Microreactors in Organic Chemistry and Catalysis, Second Edition (2013)
9. Gas–Liquid Reactions
9.4. Gas–Liquid–Solid Reactions
Shorter reaction times require for catalyst inside a microreactor to be more active than in traditional reactors. Microreactors employ less catalyst than their batch counterparts, because of their small size. The requirement for readily separable and recyclable catalysts leads often to the application of heterogeneous catalysts. Active catalysts have been coated on a microchannel wall supported by inorganic, polymer, or zeolite coatings [144, 145].
Advantages of using catalytic flow devices for solid catalyzed oxidations and hydrogenations, as well as in applying green chemistry principles and three intensifications fields are recently reviewed [24, 144].
9.4.1 Hydrogenations
Catalytic flow hydrogenations have been recently reviewed [146]. The review provides a recent update based on green chemistry principles, to present advantages of using catalytic flow devices. Various hydrogenation reactors are reviewed, among which are the falling film reactor, helical falling film reactor, 10-channel microstructured reactor, silicon/glass microreactor, mesh reactor, and H-cube reactor.
Various catalysts were applied for reactions such as the hydrogenation of double, triple, carbon–carbon bonds as well as of carbonyl, imine, and nitrile groups. This included also enantioselective hydrogenations and hydrogenation using supercritical solvents ([146] and references therein).
The chapter concluded that flow processing bears substantial advantages for unique heterogeneous catalytic hydrogenation reactions involving hydrogen gas, a substrate dissolved in a solvent, and an immobilized precious metal catalyst. The large interfacial areas generated under these conditions are not attainable in normal batch systems and can thus lead to improvements in conversions, yields, and selectivities otherwise not easily attainable. One of the many benefits of flow hydrogenation is the possibility to work with bigger scale, allowing the safe and environmentally friendly synthesis of useful chemical substances. The further evolution of this area is in catalyst development meeting the needs for better selectivities and higher turnover numbers as well as improved stability [146].
9.4.1.1 Cyclohexene Hydrogenation over Pt/Al2O3
Cyclohexene hydrogenation is a well-studied process, which serves as model reaction to evaluate performance of gas–liquid reactors since it is a fast process causing mass transfer limitations for many reactors [68, 69]. Processing at room temperature and atmospheric pressure reduces the technical expenditure for experiments so that the cyclohexene hydrogenation is accepted as simple and general method for mass transfer evaluation. Flow-pattern maps and kinetics were determined for conventional fixed bed reactors as well as overall mass transfer coefficients and energy dissipation. In this way, mass transfer can be analyzed quantitatively for new reactor concepts and processing conditions. Besides mass transfer, heat transfer is an issue, as the reaction is exothermic. Hot spot formation should be suppressed as these would decrease selectivity and catalytic activity [68].
(9.27) ![]()
Conversions ranged from 2.8% to 16.0% in a mini packed bed microreactor [68, 69]. Average reaction rates amounted from 8.6 × 10−4 to 1.4 × 10−3 mol/(min gcat) [28] (see also Ref. [147]), which is close to the intrinsic reaction rate of 3.4 × 10−3 mol/(min gcat). Cyclohexene was given in excess so that the kinetics were zero order for this species and also first order for hydrogen [68]. For this pseudo-first order reaction with the catalyst surface area of 0.57 m2/g and catalyst loading density of 1 g/cm3, a volumetric rate constant of 16 s−1 was determined.
Studies were also performed with an artificial fixed bed composed of an array of microstructured columns made by a plasma etch process. These columns were made porous to increase the surface area to 100 m2, which is not far from the porosity of catalyst particles in fixed beds, and then coated with catalyst [69]. The performance of such catalytic microcolumns was compared to a catalytic fixed bed reactor. When normalized to the metal content, the reaction rates of the columnar and the particle-containing reactor are similar with 6.5 × 10−5 and 4.5 × 10−5 mol/(min m2), respectively.
The increased interfacial area in the microreactor led to an increased pressure drop. The energy dissipation factor, the power unit per reactor volume of the microreactor process was thus higher (εV = 2–5 kW/m3) as compared to the laboratory trickle-bed reactors (εV = 0.01–0.2 kW/m3) [68]. This is, however, outperformed by the still larger gain in mass transfer, so that the net performance of the microreactor is better.
