1,001 Chemistry Practice Problems For Dummies (2014)
Part I. The Questions
Chapter 11. Thermochemistry
All processes involve energy. In this chapter, the key is the energy in chemical processes. Adding or removing heat may cause a phase change or a change in temperature. The specific heat of a substance is a useful factor relating the energy, mass, and temperature change. Calorimetry uses specific heat, temperature change, and mass to determine the energy involved in a chemical process. Heats of formation are a useful alternative to calorimetry for determining energy changes. Hess’s law allows you to predict energy changes when calorimetry or heats of formation are inadequate.
The Problems You’ll Work On
In this chapter, you work with thermochemistry in the following ways:
![]() Doing temperature conversions
Doing temperature conversions
![]() Interpreting energy and phase changes
Interpreting energy and phase changes
![]() Completing specific heat and calorimetry problems
Completing specific heat and calorimetry problems
![]() Calculating with standard heats of formation
Calculating with standard heats of formation
![]() Finding enthalpy changes with Hess’s law
Finding enthalpy changes with Hess’s law
What to Watch Out For
Remember the following when working on thermochemistry:
![]() Dimensional analysis can be exceedingly useful in thermochemistry.
Dimensional analysis can be exceedingly useful in thermochemistry.
![]() Don’t confuse exothermic and endothermic reactions.
Don’t confuse exothermic and endothermic reactions.
![]() All phase changes involve energy. A phase change is exothermic in one direction and endothermic in the reverse direction.
All phase changes involve energy. A phase change is exothermic in one direction and endothermic in the reverse direction.
![]() In calorimetry, the thermometer is part of the surroundings, not the system. When the thermometer shows a temperature increase, it’s absorbing energy (endothermic). Endothermic to a thermometer is exothermic for the system.
In calorimetry, the thermometer is part of the surroundings, not the system. When the thermometer shows a temperature increase, it’s absorbing energy (endothermic). Endothermic to a thermometer is exothermic for the system.
![]() The heat of formation of an element in its standard state is always 0.
The heat of formation of an element in its standard state is always 0.
Converting Temperatures
722–731 Convert the temperatures between the Celsius and Kelvin scales.
722. 300°C = _____ K
723. 150°C = _____ K
724. –200°C = _____ K
725. –78°C = _____ K
726. 37°C = _____ K
727. 100 K = _____°C
728. 300 K = _____°C
729. 0 K = _____°C
730. 313 K = _____°C = _____ °F
731. 233.15 K = _____°C = _____°F
Phase Changes and Energy
732–752 Answer the questions on phase changes and the energy associated with the changes.
732. Which line segment in the graph represents the process of melting?
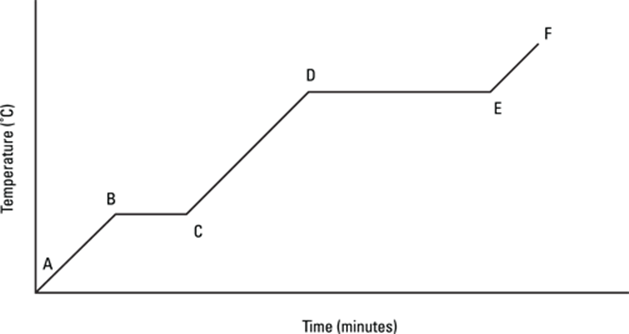
733. Which line segment in the graph represents the process of condensation?
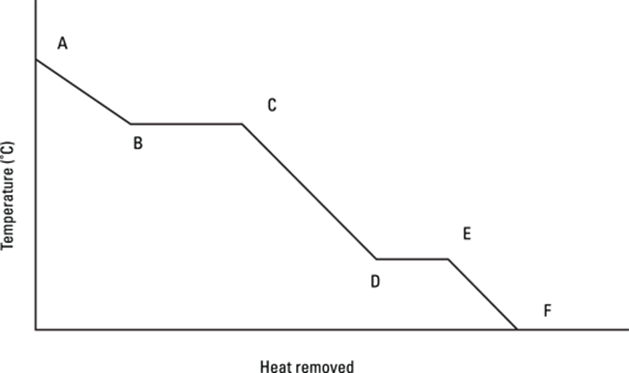
734. Which line segment in the graph represents the process of heating a liquid substance with no phase change?

