1,001 Chemistry Practice Problems For Dummies (2014)
Part II. The Answers
 Go to www.dummies.com/cheatsheet/1001chemistry to access the Cheat Sheet created specifically for 1,001 Chemistry Practice Problems For Dummies.
Go to www.dummies.com/cheatsheet/1001chemistry to access the Cheat Sheet created specifically for 1,001 Chemistry Practice Problems For Dummies.
In this part . . .
Here you get answers and explanations for all 1,001 problems. As you read the solutions, you may realize that you need a little more instruction. Fortunately, the For Dummies series offers several excellent resources. We highly recommend the following titles (all published by Wiley):
![]() Chemistry Essentials For Dummies by John T. Moore
Chemistry Essentials For Dummies by John T. Moore
![]() Chemistry For Dummies by John T. Moore
Chemistry For Dummies by John T. Moore
![]() Chemistry Workbook For Dummies by Peter J. Mikulecky, Katherine Brutlag, Michelle Rose Gilman, and Brian Peterson
Chemistry Workbook For Dummies by Peter J. Mikulecky, Katherine Brutlag, Michelle Rose Gilman, and Brian Peterson
When you’re ready to step up to more advanced chemistry courses, you’ll find the help you need in these titles:
![]() Chemistry II For Dummies by John T. Moore
Chemistry II For Dummies by John T. Moore
![]() Inorganic Chemistry For Dummies by Michael Matson and Alvin W. Orbaek
Inorganic Chemistry For Dummies by Michael Matson and Alvin W. Orbaek
![]() Organic Chemistry I For Dummies by Arthur Winter
Organic Chemistry I For Dummies by Arthur Winter
![]() Organic Chemistry II For Dummies by John T. Moore and Richard H. Langley
Organic Chemistry II For Dummies by John T. Moore and Richard H. Langley
![]() Biochemistry For Dummies by John T. Moore and Richard H. Langley
Biochemistry For Dummies by John T. Moore and Richard H. Langley
Visit www.dummies.com for more information.
Chapter 16. Answers and Explanations
Here are the answer explanations for all 1,001 chemistry questions in this book. For reference, you can find the periodic table in the Appendix.
1. a gram
A gram (g) is a common metric unit of mass used in the laboratory.
2. a centimeter
A centimeter (cm) is a common metric unit of length used to measure small objects in the laboratory. You can also measure small objects in millimeters (mm).
3. a milliliter
A milliliter (mL) is a common metric unit of volume used in the laboratory. You can also measure volume in cubic centimeters (cm3 or cc), but that unit is less common in the lab.
4. a millimeter of mercury
A millimeter of mercury (mm Hg), also known as a torr, is a common metric unit of pressure used in the laboratory when dealing with gases. Other units of pressure are pascals, atmospheres, and bars; however, they aren’t as common in the chemistry lab.
5. a joule
The joule (J) is a basic unit of energy or work in the metric system. It’s equal to a newton-meter (N·m).
6. kilo-
Kilo- is the metric prefix that represents 1,000, or 103.
7. milli-
Milli- is the metric prefix that represents ![]() , or 10–3.
, or 10–3.
8. centi-
Centi- is the metric prefix that represents ![]() , or 10–2.
, or 10–2.
9. nano-
Nano- is the metric prefix that represents 10−9, which equals ![]() , or 1 billionth.
, or 1 billionth.
10. mega-
Mega- is the metric prefix that represents 106, which equals 1,000,000, or 1 million.
11. kilogram
The kilogram (kg) is the most appropriate unit to express the mass of an adult human. A person who weighs 150 lb. (pounds) on Earth has a mass of approximately 68 kg.
12. cubic centimeters
To measure a small regularly shaped object like a child’s wooden block, you’d likely use centimeters (cm). After measuring the length, width, and height, you multiply these values together (V = lwh) and record the volume in cubic centimeters (cm3).
13. degrees Celsius
Scientists would most likely use the metric unit degrees Celsius (°C) to report the temperature on a warm autumn day. They could also use kelvins (K), the SI unit of temperature.
14. milligram
The milligram (mg) is the most common metric measurement for small doses of solid medication.
15. kelvin or ampere
The SI base unit of temperature, the kelvin, was named after Lord Kelvin, who was also known as William Thomson. The ampere, named after André-Marie Ampère, is another base unit in SI; it measures electrical current.
Other units named after people — newtons, joules, watts, and so on — are derived units, not base units.
16. a quart
One liter is about 1.06 qt. (quarts).
17. a yard
A meter is a little over 39 in. (inches), which is closest to a yard (36 in.).
18. 8 fl. oz.
One cup contains 8 fl. oz. (fluid ounces).
19. yard
The yard is an English unit for length that’s comparable to a meter.
20. centimeters
Measuring the width of the board in centimeters (cm) is appropriate.
21. 100 mg
To convert from decigrams to milligrams, use the relationship 1 dg = 100 mg. Start with the given value (1 dg) over 1 and then multiply by 100 mg/1 dg so that the decigrams cancel:
![]()
Another option is to convert decigrams to grams and grams to milligrams:
![]()
22. 10 dL
To convert from liters to deciliters, use the conversion factor 10 dL/1 L. Cancel the units that are the same in the numerator and denominator to make sure you end up with deciliters:
![]()
23. 0.001 km
To convert from meters to kilometers, recall that 1 km = 1,000 m. Divide 1 by 1,000 to get 0.001. The meters in the numerator and denominator cancel, leaving kilometers as the final unit:
![]()
24. 100 cm
To determine how many centimeters are in a meter, recall that the prefix centi- represents 1/100. You can use 1 cm/0.01 m or 100 cm/1 m as your conversion factor. Generally, it’s easier to avoid the conversions that use decimals. Multiply 1 m by 100 cm/1 m. The meters cancel, giving you 100 cm:
![]()
25. 100 g
To find out how many grams are in a hectogram, recall that the prefix hecto- represents 100. Multiply 1 hg by 100 g/1 hg. The hectograms cancel, giving you 100 g:
![]()
26. 25,000 mL
You can determine how many milliliters are in a dekaliter in two steps. Convert dekaliters to liters (1 daL = 10 L) and then convert liters to milliliters (1 L = 1,000 mL): daL → L → mL. Here’s the answer:
![]()
In scientific notation, the answer is 2.5 × 104 mL.
27. 4,900,000 cg
You can find the number of centigrams that are in 49 kg in two steps. Convert kilograms to grams (1 kg = 1,000 g) and then convert grams to centigrams (1 g = 100 cg): kg → g → cg:
![]()
In scientific notation, the answer is 4.9 × 106 cg.
28. 0.00037 GW
To get from watts to gigawatts, you need to know that giga- means 1 × 109, or 1,000,000,000, of something. Multiply 370,000 W by 1 GW/1,000,000,000 W. The watts cancel, giving you the answer in gigawatts:
![]()
Alternatively, you can move the decimal point in 370,000 W nine places to the left to convert to watts. In scientific notation, the answer is 3.7 × 10–4 GW.
29. 126,000,000,000 μg
Going from very large units to very small units can be challenging. Be sure to double-check your conversions and the number of zeros. Convert megagrams to grams (1 Mg = 1,000,000 g) and convert grams to micrograms (1 g = 1,000,000 μg): Mg → g → μg:
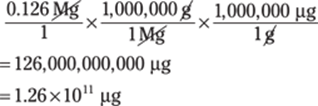
30. 0.000000000000080 km
Converting from very small units to very large units can be challenging. Think of the steps, picometers to meters (1 × 1012 pm = 1 m) and then meters to kilometers (1,000 m = 1 km): pm → m → km:
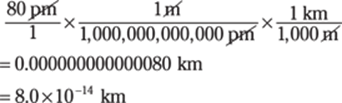
31. 0.002 m3
Conversions that involve cubic units are often challenging because people aren’t used to thinking of conversion factors in three dimensions.
To get from units used for measuring liquids (liters) to units used to measure solids (cubic meters), remember that 1 mL = 1 cm3. Your plan to get from liters to cubic meters may look like this: L → mL → cm3 → m3:
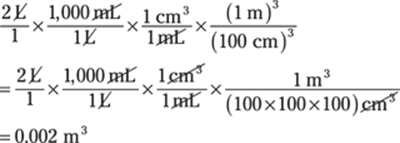
In scientific notation, the answer is 2 × 10–3 m3.
32. 640,000 mL
Conversions that involve cubic units are often challenging because people aren’t used to thinking of conversion factors in three dimensions.
