Organic Chemistry I For Dummies, 2nd Edition (2014)
Part VI. Appendixes
IN THIS PART …
See how to work multistep synthesis problems.
Recognize how to use arrows to show the mechanisms of organic reactions.
Get the definitions of chemical jargon and organic-speak that you should know.
Appendix A. Working Multistep Synthesis Problems
IN THIS APPENDIX
Understanding multistep synthesis problems
Discovering tips for working multistep synthesis problems
Multistep synthesis problems are prevalent throughout organic chemistry — and they tend to show up more and more frequently as the course goes on. Many students cite these problems, along with mechanism problems, as the most difficult types in the course. This appendix details why people bother with multistep synthesis and gives several commandments for how to work these kinds of problems so that you can avoid committing the major multistep synthesis sins.
Why Multistep Synthesis?
A good illustration of the importance of organic synthesis is drug making. Throughout history, drugs have primarily been obtained from natural sources. The kings of Mesopotamia smoked poppy flowers to get high on the hallucinogenic properties of the opium alkaloids, Native Americans chewed aspirin-containing willow bark for pain relief, and Fleming discovered penicillin in a mold that grew on his Staphylococcus bacteria plates. Even today, natural product chemists extract compounds from living things, characterize them, and test them to see if those compounds have biological activity.
But extracting drugs from natural sources has its problems. Oftentimes, the drug comes from a source that’s rare (like an exotic plant or fungus), and sometimes from a source that’s endangered or near extinction. The extraction of these compounds can also be challenging, because the active compound must be separated from the mass of inactive molecules; this can be a difficult task in some cases.
Sometimes the source doesn’t produce the drug in high quantities, and starting with thousands of pounds of a source material and extracting just a few paltry milligrams of the desired compound from it is not uncommon. Nature may be a far better synthesizer of organic molecules than humans can ever dream of becoming, but that doesn’t mean that nature produces the needed quantities of material on demand. Taking thousands of pounds of a species on the verge of extinction to make just a few small doses of a drug is usually impractical.
So, how can chemists get the desired compounds? By multistep synthesis. This process involves making the compounds from scratch. To do so, chemists take commercially available starting materials and build up the desired molecule using available reagents and techniques. A great many of the drugs that you can buy from pharmacies today were made by multistep syntheses, using enormous quantities of starting materials and reagents that were added with front-end loaders into gigantic reaction vessels. But all that was first planned out on paper by chemists, plotting the shortest, cheapest, and most elegant route available to the target compound (with the shortest and cheapest routes taking precedence in the pharmaceutical industry, and elegance being prized in academia).
Of course, that doesn’t mean that the application of multistep synthesis is limited to drugs. Multistep synthesis is used for many different purposes, and one advantage of the organic chemist’s ability to make compounds is the free reign this ability gives him or her to do pretty much anything in chemistry (which is why biochemists and physical chemists are forced to collaborate with synthetic organic chemists if they want to get anything done). In some cases, multistep synthesis is even carried out for the sake of synthesis itself. In these cases, organic chemists synthesize structurally interesting natural products that have grant proposal activity — that is, products that may show enough biological activity to fund a grant proposal, but likely do not have enough potential to be considered for clinical trials. In these cases, multistep synthesis is used as a framework to develop new reactions, as a way of testing known reaction methods, and more generally as a stomping ground for chemists to flaunt their synthesizing prowess.
And although synthetic chemists do their best to plan the syntheses of complicated molecules, steps invariably go wrong when trying them out in the laboratory. This failure is sometimes a good thing, however. When planned reactions in a synthesis don’t work, organic chemists need to figure out new ways of doing things to get around the barriers that these molecules put up. Often, new reactions and insights are discovered in this way.
 For example, the synthesis of vitamin B12 (discussed in more detail in Chapter 21) was carried out knowing ahead of time that any synthesis of this extremely complex molecule would be too expensive to perform on a commercial scale. Still, the synthesis of this molecule was extremely valuable to organic chemists. Many new synthetic techniques were discovered during this synthesis, in addition to one major theoretical breakthrough (understanding pericyclic reactions like the Diels–Alder reaction) that occurred while contemplating one of the steps in the reaction. So, although this synthesis of vitamin B12 was too impractical to carry out on a commercial scale, the total synthesis of this molecule added a great deal of knowledge to the field of organic chemistry.
For example, the synthesis of vitamin B12 (discussed in more detail in Chapter 21) was carried out knowing ahead of time that any synthesis of this extremely complex molecule would be too expensive to perform on a commercial scale. Still, the synthesis of this molecule was extremely valuable to organic chemists. Many new synthetic techniques were discovered during this synthesis, in addition to one major theoretical breakthrough (understanding pericyclic reactions like the Diels–Alder reaction) that occurred while contemplating one of the steps in the reaction. So, although this synthesis of vitamin B12 was too impractical to carry out on a commercial scale, the total synthesis of this molecule added a great deal of knowledge to the field of organic chemistry.