735. Which line segment in the graph represents the process of boiling?

736. Which line segment in the graph represents the process of heating a solid substance with no phase change?

737. Which line segment in the graph represents the process of freezing?

738. Which line segment in the graph represents the process of heating a gaseous substance with no phase change?

739. How much energy, in joules, does it take to melt 50.0 g of ice at 0°C? The ΔHfus = 334 J/g for ice at its melting point.
740. What is the energy change, in calories, when 100. g of ethanol is frozen at –112°C? The ΔHfus = 25 cal/g for ethanol at its melt-ing point of –112°C.
741. How much energy, in calories, is needed to evaporate 50.0 g of water at 100°C? The ΔHvap = 540 cal/g for water at its boiling point.
742. What is the energy change, in joules, when 20.0 g of nitrogen gas is condensed from the gaseous state to the liquid state at its boiling point? The ΔHvap = 201 J/g for nitrogen at its boiling point of 77 K.
743. How much energy, in calories, is needed to raise the temperature of 10.0 g of steam from 102°C to 112°C? The specific heat of steam is 0.500 cal/g°C.
744. How much energy, in joules, is needed to raise the temperature of 75.0 g of water from 20.°C to 80.°C? The specific heat of water is 4.18 J/g°C.
745. How much energy, in calories, does it take to change 25.00 g of ice at –10.0°C to water at 0.0°C? The specific heat of ice is 0.500 cal/g°C, and ΔHfus = 80.00 cal/g.
746. What is the energy change, in joules, when 125 g of water is cooled from 25.0°C to ice at 0.0°C? The specific heat of water is 4.18 J/g°C, and ΔHfus = 334.0 J/g.
747. What is the energy change, in calories, when 40.0 g of steam is cooled from 120.0°C to 25.0°C? The specific heat of steam is 0.500 cal/g°C, ΔHvap = 540.0 cal/g, and the specific heat of water is 1.00 cal/g°C.
748. How much energy, in joules, is necessary to raise the temperature of 200.0 g of water at 48.0°C to steam at 122.0°C? The specific heat of water is 4.18 J/g°C, ΔHvap = 2,257 J/g, and the specific heat of steam is 2.09 J/g°C.
749. How much energy, in calories, is necessary to raise the temperature of 36.0 g of ice at –40.0°C to steam at 100.0°C? The specific heat of ice is 0.500 cal/g°C, the specific heat of water is 1.00 cal/g°C, ΔHfus = 80.00 cal/g, and ΔHvap = 540.0 cal/g.
750. What is the energy change, in joules, when 72.0 g of steam at 120.0°C changes to ice at 0.0°C? The specific heat of steam is 2.09 J/g°C, the specific heat of water is 4.18 J/g°C, ΔHfus = 334.0 J/g, and ΔHvap = 2,257 J/g.
751. How much energy, in joules, is necessary to raise the temperature of 4,536 g of ice at –78.0°C to steam at 105.0°C? The specific heat of ice and steam is 2.09 J/g°C, the specific heat of water is 4.18 J/g°C, ΔHfus = 334.0 J/g, and ΔHvap = 2,257 J/g.
752. What is the energy change, in calories, when 0.500 g of steam at 150.0°C changes to ice at –50.0°C? The specific heat of steam and ice is 0.500 cal/g°C, ΔHvap = 540.0 cal/g, ΔHfus = 80.00 cal/g, and the specific heat of water is 1.00 cal/g°C.
Specific Heat and Calorimetry
753–762 Complete the calculations involving specific heat and calorimetry.
753. What is the mass of a sample if the absorption of 172 calories results in a temperature increase of 5.00°C? The specific heat of the sample is 0.573 cal/g°C.
754. The addition of 197 J to a 22.0-g sample results in a temperature increase of 2.15°C. What is the specific heat of the sample?
755. What is the temperature change for a 38.1-g sample following the addition of 153 calories? The specific heat of the sample is 0.217 cal/g°C.
756. A 17.35-g sample was heated by 1,148 J. The specific heat of the sample is 2.17 J/g°C. What is the final temperature of the sample if the initial temperature was 15.5°C?
757. A 19.75-g sample was heated by 12.35 calories. The specific heat of the sample is 0.125 cal/g°C. What was the initial temperature of the sample if the final temperature is 37.0°C?
758. A 3.75-g sample with a specific heat of 0.986 cal/g°C experienced a 2.46°C increase in temperature. What was the sample’s energy change in calories?
759. A 0.326-g sample with a specific heat of 0.896 J/g°C experienced a 1.37°C decrease in temperature. What was the sample’s energy change in joules?
760. Two samples are placed in contact, and heat flows from the warmer sample to the cooler one. Sample 1, the cooler sample, has a specific heat of 2.18 cal/g°C and exhibits a 13.9°C increase in temperature. Sample 2 has a specific heat of 1.36 cal/g°C and exhibits a 18.8°C decrease in temperature. If Sample 1 has a mass of 1.35 g, what is the mass of Sample 2? Assume no heat is lost to the surroundings.