To convert from cubic meters to milliliters, you may choose to use the relationship 1 mL = 1 cm3 as follows: m3 → cm3 → mL:
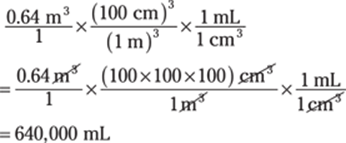
In scientific notation, the answer is 6.4 × 105 mL.
33. 22 mi.
Converting between metric units and English units mainly requires using the right conversion factor. To go from kilometers to miles, you can use the conversion factor 1 mi./1.61 km. If you set up the problem correctly, the kilometers cancel out, leaving you with miles:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well.
34. 0.079 in.
To convert between centimeters and inches, use the conversion factor 1 in./2.54 cm. The centimeters cancel out, giving you the answer in inches:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well.
35. 221 yd.
When converting between meters and yards, start with the given (202 m) and use the conversion factor 1 yd./0.914 m. The meters cancel out, giving you the answer in yards:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well.
36. 130 lb.
To convert from kilograms to pounds, you can use the conversion factors 1,000 g/1 kg and 1 lb./454 g:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well.
Note: You have to make some assumptions to do this conversion, because the kilogram is a unit of mass and the pound is a unit of weight (force). Dividing a weight by the acceleration due to gravity yields a mass. On Earth, the force of gravity is nearly constant, allowing the pound to function as a unit of mass. For clarity, if pound is a mass, the designation should be pound-mass; when pound is a weight, the designation is pound-force.
37. 7.97 qt.
Liters and quarts are similar in size, so be careful with the conversion. Use the conversion factor 1 qt./0.946 L. The liters cancel out, giving you the answer in quarts:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well.
38. 0.22 cm
To convert from inches to centimeters, multiply the inches by 2.54 cm/1 in.:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well.
39. 745 km
To find how many kilometers are in a certain number of miles, simply multiply the miles by 1.61 km/1 mi.:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well.
40. 41,000 g
To convert from pounds to grams, take the number of pounds and multiply it by 454 g/1 lb.:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well. In scientific notation, the answer is 4.1 × 104 g.
41. 1,980 L
There are 3.78 L in a gallon, so simply multiply the number of gallons, 525 gal., by 3.78 L/1 gal.:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well. In scientific notation, the answer is 1.98 × 103 L.
42. 3.0 atm
To find the number of atmospheres in 44 psi (pounds per square inch), use the appropriate pressure conversion. There are 14.7 psi in 1 atm, so multiply 44 psi by 1 atm/14.7 psi:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well.
43. 8.46 c.
Writing a short plan for the conversions may be helpful. Converting from liters to quarts and then from quarts to cups gets you through this problem: L → qt. → c. Always write the units to make sure they cancel correctly; then do the math:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well.
44. 36.1 lb.
To convert from hectograms to pounds, you can either go from hectograms to grams to pounds or from hectograms to kilograms to pounds. Here’s the hg → g → lb. conversion:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well.
45. 0.155 gal.
When converting from milliliters to gallons, I find it easiest to convert milliliters to liters and then convert liters to gallons: mL → L → gal.:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well.
46. 969,000 cm
A slightly longer problem like this requires a plan. Changing miles to kilometers and then kilometers to centimeters is the shorter way to do this. Or you can change the kilometers to meters and then the meters to centimeters, as follows: mi. → km → m → cm:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well. In scientific notation, the answer is 9.69 × 105 cm.
47. 1,020,000 dg
You can do this conversion by converting pounds to grams and grams to decigrams: lb. → g → dg. Multiply the number of pounds by 454 g/lb. and then convert the grams to decigrams by multiplying by 10 dg/1 g:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well. In scientific notation, the answer is 1.02 × 106 dg.
48. 6,400 mL
Convert quarts to liters and liters to milliliters: qt. → L → mL. Multiply the number of quarts by 0.946 L/1 qt.; then convert the liters to milliliters by multiplying by 1,000 mL/1 L:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well. In scientific notation, the answer is 6.4 × 103 mL.
49. 466 cm
Convert feet into inches and then convert inches into centimeters: ft. → in. → cm. Multiply the 15.3 ft. by 12 in./1 ft. to convert to inches; then multiply by 2.54 cm/1 in. to convert to centimeters:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well.
50. 47 L
Convert pints to quarts and then convert quarts to liters: pt. → qt. → L. Multiply 99 pt. by 1 qt./2 pt. to convert to quarts; then multiply the quarts by 0.946 L/1 qt. to convert to liters. The pints and quarts cancel out, giving you the answer in liters:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well.
51. 908 kg
To convert short tons to pounds, multiply the number of tons by 2,000 lb./1 ton. To get from pounds to kilograms (kg), multiply by 0.454 kg/1 lb.:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well.
52. 552 cm
You may be able to find a conversion factor that goes straight from yards to centimeters. If so, this conversion takes only one step. Otherwise, you may remember that 3 ft. = 1 yd., 12 in. = 1 ft., and 2.54 cm = 1 in. Set up the problem, starting with 6.04 yd. over 1, and then line up the conversions so that the units cancel correctly. Here’s the overall plan: yd. → ft. → in. → cm. And here are the calculations:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well.
53. 0.063 c.
First, remember that cc represents cubic centimeters (cm3). Cubic centimeters easily convert to milliliters because they’re in a 1:1 ratio. Then you can convert milliliters to fluid ounces and fluid ounces to cups: cc → mL → fl. oz. → c.:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well.
54. 1,610,000 mm
You can approach this conversion in many ways. One option is to convert yards to inches, inches to centimeters, and centimeters to millimeters: yd. → in. → cm → mm:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well. In scientific notation, the answer is 1.61 × 106 mm.
55. 53,000 pt.
Converting hectoliters to pints involves many steps, so put together a plan. You can go from hectoliters to liters to gallons to quarts to pints: hL → L → gal. → qt. → pt. The only division in the plan occurs when you convert liters to gallons, so you can multiply all the values in the numerator (250 × 100 × 4 × 2) and then divide by 3.78. The units cancel, giving you the answer in pints:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well. In scientific notation, the answer is 5.3 × 104 pt.
56. 5,100 g
A slug is an English mass unit that you may not encounter except when working conversion problems. First convert slugs to pounds and then convert to grams: slugs → lb. → g. Converting to grams isn’t difficult if you remember that there are 454 g in 1 lb. Here’s the conversion:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well. In scientific notation, the answer is 5.1 × 103 g.
The conversion takes an extra step if you convert pounds to kilograms and then convert the kilograms to grams, but you should get the same answer either way.
57. 0.2540 km
You can find the number of kilometers in 9,999 in. by converting inches to centimeters (by multiplying by 2.54 cm/1 in.) and then changing centimeters to meters (multiplying by 1 m/100 cm) and meters to kilometers (multiplying by 1 km/1,000 m): in. → cm → m → km.
![]()
The given measurement has four significant figures, so you round the answer to four significant figures as well.
58. 25.9 oz.
Converting from kilograms to ounces requires one metric conversion (kilograms to grams), one metric-English conversion (grams to pounds), and one English conversion (pounds to ounces): kg → g → lb. → oz. The units cancel out, leaving you with ounces:
![]()
The given measurement has three significant figures, so you round the answer to three significant figures as well.
59. 1,600,000 μL
Converting from ounces to microliters requires one English conversion (ounces to quarts), one English-metric conversion (quarts to liters), and one metric conversion (liters to microliters): oz. → qt. → L → μL:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well. In scientific notation, the answer is 1.6 × 106 μL.
60. 1,498.4 dozen
Finding the number of dozen eggs in 17,981 eggs uses the conversion factor 1 dozen/12 eggs:
![]()
The given egg count has five significant figures, so you round the answer to five significant figures as well.
61. 17.1 yr.
When dealing with a number of days that would exceed four years (1,461 days), account for the extra day in a leap year by using the relationship 365.25 days = 1 yr. Take the number of days and multiply by 1 yr./365.25 days. The days cancel out, leaving you with years:
![]()
The given length of time has three significant figures, so you round the answer to three significant figures as well.
62. 13,000 weeks
To do this conversion, you need to know that a century is 100 years. Multiply the 2.5 centuries by the number of years in a century (100); then multiply the years by the number of weeks in a year (52) to get the number of weeks: centuries → years → weeks:
![]()
The given measurement has two significant figures, so the answer has two significant figures as well.
