Organic Chemistry: Concepts and Applications - Headley Allan D. 2020
Carbon Functional Groups and Organic Nomenclature
2.5 Structure and Nomenclature of Alkanes
Alkanes are the first set of organic compounds that will be examined and named using the IUPAC rules.
2.5.1 Nomenclature of Straight Chain Alkanes
Table 2.3 shows the basic vocabulary of the IUPAC nomenclature most commonly encountered in the naming of organic compounds. These names are often referred to as the root or parent names of organic compounds.
In naming organic compounds, the IUPAC name of the molecule is based of the number of carbons that are connected in a continuous chain. Of course, not all hydrocarbons have simple structures as those shown in Table 2.3.
Table 2.3 The IUPAC names of straight chain hydrocarbons.
Number of carbons |
Structure |
Name |
1 |
CH4 |
Methane |
2 |
CH3CH3 |
Ethane |
3 |
CH3CH2CH3 |
Propane |
4 |
CH3(CH2)2CH3 |
Butane |
5 |
CH3(CH2)3CH3 |
Pentane |
6 |
CH3(CH2)4CH3 |
Hexane |
7 |
CH3(CH2)5CH3 |
Heptane |
8 |
CH3(CH2)6CH3 |
Octane |
9 |
CH3(CH2)7CH3 |
Nonane |
10 |
CH3(CH2)8CH3 |
Decane |
2.5.2 Nomenclature of Branched Alkanes
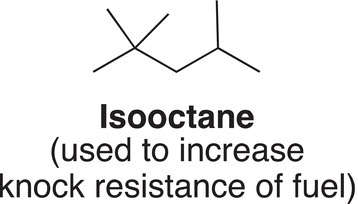
Most organic compounds encountered in organic chemistry have branches attached to a main continuous chain and they must have unique names that reflect their structures. For example, isooctane is a highly branched alkane, which is used in the petroleum industry as a fuel additive. The structure of this compound is shown below, but isooctane is not the IUPAC name, but a common name.
In naming organic molecules with branches, the following rules must be followed. The application of these rules to determine the IUPAC name of a compound is shown below.
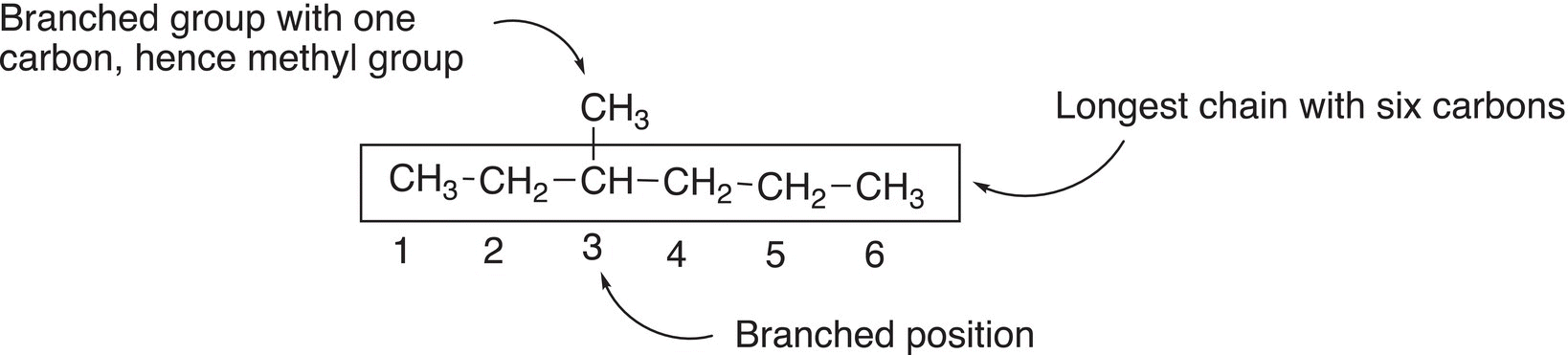
Rule #1 Find the longest continuous chain. Note that the longest continuous chain may not always be the horizontal chain of carbon atoms.
