Microreactors in Organic Chemistry and Catalysis, Second Edition (2013)
10. Bioorganic and Biocatalytic Reactions
10.4. Multienzyme Catalysis in Microreactors
Microreactors are ideal for directing complex enzymatic synthesis, such as multienzyme catalysis and cascade reactions. There have been a growing number of studies [173–178] that represent the implementation of microreactions for multistep enzymatic catalysis and a list is presented in Table 10.5. Microfluidic biocatalytic systems are particularly of interest for screening purposes since such devices allow the use of minute amount of enzymes and can easily be adapted for high-throughput analysis. A multistep enzymatic approach allows tight control of the reaction conditions and enables easy fine-tuning to get a better understanding of the role of a single enzyme within a multienzyme reaction. In addition, linking multiple enzymatic synthetic reactions also allows the production of more valuable precursor molecules, particularly, biologically active molecules for pharmaceutical purposes. In these systems, a significant consideration for enzymatic reactions has been the need for an online detection unit, which decreases processing time for high-throughput screening experiments and at the same time necessitates compatibility with the enzymatic reactions. The application of microreactors in multistep enzymatic catalysis with immobilized enzymes has been mainly focused on well-defined enzymatic conversions [123, 173–176, 178]. However, multienzyme or cascade reactions in microreactors systems are also of crucially important as an approach to performing in vitro metabolic pathway manipulation for complex bioconversions. Recently, Baganz's group reported a continuous flow immobilized enzyme microreactor platform for the evaluation of a de novo transketolase (TK)/ω-transaminase (ω-TAm)-linked asymmetric amino alcohol synthesis. The dual enzyme microreactor was based on the reversible attachment of His6-tagged enzymes via Ni-NTA linkage to two surface derivatized capillaries connected in series as illustrated in Scheme 10.16. The linked transketolase-ω-transaminase bioconversion, which involves TA reaction followed by the ω-Tam-catalyzed amination of l-erythrulose to synthesize 2-amino-1,3,4-butanetriol (ABT) is demonstrated in Scheme 10.17. The multienzyme platform based on microreactor system offers a useful approach for the screening of enzyme pairs as a tool for in vitro evaluation of multienzyme pathways toward one-pot synthesis.
Scheme 10.16 Schematic diagram of continuous flow dual reaction kinetics. The dual reaction system consists of two nickel-derivatized silica microreactors connected in series as described in Section 2.3.4. His6-enzymes transketolase and transaminase were immobilized inside each of the microreactors separately (Section 2.3.2). The proposed reaction scheme involves loading substrates for both enzymes (HPA, GA, (S)-MBA) to the first microreactor and feed these to the second containing His6-immobilized -TAm in continuous flow mode [177]. Source: Copyright 2011 Elsevier.
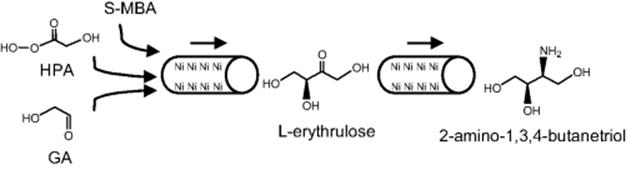
Scheme 10.17 Reaction scheme for the synthesis of 2-amino-1,3,4-butanetriol (ABT) by the TK and -TAm enzyme pathway. Dual enzyme pathway showing the TK-catalyzed production of l-erythrulose followed by the -TAm-catalyzed production of ABT from the substrates l-erythrulose and (S)-MBA [177]. Source: Copyright 2011 Elsevier.
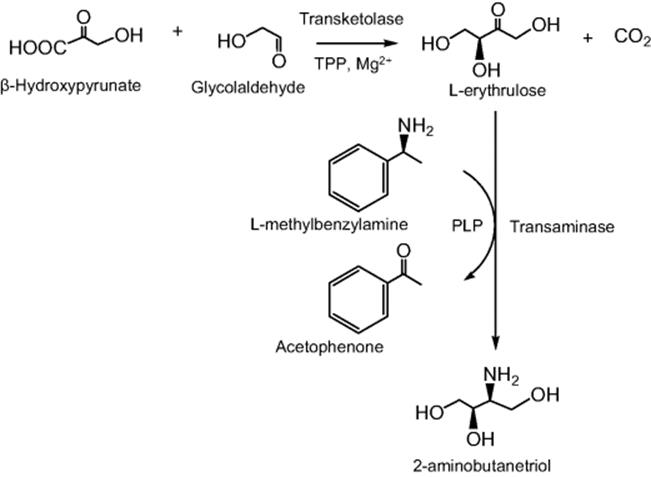
Table 10.5 Immobilized enzyme microreactors for multienzyme catalysis.