63. $2,870
First, recognize that 1 penny = 3.16 g is a conversion factor that you’ll use in your calculations. The question asks you to find the mass of 1.00 ton of pennies, so 1.00 ton is your starting point. Convert tons to pounds, pounds to grams, grams to pennies, and pennies to dollars: tons → lb. → g → pennies → $:
![]()
The given average mass has three significant figures, so you round the answer to three significant figures as well.
64. 43.8 s
The tricky part is sorting out which of the given numbers to use where. Starting with the total distance in meters, you can multiply by 1 yd./0.914 m and then use 100 yd. = 10.0 s to get to the time unit in the numerator: m → yd. → s:
![]()
The race distances (400 m and 100 yd.) are considered exact measures, so you can ignore those numbers when looking at significant figures. The given time has three significant figures, so the answer does as well.
65. 18 L
This problem is a bit like a conversion from fluid ounces to liters, but you have to take into consideration that 60 guests are each drinking 10. fl. oz. of soda. The first step is to figure out how many ounces of soda that is; then you can convert from ounces to milliliters by multiplying by 29.6 mL/1 fl. oz. Last, you convert from milliliters to liters by multiplying by 1 L/1,000 mL. Here’s the conversion plan: guests → fl. oz. → mL → L:
![]()
The given volume measurement has two significant figures, so you round the answer to two significant figures as well.
66. 8.3 sandwiches
The question gives you two relationships to use: 1 guest eats 25.4 cm of sandwich, and 1 sandwich is 6.0 ft. long. Take the amount of sandwich that each person will eat and multiply it by 60 guests to find out how many centimeters of sandwich you need. Next, you can convert centimeters to inches (by multiplying by 1 in./2.54 cm), inches to feet (by multiplying by 1 ft./12 in.), and feet to sandwiches (by multiplying by 1 sandwich/6.0 ft.): guests → cm → in. → ft. → sandwiches:
![]()
The given sub measurement has two significant figures, so you round the answer to two significant figures as well.
67. 3,780 cm3
You find the volume of a rectangular solid by multiplying the length times the width times the height. You can convert measurements before multiplying:
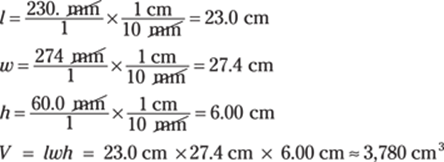
The given measurements have three significant figures, so you round the answer to three significant figures as well.
You can also find the volume in cubic millimeters and then do the conversion:
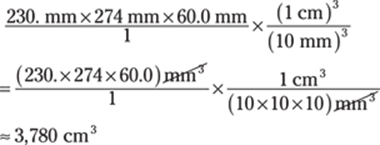
68. 0.0630 m2
The surface area of the front cover of the textbook equals to the length of the book times the width of the book: A = lw. You can convert from millimeters to meters before multiplying them together:
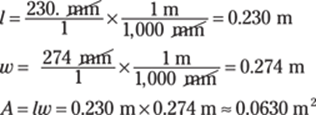
The given measurements have three significant figures, so you round the answer to three significant figures as well.
Another option is to find the surface area before converting the measurements:
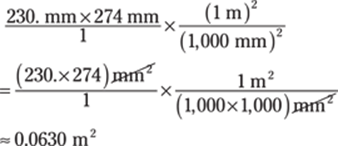
69. 72 tiles
One approach is to first find the area of the hallway in square feet. Then convert the square feet to square inches and divide by the area of one tile.
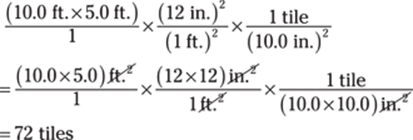
The given 5.0-ft. measurement has only two significant figures, so the answer has two significant figures as well.
Another option is to find the area of the hallway in square inches and divide by the area of a tile, also in square inches.
Or you can determine the number of tiles that fit across the 5.0-ft. hallway (by dividing the width of the hallway by the width of a tile) and multiply that number by the number of tiles that fit down the 10.0-ft. hallway:
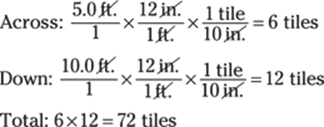
70. 54,000 cm/min.
This problem is challenging because you have to convert both the numerator and the denominator. Keeping track of the units is really important in this kind of problem. You can convert the numerator or the denominator first — the order doesn’t matter. The following equation converts hours to minutes first by multiplying by 1 hr./60 min. Then it converts miles to kilometers (by multiplying by 1.61 km/1 mi.), kilometers to meters (by multiplying by 1,000 m/1 km), and then meters to centimeters (by multiplying by 100 cm/1 m). Here’s the conversion plan: mi./hr. → mi./min. → km/min. → m/min. → cm/min.:
![]()
The given measurement has two significant figures, so you round the answer to two significant figures as well. In scientific notation, the answer is 5.4 × 104 cm/min.
As you go through a problem like this, make sure you cross out the units that cancel. If you end up with the correct units in your answer — in this case, cm/min. — you know you’ve probably set up the problem correctly.
71. 85 lb.
In this problem, you’re given a volume and a conversion factor of 1.00 cm3 = 19.3 g. So to start, this is a conversion problem from liters to cubic centimeters. You can convert liters to milliliters by multiplying by 1,000 mL/1 L. Milliliters and cubic centimeters are equivalent, so you can multiply by 1 cm3/1 mL. Next, multiply by 19.3 g/1.00 cm3 to get to mass. Last, convert grams pounds by multiplying by 1 lb./454 g. Here’s the overall plan: L → mL → cm3 → g → lb.:
![]()
The given volume measurement has only two significant figures, so you round the answer to two significant figures as well.
72. 2.5 min.
To find the number of minutes the horse takes to run a distance of 12 furlongs, first convert the distance to miles in order to use the given speed of 385.3 mph. Then convert the time from hours to minutes: furlongs → rods → yd. → mi. → hr. → min.:
![]()
The given length measurement has only two significant figures, so you round the answer to two significant figures as well.
If you don’t know how many yards are in a mile (1 mi. = 1,760 yd.), you may need to take the extra step of converting yards to feet (3 yd. = 1 ft.) and then converting feet to miles (5,280 ft. = 1 mi.), but you should get the same answer.
73. 0.43 s
Finding the number of seconds the pitch will take to travel from the pitcher to the batter requires using the distance between the two locations and the speed of the ball. Convert the distance from feet to miles by multiplying by 1 mi./5,280 ft. Next, divide by the speed (96 mi./1 hr.) and then convert the hours to minutes (by multiplying by 60 min./1 hr.) and the minutes to seconds (by multiplying by 60 s/1 min.): ft. → mi. → hr. →min. → s:
![]()
The given speed measurement has only two significant figures, so you round the answer to two significant figures as well.
74. 0.00072 mm
This problem requires that you think in three dimensions. You’re given a mass (grams), a conversion between mass and volume (grams per cubic centimeter), and a surface area (cubic meters). Volume equals length ×width × height, and surface area = length × width, so you can find the height by dividing volume by surface area:
![]()
Now for the unit conversions. Change kilograms to grams by multiplying by 1,000 g/1 kg. Multiply by 1 cm3/19.3 g to get cubic centimeters. Next, convert the cubic centimeters to cubic millimeters. That takes care of the numerator. You have to convert the square meters in the denominator to square millimeters before you can divide. Here’s the conversion plan: (kg → g → cm3 → mm3) ÷ (m2 → mm2) = mm3/mm2 = mm:
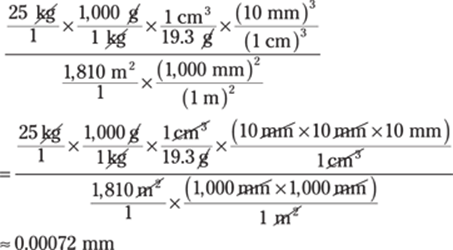
The given mass measurement has only two significant figures, so you round the answer to two significant figures as well. In scientific notation, the answer is 7.2 × 10–4 mm.
Alternatively, you can leave the numerator in cubic centimeters and convert the denominator from square meters to square centimeters. That gives you the answer in centimeters, which you can convert to millimeters by multiplying by 10 mm/1 cm.
75. 2.57 s
You’re waiting for a reply, so you have to account for the time for a signal to get to the moon and back, which means that the signal has to travel 2 × 239,000 miles. Convert miles to kilometers (multiply by 1.61 km/1 m) and kilometers to meters (multiply by 1,000 m/1 km). Then to get the time, divide by the speed:
![]()
The given distance measurement has three significant figures, so you round the answer to three significant figures as well. The speed of the radio waves is considered exact, so you can disregard it when figuring out significant figures.
