Organic Chemistry: Concepts and Applications - Headley Allan D. 2020
Synthetic Polymers and Biopolymers
20.8 Amino Acids, Monomers of Peptides and Proteins
Amino acids all have the same basic structural features, which are shown below.
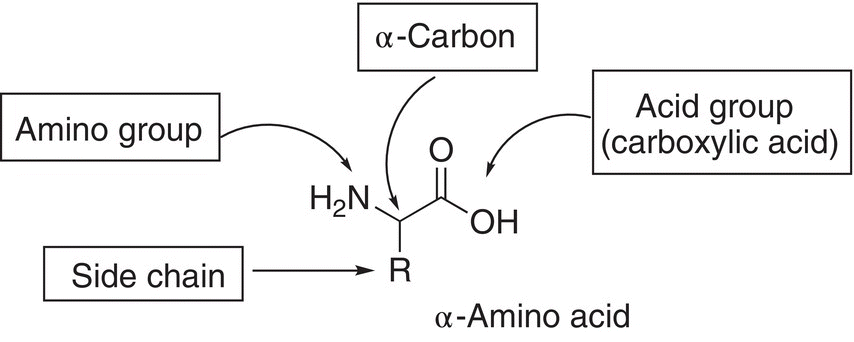
Amino acids with these features are also referred to as α-amino acids because on the α-carbon (the first carbon next to the acid functionality) is an amino functionality. There are 20 amino acids that occur naturally, and they can be classified based on the nature of the side chain. Table 20.2 shows the classification, the structure of the side chains, R and their abbreviations.
With the exception of glycine, all naturally occurring amino acids are optically active, that is, they will all rotate the plane of polarized light; they are also classified as L-configuration. The assignment of L and D for optically active amino acids is based on the system derived from the assignment of L and D for glyceraldehyde. The D and L means that the OH group that is present on the stereogenic carbon is on the right or left, respectively. The (+) and (−) means that these compounds rotate the plane of polarized light to the right or to the left, respectively. The terms dextrorotatory and levorotatory are also used to represent (+) and (−), respectively.
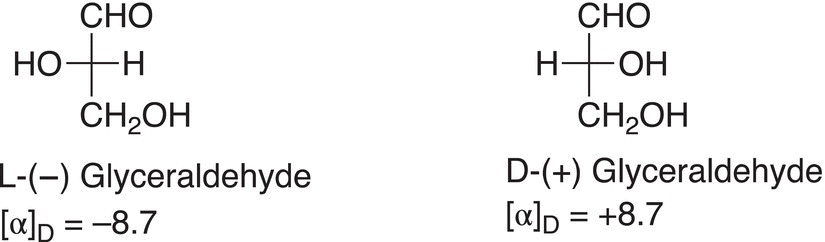
For amino acids, if the amino acid is drawn in the Fischer projection with the carboxylic acid group at the top and the R group is at the bottom, then the L and D assignment is based on the position of the amino group (NH2), similar to that of the —OH group in glyceraldehyde. If the amino group is to the left, then the assignment is the L; and if the amino group is to the right, the assignment is the D.
Table 20.2 Side-chain groups of amino acids that occur naturally.
Classification |
R |
Amino acid |
Abbreviations |
|
Alkyl side chain |
─H |
Glycine |
Gly |
─CH3 |
Alanine |
Ala |
|
─CH(CH3)2 |
Valine |
Val |
|
─CH2CH(CH3)CHs |
Leucine |
Leu |
|
─CH2CH2CH(CH3) |
Isoleucine |
Ile |
|
|
HO side chain |
─CH2CH(OH)CH3 |
Threonine |
Thr |
─CH2OH |
Serine |
Ser |
|
|
Sulfur side chain |
─CH2CH2SH |
Cysteine |
Cys |
─CH2CH2SCH3 |
Methionine |
Met |
|
|
Aromatic side chain |
p-HO-C6H4CH2 |
Tyrosine |
Tyr |
C6H5CH2 |
Phenylalanine |
Phe |
|
|
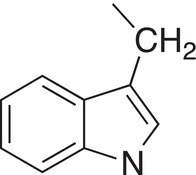
Tryptophan |
Trp |
|
|
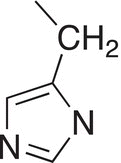
Histidine |
His |
|
|
Acidic side chain |
─CH2COOH |
Aspartic acid |
Asp |
─CH2CH2COOH |
Glutamic acid |
Glu |
|
|
Basic side chain |
CH2CONH2 |
Asparagine |
Asn |
CH2CH2CONH2 |
Glutamine |
Glu |
|
(CH2)4NH2 |
Lysine |
Lys |
|
CH2CH2CH2NH(NH)NH2 |
Arginine |
Arg |
|
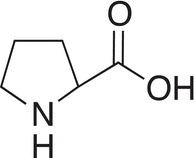
Amino group part of ring |
|
Proline |
Pro |

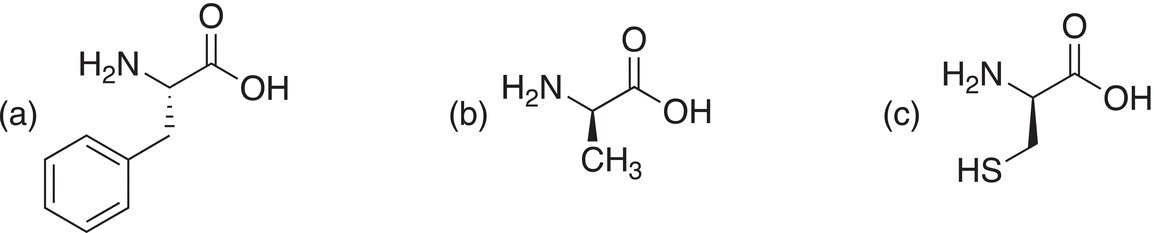
It should be pointed out that the absolute R and S configurations discussed in Chapter 5 are determined by a different method and hence there is not necessarily a direct correlation between the R and S, and the L and D assignments of these compounds. Shown below is the absolute configuration of the amino acids shown above (the assumption is made that the R group is the third priority).

Problem 20.3
Determine the absolute configuration (R or S) of the following α-amino acids.