Organic Chemistry: Concepts and Applications - Headley Allan D. 2020
Stereochemistry
5.3 Significance of Chirality
Chirality (enantiomerism) in molecules plays a key role in nature and technology. The effective functioning of the human body, for example, depends on the chiral recognition of different molecules. Many biological molecules are chiral, and the survival of living systems depends on interactions with a specific enantiomer. For example, one enantiomeric form of amino acids is not toxic to the body, yet their enantiomeric stereoisomers are toxic to the body. Just as a right-hand glove cannot fit on the left hand and vice versa; typically, only one enantiomer of a drug can fit in the receptor/drug binding site for effective biological activity. This concept is illustrated in Figure 5.9. In order for the drug to show the desired pharmacological effect, the portions B, C, and D of the drug must fit in the corresponding sockets b, c, and d of the drug-binding site (receptor). As shown in the figure, the interactions of one of the enantiomers correspond ideally to the receptor, resulting in desired biological effects. The other enantiomer also has the same groups A, B, C, and D, but is inactive since it is not able to fit into the receptor.
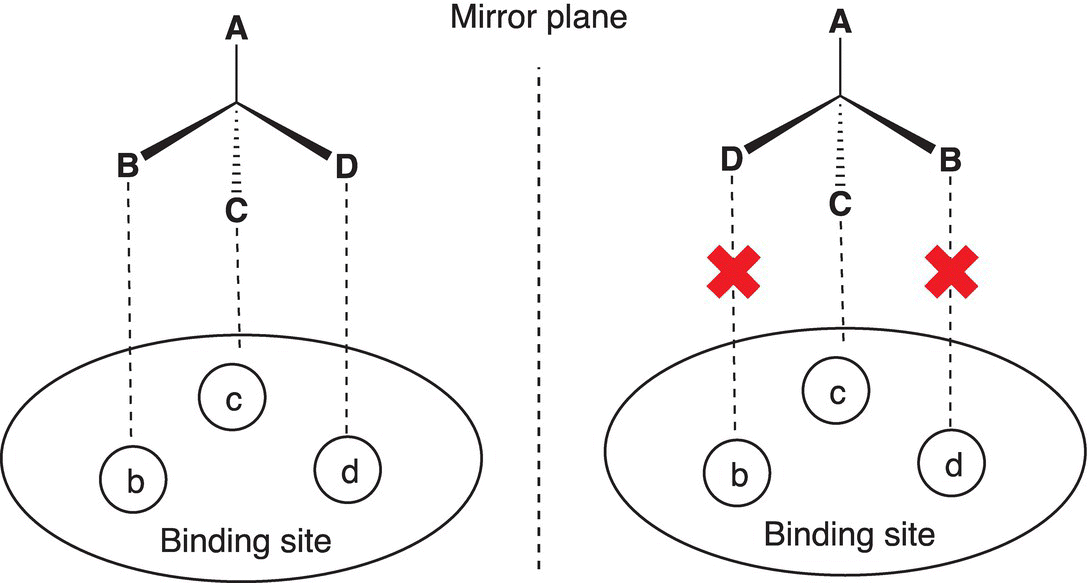
Figure 5.9 Hypothetical interaction between two enantiomers of a chiral drug and a binding site.
5.3.1 Molecular Chirality and Biological Action
In 1848, the French chemist, Louis Pasteur discovered molecular chirality in tartaric acid. In 1858, he showed that one enantiomer (the dextro form) of the ammonium tartrate was fermented by Penicillium glaucum, while its enantiomer (the levs form) was unaffected.
|
One enantiomer of ammonium tartrate is fermented by Penicillium glaucum |
The other enantiomer is unaffected by Penicillium glaucum |
Since then, scientists have recognized the significance of stereoisomerism in relationship to physiological activity. A practical importance of chirality in biological system is that of carvone and the effect of the different enantiomers on the olfactory receptors. As shown below, different odors are detected from the two enantiomers of carvone, an asterisk indicates the stereogenic carbon.