76. 8.76 × 102
Scientific notation expresses numbers with one digit to the left of the decimal point and any number of significant digits to the right of the decimal. For numbers greater than 1, the exponent on the 10 is positive. The number 876 equals 8.76 × 100, and 100 is the same as 102, so 876 is 8.76 × 102.
77. 4.000001 × 106
For numbers greater than 1, the exponent on the 10 is positive in scientific notation. The number 4,000,001 equals 4.000001 × 1,000,000, and 1,000,000 is the same as 106, so 4,000,001 is 4.000001 × 106.
78. 5.10 × 10–4
When a number is less than 1, the exponent on the 10 is negative in scientific notation. Move the decimal point in 0.000510 until you have just one digit to the left of the decimal point. The number of times you move the decimal becomes the exponent on the 10. You move the decimal point four places to the right, so 0.000510 become 5.10 × 10–4.
79. 9 × 106
The number 900 is the same as 9 × 100, or 9 × 102, so the problem becomes 9 × 102 × 104. To multiply 102 × 104, you just need to add the exponents on the 10s: You end up with 9 × 102 × 104 = 9 × 102 + 4 = 9 × 106.
80. 1 × 101
Although writing 10 in scientific notation doesn’t have much practical value, you can do it. The decimal moves one place to the left, and the 10 becomes 1 (writing .0 after the 1 isn’t necessary unless you have a decimal point after 10 in the original number). You then multiply that 1 by a power of ten. The exponent on the 10 is 1, giving you 1 × 101.
81. 200
The exponent on the 10 is positive, so the decimal point in 2.00 moves two places to the right. The answer is 200, because 2.00 × 102 is the same as 2.00 × 100.
82. 0.09
The exponent on the 10 is –2, so the value is less than 1. Move the understood decimal point in 9 two places to the left, giving you a number between 0 and 1. The answer is 0.09.
83. 4,795.2
With an exponent of +3 on the 10, the decimal point in 4.7952 moves three places to the right. Moving the decimal point three places to the right is the same as multiplying by 1,000, giving you 4,795.2 as the answer.
84. 0.0000164
With an exponent of –5 on the 10, the decimal point in 1.64 moves five places to the left, giving you 0.0000164.
Tip: When you have a large negative exponent, the easiest way to check your answer is to count the 0s to the left of your nonzero digit, including the 0 to the left of the decimal point. The number of 0s should be the same as the number of the exponent.
85. 0.083
The number 0.83 × 10–1 looks like it’s in scientific notation, but it isn’t, because you don’t have a nonzero digit in front of the decimal point. Regardless, the negative exponent on the 10 indicates that the decimal point in 0.83 needs to move one place to the left, so the number is 0.083 in standard form.
86. 5.97 × 103
When you’re adding numbers in scientific notation and the exponents on the 10s are the same, you can just add the numbers in front of the powers of ten: 1.26 + 4.71 = 5.97. The 5.97 falls between 1 and 10 — it has one nonzero digit to the left of the decimal point — so you can finish giving the answer in scientific notation by writing the × 103. The answer is 5.97 × 103.
Note: This approach works because you’re essentially factoring out the 103 before adding the decimal numbers: (1.26 × 103) + (4.71 × 103) = 103(1.26 + 4.71) = 103(5.97).
87. 6.0 × 10–1
The exponents on the 10s are the same, so you can just add the numbers in front of the powers of ten: 3.9 + 2.1 = 6.0. The sum is between 1 and 10, so you retain the power (× 10–1) from the original numbers. The answer is 6.0 × 10–1.
Note: This approach works because you’re essentially factoring out 10–1 before adding the decimal numbers: (3.9 × 10–1) + (2.1 × 10–1) = 10–1(3.9 + 2.1) = 10–1(6.0).
88. 8.6 × 102
Because the exponents on the 10s don’t match, it’s easiest to take both numbers out of scientific notation, do the subtraction, and then put the answer back into scientific notation. Round the decimal number to the tenths place to give the answer the right number of significant figures.
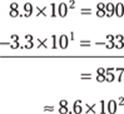
89. –5.1 × 101
The exponents on the 10s don’t match, so take the numbers out of scientific notation, do the subtraction, and then put the answer back into scientific notation. Round the decimal number to the tenths place to give the answer the right number of significant figures.
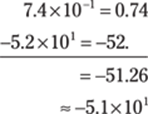
90. 1.2031 × 103
The exponents on the 10s are the same, so you can add the numbers in front of the powers of ten: 8.240 + 3.791 = 12.031. Because 12.031 is greater than 10 — it has two digits to the left of the decimal point — change it into scientific notation, giving you 1.2031 × 101.
Next, multiply that number by 102, the power of ten from the original problem; you get 1.2031 × 101 × 102. To multiply 101 × 102, add the exponents, giving you 101 + 2 = 103. The final answer in scientific notation is 1.2031 × 103.
91. 9.5 × 106
The exponents aren’t the same, so take the numbers out of scientific notation, do the subtraction, and then put the answer back into scientific notation. Round the decimal number to the tenths place to give the answer the right number of significant figures.
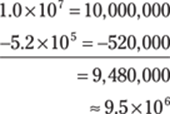
92. 6.04 × 10–3
Take the numbers out of scientific notation, do the addition, and then write the sum in scientific notation. Round the decimal number to two decimal places to give the answer the right number of significant figures.
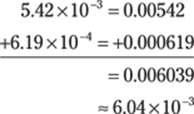
93. 8.41 × 106
In this problem, each number in scientific notation has a different exponent. Take the numbers out of scientific notation, do the addition and subtraction, and put the answer back into scientific notation. Round the decimal number to the hundredths place to give the answer the right number of significant figures.
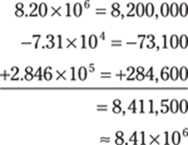
94. 4.5 × 10–2
To multiply numbers that are in scientific notation, you can multiply the numbers in front of the × 10 (here, 1.0 × 4.5 = 4.5) and then add the exponents from the powers of ten (10–7 × 105 = 10–7 + 5 = 10–2). The answer is 4.5 × 10–2.
95. 1.0 × 101
When dividing numbers that are in scientific notation, divide the numbers in front of the × 10 (here, 1.0 ÷ 1.0 = 1.0) and subtract the exponents on the powers of ten. Keep in mind that subtracting a negative number is the same as adding a positive number: 10–3 ÷ 10–4 = 10–3 – (–4) = 10–3 + 4 = 101. The answer is 1.0 × 101.
96. 6.3 × 1015
Multiply the numbers in front of the × 10 (here, 3.15 × 2.0 = 6.3) and then add the exponents (1012 × 103 = 1012 + 3 = 1015) to give you 6.3 × 1015.
97. 4.9 × 104
First, divide the numbers in front of the × 10. In this case, 4.7 ÷ 9.6 = 0.49. Because this number is less than 1, simply subtracting the exponents on the 10s isn’t enough to find the exponent in the answer. You need to account for 0.49 being less than 1 by moving the decimal one place to the right (0.49 becomes 4.9) and by subtracting another 1 from the exponent.
Dividing the powers of 10 in the problem gives you 10–2 ÷ 10–7 = 10–2 – (–7) = 10–2 + 7 = 105. Subtracting 1 from the exponent (to account for moving the decimal point to the right) gives you 105 – 1 = 104, so the complete answer is 4.9 × 104.
98. 1.68 × 1011
To multiply numbers that are in scientific notation, you can multiply the numbers in front of the × 10 and then add the exponents from the powers of ten. When you multiply 8.40 by 2.00, you get 16.8. To put this in scientific notation, you need to move the decimal point one place to the left, so 16.8 becomes 1.68 × 101.
To multiply powers of ten, add the exponents together: 1015 × 10–5 = 1015 + (–5) = 1010. Next, add another 1 to the exponent to account for moving the decimal point in 16.8 to the left: 1010 + 1 = 1011. The final answer is 1.68 ×1011.
99. 3.1 × 105
To divide numbers that are in scientific notation, you can divide the numbers in front of the × 10 and then subtract the exponents from the powers of ten. When you divide 1.0 by 3.2, you get 0.31, a number less than 1. To put the number in scientific notation, you need to move the decimal one place to the right (0.31 becomes 3.1) and decrease the exponent on the 10 by 1.
