Organic Chemistry: Concepts and Applications - Headley Allan D. 2020
Reduction Reactions in Organic Chemistry
10.5 Reduction of Oxiranes
Oxiranes are cyclic ethers, and as you observe from the structure below, they are very similar to carbonyl compounds in that there are polar bonds, in which the carbons bonded to the oxygen are partially positive and the oxygen is partially negative.
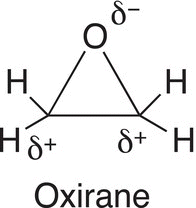
Based on the partial charges of oxiranes, reducing agents will attack either of the partially positive carbons to generate an alkoxide anion salt as shown in Reaction (10-51), which is similar to the alkoxide anion salt that we saw in the section with carbonyl compounds.
(10-51)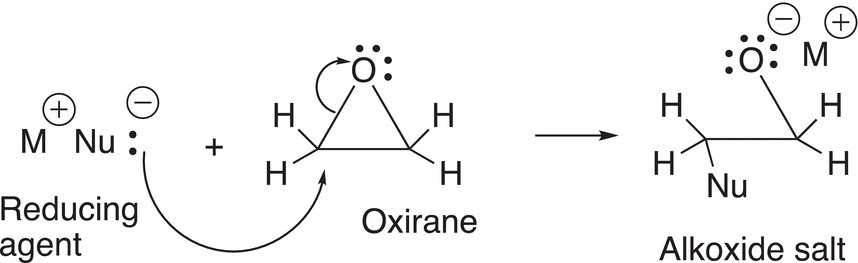
In a separate step, the alkoxide salt can be hydrolyzed under acidic conditions to produce an alcohol as shown in Reaction (10-52).
(10-52)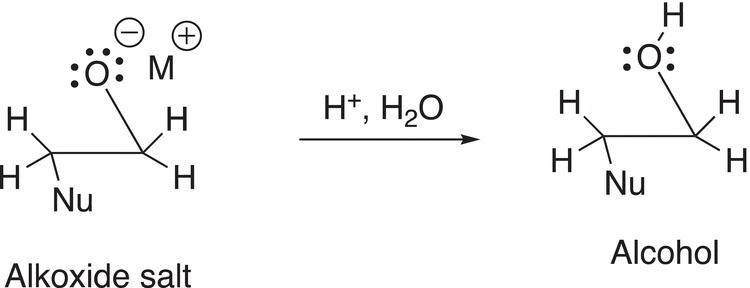
Note that via this transformation, two carbons are added to the reducing agent, such as a Grignard reagent, to form the alcohol product. A summary of this concept is shown in Reaction (10-53).
(10-53)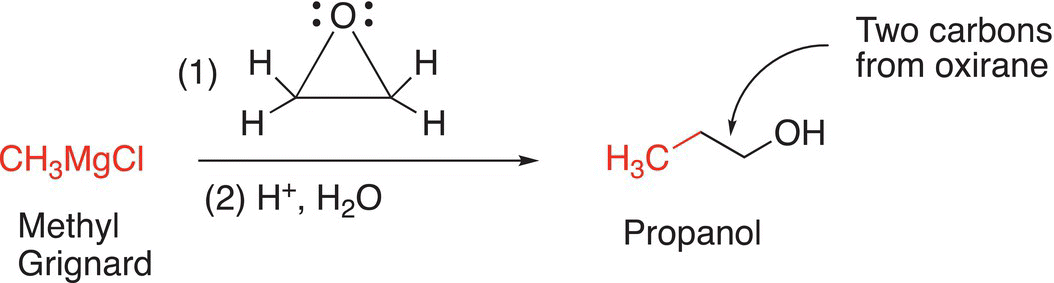
Thus, it is possible to use methyl chloride as a starting compound to synthesize propanol by this method as shown in Reaction (10-54).
(10-54)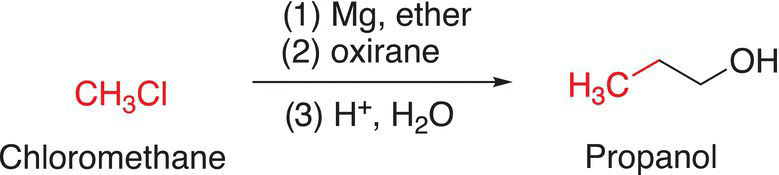
Students should be able to apply analytical thinking skills to determine the type of reactions needed for the above transformation and equally important the specific reactions needed. Problem 10.11 is designed to help students apply their analytical thinking skills to determine possible starting compounds and possible reactions in order to synthesize the compounds shown.
Problem 10.11
Show how you could use ethylene oxide and appropriate organohalides to synthesize the alcohols shown below.