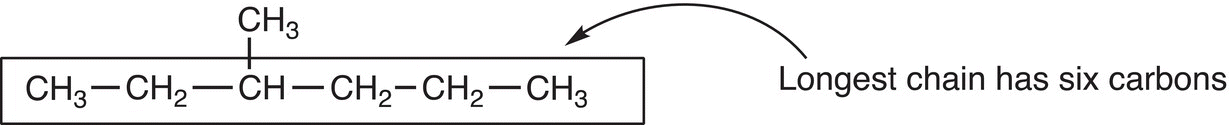
Rule #2 Based on the number of carbons in the longest chain, use Table 2.3 to determine the parent or root name. Based on the number of carbons that are present in the compound, a root or parent name is assigned such as shown below.

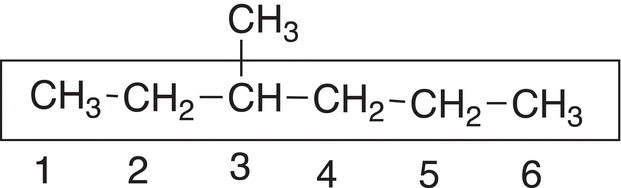
Rule #3 In order to determine the positions of branches, assign numbers to each carbon of that longest continuous chain. Start the assignment so that the end closest to a branch gets the number 1, as shown below.
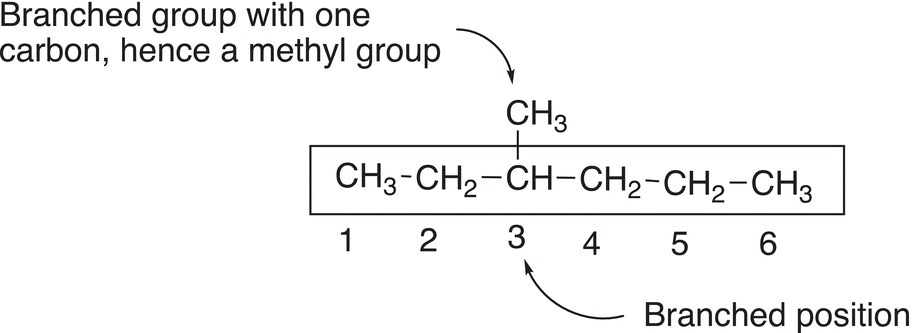
Rule #4 Identify the branched position by the number assigned in rule 3.
Rule #5 Name the branch based on the number of carbons of that branch. Determine the number of carbons present in the branch and use the names in Table 2.3 to derive a name. Derive the name of the branch by converting the corresponding alkane from Table 2.3 to alkyl. That is, if one carbon is present, methane becomes methyl; if two carbons are present, ethyl is used, etc.
Rule #6 Write the complete IUPAC name so that the root name is at the end, the name of the branch is next to the root name, and the number that indicates the position of the branch is next to the name of the branch and separated by a hyphen. Remember that the parent or root name is at the end of the IUPAC name as shown in the example below.
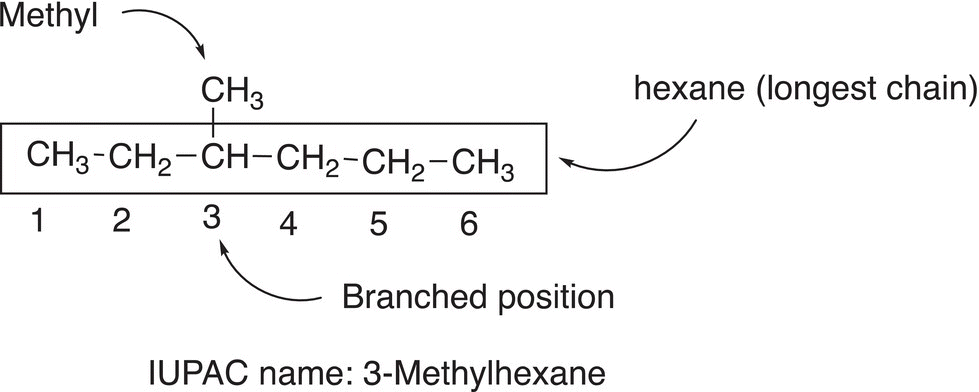
Note that the number is separated from a letter by a hyphen.