You’re dividing in this problem, so subtract the exponents from the powers of ten: 108 ÷ 102 = 108 – 2 = 106. Then subtract another 1 from the exponent to account for moving the decimal point in 0.31 one place to the right: 106 – 1 = 105. The answer becomes 3.1 × 105.
100. 1.9 × 10–6
Start by doing calculations with the numbers in front of the × 10: 9.76 × 3.55 ÷ 1.8 = 19.25. Round to two digits to give the answer the right number of significant figures: That means 19.25 becomes 1.9 × 101.
Now deal with the powers of ten from the problem. You add exponents to multiply powers of ten and subtract exponents to divide by powers of ten: 10–9 × 10–3 ÷ 10–5 = 10–9 + (–3) – (–5) = 10–9 – 3 + 5 = 10–7. Finally, multiply the 1.9 × 101 by the 10–7 to get the answer: 1.9 × 101 × 10–7 = 1.9 × 101 + (–7) = 1.9 × 10–6.
101. 1.1 × 10–1
Start by doing the math with the numbers in front of the × 10: 2.48 × 4.756 × 9.1 = 107.33. Round to two digits to give the answer the right number of significant figures: That means 107.33 becomes 110, or 1.1 × 102.
Now add the exponents to multiply the powers of ten: 103 × 10–4 × 10–2 = 103 + (–4) + (–2) = 10–3. Multiply the 1.1 × 102 by the 10–3, and you get 1.1 × 102 × 10–3 = 1.1 × 102 – 3 = 1.1 × 10–1.
102. 3.6 × 10–4
Using the rules for order of operations, complete the multiplication portion first. To multiply numbers that are in scientific notation, you can multiply the numbers in front of the × 10 and then add the exponents from the powers of ten:
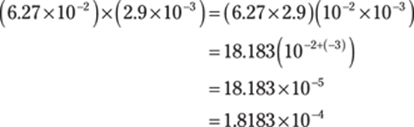
Then do the addition. The powers of ten are the same, so you can simply add the numbers in front of the × 10. (Mathematically, you’re factoring out 10–4.) Round the answer to one decimal place to give the answer the right number of significant figures.
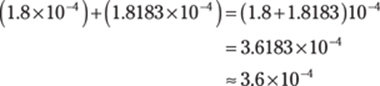
103. 1.526 × 10–5
Here, the order of operations applies. Start with the division. To divide numbers that are in scientific notation, you can divide the numbers in front of the × 10 and then subtract the exponents from the powers of ten:
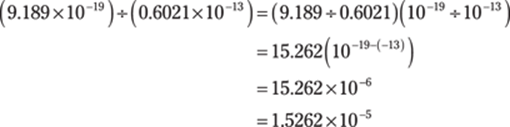
Then do the addition. The powers of ten are different, so take the numbers out of scientific notation, do the addition, and then put the answer back in scientific notation. Round the answer to three decimal places to give the answer the right number of significant figures:
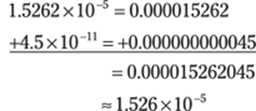
The answer has four significant figures because the –11 exponent makes the second value too small to make a significant difference in the first value. It would be important only if it affected one of the significant figures in the first number.
104. 4.48 × 102
Based on order of operations, do the division from left to right before doing the addition. Start with (1.1 × 101) ÷ (3.68 × 10–6). To divide numbers that are in scientific notation, divide the numbers in front of the × 10 and then subtract the exponents on the powers of ten:
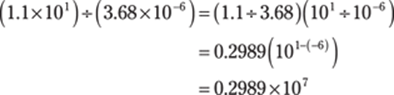
Then divide that answer by 8.2 × 104:
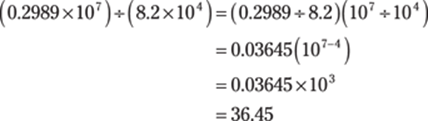
Now add 4.115 × 102 = 411.5 and put the answer back into scientific notation (round to 448 to give the answer the right number of significant figures):
![]()
All the digits in 411.5 are significant; however, due to the division by a two-significant-figure value (8.2 × 104), only the first two digits in 36.45 are significant. Because 36.45 is understood to have no significant figures after the ones place, neither can the sum 448.
105. 5 × 10–2
In this problem, follow the order of operations. Solve the numerator and the denominator before dividing.
In the numerator, do the multiplication before doing the addition. In the denominator, take the numbers out of scientific notation before subtracting. Here are the first couple of steps:
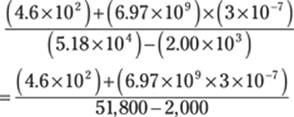
Remember that to multiply powers of ten by each other, you add the exponents:
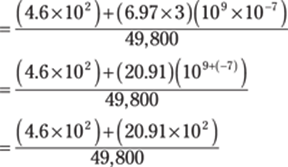
To do the addition in the numerator, you can take the numbers out of scientific notation:
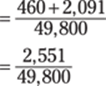
Now you’re ready to divide. You can put the numbers back into scientific notation to help you with the calculations:
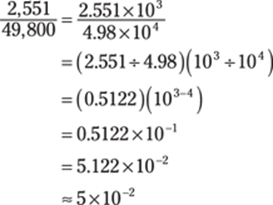
The answer has one significant figure. Why? Multiplying by (3 × 10–4) in the first step would give you a one-significant-figure answer; when you then add (4.6 × 102) to the product, the sum would likewise have one significant figure. Finally, dividing a one-significant-figure number by a three-significant-figure number yields a one-significant-figure answer.
106. 3
All nonzero digits are significant, so 343 has three significant figures.
107. 4
All nonzero digits are significant, so 0.4592 has four significant figures.
108. 6
All nonzero digits and zeros that are sandwiched between two nonzero digits are significant. In 705,204, the 7, 5, 2, and 4 are nonzero digits. The zeros trapped between the 7 and 5 as well as the 2 and 4 are also significant, so 705,204 has a total of six significant figures.
109. 2
Leading zeros (zeros to the left of a nonzero digit and to the right of the decimal point) aren’t significant — they’re just placeholders. The 7 and 5 are significant, so 0.0075 has two significant figures.
110. 3
Zeros to the right of nonzero digits but before an understood (not written) decimal point are not significant. The 2, 4, and 8 are significant, so 248,000 has three significant figures.
111. 5
The nonzero digits and the trapped zeros are significant, but the trailing zeros are not. So in 9,400,300, the five digits from the 9 to the 3 are significant.
112. 5
The nonzero digits, the trapped zeros, and the trailing zero are all significant. Zeros to the right of a nonzero digit and to the right of the decimal point are significant. So in 1.0070, all five digits are significant.
113. 8
Zeros to the right of a nonzero digit and to the right of the decimal point are significant. This traps the zeros between the 3 and the final 0, making all eight digits in 3,000,000.0 significant.
114. 5
The nonzero digits, the trapped zeros, and the trailing zeros are all significant. The leading zeros are not significant. In 0.0040800, the five digits from the 4 to the final 0 are significant.
115. 3
The nonzero digits and the trailing zero are significant; the leading zero before the decimal point is not. The number 0.870 has three significant figures: the 8, 7, and final 0.
116. 5,500
Adding 5,379 + 100 gives you 5,479. After adding, round the final answer with the least-accurate decimal place in mind. The first number (5,379) ends with the ones place, and the second number (100) is significant only to the hundreds place, so round 5,479 to the hundreds place: 5,500. (You can also write the answer in scientific notation: 5.5 × 103.)
117. 13.0
First, do the addition: 12.4 + 0.59 = 12.99. Now determine the least-accurate decimal place: 12.4 ends with the tenths place, and 0.59 ends with the hundredths place. The tenths place is less accurate than the hundredths place, so round 12.99 to 13.0. (You can also write the answer in scientific notation: 1.30 × 101.)
118. 27.56
Do the subtraction first: 61.035 – 33.48 = 27.555. Round the answer with the least-accurate decimal place in mind. In this question, the first number (61.035) ends with the thousandths place, and the second number (33.48) ends with the hundredths place, so round the answer to the hundredths place: 27.555 ≈ 27.56. (You can also write the answer in scientific notation: 2.756 × 101.)
119. 96
Do the math and then round the answer with the least-accurate decimal place in mind. The addition gives you 71 + 24.87 + 0.0003 = 95.8703. The ones place from the 71 is the least accurate for the three numbers, so round the answer to 96. (You can also write the answer in scientific notation: 9.6 × 101.)
