Microreactors in Organic Chemistry and Catalysis, Second Edition (2013)
7. Heterogeneous Reactions
7.5. Flow Reaction with an Immobilized Catalyst: Transition Metal Catalysts Dispersed on Polymer
Transition metal catalysis has received significant attention in the synthetic chemistry community, providing access to a variety of molecular transformations based on its high and unique catalytic performance. The number of publications that have appeared in this area has become truly enormous. Despite the usefulness of the transition metal catalysts, their toxicity is often problematic. For pharmaceutical molecules, this is a major problem as there are strict guidelines that limit the contamination of heavy metals. Sometimes, the cost of precious metal catalysts such as Pd, Pt, and Au might be a drawback in the synthetic process. These problems can be overcome by employing reusable and recyclable solid-support catalysts [58, 59]. Though metal leaching from the solid phase still merits attention, the toxic waste can be minimized. Moreover, flow synthesis may allow efficient use of the metal catalyst with a high turnover number (TON). This section focuses on catalytic heterogeneous reactions using metals and metal oxides themselves immobilized in the flow reactor. The reactions using metal catalysts bound to solid-supported ligands are summarized in the next section.
7.5.1 Catalytic Hydrogenation
Over the last decade, great progress has been made using commercially available H-Cube® systems from ThalesNano Inc. for laboratory-scale flow hydrogenation reactions. The H-Cube instrument is equipped with an endogeneous continuous hydrogen gas generator, an immobilized metal catalyst cartridge, and a T-piece mixer based on an HPLC-like platform for substrate delivery and product collection (Figure 7.4). The catalyst cartridge can be packed with various metal catalysts and can hold approximately 140 mg of catalyst. Some of the commercially available catalyst cartridges for H-Cube, including CatCart®, are summarized in Table 7.1. The reaction mixture can be heated and pressurized up to 100 °C and 100 bar, respectively. The mixture is then passed through the packed-bed catalyst cartridge, where the reaction takes place, and the product continuously elutes into a collector. A required amount of hydrogen gas is generated in situ by electrolysis of distilled water. Hydrogen gas is fed into the reaction mixture as a series of bubbles, which ensures efficient mixing with large surface area-to-volume ratios. The gas can be fully consumed in the hydrogenation process, thereby preventing potential hazardous events. Reductions varying in scale from 10 mg to 10 g can be performed on the H-Cube reactor. Further scale-up is possible by using a series line-up of H-Cube Midi or H-Cube Maxi. This permits the use of another gas molecule in place of hydrogen – such as carbon monoxide, ethylene, nitric oxide, or oxygen – at up to 100 bar by adding an optional gas module, which ensures a variety of metal-catalyzed reactions under the flow conditions [60].
Figure 7.4 The H-Cube® system.

Table 7.1 Commercially available catalyst cartridges for the H-Cube®
|
Ru |
Ru/C, Ru/Al2O3, RuO2 |
|
Rh |
Rh/C, Rh/SiO2, Rh/Al2O3, Rh(cod)(S,S)-BDPP/Al2O3, RhCl(PPh3)3 |
|
Pd |
Pd/C, Pd/Al2O3, Pd/CaCO3, Pd/CaCO3 (Pb), Pd/BaSO4, Pd/SrCO3, Pd(OH)2/C, Pd encat, PdO |
|
Ir |
Ir/C, Ir/CaCO3, IrO2 |
|
Pt |
Pt/C, Pt/SBA, Pt/SiO2 [63], Pt/Al2O3, Pt/CaCO3, Pt encat |
|
Misc. |
Pd-Pt/C, Pd-Cu/Al2O3, Ru-Pd/C, Re/C, Au/C, Au/TiO2, Ni-sponge, Raney Ni, Raney Co, |
The first report using the H-Cube system appeared in 2005. A ThalesNano team reported the reduction of nitro compounds into the corresponding amines under flow conditions using a Pd/C cartridge [61]. They also demonstrated the flow reduction of a variety of functional groups (Scheme 7.15) [62].