Problem 2.4
i. Give the IUPAC names for the following compounds.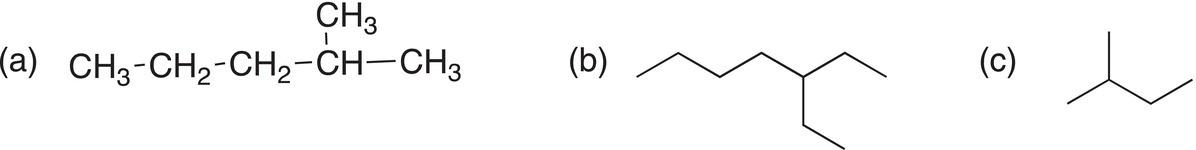
ii. Give the line-angle formula for the following compounds.
a) 3-Methylhexane |
b) 3-Ethylhexane |
c) 2-Methylheptane |
Rule #7 If there are two or more of the same branches that are present in a molecule, the prefix di, tri, tetra, etc., along with the corresponding numbers are used as shown in the example below.
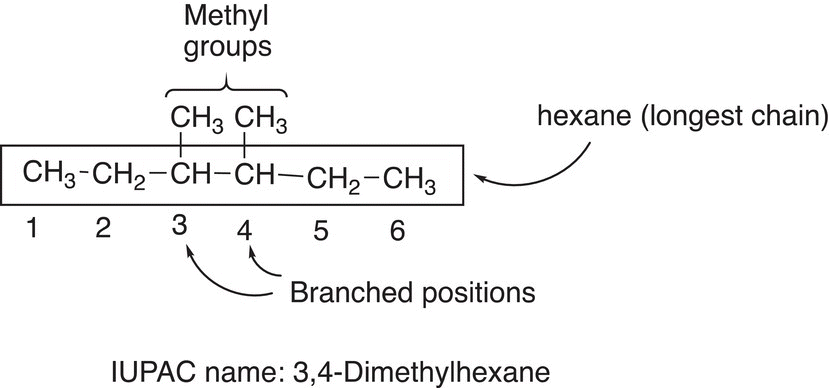
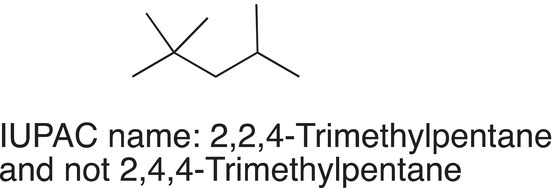
Note that a comma is used to separate numbers, and a hyphen is used to separate a number from a letter. If there is a choice, start at the end with the most branches so that numbers used in the prefix are the lowest combination, as shown in the example below.
Problem 2.5
i. Give the IUPAC names for the following compounds.
ii. Give the line-angle formula for the following compounds.
a) 2,3-Dimethylhexane |
b) 3,3-Dimethyloctane |
c) 2,2,4-Trimethylheptane |
If there are different types of alkyl branches, identify each branch by their location on the longest chain and list the branches in alphabetical order as part of the name as shown below.
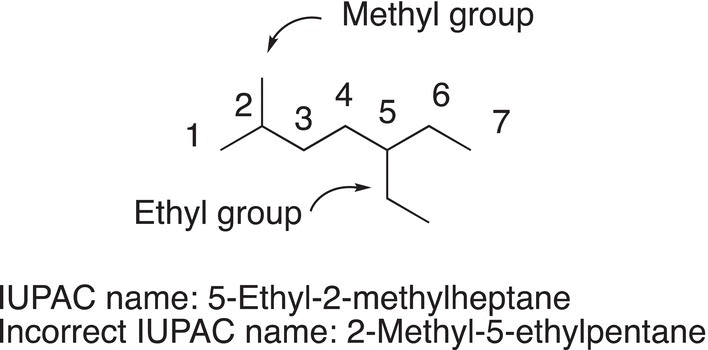
Note that the IUPAC name is 5-ethyl-2-methylheptane and not 2-methyl-5-ethylheptane. The groups bonded to the main longest chain must be listed in alphabetical order. As a result, ethyl, which starts with an “e” comes before methyl, which starts with “m”. It should be pointed out that the prefixes, di, tri, tetra, etc., that are used to describe the number of similar groups do not play a role in the alphabetical ordering of the names. That is, if there is a dimethyl in the name, the “m” of the methyl and not the “d” of the dimethyl is used to determine the alphabetical order. Similarly, for trimethyl and tetramethyl, the methyl will dictate which comes first in the nomenclature.