120. –467
Doing the subtraction gives you 0.387 – 467 = –466.613. When subtracting, round the final answer to the least-accurate decimal place that both numbers have in common. In this case, the first number is accurate to the thousandths place and the second number, 467, is accurate only to the ones place, so round the answer to the ones place. The answer is –467. (You can also write the answer in scientific notation: –4.67 × 102.)
121. 0.0467
When adding, you want to round your final answer to the least-accurate decimal place. Sometimes it’s easier to see which digit to round your answer to if you line up the numbers vertically:

The least-accurate place is the 0 in the ten-thousandths place of 0.0410, so round the final answer to the ten-thousandths place, giving you 0.0467. (You can also write the answer in scientific notation: 4.67 × 10–2.)
122. –60.405
First, line up the numbers based on the location of the decimal point. Then do the addition and subtraction and round the answer to the least-accurate decimal place.
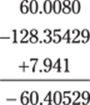
The least-accurate decimal place is the thousandths place in 7.941, so round the answer to –60.405. (You can also write the answer in scientific notation: –6.0405 × 101.)
123. 5,100
Doing the addition gives you 130 + 4,600 + 395.2 = 5,125.2. When adding, round the answer to the least-accurate decimal place. The least-accurate place in 130 is the tens, the least-accurate place in 4,600 is the hundreds, and the least-accurate place in 395.2 is the tenths. Of the three, the hundreds place is the least accurate, so round the answer, 5,125.2, to the hundreds place: 5,100. (You can also write the answer in scientific notation: 5.1 ×103.)
124. 210
The division gives you 0.0074 ÷ 0.000035 ≈ 211.4285714. When dividing, the answer should have the same number of significant figures as the number with the fewest significant figures, regardless of where the decimal point is located. In this problem, each number has only two significant figures, so the answer should have two significant figures. You round the answer to 210. (You can also write the answer in scientific notation: 2.1 ×102.)
125. 26,000
The multiplication gives you 75 × 349 = 26,175. In multiplication problems, identify the number that has the fewest significant figures to determine how many significant figures the answer should have. The number 75 has two significant figures, and 349 has three significant figures. The final answer should have the smaller number of significant figures, which is two, so round the answer to 26,000. (You can also write the answer in scientific notation: 2.6 × 104.)
126. 41.6
The multiplication gives you 7.98 × 5.21 = 41.5758. For a multiplication problem, your answer should have the same number of significant figures as the given number with the fewest significant figures. Both 7.98 and 5.21 have three significant figures, so round the answer to 41.6. (You can also write the answer in scientific notation: 4.16 × 101.)
127. 2.0 × 103
The division gives you 5.00 ÷ 0.0025 = 2,000. With division, the answer should have the same number of significant figures as the number with the fewest significant figures. In this question, 5.00 has three significant figures and 0.0025 has two, so the answer should have two significant figures. The easiest way to express 2,000 with two significant figures is to put it into scientific notation, with one zero after the decimal point: 2.0 ×103.
128. 50 cm2
The multiplication gives you 7.0 cm × 7 cm = 49 cm2. Each measurement has a different number of significant figures: 7.0 has two, and 7 has one. One is the lower number, so the answer should have one significant figure. Round 49 to 50. (You can also write the answer in scientific notation: 5 × 101 cm2.)
129. 0.0334
The division gives you 6.48 ÷ 194.21 ≈ 0.033365944. When dividing, you record the answer with the same number of significant figures as the number with the fewest number of significant figures. In this case, you have only three significant figures coming from 6.48, so round the answer to 0.0334. (You can also write the answer in scientific notation: 3.34 × 10–2.)
130. 2.2 × 10–13
The multiplication gives you 0.000000029 × 0.00000745 = 0.00000000000021605 = 2.1605 × 10–13. The first number you’re multiplying has two significant figures, and the second number has three. Because the answer should have the lower number of significant figures, 2.1605 rounds to 2.2, and the 10–13 remains. The answer is 2.2 × 10–13.
131. 0.20
For a multiplication and division problem, the answer should have the same number of significant figures as the given number with the fewest number of significant figures. The calculations give you the following:
![]()
Here, 0.0034 has two significant figures, 518.27 has five significant figures, and 9.00 has three significant figures. The fewest number of significant figures is two, so round the answer to 0.20. (You can also write the answer in scientific notation: 2.0 × 10–1.)
132. 2,070
Complete the multiplication first: 2,300.00 × 0.854 = 1,964.2. Then add 110, giving you 2,074.2, and round to the least-accurate decimal place, which is the tens place in 110. The answer is 2,070. (You can also write the answer in scientific notation: 2.07 × 103.)
133. 9.0 g/mL
Do the subtraction first to see how many significant figures would be in the denominator: 25.0 mL – 23.8 mL = 1.2 mL. Then do the division: 10.78 ÷ 1.2 ≈ 8.983333333. The final answer should have the same number of significant figures as the 1.2 mL, so you round the answer to 9.0 g/mL.
134. 4.39
At first, you may think this answer should have two significant figures. But think about doing the addition in the numerator: 8.1 + 2.32 + 0.741 = 11.161. If you were to round this answer before dividing by the denominator, it would have three significant figures, because with addition, you round to the least-accurate decimal place: in this case, the tenths place. When you’re ready to divide, both the numerator and denominator would have three significant figures, so the final answer should have three significant figures as well.
The usual practice is to do all the calculations and round at the end to avoid cumulative rounding errors. So here are the calculations:
![]()
Rounded to three significant figures, the answer is 4.39.
Note that if you round and then divide, your answer will be slightly different: 4.4094, which rounds to 4.41.
135. 55
Do the math first:
![]()
After doing the addition and subtraction, you can determine the number of significant figures that need to be in the answer.
The numerator in the first fraction is accurate only to the tens place (due to the 250), and the denominator in the second fraction is accurate only to the tenths place (due to the 1.2). If you were to round 262 and 2.37 at this point, the resulting numbers (260 and 2.4) would have two significant figures.
The numbers you’re multiplying and dividing by also have two significant figures, so the final answer should have two significant figures as well. The final answer is 55. (You can also write the answer in scientific notation: 5.5 × 101.)
136. gas
Think of air in a balloon. Gases take the shape of their containers, and they’re easily compressed or expanded (they don’t have a definite volume).
137. solid
Think of a solid like ice or a block of wood. A solid has a definite shape and a definite volume.
138. liquid
Matter that takes the shape of its container but has a definite volume is a liquid. Think of room-temperature water. One liter of water is 1 liter of water, whether it’s in a vase or spilled all over the kitchen table — same volume, different shape.
139. freezing
When a liquid (such as water) is becoming solid (ice), it’s going through a phase change called freezing.
140. condensation
When a gas cools, it changes into a liquid. This phase change is called condensation.
141. vaporization
When you add enough energy to a liquid that it changes in phase to a gas, it’s said to vaporize or go through vaporization. This phase change is called evaporation when it occurs below the liquid’s boiling point.
142. sublimation
When matter goes from a solid phase to a gas phase without becoming a liquid, it’s said to sublime. You can observe sublimation in substances such as dry ice, moth balls, and iodine crystals.
143. deposition
Deposition is the opposite of sublimation. Deposition occurs when a substance goes from the gaseous phase to the solid phase without becoming a liquid in between. You can see the result of deposition as frost in a freezer; the water vapor in the air turns to ice.
144. pure substance; element
Gold (Au), a pure substance, is element number 79 on the periodic table of the elements.
145. pure substance; compound
Table sugar is a pure substance made of the compound sucrose. Sucrose is a carbohydrate that contains fixed amounts of carbon, hydrogen, and oxygen atoms bonded together.
146. mixture; homogeneous
Fresh air is a mixture of colorless gases containing mostly nitrogen gas and oxygen gas. Each breath of air contains the same composition of gases, so the mixture is homogeneous.
147. pure substance; element
Oxygen (O), a pure substance, is element number 8 on the periodic table of the elements.
148. mixture; heterogeneous
Vegetable soup is a mixture of water, salt, and assorted vegetables. Each bowl you serve has a slightly different composition of ingredients — that’s what makes it heterogeneous.
149. mixture; heterogeneous
Each serving of fruit salad that you take from a serving bowl generally has a different number of each kind of fruit. This variety in each sample makes fruit salad heterogeneous.
150. pure substance; element
Calcium (Ca), a pure substance, is element number 20 on the periodic table of the elements.
151. mixture; heterogeneous
Concrete is made of a variety of rocks and cement, which holds the concrete together. The variety of rocks depends on where you live or what’s in the mix. You can see the rock differences — although they’re sometimes subtle — when you break off a piece of concrete. These differences are what make concrete a heterogeneous mixture.