Scheme 7.15 Hydrogenation conducted on H-Cube® as demonstrated by the ThalesNano group.
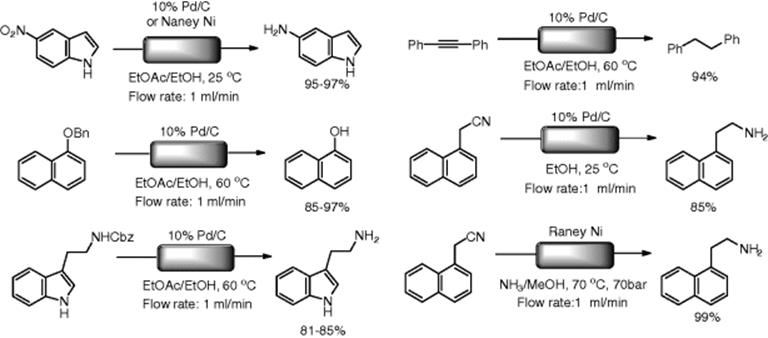
Kappe's group demonstrated hydrogenation reactions of functionalized pyridines under flow conditions utilizing the H-Cube (Scheme 7.16) [64]. Picolinic acid was fully hydrogenated into pipecolic acid by using a standard Pd/C catalyst at 80 °C under normal pressure. In contrast, under batch conditions, the transformation requires significant hydrogen pressure, elevated temperature, and/or microwave irradiation. It was found that 2,6-pyridinedicarboxylate (17) was stereoselectively hydrogenated into the corresponding cis-piperidinedicarboxylate (18). Flow reactors are usually suitable for the rapid optimization of reaction conditions. Therefore, Kappe and coworkers examined the suitable conditions for partial and full hydrogenation of ethyl nicotinate. The partial hydrogenation product (19), vinylogous carbamnate, was selectively obtained in EtOH at room temperature and 30 bar of hydrogen pressure. On the other hand, complete hydrogenation into (20) was achieved in acetic acid by using a Pt/C cartridge at higher pressure (100 bar) and temperature (100 °C). Schaus et al. reported the diastereoselective reduction of dihydropyrimidone with high stereoselectivity using an H-Cube equipped with a Raney-Ni cartridge [65].
Scheme 7.16 Hydrogenation of substituted pyridines.
The hydrogenation of a series of functional groups was performed using the H-Cube flow system [66–77]. The reduction of the carbonyl function was investigated. Sato reported continuous hydrogenation reactions in a tube reactor packed with Pd/C in 2005. They demonstrated that their flow system was more efficient for hydrogenation of 4-cyanobenzaldehyde than a batch reactor, although their system required a hydrogen gas cylinder [78]. A Cu/Fe-containing packed-bed reactor was prepared by Lessard's group. The system continuously produced 2-methylfuran from 2-furfural for 20 h without loss of activity [79]. Chen et al. reported that 2,2,6,6-tetramethyl-4-piperidinone was efficiently reduced into the corresponding alcohol thorough a Cu-Cr/γ-Al2O3 packed-bed reactor [80].