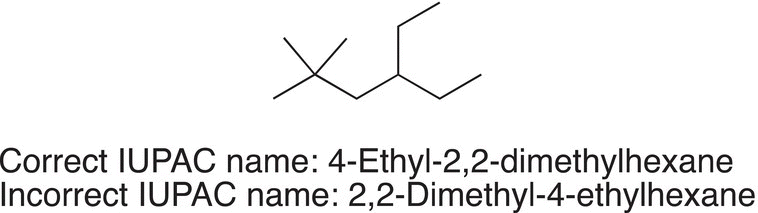
Problem 2.6
i. Give the IUPAC names for the following compounds.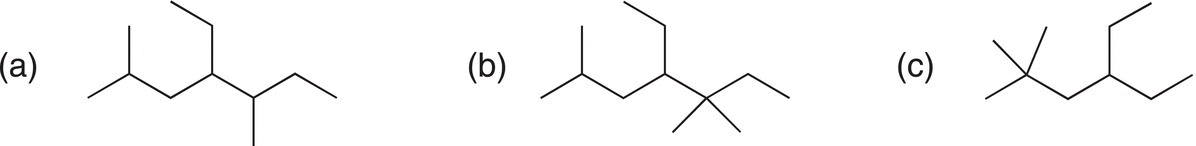
ii. Give the line-angle formula for the following compounds.
a) 3-Ethyl-2-methylheptane |
b) 4-Ethyl-2,2,3-trimethyloctane |
2.5.3 Nomenclature of Compounds that Contain Heteroatoms
As pointed out earlier, some organic compounds have heteroatoms; that is, some organic compounds have carbons, hydrogens, and other types of atoms, such as oxygen, nitrogen, or halogens present. In naming such compounds using the IUPAC naming system, rules 1 through 7 apply, except the heteroatomic substituent is named as shown in Table 2.4.
For example, if a compound has a chlorine atom bonded to the longest chain, chloro is used in the IUPAC name of such a compound, as shown in the example below.
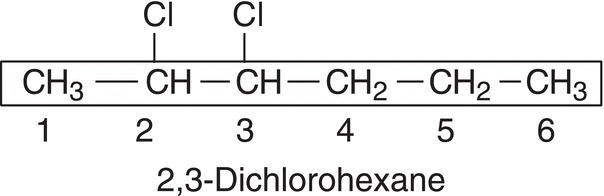
Table 2.4 IUPAC names for selected heteroatomic groups.
Group |
Name |
Cl |
Chloro |
Br |
Bromo |
I |
Iodo |
F |
Fluoro |
NO2 |
Nitro |
For compounds that have different substituents, including heteroatomic and alkyl substituents, the groups are listed in an alphabetical order as shown in the compound below.
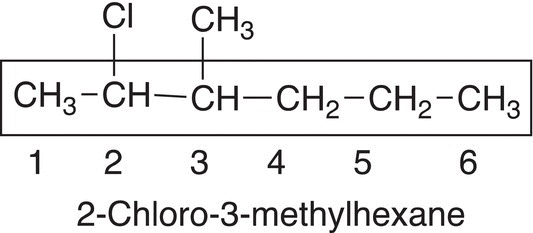
The longest chain contains six carbons and hence the root name is hexane; there are two different types of substituents, an alkyl substituent and a chloro substituent. Since chloro starts with the letter “c” and comes before the letter “m” for the methyl group in the alphabet, the chloro group is listed first for the IUPAC name. Note that numbers are separated from letters by a hyphen.
Problem 2.7
i. Give the IUPAC names for the following compounds.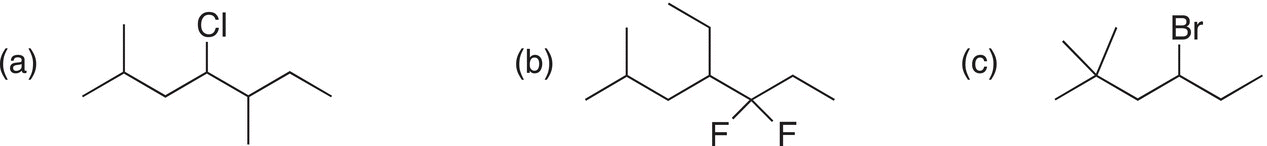
ii. Give the line-angle formulas for the following compounds.
a) 3-Chloro-2,2-dimethylpentane |
b) 2,2-Dibromo-4,4-dimethylheptane |
2.5.4 Common Names of Alkanes
Throughout this course, common names of some organic compounds are used and examples are shown in Figure 2.6. In this text, however, IUPAC names will be used routinely.
