MCAT General Chemistry Review - Alexander Stone Macnow, MD 2019-2020
Equilibrium
Equilibrium
LEARNING GOALS
After Chapter 6.1, you will be able to:
· Determine the sign of ΔG and the direction of a reaction given its Keq constant
· Calculate Keq for a reaction
· Write the equilibrium constant expression for a reaction: 3 H2 (g) + N2 (g) ⇌ 2 NH3 (g)
In the previous chapter, we danced around the term equilibrium. We warned you not to confuse the chemical equilibrium expression for the rate expression. We stressed that catalysts make reactions go faster toward their equilibrium position, but that they can’t actually change the equilibrium position or alter the value of Keq. The principles and concepts that are the focus of this chapter will direct our discussion in the upcoming chapters about some of the most important general chemistry topics for the MCAT: solutions, acids and bases, and oxidation—reduction reactions.
DYNAMIC EQUILIBRIA AND REVERSIBILITY
So far, most of the reactions we’ve covered are irreversible; that is, the reaction proceeds in one direction only, the reaction goes to completion, and the maximum amount of product formed is determined by the amount of limiting reagent initially present. Reversible reactions are those in which the reaction can proceed in one of two ways: forward (toward the products or “to the right”) and reverse (toward the reactants or “to the left”). Reversible reactions usually do not proceed to completion because the products can react together to reform the reactants. When the reaction system is closed and no reactants or products are added or removed, the system will eventually settle into a state in which the rate of the forward reaction equals the rate of the reverse reaction and the concentrations of the products and reactants remain constant. In this dynamic equilibrium, the forward and reverse reactions are still occurring—they haven’t stopped, as they do in a static equilibrium—but they are going at the same rate; thus, there is no net change in the concentrations of the products or reactants, as shown in Figure 6.1.
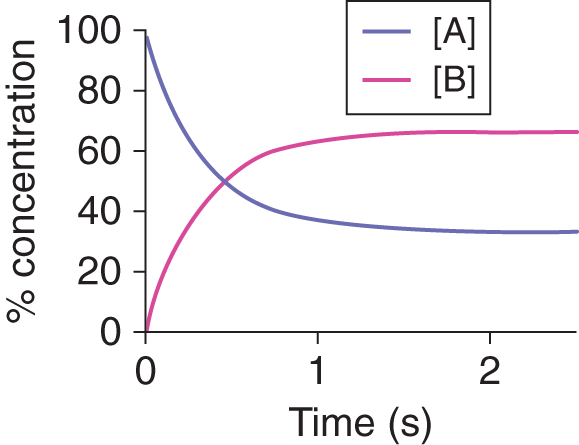 Figure 6.1. Dynamic Equilibrium Occurs when Forward and Reverse Rates Are Equal
Figure 6.1. Dynamic Equilibrium Occurs when Forward and Reverse Rates Are Equal
Bridge
Many biochemical reactions can be classified as reversible, and their activation energies are lowered by enzymes. Irreversible biochemical steps are sometimes termed “committed” because they cannot be reversed in their pathways. These steps also tend to be the rate-limiting steps of metabolic pathways, which are discussed in Chapters 9 through 11 of MCAT Biochemistry Review.
Consider the generic reversible reaction illustrated in Figure 6.1:
A ⇌ B
At equilibrium, the concentrations of A and B are constant (although not necessarily equal), and the reactions A → B and B → A continue to occur at equal rates.
Equilibrium can be thought of as a balance between the forward and reverse reactions. Better still, equilibrium should be understood on the basis of entropy, which is the measure of the distribution of energy throughout a system or between a system and its environment. For a reversible reaction at a given temperature, the reaction will reach equilibrium when the system’s entropy—or energy distribution—is at a maximum and the Gibbs free energy of the system is at a minimum.
Bridge
In Chapter 7 of MCAT General Chemistry Review, we will explore the more “classic” MCAT definition of entropy—a measure of the disorder of a system. It is important to realize, though, that the units of entropy ![]() imply a distribution of energy in a system.
imply a distribution of energy in a system.