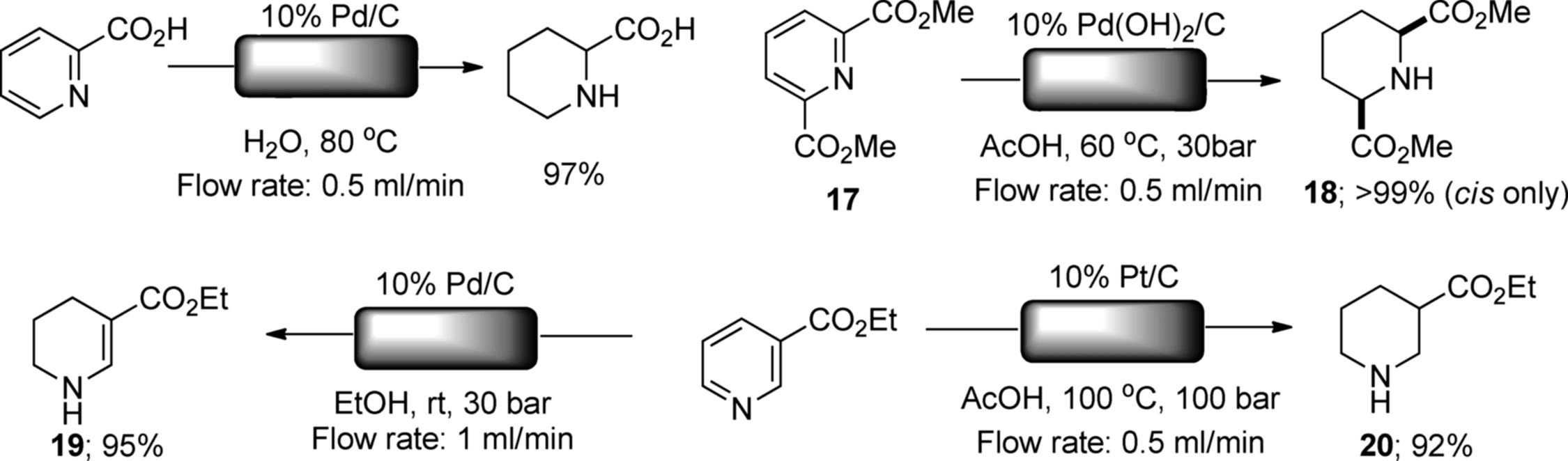
Asymmetric hydrogenation using an H-Cube reactor has also been investigated. Bartók reported the enantioselective hydrogenation of α-ketoesters by using a 5% Pt/Al2O3 cartridge [81, 82]. In the presence of cinconidine and cinconine as chiral sources, ethyl pyruvate was reduced into (R)- and (S)-lactate in about 90% ee (Scheme 7.17). Enantioselective reduction of ketopantolactone and methyl benzoylformate was also examined by the same authors. Quite recently, Bakos and coworkers reported an enantioselective hydrogenation of dehydroalanine using an in situ-produced [Rh(COD)(S)-MonoPhos)2]BF4 complex on Al2O3 [83, 84]. After the optimization of the flow reaction conditions, a highly enantioselective production of (R)-acetylalanine methyl ester (>99% conversion and 96–97% ee) was achieved.
Scheme 7.17 Asymmetric hydrogenation of pyruvate in H-Cube®.
Polymers provide stabilization for metal nanoparticles through the framework or by binding weakly to the nanoparticle surface through a heteroatom that plays the role of a ligand. Kirschning and coworkers developed Pd(0) nanoparticle-functionalized glass–polymer monoliths (Scheme 7.18). Nanoparticles of the metal catalyst were formed by passing a metal salt (Na2PdCl4) thorough an ion-exchange monolith displacing the chloride anion. Next, reduction of the ion-exchanged palladium(II) by sodium borohydride produced Pd(0) nanoclusters with a narrower particle size distribution (7 to 10 nm in size) and a better dispersion on the surface of the monolith (palladium content of 0.003 wt.%) [48]. In this paper, the authors demonstrated transfer hydrogenation of various alkenes (Scheme 7.19) and Suzuki–Miyaura coupling reactions. The activity of the monolithic catalyst was compared under batch and continuous-flow conditions. Monoliths doped with other metal particles can be prepared by the same protocol [85, 86].
Scheme 7.18 Formation of Pd(0) nanoparticles inside the monolithic reactor.
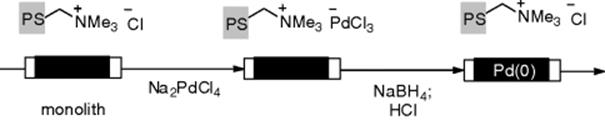
Scheme 7.19 Transfer hydrogenation using a Pd(0)-doped monolithic reactor.