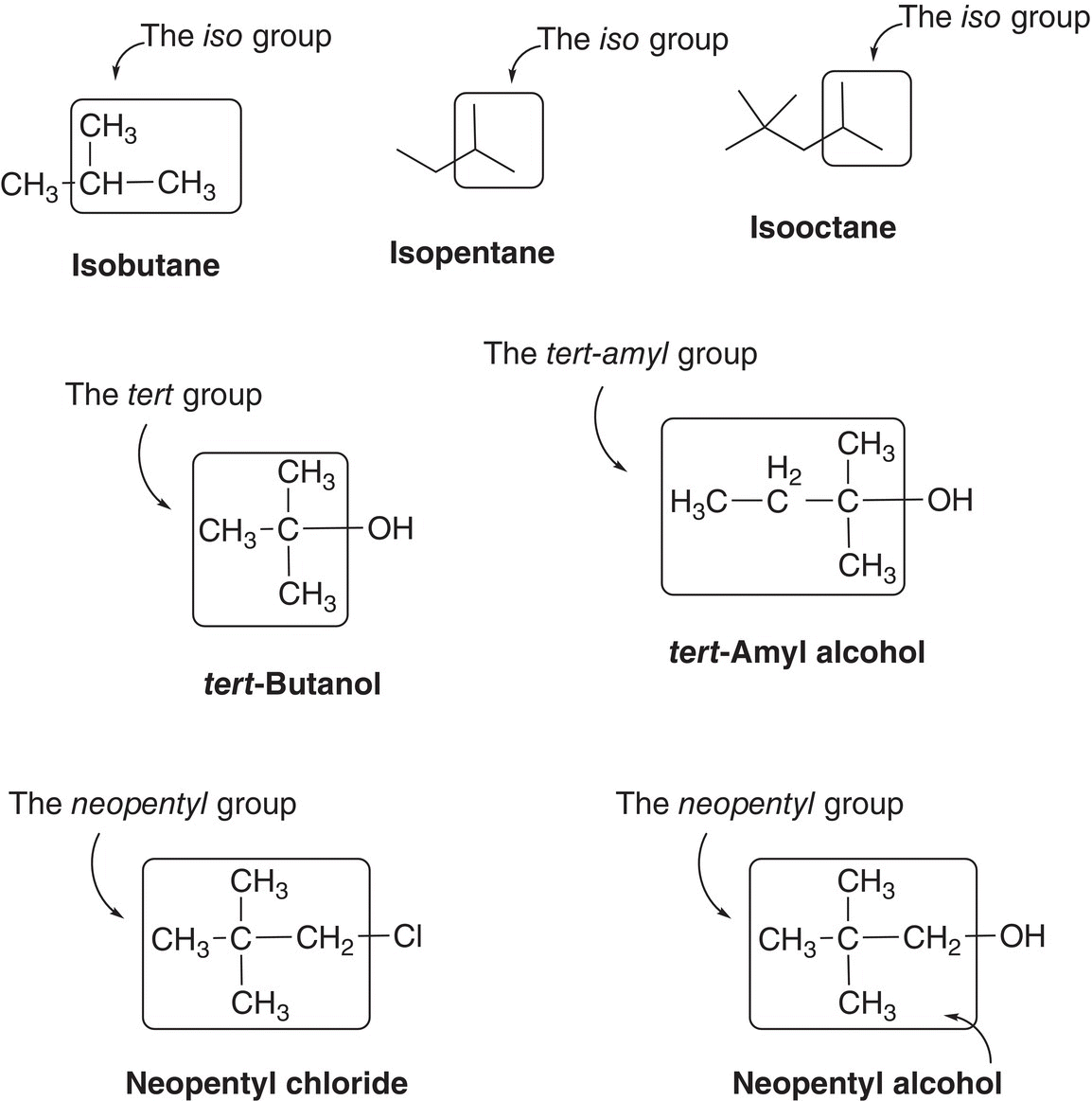
Figure 2.6 Examples of using common names for some common compounds.