The same reaction was performed in a packed bed microreactor using supercritical CO2 [148]. The phase studies confirmed a single phase reaction mixture at 25 and 50 °C during isobaric reaction conditions of 136 bar. The single phase behavior of the reaction mixture avoids the gas/liquid mass transfer resistance. This further enhances productivity, so that the comparison with larger scale systems indicated an increase of about one order of magnitude in space time yield [148].
9.4.1.2 Hydrogenation of p-Nitrotoluene and Nitrobenzene over Pd/C and Pd/Al2O3
Nitroaromatics are frequently used in organic synthesis as intermediates for the corresponding aniline derivatives by hydrogenation [76, 147, 149]. Among other uses, pharmaceuticals are produced via that route [147]. The high intrinsic reaction rate in hydrogenations of nitrobenzene derivatives cannot be exploited by conventional reactors, because of to the large reaction enthalpies (500–550 kJ/mol) much heat would be released [76, 147, 150]. Another reason is the loss in selectivity under such conditions by decomposition of the nitroaromatics or by formation of partially hydrogenated intermediates [150]. Therefore, hydrogen supply is restricted to slow down the reaction.
Hydrogenations of nitrobenzene and p-nitrotoluene over supported noble metal catalysts are often investigated as model reactions, as they consist of various elemental reactions with different intermediates, which can react with each other, as depicted in the scheme below for p-nitro toluene [150]. At short reaction times, the intermediates are predominantly formed, while complete conversion to p-methyl aniline is achieved at long reaction times. Aniline itself can react further to form side products such as cyclohexanol, cyclohexyl amine, and other species.
(9.28) 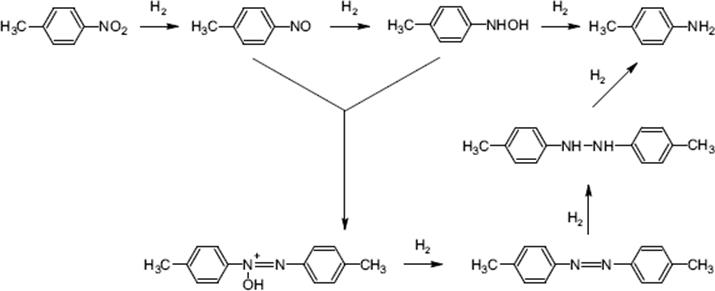
The hydrogenation of p-nitrotoluene in the presence of a supported Pd catalyst was carried out in a stacked plate microreactor with complete selectivity [76, 150]. The Pd catalyst was prepared in three different ways. Conversions were 58–98% for an impregnated aluminum oxide wash-coat catalyst, depending on the process conditions. The conversions for an electrodeposited catalyst and an impregnated catalyst on electro-oxidized nano-porous substrate were 58% and 89%, respectively. The best latter result is similar to that of a conventional fixed bed reactor (85%), while the maximum yield of 30% in a microreactor was superior due to the high selectivity.
Computational fluid dynamics (CFD) calculations were performed to give the Pd concentration profile in a nano-pore of the oxide catalyst carrier layer [76]. For wet-chemical deposition, most of the catalyst was deposited in the pore mouth, in the first 4 μm of the pore. Thus, most of the hydrogenation reaction is expected to occur in this location. For electrochemical deposition, large fractions of the catalyst are located both in the pore mouth and base. Since the pore base is not expected to contribute to large extent to hydrogenation, a worse performance was proposed for this case. This was corroborated by experimental evidence. Higher conversions were indeed found for the catalyst prepared by wet chemical deposition.
An even wider variation of preparation procedures for the palladium catalyst was investigated for the hydrogenation of nitrobenzene using a falling film microreactor [150, 152].
(9.29) 
A sputtered palladium catalyst exhibited low conversion and large deactivation of the catalyst was found initially (60 °C; 4 bar) [150]. The corresponding selectivity was also low. A slightly better performance was obtained after an oxidation/reduction cycle. Following a steep initial deactivation, the catalyst activity stabilized at 2–4% conversion and at about 60% selectivity. After reactivation, selectivity approached initially 100%. A palladium catalyst made by UV-decomposition had a conversion somewhat higher than the sputtered one. When stopping and restarting microreactor operation, activity changed stepwise. A similar pattern of side products as for the sputtered catalyst was found. An improved performance was found for an impregnated palladium catalyst. Complete conversion was found after 6 h, and selectivity decreased slowly to a level, which still was high. For an incipient-wetness palladium catalyst, the best performance of all catalysts investigated was found. Starting from more than 90% conversion, a 75% conversion at selectivity of 80% was reached and maintained for a long time (Figure 9.36).
