Why Is Milk White?: & 200 Other Curious Chemistry Questions (2013)
8. Chemistry in the World
PROJECT: EXTRACTING IODINE
Adult
supervision
required
Materials
Test tube with stopper or similar glass container, such as a small glass jar with lid
Tincture of iodine (available at drugstores and pharmacies)
Water
Toluene solvent (available at hardware stores) or naphtha cigarette lighter fluid (available at drugstores)
Eyedropper or pipette
Glass jar with lid
Hair dryer with no-heat setting
One of the projects coming later uses crystals of the element iodine.
For someone like me, finding iodine crystals is as simple as reaching up to the top shelf in the lab. But for most of the readers of this book, the pure element is not so easy to come by.
However, there is a tiny bit of iodine in that bottle of tincture of iodine you can find at the pharmacy. There is about half a gram in each fluid ounce of the disinfectant.
Extracting the pure element is simple and demonstrates how clever chemists can be using simple supplies and materials. The principle is called partitioning, or sometimes liquid-liquid extraction. It takes advantage of the fact that some things dissolve more easily in one liquid than another.

The liquid in tincture of iodine is alcohol. Iodine does not dissolve well in alcohol, and dissolves even less well in water. To get the elemental iodine to dissolve in either of these solvents, a little potassium iodide is added. The iodide ion helps more of the pure iodine dissolve.

Pour a small amount of the tincture of iodine into a test tube or a similar glass container with a stopper or lid, so it’s no more than about one-sixth of the way full. (Do not use a plastic container, because the toluene we’ll be adding in the next step dissolves plastic.) Then add an equal amount of water and an equal amount of toluene (available in stores either as a pure solvent or as naphtha cigarette lighter fluid). The water dilutes the alcohol and makes the iodine less soluble in the mixture. The toluene will float to the top, since it does not mix well with either alcohol or water.

Iodine dissolves easily in toluene. In the photo on the previous page you can see the bottom layer of 50 percent water and 50 percent tincture of iodine, and above it the layer of toluene, which has already started to get pinkish-violet from the iodine it has absorbed.
The next step is to stopper the test tube (or close the glass jar, etc.) and shake well for 15 to 20 seconds. The two layers will mix, and there will be some froth at the top, but when you stop shaking, the two layers will fairly quickly separate again. After 30 seconds of sitting still, the top layer of toluene can be seen stained a dark purple by the iodine it has extracted from the tincture, as in the photo above.

The next step is to use an eyedropper or a pipette to carefully remove as much of the top layer of dark purple toluene as you can into a glass jar, without getting any of the alcohol and water mixture in with it. There will be a little toluene left in the test tube, but that is OK.
Now you have a jar with dark purple toluene in it. You need to evaporate the toluene while losing as little of the iodine as possible. Iodine slowly vaporizes at room temperature, but if you give the toluene a lot of flowing air, it will evaporate faster than the iodine does. I used a blow dryer that had a setting that allowed the air to flow without any heat (heat would evaporate the iodine quickly). In a few minutes, the toluene had evaporated, and I had pure iodine crystals in the bottom of the jar.

Save the crystals by tightly closing the jar. You will need them for a later project, “Latent Fingerprints” (page 202).
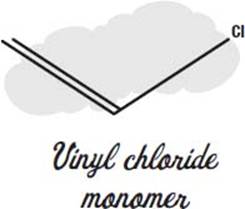
Plastic plumbing pipes are made from monomers of vinyl chloride, joined together to make polyvinyl chloride plastic.
The carbon atoms join together into long chains in which every second carbon has a hydrogen and a chlorine atom instead of two hydrogens.
Another common plastic monomer is vinyl acetate.
Polyvinyl acetate, made from this monomer, is familiar as white school glue. It forms a flexible, translucent plastic that can be heated to change its shape.
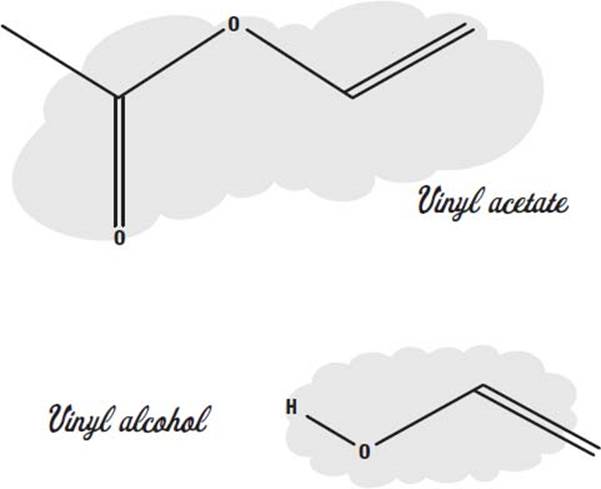
Between 100 and 5,000 vinyl acetate molecules join up to form the long chains in the polymer.
A related polymer is polyvinyl alcohol. It is made of monomers of vinyl alcohol. This plastic is often combined with other monomers to make what are called copolymers. Changing the type of copolymer or the proportion of the two monomers can change the nature and properties of the plastic produced.