2.5.5 Nomenclature of Cyclic Alkanes
For cyclic saturated hydrocarbons, Table 2.5 shows the names of the most common cyclic compounds that will be encountered throughout this course. As is obvious, there are no cyclic compounds for compounds that contain one or two carbons.
It is obvious that the prefix cyclo is added to the names of the straight chain alkane given in Table 2.3 to determine the names of cycloalkanes. Thus, if the cyclic molecule has six carbons, its root name is cyclohexane.
Problem 2.8
Give the molecular formulas for (a) cyclononane and (b) cyclodecane.
2.5.6 Nomenclature of Branched Cyclic Alkanes
Most cycloalkanes that will be encountered throughout this course contain the basic structural features as shown in Table 2.5, but they will have branches. In naming cyclic compounds, the root or parent name is that of the cyclic structure shown in Table 2.5, but the numbering system is a little different from that of a straight chain, the position that gets the number 1 is the carbon that has a branch.
Rule #8 In naming cyclic compounds, start numbering at the carbon that has a branch and continue in the direction that has the most branches. Below is an example of a compound with two numbering possibilities.
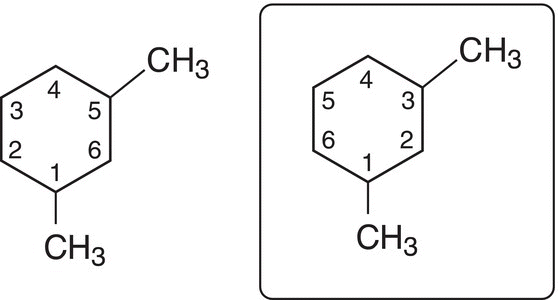
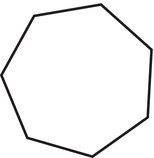
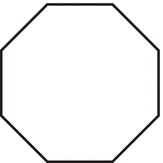
Table 2.5 The IUPAC names of cyclic hydrocarbons.
Number of carbons |
Structure |
Name |
3 |
|
Cyclopropane |
4 |
|
Cyclobutane |
5 |
|
Cyclopentane |
6 |
|
Cyclohexane |
7 |
|
Cycloheptane |
8 |
|
Cyclooctane |
Note that the numbering system used for the molecule on the right gives the branches on the ring the numbers 1 and 3, compared to the numbering system for the molecule on the left, which gives the numbers 1 and 5. For IUPAC names of cyclic molecules with branches, the lower number combination is always used. Thus, the correct IUPAC name of the molecule shown above is 1,3-dimethylcyclohexane.
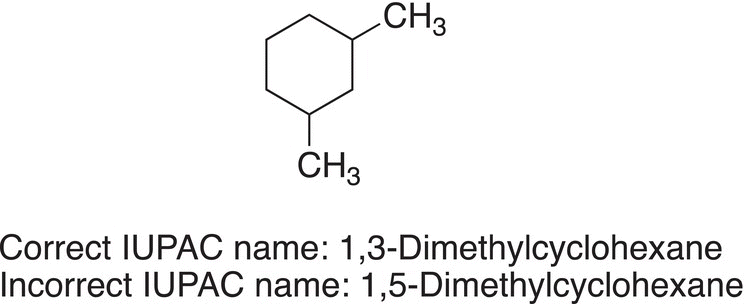
Rules 2 through 7 also apply in the naming of cyclic compounds that have more than two branches or heteroatomic substituents.
Problem 2.9
i. Give the IUPAC names for the compounds shown below.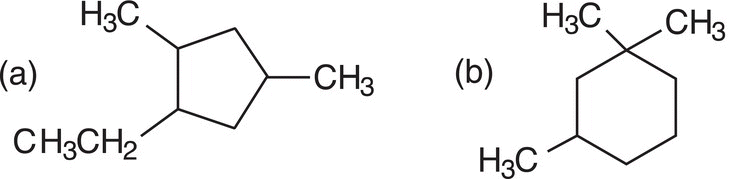
ii. Give the structures of the compounds shown below.
a) 1,1-Dichloro-2-methylcyclohexane |
b) 1-Chloro-3-methylcyclopentane |
2.5.7 Nomenclature of Bicyclic Compounds
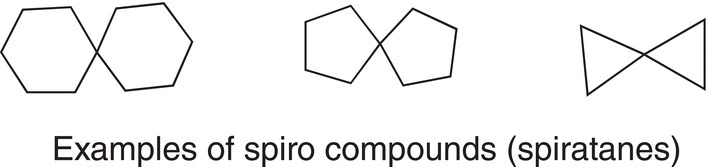
Some alkanes have two rings that are fused at different points. Compounds that have the rings bonded at the same carbon are the simplest and are called spiro compounds, spiranes, or spiratanes, which is derived from a Latin term, which means twisted or coiled. Examples of spiranes are shown below.