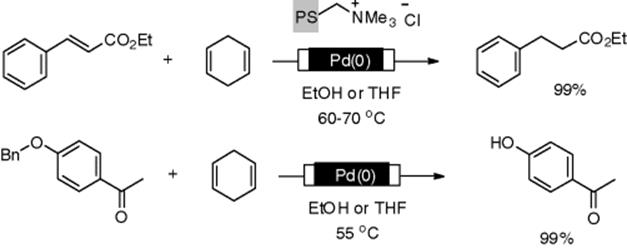
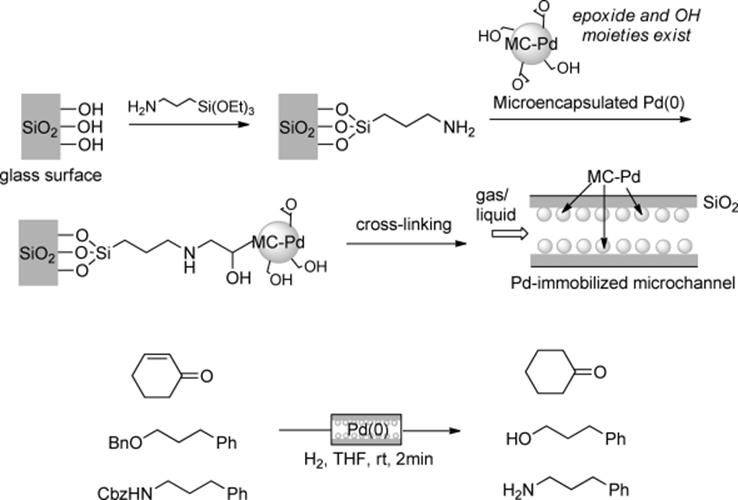
Kobayashi et al. developed a different type of reactor. They immobilized microencapsulated (MC) palladium(0) on the surface of the glass wall of a microchannel reactor [87]. Amino functions were introduced onto the surface of the glass. Then, a colloidal solution of the MC-Pd, in which cross-linking moieties were introduced, was passed through the microchannel. Cross-linkage of the MC-Pd gave the desired Pd-immobilized microchannel (Scheme 7.20). The reactor enables effective interaction between hydrogen, a substrate, and a catalyst (triphasic reaction) using extremely large interfacial areas in the very narrow channel space [88]. This reactor is applicable to the reactions using supercritical carbon dioxide (scCO2) as a solvent [89]. Ley's group developed a multichannel microreactor functionalized with Pd(0) on the internal surface. The immobilization method was similar to that for the Pd(0)-monolith by Kirschning [46]. Using this reactor, the authors demonstrated the reduction of several functional groups using triethylsilane as a hydride source [29]. Kreutzer and coworkers reported continuous-flow hydrogenation using the segmented flow method in a Pd(0)-coated capillary column. A γ-Al2O3coated fused-silica capillaries was prepared, on which nanosized Pd(0) particles were dispersed. The authors demonstrated the kilogram-scale synthesis of amine 3-phenylpropylamine from 3-phenylpropyl azide using the capillary under segmented flow conditions [90].
Scheme 7.20 Pd-immobilized glass microchannels developed by Kobayashi.
7.5.2 Catalytic Cross-Coupling Reactions and Carbonylation Reactions
Cross-coupling reactions and related reactions have received significant attention as powerful synthetic tools. The chemistry has impacted the pharmaceutical and fine-chemical areas, as well as academic researchers studying natural product chemistry and supramolecular chemistry. The development of metal catalysts and ligands continues to progress; currently, a variety of homogeneous and heterogeneous catalysts can be utilized. This section summarizes the continuous-flow cross-coupling and carbonylation reactions using heterogeneous transition catalysts [91].