LAW OF MASS ACTION
For a generic reversible reaction aA + bB ⇌ cC + dD, the law of mass action states that, if the system is at equilibrium at a constant temperature, then the following ratio is constant:
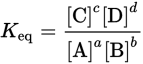
Equation 6.1
The law of mass action is actually related to the expressions for the rates of the forward and reverse reactions. Consider the following one-step reversible reaction:
2 A ⇌ B + C
Because the reaction occurs in one step, the rates of the forward and reverse reactions are given by:
ratef = kf[A]2 and rater = kr[B][C]
When ratef = rater, the system is in equilibrium. Because the rates are equal, we can set the rate expressions for the forward and reverse reactions equal to each other:
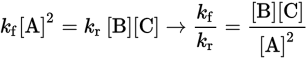
Key Concept
At equilibrium, the rate of the forward reaction equals the rate of the reverse reaction, entropy is at a maximum, and Gibbs free energy is at a minimum. This links the concepts of thermodynamics and kinetics.
Because kf and kr are both constants, we can define a new constant Kc, where Kc is called the equilibrium constant and the subscript c indicates that it is in terms of concentration. When dealing with gases, the equilibrium constant is referred to as Kp, and the subscript p indicates that it is in terms of pressure. For dilute solutions, Kc and Keq are used interchangeably. The new equation can thus be written:
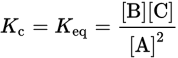
Key Concept
For most purposes, you will not need to distinguish between different K values. For dilute solutions, Keq = Kc and both are calculated in units of concentration.
While the forward and the reverse reaction rates are equal at equilibrium, the concentrations of the reactants and products are not usually equal. This means that the forward and reverse reaction rate constants, kf and kr, respectively, are not usually equal to each other. The ratio of kf to kr is Kc:
![]()
When a reaction occurs in more than one step, the equilibrium constant for the overall reaction is found by multiplying together the equilibrium constants for each step of the reaction. When this is done, the equilibrium constant for the overall reaction is equal to the concentrations of the products divided by the concentrations of the reactants in the overall reaction, with each concentration term raised to the stoichiometric coefficient for the respective species. The forward and reverse rate constants for the nth step are designated kn and k—n, respectively. For example, if the reaction aA + bB ⇌ cC + dD occurs in three steps, each with a forward and reverse rate, then:
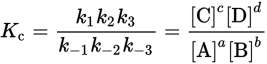
Example:
What is the expression for the equilibrium constant for the following reaction?
3 H2 (g) + N2 (g) ⇌ 2 NH3 (g)
Solution:
![]() The Kp of this reaction would be:
The Kp of this reaction would be: 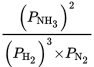
Key Concept
Remember the warning in Chapter 5 of MCAT General Chemistry Review about confusing equilibrium expressions and rate laws? In equilibrium expressions, the exponents are equal to the coefficients in the balanced equation. In rate laws, the exponents must be determined experimentally and often do not equal the stoichiometric coefficients.
REACTION QUOTIENT
The law of mass action defines the position of equilibrium; however, equilibrium is a state that is only achieved through time. Depending on the actual rates of the forward and reverse reactions, equilibrium might be achieved in microseconds or millennia. What can serve as a “timer” to indicate how far the reaction has proceeded toward equilibrium? This role is served by the reaction quotient, Q. At any point in time during a reaction, we can measure the concentrations of all of the reactants and products and calculate the reaction quotient according to the following equation:
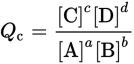
Equation 6.2
This equation looks identical to the equation for Keq. It is the same form, but the information it provides is quite different. While the concentrations used for the law of mass action are equilibrium (constant) concentrations, the concentrations of the reactants and products are not constant when calculating a value for Q of a reaction. Thus, the utility of Q is not the value itself but rather the comparison that can be made between Q at any given moment in the reaction to the known Keq for the reaction at a particular temperature. Le Châtelier’s principle, which will be elaborated upon shortly, will then guide the reaction. For any reaction, if:
· Q < Keq, then the forward reaction has not yet reached equilibrium.
o There is a greater concentration of reactants (and smaller concentration of products) than at equilibrium.
o The forward rate of reaction is increased to restore equilibrium.