Figure 9.36 Arrhenius plot for the Pd/Al2O3 (open circles) and Pt/Al2O3 (filled rhombs) catalysts. Continuous microreactor operation; 2–3 bar; 1 M reactant solution in methylcyclohexane; 0.1 ml/min liquid flow rate. Source: By courtesy of Royal Society of Chemistry [58].
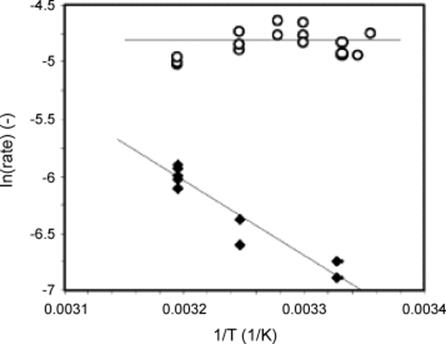
It was concluded that the catalyst lifetime is a function of the catalyst loading and does not relate to its way of synthesis [150]. With larger loadings, catalysts are longer active before they need reactivation. With regards to lifetime and activity the four catalysts were ranked as follows: wet-impregnation ![]() incipient wetness > UV-decomposition of precursors > sputtering. In case of loss of performance, two reactivation routes for the catalyst were recommended. First, to try to dissolve the organic residues on the catalyst by dichloromethane or, if this is not successful, to burn these coats by heating in air. In this way, initial activity was gained back showing again complete conversion. Deactivation of such catalysts did still occur, however, could be slowed down by suitable reactivation.
incipient wetness > UV-decomposition of precursors > sputtering. In case of loss of performance, two reactivation routes for the catalyst were recommended. First, to try to dissolve the organic residues on the catalyst by dichloromethane or, if this is not successful, to burn these coats by heating in air. In this way, initial activity was gained back showing again complete conversion. Deactivation of such catalysts did still occur, however, could be slowed down by suitable reactivation.
From the list of side products mentioned for the reduction of nitrobenzene, all intermediates except phenylhydroxylamine were identified [150]. Their relative amounts allowed a judgment on the route by which the hydrogenation proceeds. As a result, it was concluded that species containing nitroso-, azo-, and azoxy-groups have strong interaction with the catalyst and so are preferably involved in the reaction course. On the contrary, reduction of the hydrazo-species was hindered. These assumptions are in agreement with previous reports.
The hydrodynamics of the falling film governs, as to be expected, the reaction behavior [151, 152]. When increasing flow rate, the reaction time was reduced and the film thickness was increased. For both reasons, the conversion decreased and exhibited in a linear dependence on log Re, Re being the Reynolds number. The hydrogenation conversion had only a weak dependence on hydrogen pressure.
9.4.1.3 Hydrogenation of Azide
The selective catalytic hydrogenation of nitrogen containing compound is investigated using segmented flow in capillary columns [153]. Commercially available fused-silica capillaries coated with a 6 μm thick layer of high surface area γ-Al2O3 that were pretreated and impregnated with a [Pd(OAc)2] solution were used. Nanosized Pd particles were evenly dispersed on the γ-Al2O3 coating layer.
The hydrogenation of the nitrogen-containing compounds over Pd catalysts is a good example of strongly adsorbing, or even poisoning, reactants and products, that are much easier analyzed in a continuous flow. Moreover, azide chemistry benefits greatly from miniaturized synthesis, because the toxic and explosive properties of azides complicate the handling of large quantities.
(9.30) 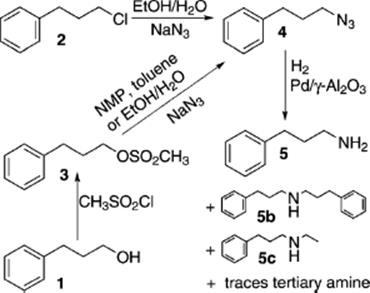
The azide was synthesized from materials shown in the scheme, in various solvents, and subsequently hydrogenated without intermediate workup, that is, the purification was postponed until the toxic and explosive azide was converted. The goal was to keep the hydrogenation to the primary amine going as long as possible until deactivation of the catalyst by strongly adsorbing compounds in the synthesis mixture sets in.
An optimum of 2 kg of the corresponding amine could be synthesized using 1 g Pd in ethanol/water without catalyst regeneration. Therewith, it was shown that continuous-flow analysis allows a fast optimization of various aspects of heterogeneous catalysis and synthesis routes, such as solvent effects, competitive adsorption, and irreversible poisoning.