The Five Commandments
Multistep syntheses are the kind of problems in which you’re told what the starting material and the product are, and you’re asked to figure out a synthetic route that’ll convert the starting material into that product. For these types of problems, you must provide reagents to convert the starting materials into one or more intermediate compounds that you’ll then further react in order to make the final product. Often, there will be several steps in the synthesis. However, no mechanisms (arrow pushing) need to be shown for the individual reactions in a multistep synthesis problem. This is a common mistake. For these problems, simply provide the reagents and the products of each step as you go along.
For example, suppose the question asked how to convert Compound W into Compound Z (see Figure A-1) using multistep synthesis.
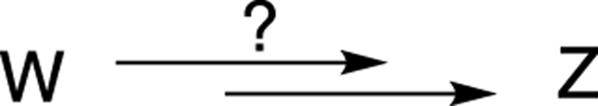
FIGURE A-1: Multistep synthesis.
Here, an acceptable answer would show the individual reactions that would transform Compound W into Compound Z (an example is given in Figure A-2). Note that no mechanisms for these reactions are shown, just the reagents and the intermediates along the way. In many cases, more than one route is possible for the conversion of the starting material into the product, so these types of problems often have several acceptable answers (the shortest route is often preferable, however).
![]()
FIGURE A-2: Answering multistep synthesis questions.
Here are five commandments to help you solve these synthesis problems.
Commandment 1: Thou shalt learn thy reactions
Learning the reactions is the most basic requirement for multistep synthesis. No matter how smart you are, you don’t stand a chance of getting synthesis questions right unless you know the reactions. Memorize the reagents, use flash cards, or use whatever other techniques you find most helpful, but learn the reactions cold. This memorization is not something that you can do overnight. Learning the reactions takes time. A good technique is to add reagent learning as a portion of your daily study time.
Because organic chemistry is a cumulative course, you can’t afford to forget any reactions that have been covered in previous chapters. If you use flashcards, you should never throw away your stack of old flash cards that contain the reactions from previous chapters. Instead, keep adding to your pile. (Yes, the deck will be thick enough to choke a small whale by the end of the course!) Most textbooks have convenient end-of-chapter reaction summaries that give an overview of all the reactions that were covered in the chapter, and these overviews can be extremely helpful in making flash cards.
Commandment 2: Thou shalt compare carbon skeletons
The first thing to look for in a multistep synthesis is to compare the carbon skeleton of the starting material to the carbon skeleton of the product. Were any carbons lost or added? If so, can you identify where they were added or lost? A count of the number of carbons in the reactant and the product doesn’t take long, but it can help you determine what kinds of reactions you’re dealing with.
Take the following simple example shown in Figure A-3. The lighter-shaded portion of the molecule identifies where the likely carbon skeleton of the reactant is found in the product. Looking at the molecules in this way allows you to clearly see what portion needs to be added or lost during your synthesis. (This step may seem trivial in this obvious example, but in tougher problems, this process can help you organize your thoughts.)
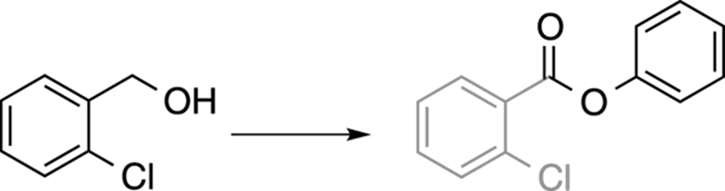
FIGURE A-3: Comparing carbon skeletons.
Commandment 3: Thou shalt work backward
Have you ever worked through mazes and found that you can more easily get through a maze by starting at the finish and working back to the start than you can by working from the start? The same thing applies to multistep synthesis problems. Working backward in synthesis problems is called retrosynthesis.
The first thing you want to look at is your product, thinking of all the reactions that you know of that could form it. For the moment, ignore your starting material. If your product is an alkene, for example, think of alkene-forming reactions like elimination reactions or the Wittig reaction. (See Chapter 10 for a summary of alkene-making reactions.) Write all of these reactions out and look at what reactant would be required for each.
After you’ve written out all the potential reaction candidates, return to your starting material. Which reaction has a reactant that most resembles your starting material? That one is probably the best one to select as a potential candidate.
For example, suppose you were asked to do the synthesis shown in Figure A-4.
