Microreactors in Organic Chemistry and Catalysis, Second Edition (2013)
7. Heterogeneous Reactions
7.4. Flow Synthesis with Immobilized Catalysts: Solid Acid Catalysts
Solid acid catalysts, such as ion exchange resins, montmorillonites, zeolites, heteropoly acids, and silica gels, are widely used in heterogeneous reactions. Some of them are used in industrial processes and can be employed under the flow systems. In 1932, Karnatz and Whitmore reported dehydration of 3-pentanol with a phospholic acid catalyst on silica gel at high temperature under the flow conditions, which might be the first example of a heterogeneous flow reaction [55]. Although they made clear that the chemical transformation could be completed, the efficiency of the reaction was fairly limited.
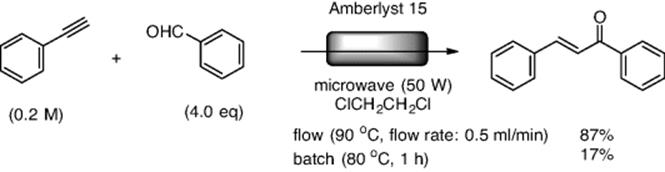
Rueping and his group reported a continuous-flow hydration–condensation reaction giving α,β-unsaturated ketones from alkynes and aldehydes by using Amberlyst™ 15 (a strongly acidic ion exchange resin) as a heterogeneous solid catalyst [56]. The flow apparatus for this experiment was set up like an overflow-type column coupled with a microwave system. Running the flow reaction of phenylacetylene with benzaldehyde at 90 °C under microwave irradiation (50 W) resulted in an efficient production of chalcone (Scheme 7.13). In contrast, the reaction under batch conditions resulted in low yield. They also evaluated the potential of the flow system by performing the reactions on a multigram scale. The reaction proceeded smoothly, providing the desired product in excellent isolated yields. They demonstrated 21 examples of the flow reactions, whose yields were within the range of 68–98%.
Scheme 7.13 Microwave-irradiated flow synthesis of chalcone.
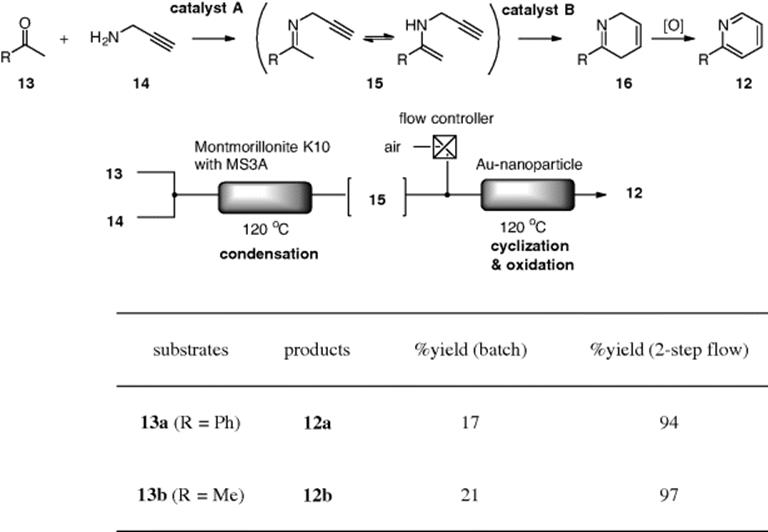
Two-step synthesis of pyridines (12) from methylketones (13) and propargylamine (14) was investigated using a combination of two flow reactors [57]. The chemical process consists of three reaction states. First, condensation of (13) with (14) gives the corresponding imine/enamine (15). Secondly, 6-exo-dig cyclization of the enamine intermediate occurs to form the dihydropyridine (16). And, finally, oxidative aromatization of (16) is required to form the desired pyridines (12). Montmorillonite (solid acid catalyst) and gold nanoparticles impregnated on alumina were found to be the best heterogeneous catalysts for the first and second/third steps, respectively. The authors demonstrated that the reactions under the two-step microflow conditions yielded better results compared with those in a batch reactor (Scheme 7.14).
Scheme 7.14 Two-step flow synthesis of 2-substituted pyridines.