152. mixture; heterogeneous
Smog is air that contains a variety of particulates (dirt, dust, and so on) and pollutants. Smog can be different shades of color, depending on the types and concentrations of contaminants. This variety of substances mixed together is rarely homogeneous.
153. intensive
An intensive property is independent of the amount of the substance that’s present. Intensive properties are helpful in identifying an unknown substance.
The boiling point of water is an intensive property. If you have a large pot and a small pot full of boiling water on the stove, the water will be boiling at the same temperature in both.
154. physical
A physical change involves a change in the physical properties of a substance, not a change in chemical composition. The shape of a substance is a physical property.
155. chemical
In a chemical change, a substance changes into a new substance. The burning of a match illustrates a chemical change, and the match’s ability to burn is a chemical property of the match.
156. extensive
An extensive property depends on exactly how much of a substance is present. Mass is an example of an extensive property.
157. intensive physical
Density is the ratio of an object’s mass to its volume. This physical property can help chemists identify a substance, especially because density is also an intensive property (one that stays the same no matter how much of the substance you have).
158. extensive physical
Length is a physical property that describes size. Length is extensive because it depends on exactly how much of a substance you have.
159. intensive physical
Color is a physical property that’s intensive. A piece of paper that’s green is still green if you cut the paper in half; the color is independent of the size of the paper.
160. chemical
Flammability is a chemical property because if a flammable substance catches fire, the substance changes into new substances. For example, as wood burns, carbon dioxide and water escape, and a residue of ash remains behind.
161. extensive physical
Mass is a physical property that describes how much of a substance you have. Mass is extensive because it depends on exactly how much is present — more matter, more mass.
162. intensive physical
Odor is a physical property that’s intensive. A small orange has a distinctive odor that’s essentially the same as the odor of a large orange.
163. intensive physical
Ductility is a physical property that describes how a substance can be drawn into thin wires. A small block of copper is just as ductile as a large block of copper. Ductility is based on the identity of the substance, not how much is present, so it’s an intensive property.
164. intensive physical
Conductivity is a physical property that’s intensive. A piece of silver metal will conduct electricity whether it’s 10 cm long or 30 cm long. The fact that silver is a good conductor is based on its identity.
165. intensive physical
Solubility is a physical property because you can evaporate away the solvent and be left with the solute. Solubility is intensive; whether something dissolves or not depends more on the nature of the substance and the solvent than on the amount.
166. 12 g/cm3
Density is the ratio of the mass of a substance to its volume. Enter the numbers in the formula and do the math:
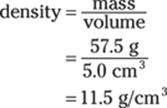
The 5.0 cm3 measurement limits the answer to two significant figures. Rounded to two significant figures, the answer is 12 g/cm3.
167. 0.884 g/mL
To find the density in grams per milliliter, divide the mass (in grams) by the volume (in milliliters):
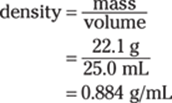
Both the mass and the volume measurements have three significant figures, so the answer has three significant figures as well.
168. 1.98 kg/m3
First, convert the number of grams to kilograms; you can do this by multiplying 3,960 g by 1 kg/1,000 g:
![]()
To get the density, divide the mass by the volume:
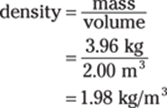
Both the mass and the volume measurements have three significant figures, so the answer has three significant figures as well.
169. 240 g
Because density = mass/volume, you can multiply density by volume to find the mass. Here’s the initial setup:
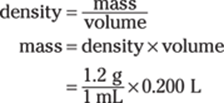
Before you do the math, you need to make the volume units — milliliters and liters — consistent so they’ll cancel out, leaving you with the mass in grams. Because 1 L = 1,000 mL, you can multiply by the conversion factor 1,000 mL/1 L. Here are the calculations, with the conversion factor in parentheses:
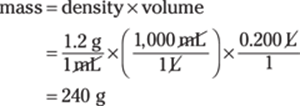
The density measurement, 1.2 g/mL, has only two significant figures, limiting your answer to two significant figures. The answer is 240 g.
170. 73 g
Because density = mass/volume, you can multiply density by volume to find the mass:
![]()
You have a cube with a side that’s 3.00 cm long. The volume of a cube is equal to length × width × height, or side3, so the cube’s volume is V = (3.00 cm)3 = 27.0 cm3. To find the mass, multiply the density by the volume:
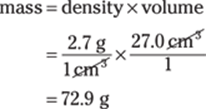
The density measurement (2.7 g/cm3) has only two significant figures, limiting the answer to two significant figures, so round the answer to 73 g.
171. 4.0 g/cm3
Density is the mass divided by the volume. You need both the mass of the block and its volume, so first solve for the volume of the block:
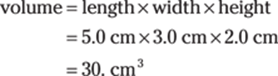
Then enter the mass and volume in the density formula:
![]()
The given volume measurement (30. cm3) has only two significant figures, so you include two significant figures in your answer.
172. 29 kg
You need to convert several units in this problem, so make a plan. Start by putting the quantity that has only one unit (the 1.5 L) over 1; writing the number this way keeps it from falling into the denominator.
Note that the density gives you a conversion factor (19.3 g = 1 cm3) that lets you relate the given volume to the mass you want to find. Your plan for the conversions in this problem may look something like this: L → mL → cm3 → g → kg. Here are the calculations with the appropriate units canceled out:
![]()
The given volume (1.5 L) has only two significant figures, so your answer should have two significant figures as well. Rounding to two significant figures gives you an answer of 29 kg.
173. 110 mL
Density is the mass divided by the volume, and you want to find the volume. You can start by solving the density equation for volume:
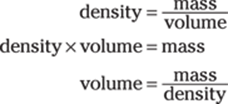
Now just enter the numbers and do the math:
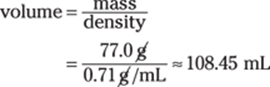
The given density measurement (0.71 g/mL) has only two significant figures, so the answer should have two significant figures as well; 108.45 mL rounds to 110 mL.
174. 4.00 cm
You’re given the cube’s mass and density. Density is the mass divided by the volume, so you can solve for the volume of the cube using the density formula:
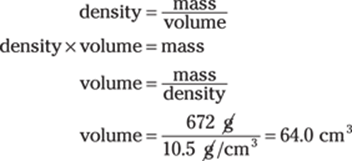
Then solve for the length of a side of the cube. The volume, V, of a cube is side × side × side, or s3. Enter the volume in the formula and solve for s:
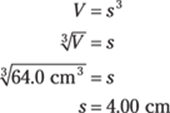
All the measurements in this problem have three significant figures, so the answer also has three significant figures.
175. calorie
A calorie is the amount of energy required to raise the temperature of 1 g of water by 1°C. A calorie equals 4.184 joules (J).
176. kilojoules/mole
The heat content of chemicals is measured in kilojoules per mole (kJ/mol).
177. Calorie (kilocalorie)
The food calorie is really a kilocalorie (kcal), or 1,000 calories. You can also indicate a food calorie by writing Calorie with a capital C.
A calorie (small c) is the amount of heat necessary to raise the temperature of 1 g of water by 1°C.
178. potential; position
Objects stored above ground level have energy based on their position, which is potential energy.
179. kinetic; motion
A rolling ball is moving, so it has kinetic energy, the energy of motion.
Note: If the ball is rolling downhill, it has a combination of potential and kinetic energy until it reaches the bottom of the hill. At the bottom, all the potential energy (energy of position) will have changed to kinetic energy.
180. stored; potential
Fuels contain chemical energy stored in the bonds between elements. This chemical energy is potential energy.
181. 6.207 kcal
In this problem, start by writing the number of joules over 1. A calorie equals 4.184 J, so convert joules to calories by multiplying the number of joules by 1 cal/4.184 J. Then convert to kilocalories by multiplying by 1 kcal/1,000 calories: J → cal → kcal:
![]()
The answer has four significant figures because the given measurement, 25,970 J, has four significant figures. The conversions here are exact numbers, so they have no bearing on the number of significant figures in the answer.
182. 1.3 × 1012 J
To find the number of joules from a given number of kilocalories, first convert kilocalories to calories by multiplying by 1,000 kcal/1 cal. Then convert calories to joules by multiplying by 4.184 J/1 cal: kcal → cal → J:
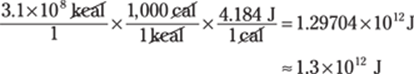
The answer has two significant figures because the given energy measurement has two significant figures. The conversions involve exact numbers, so they have no bearing on the number of significant figures in the answer.