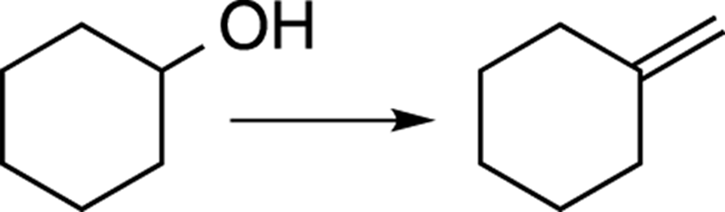
FIGURE A-4: A sample synthesis problem.
After completing the first two commandments, you would want to think of ways to make the alkene in the product. Ignore the starting material for the moment. Just brainstorm all the ways you can think of to make the alkene and write them down on your scratch paper. You should get something that looks like the following list (see Figure A-5).
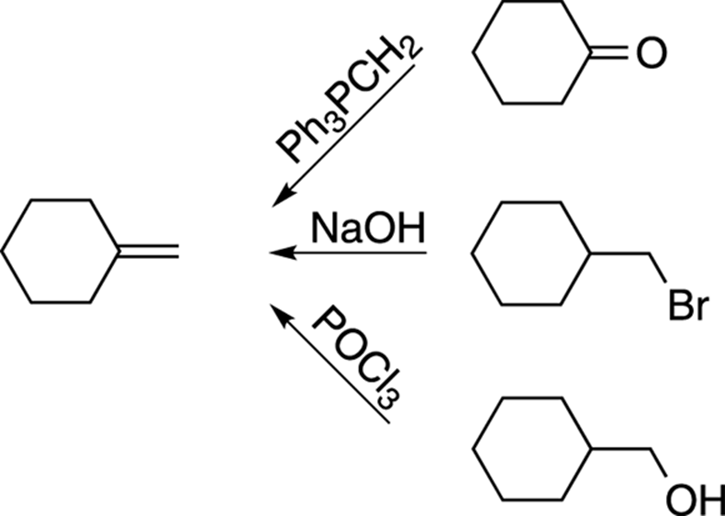
FIGURE A-5: Alkene-forming reactions.
Now you have three possible routes to choose from. The route to choose is the one that uses a reactant that most resembles the original starting material. If you did Step 2 (accounting for the carbon skeleton), you would know that the product has one carbon more than the starting material. Only the first reaction, the Wittig reaction, accounts for this additional carbon, so the Wittig reaction would be the reaction you would tentatively choose. If this choice turns out to be wrong, you can always go back and try another route.
Looking at the reaction scheme, you now have something that looks like Figure A-6.
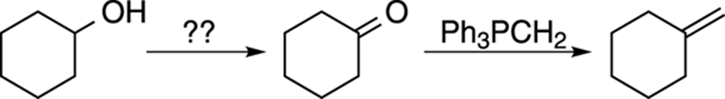
FIGURE A-6: Completing a retrosynthesis.
Now repeat the same procedure for cyclohexanone, thinking of all the different ways you could make this ketone. One pointer here is that the closer you get to completing a retrosynthesis — in other words, the closer you get back to your starting material — the more you should reference the starting material in your thinking. At this point, for example, you may want to modify your thinking from, “I need to think of all the ways I can make cyclohexanone,” to something more like, “I need a reaction that converts an alcohol to a ketone.” If you did Step 1, you would know several ways of producing a ketone from an alcohol (for example, using Jones’s reagent, PCC, and so forth). Choosing one of these methods would complete your synthesis.
If you get stuck, go back and try one of the other pathways. If the Wittig reaction in our example had lead to a dead end, you could’ve gone back and tried one of the elimination reactions. Choosing the correct way back is often a matter of intuition, and that only comes after working a lot of problems (see Commandment 5).
Commandment 4: Thou shalt check thyne answer
After you have a potential synthesis, go back and make sure that all your reagents are compatible with the functional groups on your molecule. Make sure, for example, if you’re proposing a Grignard reaction, that there are no alcohols or other incompatible functionalities on your reagent. Undergraduate organic professors often seem to take delight in creating challenging (tricky) exam questions, so double-check every detail of your synthesis for correctness.
Commandment 5: Thou shalt work many problems
This is the greatest commandment of all. As a mere mortal, you have no way around this; you have no magic formula at your disposal to avoid this commandment. A good textbook will have plenty of multistep synthesis problems to practice on. Start with easy synthesis problems to get the feel of what’s required, and then work your way to harder problems.
If you have a solutions manual to your text (something that I highly recommend you have), don’t refer to it until after you’ve completed the problem. Just as it would be unreasonable to expect to be able to play the piano after only listening to someone else play and without ever practicing yourself, you can’t expect to look at the study guide and be able to do synthesis problems on the exam without ever having practiced these problems by yourself. Looking at the solution manual and thinking, “Yeah, I could do this problem,” is no substitute for actually doing that problem. You need experience. Get lots of it!