This next section concentrates on compounds in which two rings are bonded at different carbons and not at the same carbon as discussed for spiranes. Such compounds are called bicyclic compounds and examples are shown below.
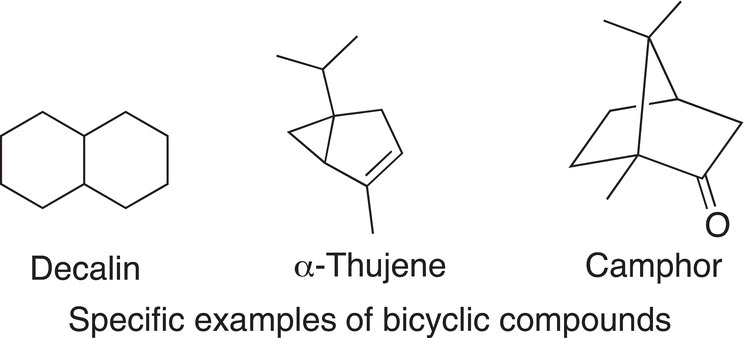
Decalin is a bicyclic compound, which is used as an industrial solvent in the synthesis of resin and also used as fuel additive, and this type of bicyclic framework is found in many important organic compounds. Another type of bicyclic compound frequently found in many natural products is shown in the structure of α-thujene, which is found in nature and is an essential oil of some plants. Camphor contains another type of bicyclic system, and it is frequently found in many natural products. Camphor has a distinct odor and is found in the wood of the camphor laurel and kapur trees, which are found primarily in different regions of Asia.
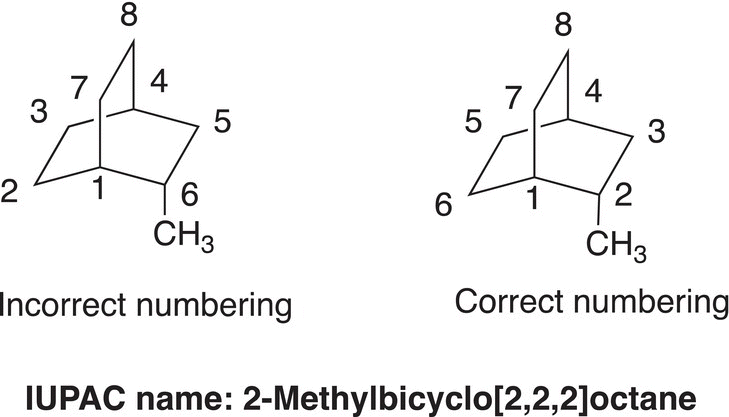
In naming bicyclic compounds, the total number of carbons must first be determined, which will become the root name of the compound. Next, the points where the rings are bonded must be identified (also known as the bridgehead carbons), as shown below.
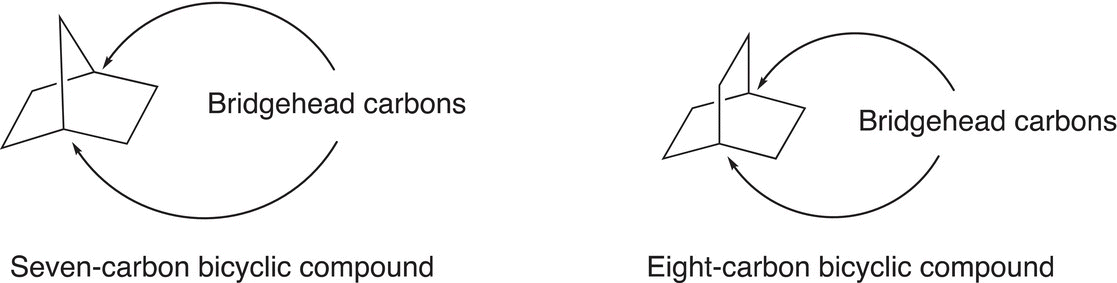
Problem 2.10
For the compounds shown below that are used in the treatment of different forms of cancer, circle the carbons of bicyclic systems.
