Microreactors in Organic Chemistry and Catalysis, Second Edition (2013)
8. Liquid–Liquid Biphasic Reactions
8.3. Kinetics of Biphasic Systems
Reactions in biphasic systems can take place either at the interface or in the bulk of one of the phases. The reaction at the interface depends on the reactants meeting at the interface boundary. This means, the interface area as well as the diffusion rate across the bulk of the phase plays an important role. On the other hand, in reactions that take place in the bulk phase, the reactants have to be transferred first through the interface before the reaction takes place. In this case, the rate of diffusion across the interface is an important factor. Diffusion across the interface is more complicated than the diffusion across a phase, as the mass transfer of the reactant across the interface must be taken into an account. Hence, the solubility of the reactants in each phase has to be considered, as this has an effect on diffusion across the interface. In a system where the solubility of a reactant is the same in both phases, the reactant diffuses from the concentrated phase to the less concentrated phase across the interface. This takes into account the mass transfer of the reactant from one phase into the other through the interface. The rate of diffusion J in such systems is described in Equation 8.1, where D is the diffusion coefficient, x is the diffusion distance, and l is the interface thickness (Figure 8.1).
(8.1) ![]()
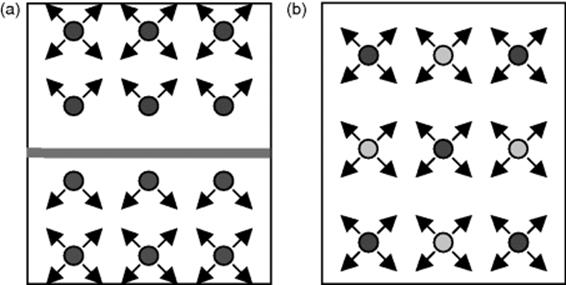
Figure 8.1 (a) Formation of an interface in a biphasic system of two solvents as result of unbalanced forces at the boundary; in contrast to (b) single phase system of two solvents without any boundary as a homogenous mixture.
A reactant with different solubility in both phases will lead to diffusion in the direction of the phase in which the reactant is more soluble. Hence, the diffusion is affected by the concentration relative to the saturation of the solution and not the absolute concentration of the reactant. The ratio of distribution of solute between the two phases is known as partition coefficient, which can be determined by measurements of the relative solubility of the reagent in each phase of the system under identical physical conditions.