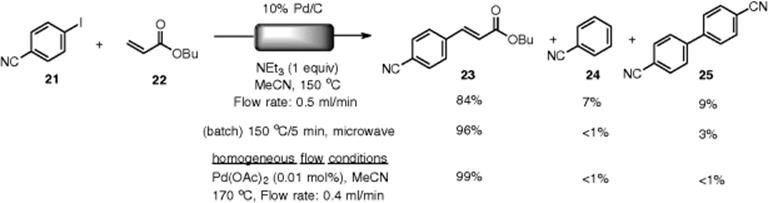
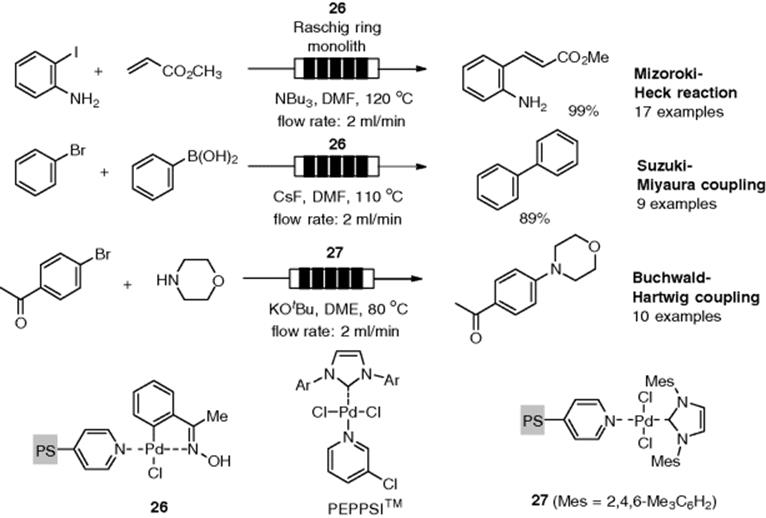
Kappe and coworkers demonstrated a Mizoroki–Heck reaction catalyzed by a ligand-free palladium, Pd/C. Mizoroki–Heck reactions of both aryl iodides and bromides with an acrylate through a Pd/C packed-bed cartridge were successfully performed under continuous-flow conditions [92]. The reaction of aryliodide (21) and butyl acrylate (22) under optimized flow conditions (at 150 °C, 0.5 ml/min flow rate) gave the desired adduct (23) in 84% yield along with the dehalogenated byproduct (24) and homocoupling product (25), while selective formation of (23) with full conversion was observed in a batch reactor (Scheme 7.21). The increased formation of the by-products under flow conditions was attributed to the fact that the catalyst quantity in the flow experiments would be significantly higher than in the batch environment. This heterogeneous flow system indicated another drawback – namely, they observed significant leaching of the Pd metal from the support. De Vries mentioned that deposition of Pd black was observed in the ligand-free Pd-catalyzed Mizoroki–Heck reaction at temperature higher than 100 °C. [93]. Under their optimized flow conditions, the leaching of Pd (1370 μg) from a reactor (310 mg Pd/C) during the Mizoroki–Heck coupling was detected. It was concluded that a flow reaction using a homogeneous pre-catalyst would be the preferred option. By using 0.01 mol.% of Pd(OAc)2, the desired coupling product was obtained with full conversion and high selectivity.
Scheme 7.21 Comparison of the heterogeneous/homogeneous flow Mizoroki–Heck reaction.
A continuous-flow Mizoroki–Heck reaction using monolithic Pd(0) nanoparticles was investigated [46, 94]. Ley's group reported that the reactor afforded the desired product in high yield with high selectivity (Scheme 7.22). The reactor was reused more than 25 times without regeneration. However, Pd leaching with an unacceptable average metal content of approximately 270 ppm was observed. Finally, the problem could be eliminated by the in-line use of a metal-scavenger column (Quadrapure TU [95]). Pd(0)-immobilized monoliths were available for copper-free Sonogashira coupling [46] and Suzuki–Miyaura coupling [96] under flow conditions.
Scheme 7.22 Flow Mizoroki–Heck reaction through Pd-monolith and metal scavenger columns.