|
One enantiomer has an odor of caraway seeds |
The other enantiomer has an odor of spearmint |
Problem 5.3
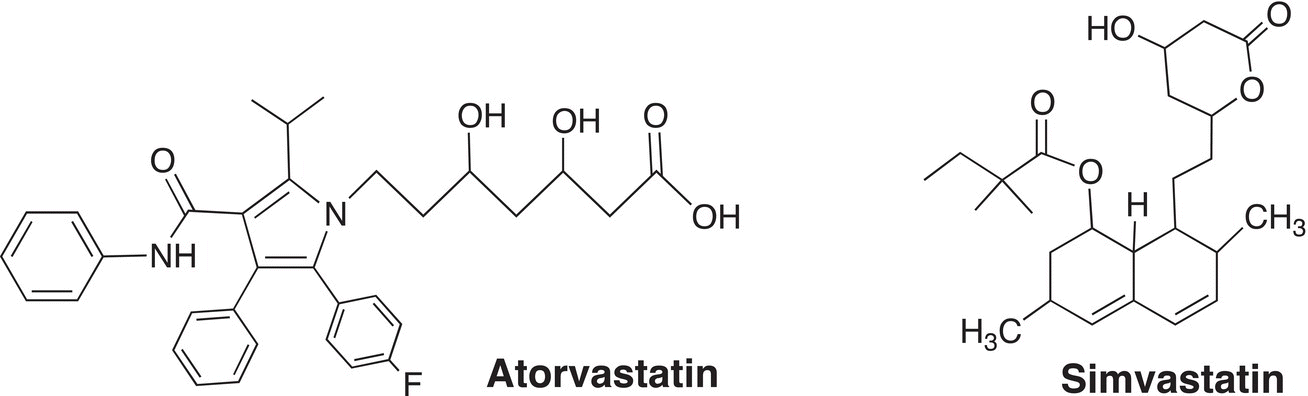
Atorvastatin and simvastatin are the active ingredients in the lipid-lowering drugs, Lipitor and Zocor, respectively. Identify chiral carbons that are present in atorvastatin and simvastatin.
The urgency of the importance of stereochemistry and chirality, especially in the pharmaceutical industry, lies in the history of the thalidomide tragedy. Thalidomide was a drug first used in the market in 1957 in Germany. It was prescribed as a sedative and was claimed to cure anxiety, insomnia, gastritis, and tension. Later, in the 1960s, it was widely used against nausea for morning sickness for pregnant women. Shortly after the drug was introduced in the market, many infants were born with malformation of limbs. The research led to the discovery of two enantiomers of thalidomide and found that the one enantiomer has the desired effect in treating morning sickness, whereas the other enantiomer is teratogenic.
|
One enantiomer is a sedative and used in the cure of morning sickness in pregnant women |
The other enantiomer is a teratogenic and causes severe deformity in babies |
Problem 5.4
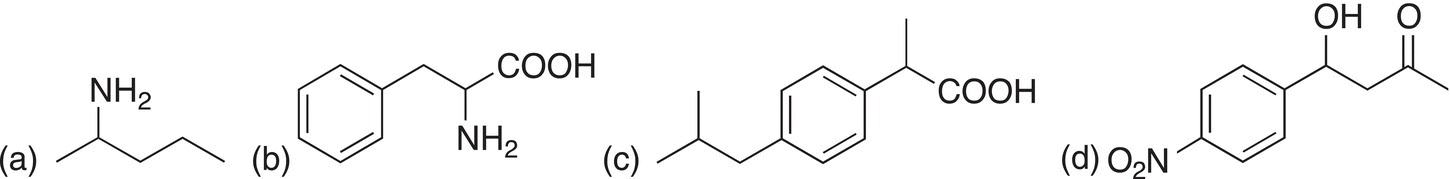
Identify the chiral centers and give a dashed-wedge representation around the chiral carbon for each of the molecules shown below.
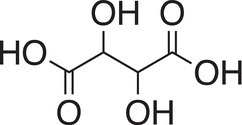 Tartaric acid
Tartaric acid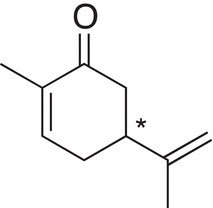 Carvone
Carvone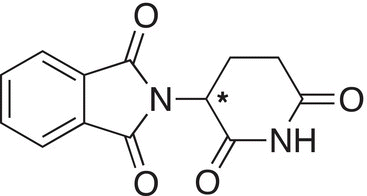 Thalidomide
Thalidomide