
|
Al(OH)3 + H3PO4 |
→ |
3 H2O + AlPO4 |
Neutralization (a type of double-displacement) |
|
2 H2O |
|
2 H2 + O2 |
Decomposition |
|
NaNO3 + CuOH |
→ |
NaOH + CuNO3 |
Double-displacement (metathesis) |
|
Zn + AgCl |
→ |
ZnCl + Ag |
Single-displacement |
· 4.4
o 2 Fe + 3 Cl2 → 2 FeCl3
o Zn + 2 HCl → ZnCl2 + H2
o C5H12 + 8 O2 → 5 CO2 + 6 H2O
o 3 Pb(NO3)2 + 2 AlCl3 → 3 PbCl2 + 2 Al(NO3)3
· 4.5
0. 4 Na (s) + O2 (g) → 2 Na2O (s)
1. ![]()
![]()
Because 4 sodium atoms are needed for every oxygen molecule, sodium will run out first. To determine the amount of Na2O formed:
![]()
2. The limiting reagent is Na because 4 sodium atoms are needed for every oxygen molecule.
![]()
will be used, so 1.0 — 0.5 mol O2 = 0.5 mol O2 will remain. In grams, this is:
![]()
excess O2
3. Reaction: BeO + H2O → Be(OH)2
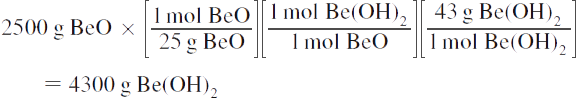
· 4.6
0. Electrolytes: HCl, MgBr2; Nonelectrolytes: sucrose, CH4
1.
|
Ion |
Cation or Anion |
Formula |
|
Phosphate |
Anion |
PO43− |
|
Hypochlorite |
Anion |
ClO− |
|
Ammonium |
Cation |
NH4+ |
|
Phosphide |
Anion |
P3− |
|
Bicarbonate |
Anion |
HCO3− |
|
Nitrite |
Anion |
NO2− |
|
Chromium(II) |
Cation |
Cr2+ |