MCAT General Chemistry Review - Alexander Stone Macnow, MD 2019-2020
Equilibrium
Answers to Concept Checks
6.1
1. First calculate the value of Q from the given concentrations:
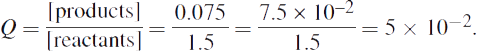
Q can now be compared to each value of Keq to predict the direction of the reaction.
|
Keq |
Direction of Reaction |
ΔG |
|
5.0 × 10−2 |
At equilibrium: no net reaction |
0 |
|
5.0 × 10−3 |
Qc > Keq: proceeds toward reactants (left) |
Positive |
|
5.0 × 10−1 |
Qc < Keq: proceeds toward products (right) |
Negative |
2. ![]()
![]()
3. The concentration of a reactant that converts to product can be considered negligible if it is two or more orders of magnitude less than the initial concentration of the reactant.
|
|
Initial |
Is the amount |
|
1 x 10-12 |
1 |
Yes |
|
1 x 10-2 |
0.1 |
No |
|
1 x 10-3 |
0.001 |
No |
|
1 x 10-15 |
0.001 |
Yes |
4. The first step in solving is to write the equation for Keq for the reaction: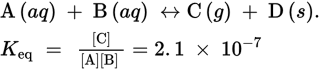
Note that the equation for Keq does not include product D because D is a solid.
Next, using the initial concentrations for A and B and x for the amount that has reacted, plug into the equation for Keq: ![]()
Given that Keq = 2.1 x 10—7, the concentrations of A and B are sufficiently large that x can be considered negligible in comparison to both. This allows the equation for Keq to be simplified and solved:
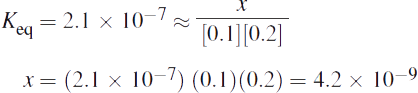
The value of x = 4.2 x 10—9 is equal to both the equilibrium concentration of C and the amount of A and B that have reacted. The approximation that x is negligible compared to the initial concentrations of A and B is valid.
· 6.2
1.
§ Increasing pH of H2SO4 (aq) ⇌ H+ (aq) + HSO4− (aq): [H+] decreases, shifting reaction to the right.
§ Decreasing pressure of 2 C (s) + O2 (g) ⇌ 2 CO (g): Reaction shifts right, favoring the side with more moles of gas.
§ Warming CH4 (g) + 2O2 (g) ⇌ CO2 (g) + 2 H2O (l) + heat: Reaction shifts left, using the additional heat energy to produce more reactants.
§ Removing water from H3PO4 (aq) + H2O (l) ⇌ H3O+ (aq) + H2PO4− (aq): Reaction shifts left. All concentrations would increase proportionately; because there are more products than reactants (and the stoichiometric coefficient is 1 for each reactant and product), the value of Q will increase.
· 6.3
1. Kinetic products are favored at low temperatures with low heat transfer. Thermodynamic products are favored at high temperatures with high heat transfer.
2. Kinetic pathways require a smaller gain in free energy to reach the transition state. They also have a higher free energy of the products, with a smaller difference in free energy between the transition state and the products.