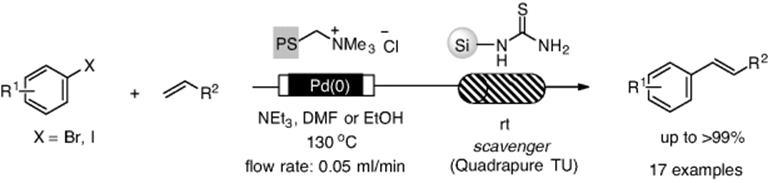
In a further study, Kirschning et al. developed a ring-type monolith reactor, which has an advantage over the rod-type reactor because it can be easily unscrewed and functionalized Raschig rings can be removed or fresh rings can be easily inserted. They prepared pyridine-functionalized monoliths by copolymerization of 4-vinylpyridine cross-linked with divinylpyridine within Raschig rings [97]. The pyridine function can coordinate with cationic metal to create a monolith doped with metal within microchannels. The monolithic reactor was used to promote the Mizoroki–Heck reaction and Suzuki–Miyaura coupling. A polymer-supported PEPPSI-type catalyst, which was developed by Organ [98], was utilized under continuous-flow Buchwald–Hartwig coupling between aryl bromides with morpholine, giving C-N-coupled adducts (Scheme 7.23) [99]. Buchwald's group achieved the C-N coupling using a packed-bed reactor under continuous-flow conditions [100].
Scheme 7.23 Cross-coupling reactions under continuous-flow conditions using poly(vinyl pyridine) doped with palladium.
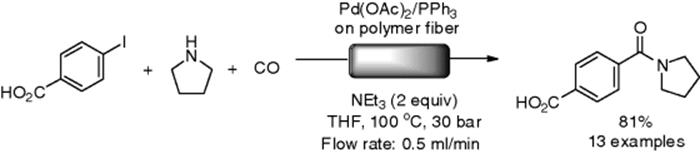
The X-Cube® reactor from ThalesNano Inc. is a suitable system for continuous-flow reactions at high temperature and high pressure without a hydrogen generator. However, gas substrates such as carbon monoxide (CO) can be easily introduced in a packed-bed reactor. ThanesNano researchers reported aminocarbonylation of aryl halides with amines and CO gas at high pressure and high temperature under flow conditions, which afforded the corresponding amides in moderate-to-high yield (Scheme 7.24) [101]. In this paper, they described that reaction parameters (solvent, base, catalyst, pressure, temperature, and so on) were rapidly optimized in the reactions, which required less than 2 min. As a continuous study, the authors reported a double carbonylation reaction of iodobenzene to give α-ketoamides in a flow reactor [102].
Scheme 7.24 Flow aminocarbonylation using X-Cube®.
Noël and coworkers reported flow Suzuki–Miyaura coupling using the X-cube reactor with a Pd-immobilized packed-bed column. A cross-coupling of heteroaryl halides and (hetero)arylboronic acids with high efficiency in a continuous flow was achieved [103]. Hiyama coupling of aryl bromide with trimethoxysilylbenzene has also been applied for flow reaction by using a SiO2-supported Pd [104]. A copper tube flow reactor enables Ullmann coupling, Sonogashira coupling, and decarboxylative aryl coupling in greater than 90% yield. The crude products could be further used in multistep processes without additional purification [105].
For transformations using a heterogeneous catalyst, microwave-assisted reactions appear to be particularly attractive, since heterogeneous catalysts general exhibit a high level of microwave absorption. Microwave-assisted flow synthesis has been well investigated, and several flow devices combined with an external microwave irradiator have been developed [3]. In 2004, Haswell and coworkers reported a flow reactor equipped with a microwave system. They proved its utility in a continuous-flow Suzuki–Miyaura coupling of aryl halides with phenylboronic acid using a Pd/Al2O3 packed-bed reactor [106]. Organ developed a microwave-assisted flow system consisting of capillaries coated with a thin film of palladium [107]. The capillary reactor with microwave system allowed for the efficient promotion of Suzuki–Miyaura coupling and the Mizoroki–Heck reaction, while without microwave irradiation only a lower production of the desired adduct was observed.