9.4.1.4 Hydrogenation of Pharmaceutical Intermediates
A significant portion of the reaction steps in a typical fine chemical synthesis are catalytic hydrogenations, generally limited by resistances to mass and heat transport. Large surface-to-volume ratios of microreactors would greatly benefit chemical processing in the pharmaceutical industry [154]. Hydrogenation of o-nitroanisole to o-anisidine was investigated in a silicone microreactor showing that the highest conversion is achieved in the transitional flow regime, where competition between phases induces the most favorable gas–liquid mass transfer.
(9.31) 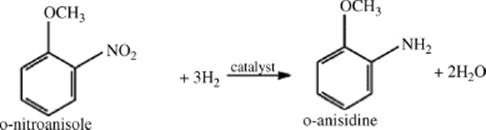
The reactor design was such that solid catalyst is suspended in the reaction channel by an arrangement of catalyst traps. The design enables use of commercial catalyst and allows control of pressure drop across the bed by adjusting the packing density. An optimized design of a microreactor may result in the possibility of microreactors as a beneficial alternative to conventional reactors in fine chemical and other industries.
9.4.1.5 Selective Hydrogenation of Acetylene Alcohols
The selective hydrogenation of acetylene alcohols is an important step in the synthesis of fine chemicals, especially vitamins A and E [153, 155].
Continuous flow capillary microreactors with embedded monometallic (Pd) or bimetallic (Pd25Zn75) catalysts have been tested in the selective hydrogenation of alkyne reagents, among which was 2-methyl-3-butyne-2-ol [155]. Under conventional reaction conditions a number of side products can be formed.
Under nitrogen atmosphere, oxidative dimerization dominates, and 2-methyl-3-butyn-2-ol (MBY) is converted over a Pd catalyst into 2,7-dimethyl-3,5-octadiyne-2,7-diol (1) which can further be hydrogenated to 2,7-dimethyl-5-octen-3-yne-2,7-diol (2).
(9.32) 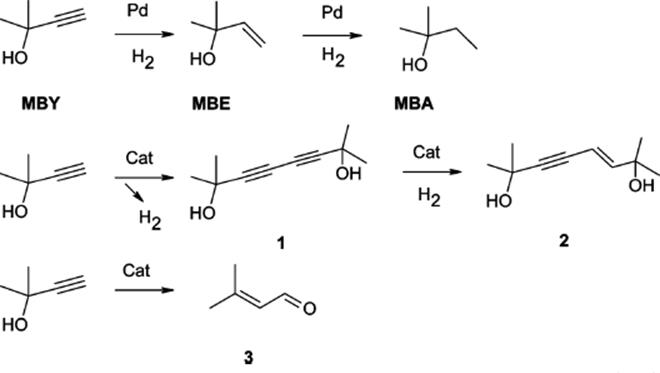
The highest selectivity toward the alkene product of 90% was obtained at 99.9% conversion on the Pd25Zn75/TiO2 catalyst. The selectivity was further increased to 97% by addition of pyridine into the reactant solution. The challenge here is to find an optimum Pd/Zn ratio, which would still provide a high activity of the Pd catalyst while maintaining a high selectivity observed on the Pd25Zn75 catalyst. No deactivation of the wall-coated catalysts was observed during one month or continuous operation at 333 K with pure hydrogen.
Further, selective Pd-catalyzed hydrogenation of 3-methyl-1- pentyn-3-ol to P1 without over-hydrogenation to P2 was used to prove the advantageous characteristics of segmented flow in eliminating diffusional effects and axial dispersion.
(9.33) 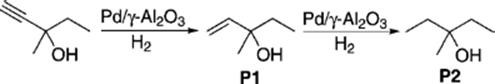
The theoretically maximum yield of P1 (78 ± 2)% was easily obtained. Without H2 bubbles, the yield of P1 would have been about 57% [153].
9.4.1.6 Hydrogenation of α-Methylstyrene over Pd/C
The hydrogenation of α-methylstyrene is another standard process for studying mass-transfer effects in catalyst pellets and in fixed bed reactors since the intrinsic kinetics is known [156]. The reaction is relatively fast at room temperature and 1 atm hydrogen pressure.