761. Two samples are placed in contact, and heat flows from the warmer sample to the cooler one. Sample 1, the cooler sample, exhibits a 20.1°C increase in temperature. Sample 2 has a specific heat of 2.15 J/g°C and exhibits a 12.8°C decrease in temperature. If Sample 1 has a mass of 1.42 g and Sample 2 has a mass of 2.70 g, what is the specific heat of Sample 1? Assume no heat is lost to the surroundings.
762. Two samples are placed in contact, and heat flows from the warmer sample to the cooler one. Sample 1, the warmer sample, exhibits a 15.0°C decrease in temperature. The initial temperature of Sample 2 was 25.1°C. Sample 1 has a specific heat of 0.581 cal/g°C, and Sample 2 has a specific heat of 0.381 cal/g°C. If Sample 1 has a mass of 5.13 g and Sample 2 has a mass of 4.19 g, what is the final temperature of Sample 2? Assume no heat is lost to the surroundings.
Heats of Formation
763–771 Complete the calculations related to heats of formation. Use the following table for these problems.
Standard Heats of Formation of Selected Substances
|
Substance |
Formula |
ΔH°f (kJ/mol) |
|
Ammonia |
NH3(g) |
–46 |
|
Boron oxide |
B2O3(s) |
–1,274 |
|
Carbon dioxide |
CO2(g) |
–394 |
|
Carbon monoxide |
CO(g) |
–111 |
|
Chlorine |
Cl2(g) |
0* |
|
Diamond |
C(dia) |
2 |
|
Ethyl alcohol |
C2H5OH(l) |
–277 |
|
Glucose |
C6H12O6(s) |
–1,273 |
|
Graphite |
C(gr) |
0* |
|
Nitrogen oxide |
NO(g) |
91 |
|
Oxygen |
O2(g) |
0* |
|
Water |
H2O(l) |
–286 |
|
Water vapor |
H2O(g) |
–242 |
*The heat of formation for all elements in their standard state is, by definition, exactly zero.
763. Use standard heats of formation to determine the heat of reaction (ΔH°rxn) for the following reaction:
![]()
764. Use standard heats of formation to determine the heat of reaction (ΔH°rxn) for the following reaction:
![]()
765. Use standard heats of formation to determine the heat of reaction (ΔH°rxn) for the following reaction:
![]()
766. Use standard heats of formation to determine the heat of reaction (ΔH°rxn) for the combustion of ammonia:
![]()
767. Use standard heats of formation to determine the heat of reaction (ΔH°rxn) for the combustion of ethyl alcohol:
![]()
768. Use standard heats of formation to determine the heat of reaction (ΔH°rxn) for the combustion of glucose:
![]()
769. The heat of reaction for the following reaction is –76 kJ:
![]()
Determine the standard heat of formation for nitrosyl chloride (NOCl).
770. The heat of reaction for the combustion of propane is –2,045 kJ. This reaction is
![]()
Determine the standard heat of formation of propane.
771. If the heat of reaction for the following reaction is –9,090. kJ, what is the standard heat of formation of pentaborane-9 (B5H9)?
![]()
Enthalpy Changes with Hess’s Law
772–781 Practice using Hess’s law. This law says that the overall enthalpy change of a process is the sum of the individual enthalpy changes of the steps involved in the process.
772. Determine the heat of reaction for the following reaction using Hess’s Law:
![]()
Use these thermochemical equations to find the answer:
![]()
773. Determine the heat of reaction for the following reaction using Hess’s law:
![]()
Use these thermochemical equations to find the answer:
![]()
774. Determine the heat of reaction for the following reaction using Hess’s law:
![]()
Use these thermochemical equations to find the answer:
![]()
775. Determine the heat of reaction for the following reaction using Hess’s law:
![]()
Use these thermochemical equations to find the answer:
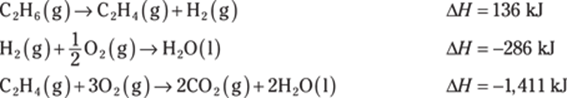
776. Determine the heat of reaction for the following reaction using Hess’s law:
![]()
Use these thermochemical equations to find the answer:
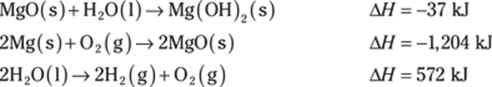
777. Determine the heat of reaction for the following reaction using Hess’s law:
![]()
Use these thermochemical equations to find the answer:
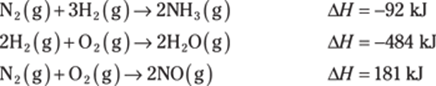
778. Determine the heat of reaction for the following reaction using Hess’s law:
![]()
Use the following thermochemical equations to find the answer:
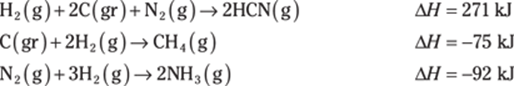
779. Determine the heat of reaction for the following reaction using Hess’s law:
![]()
Use the following thermochemical equations to find the answer:
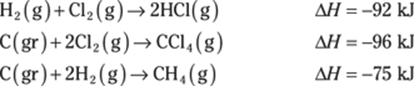
780. Determine the heat of reaction for the following reaction using Hess’s law:
![]()
Use the following thermochemical equations to find the answer:
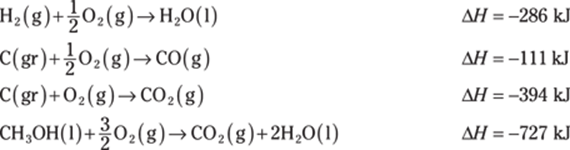
781. Determine the heat of reaction for the following reaction using Hess’s law:
![]()
Use these thermochemical equations to find the answer:
