Why Is Milk White?: & 200 Other Curious Chemistry Questions (2013)
3. Household Chemistry
Why do we use chemicals as cleaning supplies?
You have seen how chemists can make better cleaning molecules than soap by changing part of the molecule so that it can dissolve in hard water. But there are other chemicals in cleaning products that can help in other ways.
Bleach is used to remove stains and kill germs. Bleach releases oxygen, which reacts with the stain or the germ. In effect, it slowly burns them. One kind of bleach is hydrogen peroxide, which can bleach clothes and disinfect cuts and scrapes but is also used to bleach hair.
Phosphoric acid is added to some cleaning products to dissolve hard-water films on glass and metal surfaces. It removes lime scale—the chalky substance left behind on coffeepots as hard water is boiled—by dissolving the calcium and magnesium compounds that form it. Phosphoric acid is a strong acid, but in dilute form it is added to cola drinks to give them a tart taste. Acids taste sour.
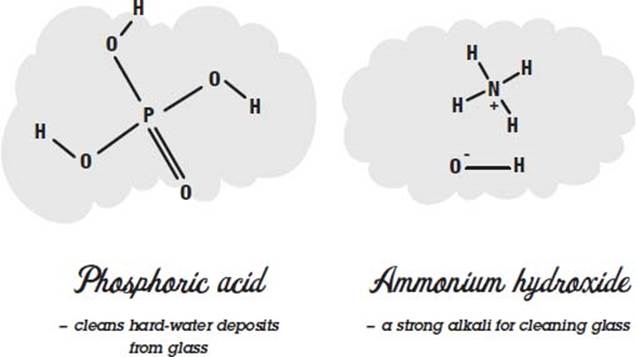
Ammonium hydroxide is a strong alkali that reacts with fats and oils in much the same way lye does, forming water-soluble soap. It is used in glass cleaning products to remove grease and oils from windows.
Alcohols dissolve grease and fats, and one alcohol, isopropanol, is used to remove grease from skin and to disinfect the skin (it is sometimes called rubbing alcohol). It is added to window cleaners to help remove oils and grease.