(9.34) 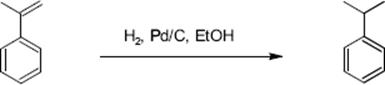
The hydrogenation of α-methylstyrene was investigated to demonstrate the performance of a mini packed bed microreactor with a palladium catalyst supported on activated carbon [156]. The microreactor was operated at 50 °C and conversions from 20% to 100% were measured. It was determined that the reaction is first order for hydrogen and zero order for α-methylstyrene. Initial reaction rates were close to 0.01 mol/min per reaction channel and were achieved without additional activation of the catalyst. This is in agreement with literature data on intrinsic kinetics.
Similar studies were made in a mesh microreactor comprising Pd/Al2O3 and Pt/Al2O3 catalysts coated on a microstructured mesh [58, 59]. The global rate constants were 56 and 1.4 1/s for the Pd and Pt catalysts, respectively [58]. An activation energy of 46 ± 5 kJ/mol was found for the Pt catalyst in the mesh reactor, which corresponds to the value of 39 kJ/mol determined for a commercial Pt/Al2O3 powder catalyst in a well-behaved batch reactor (Figure 9.37).
Figure 9.37 Kinetic study for the hydrogenation of a cinnamonic acid derivative: parity plot comparing experimental and modeling data. Open and closed circles correspond to data outside or within a match of experiments and modeling. Source: By courtesy of Elsevier [162].
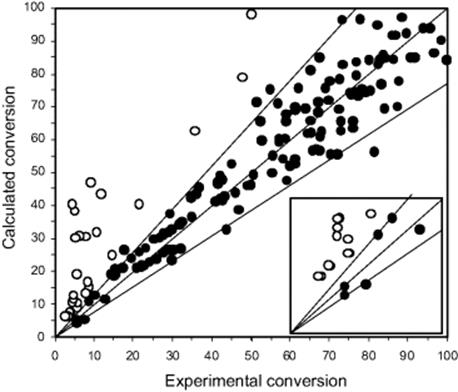
Accordingly, operation was in the chemical regime, that is, governed by kinetics, and not controlled by mass transfer [58]. For the Pd catalyst, however, the activation energies measured in the mesh reactor and in a well-behaved batch reactor with commercial Pd/Al2O3 powder catalyst were different, being about 0 and 41 kJ/mol, respectively. The activity of the Pd catalyst is much higher so that mass transfer limitations are given [58].
The hydrogenation of biphenyl over a Pt-Pd/Al2O3 catalyst was carried out in a three-phase plug-flow microreactor [157]. The reactor consisted of six parallel reactor tubes with an inner diameter of 2.2 mm and a catalyst-bed of maximum 200 mm long.
Biphenyl hydrogenation served as a model reaction for developing a fixed bed test to evaluate long-term performance of powder catalysts, such that it gives the reliable rates that are, for the fresh catalyst, the same as those obtained in an autoclave over the whole range of conversion. Gas–liquid flow distribution over a packed bed needs more care than running only a liquid or a gas. Hydrodynamic anomalies, stagnant zones of gas and liquid where diluent and catalyst were unevenly distributed, were observed. This causes low reproducibility of such reactor design. An optimization and careful control of reactor parameters was needed to get the same results in each reactor tube. However, authors advice to use one of the reactor tubes with a reference catalyst for checking reproducibility.
9.4.2 Oxidations
Liquid phase oxidation reactions in flow are described in Section 7.5.2. It is much more difficult to control gas–liquid flow while performing fast and exothermic reactions such as oxidations. A microstructured reactor used for the investigation of the oxidation of cyclohexane under pure oxygen at elevated pressure and temperature was investigated [157]. The reaction is performed at high temperature (>200 °C) and pressure (up to 25 bar) using pure oxygen in a Pyrex capped silicon etched microreactor, which allows reaction conditions well above the flammability limit. The test reaction was the oxidation of cyclohexane (CyH) into a mixture of cyclohexanone (K), cyclohexanol (A), and cyclohexylhydroperoxide (CHHP).
(9.35) 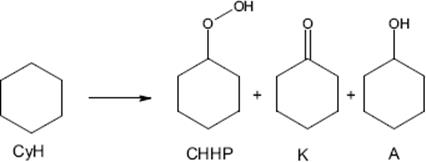
In industry, this reaction is conducted at elevated pressure (10–30 bar) and temperature (80–250 °C). Under such conditions, the content of oxygen must be kept low to avoid the explosive regime. Also, the cyclohexane conversion is kept low (<5%) to avoid the formation of side products. In a microreactor, composition and temperature of the gas phase far above the flammability limit of 4% are possible. A selectivity of 88% was observed. However, it is shown that despite the narrow channels, gas–liquid mass transfer still limits the rate of reaction so that further engineering of a reactor is required that will enable mass transfer free operations and oxygen refilling.