· Q = Keq, then the reaction is in dynamic equilibrium.
o The reactants and products are present in equilibrium proportions.
o The forward and reverse rates of reaction are equal.
· Q > Keq, then the forward reaction has exceeded equilibrium.
o There is a greater concentration of products (and smaller concentration of reactants) than at equilibrium.
o The reverse rate of reaction is increased to restore equilibrium.
Key Concept
· Q < Keq: ΔG < 0, reaction proceeds in forward direction
· Q = Keq: ΔG = 0, reaction is in dynamic equilibrium
· Q > Keq, ΔG > 0: reaction proceeds in reverse direction
Any reaction that has not yet reached the equilibrium state, as indicated by Q < Keq, will continue spontaneously in the forward direction (consuming reactants to form products) until the equilibrium ratio of reactants and products is reached. Any reaction in the equilibrium state will continue to react in the forward and reverse directions, but the reaction rates for the forward and reverse reactions will be equal, and the concentrations of the reactants and products will be constant, such that Q = Keq. A reaction that is beyond the equilibrium state, as indicated by Q > Keq, will proceed in the reverse direction (consuming products to form reactants) until the equilibrium ratio of reactants and products is reached again. Once a reaction is at equilibrium, any further movement in either the forward direction (resulting in an increase in products) or in the reverse direction (resulting in the reformation of reactants) will be nonspontaneous. This trend is illustrated in Figure 6.2.
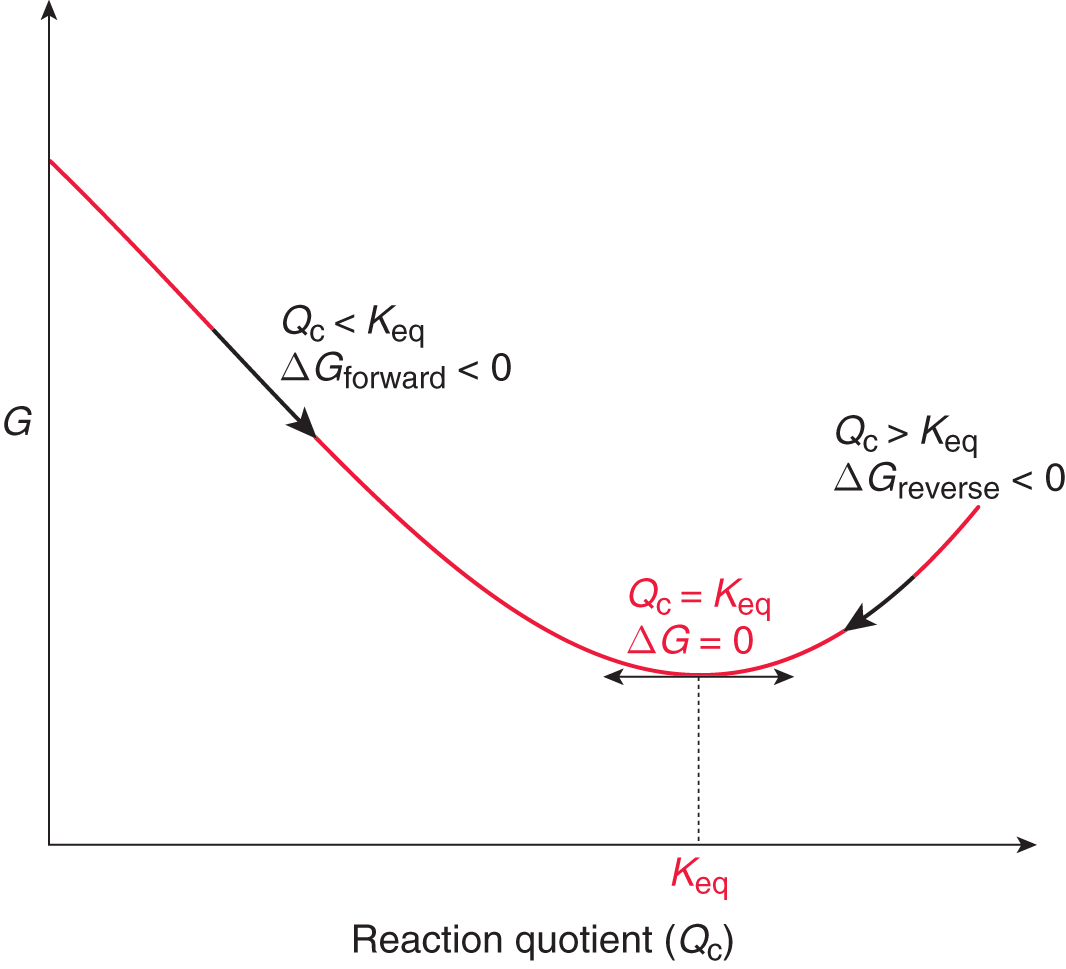 Figure 6.2. Gibbs Free Energy vs. Reaction Quotient
Figure 6.2. Gibbs Free Energy vs. Reaction Quotient
In Chapter 7 of MCAT General Chemistry Review, we’ll further discuss how the spontaneity of these systems is related to enthalpy and entropy.
