Microreactors in Organic Chemistry and Catalysis, Second Edition (2013)
5. Homogeneous Reactions
5.6. High Temperature Reactions
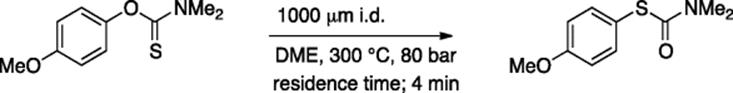
Due to the excellent heat-transfer capability and confined nature of the microreactors, high-temperature organic synthesis can be easily carried out in a microflow system. Generally, back pressure regulators are used to enable the reactions to occur at a temperature higher than the boiling point of a solvent. Kappe and coworkers showed that the Diels–Alder reaction, Newman–Kwart rearrangement, Claisen rearrangement, and Fisher indole synthesis can be efficiently carried out in a continuous flow under high temperature/pressure conditions [60]. For example, the Newman–Kwart rearrangement of O-thiocarbamate using DME as the solvent proceeded at 300 °C and 80 bar with 4 min residence time to give the rearranged product in high yield (Scheme 5.43).
Scheme 5.43
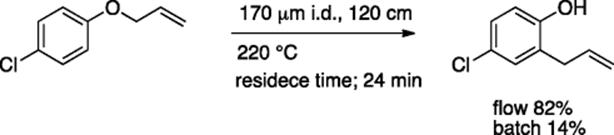
Jia, Zhou, and coworkers reported flow Claisen rearrangement without solvent [61]. The reaction of allyl p-chlorophenyl ether at 220 °C proceeded well with 24 min residence time to give 82% yield of product in a microflow system, while the same reaction in a batch reactor gave 14% yield of the product (Scheme 5.44).
Scheme 5.44
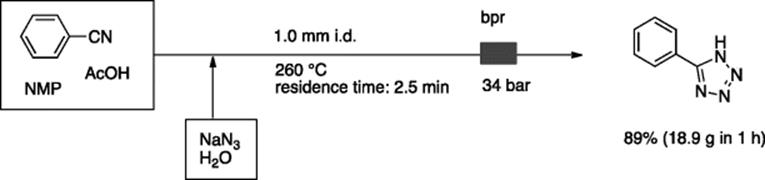
Roberge, Kappe, coworkers [62], and the Jamison's group [63] independently reported tetrazole synthesis from nitriles and azide using a high temperature/pressure reactor system. In the former case, the in situ formation of HN3from NaN3 (2 equiv.) and AcOH, reacted with benzonitrile at 260 °C and 34 bar with a 2.5 min residence time, gave 89% of 5-phenyltetrazole (18.9 g in 1 h) (Scheme 5.45) [62]. In the latter case, the reaction proceeded without the addition of AcOH, where nearly 1 equivalent of azide was employed [63]. A variety of tetrazoles were synthesized in good to high yield at 170–190 °C and 250 psi with 20–30 min residence time.
Scheme 5.45
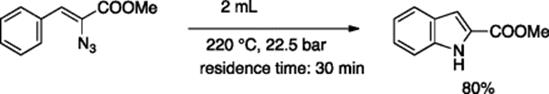
Seeberger and coworkers reported the synthesis of indoles and related heterocycles by continuous flow thermolysis of azidoacrylates under high temperature and pressure [64]. 2-Methoxycarbonyl-substituted indole was obtained in a good yield at 220 °C and 22.5 bar (Scheme 5.46). Gram scale synthesis of a precursor of d-amino acid oxidase (DAAO) inhibitor was also demonstrated.
Scheme 5.46
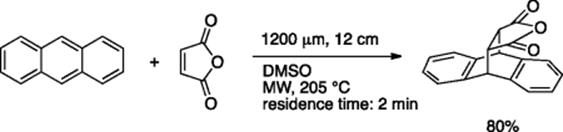
Flow reaction under microwave irradiation is also used for high temperature reactions. Shore and Organ reported the Diels–Alder reaction under MW irradiation [65]. The use of metal-coated capillaries dramatically accelerated the reaction to give the cycloaddition product (Scheme 5.47).
Scheme 5.47
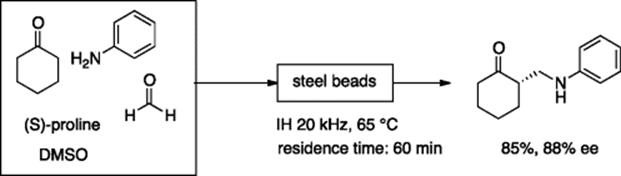
Kirschning and coworkers developed a new heating method, inductive heating (IH), for chemical reactions, which is comparable to microwave heating with respect to rate acceleration [66]. Their IH flow reactor is filled with magnetic nanoparticles (MAGSILICA), steel beads, or Pd/C, available for a variety of organic reactions. For example, proline-catalyzed Mannich reaction using steel bead-filled IH flow reactor gave the condensation product in good yield and high enantioselectivity (Scheme 5.48).
Scheme 5.48