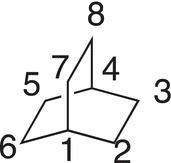
Once the points of fusion are identified, number the carbons starting at a point of fusion as shown below. Note that number 1 is a bridgehead carbon, and the numbering continues in the direction so that an entire ring is numbered. Then, include the carbons of the other ring, carbons 7 and 8. Also, note that only carbons 1 and 4 are the bridgehead carbons, and carbons 2 and 3; 5 and 6; and 7 and 8 are not.
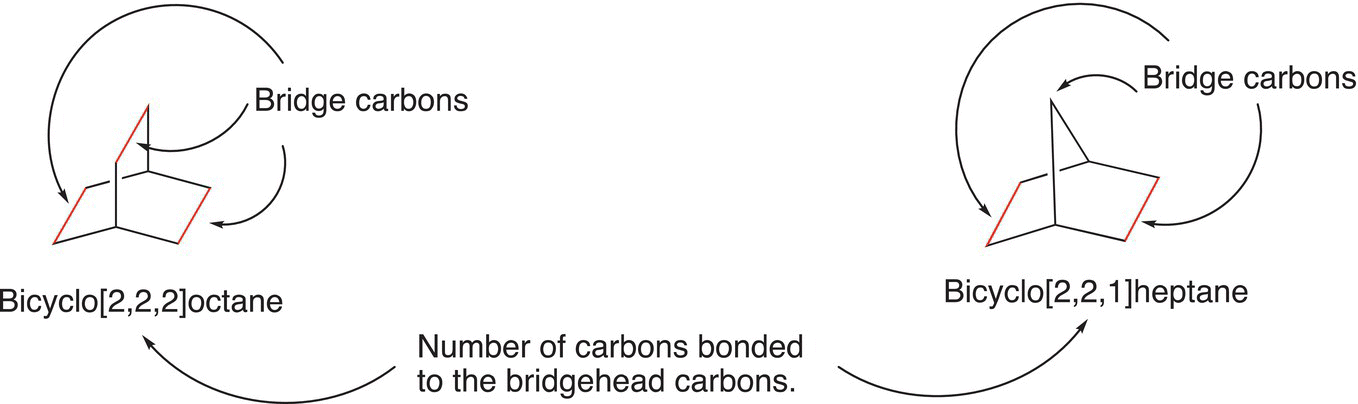
For the IUPAC name of the above molecule, the root is octane since there are eight carbons in the molecule. Next, the numbers of carbons that are not bridgehead carbons, but bonded to the bridgehead carbons, are indicated as shown in the name of the molecules below.
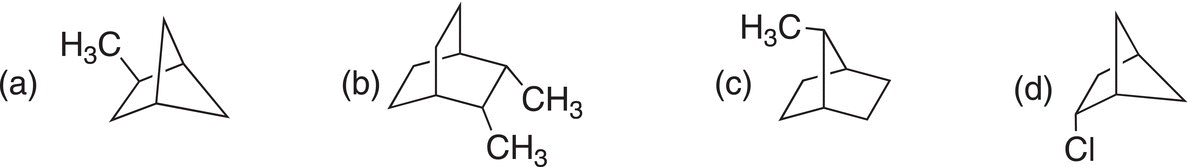
Problem 2.11
Give the IUPAC names for the bicyclic molecules shown below.
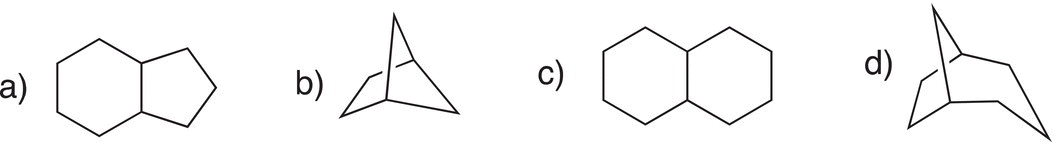
If there are substituents present, the molecule must be numbered so that the lowest number combination, which describes the positions of the substituents, is achieved. The starting point in numbering a bicyclic compound is always at a point of a bridgehead carbon. Shown below are some examples.
Problem 2.12
Give the IUPAC names for the bicyclic molecules shown below.