PROPERTIES OF THE LAW OF MASS ACTION
Make sure to remember the following characteristics of the law of mass action and equilibrium constant expressions:
· The concentrations of pure solids and pure liquids do not appear in the equilibrium constant expression. This is because the equilibrium expression is technically based on the activities of compounds, not concentrations; the activities of pure solids and liquids are defined to be 1. For the purposes of the MCAT, there is a negligible difference between concentration and activity.
· Keq is characteristic of a particular reaction at a given temperature; the equilibrium constant is temperature-dependent.
· The larger the value of Keq, the farther to the right the equilibrium position.
· If the equilibrium constant for a reaction written in one direction is Keq, the equilibrium constant for the reverse reaction is ![]() .
.
EQUILIBRIUM CALCULATIONS
Calculations involving the equilibrium constant can take several forms but are also highly repetitive. Although we have discussed Keq in this chapter, equilibrium constants and related calculations appear in solutions (Ksp), acids and bases (Kw, Ka, and Kb), and enzyme kinetics (Kd, Kb, and Ka). Thus, learning the “ins and outs” of equilibrium calculations has a large payoff on the MCAT.
One of the first concepts to grasp is the scale of the equilibrium constant. In an ideal situation, the concentrations of products and reactants would all be the same, and regardless of their actual concentrations would reduce to 1:1 ratios. In this case, Keq would equal 1. In the real world, this situation doesn’t exist, as it is unlikely that any reaction would have exactly equal concentrations of products and reactants at equilibrium. However, a Keq of 1 can be a valuable reference point, given that ![]()
If the concentration of products is greater than the concentration of reactants, Keq becomes a “top heavy” fraction and must be greater than 1. On the other hand, if the concentration of reactants is greater than that of products, Keq becomes a “bottom heavy” fraction and must be less than 1. Keep in mind, however, that Keq is often expressed as a single value using exponents, and the sign and scale of these exponents gives even more information about the relative quantities of reactants and products. A reaction that strongly favors products will have a large, positive exponent, and the larger the exponent, the less reactant that will be present at equilibrium. In other words, a large positive exponent indicates a reaction that goes almost to completion.
On the other hand, a large negative exponent indicates a reaction that strongly favors reactants at equilibrium. In this case, only a small amount of reactant is converted to product. When performing equilibrium calculations, a Keq with a large negative exponent allows a very convenient and very necessary shortcut to be used: the amount that has reacted can be considered negligible compared to the amount of reactant that remains.
Consider the reaction ![]() with Keq = 10-12 and a starting concentration of [A] = 1 M. The Keq expression can be written as:
with Keq = 10-12 and a starting concentration of [A] = 1 M. The Keq expression can be written as:
![]()
MCAT Expertise
You may have been previously taught to solve these types of equilibrium problems using a technique referred to as an ICEbox. On Test Day, generating an entire ICEbox table takes valuable time and effort, and this technique can be shortcut using logic alone. Practice solving equilibrium problems without the ICEbox technique, using the methods described in this chapter, for a faster solution on Test Day.
