MCAT General Chemistry Review - Alexander Stone Macnow, MD 2019-2020
Thermochemistry
Answers to Concept Checks
· 7.1
1. The boundary between system and surroundings could be placed anywhere. Most commonly, the ice pack would be considered the chemical system using up energy, and the person (and the remainder of the universe) constitutes the surroundings that are providing the heat for the ice pack to function.
2.
§ Isothermal: no change in temperature; ΔU = 0, Q = W
§ Adiabatic: no heat exchange; Q = 0, ΔU = —W
§ Isobaric: no change in pressure; line appears flat in a P—V graph
§ Isovolumetric (isochoric): no change in volume; W = 0, ΔU = Q
· 7.2
1. Kinetics, equilibrium, and thermodynamics calculations use standard conditions, which are 25°C (298 K), 1 atm pressure, and 1 M concentrations.
2. State functions are properties of a system at equilibrium and are independent of the path taken to achieve the equilibrium; they may be dependent on one another. Process functions define the path between equilibrium states and include Q (heat) and W (work).
3. State functions include pressure (P), density (ρ), temperature (T), volume (V), enthalpy (H), internal energy (U), Gibbs free energy (G), and entropy (S).
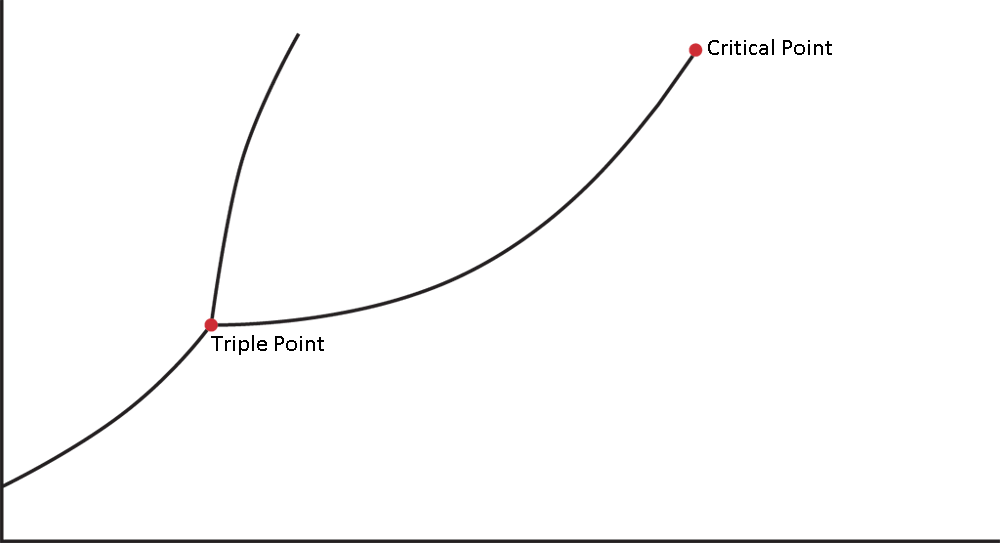
4. The triple point is the specific combination of temperature and pressure at which all three phases are in equilibrium. The critical point is the temperature and pressure above which the liquid and gas phases are indistinguishable and the heat of vaporization is zero.
· 7.3
1. Temperature is an indirect measure of the thermal content of a system that looks at average kinetic energy of particles in a sample. Heat is the thermal energy transferred between objects as a result of differences in their temperatures.
2. Specific heat (c) is the energy required to raise the temperature of one gram of a substance by one degree Celsius. Heat capacity (mc) is the product of mass and specific heat and is the energy required to raise any given amount of a substance one degree Celsius.
3. A constant-pressure calorimeter (coffee cup calorimeter) is exposed to constant (atmospheric) pressure. As the reaction proceeds, the temperature of the contents is measured to determine the heat of the reaction. A constant-volume calorimeter (bomb calorimeter) is one in which heats of certain reactions (like combustion) can be measured indirectly by assessing temperature change in a water bath around the reaction vessel.
4. ![]()
· 7.4
1. Endothermic reactions involve an increase in heat content of a system from the surroundings (ΔH > 0), while exothermic reactions involve a release of heat content from a system (ΔH < 0).
2. To reach the net equation ![]() , the second reaction must be reversed along with the sign of its enthalpy of reaction.
, the second reaction must be reversed along with the sign of its enthalpy of reaction.
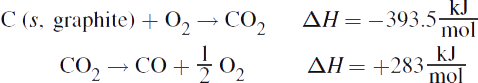
Adding the enthalpies gives:
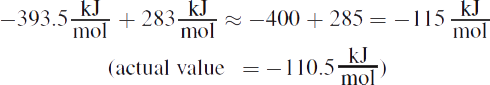
3. Enthalpy of reaction = bonds broken — bonds formed. There are four O—H bonds broken, two H—H bonds formed, and one O=O bond formed. Therefore,
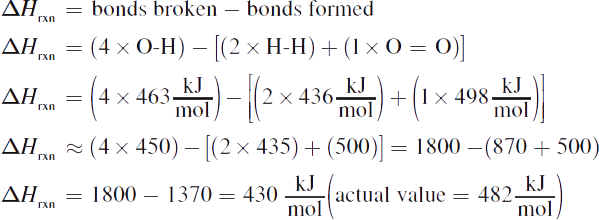
· 7.5
1. Solids have the lowest entropy, followed by liquids, with gases having the highest entropy.
2. Entropy increases as a system has more disorder or freedom of movement, and energy is dispersed in a spontaneous system. Entropy of the universe can never be decreased spontaneously.
3.
|
Reaction |
ΔS |
|
H2O (l) → H2O (s) |
Decrease (freezing) |
|
Dry ice sublimates into carbon dioxide |
Increase (sublimation) |
|
NaCl (s) → NaCl (aq) |
Increase (dissolution) |
|
N2 (g) + 3 H2 (g) → 2 NH3 (g) |
Decrease (fewer moles of gas) |
|
An ice pack is placed on a wound |
Increase (heat is transferred) |
· 7.6
1. At standard conditions:
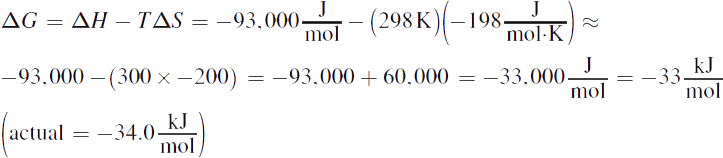
At 500 K:
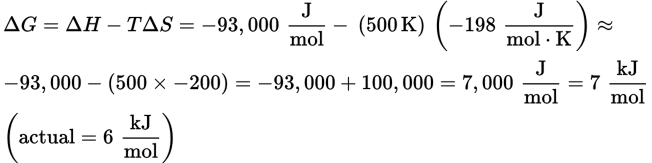
2. The system is at equilibrium when ![]()
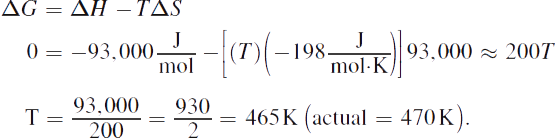
3. The value of Q would increase significantly, causing the system to shift left, forming more reactants until the system again reached equilibrium.