Ley et al. reported the development of a recyclable heterogeneous palladium catalyst by using microencapsulation technology [108]. Pd EnCat is a polyuria-encapsulated Pd(OAc)2 catalyst, which is commercially available and widely applicable for a variety of cross-coupling reactions under batch conditions. The autors have also investigated the use of Pd EnCat for continuous-flow Suzuki–Miyaura coupling. Similar to Haswell's reactor [106], the heterogeneous catalyst was packed within a U-tube vessel and placed within a microwave cavity. The preparation of biaryl compounds on a scale of more than 40 mmol was demonstrated by using this reactor [109].
7.5.3 Miscellaneous
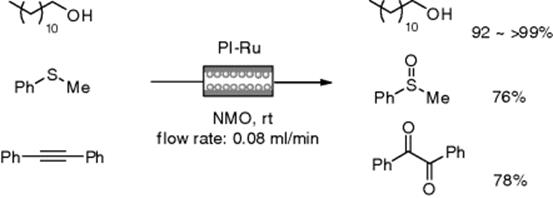
Oxidation is one of the fundamental and important transformations in organic synthesis. Ley's group reported a flow oxidation of 3,4-dimethoxybenzyl alcohol through a pre-packed column of tetra-N-alkylammonium perruthenate (TPAP) [110]. Although the corresponding aldehyde was obtained in quantitative yields, a stoichiometric amount of the immobilized reagent was required. Kobayashi et al. reported catalytic oxidation in the presence of N-methylmorpholine N-oxide (NMO) as a co-oxidant under flow conditions using a polymer-incarcerated ruthenium (PI-Ru) catalyst. The PI-Ru was prepared using basically the same procedure as used for the preparation of microencapsulation-Pd (Scheme 7.20). The Ru-immobilized catalyst was found to be highly active for oxidation under flow conditions. Primary alcohols, sulfides, and acetylenes were oxidized into aldehydes, sulfoxides (occasionally sulfones), and 1,2-diketones, respectively, with high yields (Scheme 7.25). Moreover, no leaching of Ru was observed in any case [111]. Flow oxidation of hydroquinones to benzoquinones with hydrogen peroxide through a chromatographic column packed with a mixture of Ag2O catalyst and silica gel was demonstrated by Sartoni et al. The reactor could be reused at least five times, giving the same results (>95% yield, >98% selectivity) [112]. Aerobic oxidation of alcohols to aldehydes and ketones has been developed using an X-Cube and an Ru/Al2O3 cartridge under flow conditions [113].
Scheme 7.25 Flow catalytic oxidations using a PI-Ru catalyst.
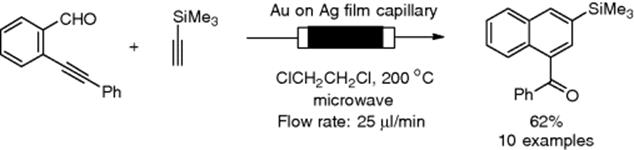
Cu(I)-catalyzed Huisgen cycloaddition of benzyl azide with phenylacetylene was carried out under flow conditions using an X-cube reactor through a Cu/C immobilized catalyst cartridge. To remove leached Cu metal, a scavenger Quadrapure-TU cartridge was in-line coupled with the flow system [114]. A three-component reaction giving triazoles from benzyl bromide, sodium azide, and phenylacetylene under flow conditions was reported by Kirschning's group [115]. Organ also developed a gold-silver-coated capillary and demonstrated its utility in microwave-assisted benzannulation under flow conditions (Scheme 7.26) [116]. The gold-coated reactor could be reused several times, but it was found that the gold film was gradually decomposed.
Scheme 7.26 Microwave-assisted benzannulation using a gold-coated capillary reactor.