If x amount of A has reacted, x amount of C and x amount of B have been produced at equilibrium, and the equilibrium concentration of A will be [1-x] M. Substituting these values into the Keq expression gives:
![]()
Unfortunately, performing the calculations required by this equation would give us a polynomial function that would be extremely burdensome to solve. However, the value of Keq has a large negative exponent, allowing us to use the “x is negligible” shortcut. Relative to the 1 M starting concentration, the amount that has reacted is so small, based on Keq = 10-12, that we can assume x is negligible and round the denominator to the starting concentration:
![]()
The problem is much more readily solved, and the value for x is found to be 10-6. This confirms our estimate that x is negligible compared to 1, since x = 0.000001, and [1- (0.00001)] ≈ 1.
If the value for Keq is within one to two orders of magnitude of one, or if the concentration of reactant that goes to product is within two orders of magnitude of the initial concentration of reactant, this estimation will not be valid. Likewise, if the value of Keq is significantly larger than one, this estimation cannot be used. In both cases, the amount that reacts will be significant compared to the starting concentration of reactant. However, these situations are unlikely to be tested on the MCAT.
Bridge
Equilibrium constants are calculated for many types of reactions, and go by many different names. For solubility problems (MCAT General Chemistry Review Chapter 9), Keq is known as Ksp. For acids and bases (MCAT General Chemistry Review Chapter 10), Keq is known as Ka, Kb, or Kw. For enzyme kinetics (MCAT Biochemistry Review Chapter 2), Keq is known as Kd, Kb or Ka.
Example:
3 moles of N2O4 is placed in a 0.5 L container and allowed to reach equilibrium according to the following reaction: ![]()
What is the equilibrium concentration of NO2, given Keq for the reaction is 6 × 10-6?
Solution:
Start by writing the expression for Keq: ![]()
Note that the concentration of NO2 is squared due to its coefficient of 2 in the balanced reaction. Next, determine the starting concentration of N2O4, taking into account that the initial volume is 500 mL = 0.5 L: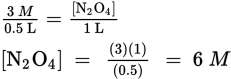
Thus, the starting concentration of N2O4 = 6 M. Next, using x to represent the amount of N2O4 that reacts, 2x to represent the amount of NO2 that is produced, and 10-6 for the value of Keq, plug into the expression for the equilibrium constant: ![]()
Note that the small negative exponent in the value of Keq indicates that x will be negligible in comparison to 6 M, allowing the Keq expression to be simplified, and x to be determined as follows:
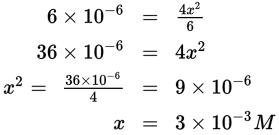
However, be careful to note that this is the value of x, which represents the amount of N2O4 that reacts. The final answer must represent the amount of NO2 produced, which is twice the amount of N2O4 that reacts, or 2x. Thus the final answer is the concentration of NO2, which is 6 × 10—3 M.
MCAT Concept Check 6.1:
Before you move on, assess your understanding of the material with these questions.
1. Given that [product] = 0.075 M and [reactant] = 1.5 M, determine the direction of reaction and the sign of the free energy change for reactions with the following Keq values: (Note: Assume that the reaction has only one product and one reactant, and that the stoichiometric coefficient for each is 1.)
|
Keq |
Direction of Reaction |
ΔG |
|
5.0 × 10−2 |
||
|
5.0 × 10−3 |
||
|
5.0 × 10−1 |
2. Write the equilibrium constant expression for the following reactions:
o CO (g) + 2 H2 (g) ⇌ CH3OH (g):
o H3PO4 (aq) + H2O (l) ⇌ H2PO4− (aq) + H3O+ (aq):
3. Consider the hypothetical reaction ![]()
For each of the following, determine if the amount of reactant A that has converted to product at equilibrium will be negligible compared to the starting concentration of A.
|
Keq |
Initial |
Is the amount reacted |
|
1.0 × 10−12 |
1 |
|
|
1.0 × 10−2 |
0.1 |
|
|
1.0 × 10−3 |
0.001 |
|
|
1.0 × 10−15 |
0.001 |
4. The following reaction has a Keq of 2.1 × 10—7. Given an initial concentration for A equal to 0.1 M and an initial concentration of B equal to 0.2 M, what is the equilibrium concentration of C? Is the approximation that x is negligible valid for this calculation?
![]()