The heterogeneously catalyzed selective gas phase oxidation of o-xylene in a micropacked bed reactor was investigated, using the benefits of explosive regime [158].
The reaction was carried out with high amounts of o-xylene in air and stoichiometric with oxygen using a V2O5/TiO2-catalyst prepared through grafting. There were no significant losses in the selectivity to phthalic anhydride. The space-time yield was up to 2.3 times higher in comparison to conventional reaction conditions.
The work has shown that a strong exothermic reaction such as the o-xylene oxidation to phthalic anhydride can be operated in the explosion regime using a micropacked bed reactor, even with high adiabatic temperature rise of several 1000 K. An increase of the selectivity to total oxidation products was observed at higher o-xylene concentrations between 10 and 25 vol% o-xylene, which possibly was caused by the formation of a hotspot.
Catalyst deactivation was also studied. It was shown that crystalline vanadium oxide was formed due to phase transition of anatase into rutil [158].
An upscaling of the reactor to industrial dimensions is influenced by the particle size. Since reducing the particle size could not be an option with respect to pressure drop at high flow rates, applying catalyst coatings to the reactor walls is a possible alternative for industrial applications.
9.4.2.1 Oxidation of Alcohols
Oxidation of benzyl alcohol in the absence of solvent on 1% (Au–Pd)/TiO2 catalyst with pure oxygen was performed in silicon-glass micropacked bed reactors [159].
A pillar structure of small rectangular posts was incorporated near the outlet of the reaction channel to retain the catalyst. The reaction was studied in the temperature range of 80–120 °C and at inlet pressures up to 5 bar. Benzyl alcohol conversion and benzaldehyde selectivity at 80 and 120 °C were very close to those from conventional glass stirred reactors. The best conversion of benzyl alcohol of 95.5% with selectivity to benzaldehyde of 78% was obtained for a micropacked bed reactors with catalyst sizes of 53–63 μm and a catalyst bed length of 48 mm at 120 °C and 5 bar. The effect of catalyst particle size on the reaction was examined with two ranges of particle size: 53–63 μm and 90–125 μm. Lower conversion was obtained with particle sizes of 90–125 μm, indicating the presence of internal mass transfer resistances. In situ Raman measurements were performed and could be used to obtain the benzaldehyde concentration profile along the catalyst bed.
Photocatalytic oxidation of p-chlorophenol and toluene under gas–liquid–solid multiphase flow conditions was investigated using a photocatalytic microreaction system [160]. By loading both gaseous and liquid samples simultaneously into a microchannel with a photocatalytic titanium dioxide thin layer therein, a gas–liquid–solid multiphase annular flow was generated. The reaction yield was greatly enhanced with decreasing thickness of liquid layer because of improved efficiency of interaction and mass transfer between different phases.
9.4.2.2 Oxidation of Sugars
An efficient oxidation of glucose to gluconic acid was performed using a porous gold catalyst in a low-cost microreactor designed from Pyrex glass capillary tubing [161]. A reaction yield of 99% was achieved in the capillary-microreactor, whereas the conventional method yielded only 48% of gluconic acid. In the capillary microreactor, the higher surface area of the porous sponge catalyst is responsible for a high reaction yield.
(9.36) 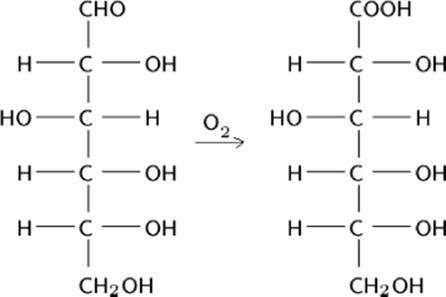
Moreover, no isomerization of glucose to fructose was observed during the reaction and total selectivity to d-gluconic acid was obtained. The low cost of fabrication, disposable nature of the capillary tube, and availability of a wide range of porous catalysts make this reactor versatile and cost effective.
Gas–liquid–solid oxidation of glucose was also studied as a model reaction to show the methodology for the ex-ante evaluation of different processing options [95]. Possible improvements using microreactor technology are evaluated.