Microreactors in Organic Chemistry and Catalysis, Second Edition (2013)
8. Liquid–Liquid Biphasic Reactions
Matthew J. Hutchings, Batool Ahmed-Omer, and Thomas Wirth
8.1. Introduction
Organic solvents play a key role in many chemical processes within the pharmaceutical and chemical industry. Solvent loss during the process is often inevitable because of their volatility causing environmental concerns and adverse health effects. Hence, the search for cleaner chemical processes that reduce the release of harmful chemicals into the environment is a great challenge in organic synthesis. The use of two immiscible solvents in the so-called biphasic reactions can be a possible solution to solvent loss or recycling as different solubility of substrate, reagent/catalyst, and product can be used advantageously to setup an economically and environmentally friendly process. There are many applications of biphasic reactions in different areas of chemistry [1–3].
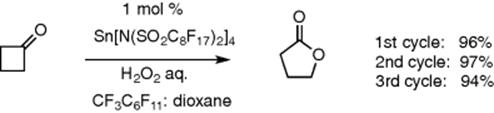
Examples of applying biphasic system to catalyzed reactions, such as phase-transfer catalysis, show the efficiency over stoichiometric reactions. In a typical catalytic biphasic system, one phase contains the catalyst while the substrate is in the second phase. In some systems, the catalyst and substrates are in the same phase while the product produced is transferred to the second phase. In a typical reaction, the two phases are mixed during the reaction and after completion, the catalyst remains in one phase ready for recycling while the product can be isolated from the second phase. The most common solvent combination consists of an organic solvent combined with another immiscible solvent, which, in most applications, is water. However, there are few examples of suitable water-soluble and stable catalysts, therefore various applications are limited to some extent [4]. Immiscible solvents other than water are recently becoming more applicable in biphasic catalysis due to the better solubility and stability of various catalysts in such solvents. For example, ionic liquids and fluorous solvents have many successful applications in liquid–liquid biphasic syntheses such as Heck reactions and hydroformylations using ionic liquid media, or Baeyer–Villiger reactions using Lewis acidic catalysts in fluorous biphasic systems as shown as an example in Scheme 8.1[5–8].
Scheme 8.1 Baeyer–Villiger reaction of cyclobutanone in fluorous media [8].