183. degrees Celsius, degrees Fahrenheit, and kelvins
Average kinetic energy is the definition of temperature. You can measure temperature in kelvins, degrees Celsius (°C), or degrees Fahrenheit (°F).
184. 353.2 K
To find kelvins from degrees Celsius, add 273.15.
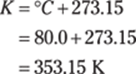
The given measurement (80.0°C) has only one decimal place, so round the answer to the tenths place. The answer is 353.2 K.
185. 801°C
To solve for degrees Celsius from kelvins, use the Kelvin temperature equation:
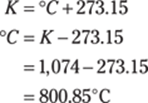
Rounded to the ones place, the answer is 801°C.
186. –297°F
To convert from degrees Celsius to degrees Fahrenheit, use the formula ![]() . Be sure to follow the order of operations by doing the multiplication and division before the addition:
. Be sure to follow the order of operations by doing the multiplication and division before the addition:
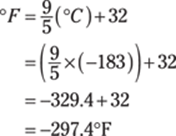
The answer should have three significant figures because the original measurement (183°C) has three significant figures; the conversions use exact numbers, so they have no bearing on the number of significant figures in the answer. Rounded correctly, the answer is –297°F.
187. 37.0°C
To go from degrees Fahrenheit to degrees Celsius, use the formula ![]() . Follow the order of operations by completing the subtraction in the parentheses before multiplying by 5 and dividing by 9:
. Follow the order of operations by completing the subtraction in the parentheses before multiplying by 5 and dividing by 9:

Write the answer with three significant figures, just like the original number. The answer is 37.0°C.
188. –110°F
To go from degrees Fahrenheit to degrees Celsius, use the formula ![]() . Plug in –78°C for the °C and solve.
. Plug in –78°C for the °C and solve.
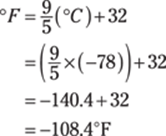
Rounded to two significant figures, the answer is –110°F. The answer has two significant figures because the given measurement (–78°C) has two significant figures; the conversions are exact numbers, so they don’t have any bearing the on the significant figures in the answer.
189. 318 K
To find the Kelvin temperature from degrees Fahrenheit, first convert from Fahrenheit to Celsius:
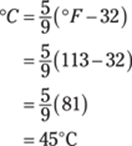
Then you can convert from degrees Celsius to kelvins by adding 273.15:
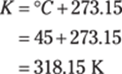
Rounded to the ones place, the answer is 318 K.
190. 80.°F
To find the temperature in degrees Fahrenheit, first convert the temperature from kelvins to degrees Celsius by subtracting 273.15 from the Kelvin temperature:
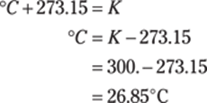
Next, use the formula ![]() to convert to degrees Fahrenheit:
to convert to degrees Fahrenheit:
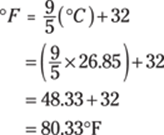
This answer rounds to 80.°F.
191. 11 protons
The atomic number of an element is equal to the number of protons. Sodium, Na, is atomic number 11, so it has 11 protons.
192. 35 electrons
The number of protons is equal to the number of electrons in an atom, which is neutral. Bromine, Br, is atomic number 35. It has 35 protons, so it has 35 electrons.
193. 28 electrons
Nickel, Ni, has 28 protons. Because the number of protons equals the number of electrons in an atom, an atom of nickel contains 28 electrons.
194. 86 protons
The number of protons in an atom is equal to the element’s atomic number. Radon, Rn, is atomic number 86, so it has 86 protons.
195. 21 neutrons
To find the number of neutrons in an atom, subtract the atomic number (which equals the number of protons) from the atom’s mass number (which equals the number of protons + the number of neutrons). Here are the calculations for potassium-40:
![]()
196. The atomic number is equal to the number of protons.
The general definition of atomic number is the number of protons in an atom.
197. The mass number is equal to the number of neutrons plus the atomic number (the number of protons).
The majority of the mass of the atom is in the nucleus, where you find the protons and neutrons, collectively called nucleons. By adding the number of neutrons to the atomic number (which equals the number of protons), you get the mass number.
198. 29 protons, 29 electrons, and 34 neutrons
The number of protons is equal to the atomic number; therefore, copper, Cu, has 29 protons. Because the atom is neutral, the number of protons equals the number of electrons.
To get the number of neutrons, subtract the number of protons (29) from the mass number. The mass number, 63, follows the name of the element in copper-63, so the number of neutrons is 63 – 29 = 34.
199. 6 protons and 8 neutrons
If the atom has 6 electrons, then it has 6 protons, because the positive and negative charges have to cancel each other out. The number of neutrons is equal to the mass number minus the number of protons: 14 – 6 = 8.
200. mass number 91, 40 protons
The number of protons equals the number of electrons in an atom, so 40 electrons means 40 protons. The element is atomic number 40, zirconium (Zr).
The mass number is equal to the number of protons plus the number of neutrons. The atom contains 51 neutrons, so the mass number is 40p + 51n = 91.
201. the mass number
In isotope notation ![]() , the top number A represents the mass number, which is the sum of the number of protons and neutrons in the nucleus of the atom.
, the top number A represents the mass number, which is the sum of the number of protons and neutrons in the nucleus of the atom.
202. the number of protons
In isotope notation ![]() , the bottom number Z represents the atomic number, which is the same as the number of protons in the nucleus of the atom.
, the bottom number Z represents the atomic number, which is the same as the number of protons in the nucleus of the atom.
203. 9 protons, 9 neutrons
In isotope notation, the bottom number is equal to the number of protons, so ![]() has nine protons. The number of neutrons is equal to the top number (mass number) minus the bottom number (atomic number): 18 – 9 = 9 neutrons.
has nine protons. The number of neutrons is equal to the top number (mass number) minus the bottom number (atomic number): 18 – 9 = 9 neutrons.
204. 11 protons, 14 neutrons
The bottom number in isotope notation is the atomic number, which is equal to the number of protons, so ![]() has 11 protons. The number of neutrons is equal to the mass number (the top number) minus the atomic number (the bottom number): 25 – 11 = 14 neutrons.
has 11 protons. The number of neutrons is equal to the mass number (the top number) minus the atomic number (the bottom number): 25 – 11 = 14 neutrons.
205. ![]()
In isotope notation, carbon-12 has the atomic symbol C with a mass number of 12 on top. The bottom number is the atomic number of carbon, 6. The mass number follows the name of the element in the isotope name (carbon-12), and you can find the element’s symbol and atomic number on the periodic table.
206. ![]()
In isotope notation, chlorine-37 has the atomic symbol Cl with a mass number of 37 on top. The bottom number is the atomic number of chlorine, 17. The mass number follows the name of the element in the isotope name (chlorine-37), and you can find the element’s symbol and atomic number on the periodic table.
207. argon-35
Ar is the symbol for argon. When you write the isotope name, the top number in isotope notation (the mass number) becomes the number that follows a hyphen and the element’s name: argon-35.
208. An ion has a different number of electrons from the atom.
An ion is an atom that has either gained or lost electrons.
If, instead, the number of protons changes, then the element’s identity is changed (transmutation). If the number of neutrons changes, you have an isotope.
209. isoelectronic
The prefix for same is iso-, so two atoms or ions with the same number of electrons are said to be isoelectronic.
210. The number of protons is greater than the number of electrons.
Protons are positive and electrons are negative, so for an ion to be positive, it needs more protons than electrons. An ion with a positive charge has lost electrons.
211. The number of protons is less than the number of electrons.
For an ion to have a negative charge, it must have more electrons than protons. An ion with a negative charge has gained electrons.
212. 20 protons and 18 electrons
![]() has a positive charge, so the number of protons must be greater than the number of electrons. The atomic number of Ca is 20 (the bottom number in isotope notation), giving you 20 protons. To find the number of electrons, simply subtract the charge from the number of protons: 20 – 2 = 18 electrons.
has a positive charge, so the number of protons must be greater than the number of electrons. The atomic number of Ca is 20 (the bottom number in isotope notation), giving you 20 protons. To find the number of electrons, simply subtract the charge from the number of protons: 20 – 2 = 18 electrons.
213. 53 protons and 54 electrons
For an ion to be negative, it needs to have more electrons than protons. Iodine has an atomic number of 53, so it has 53 protons. Because the iodine ion has a –1 charge, the ion must have one more electron than proton, giving you a total of 54 electrons.
214. 13 protons and 10 electrons
For an ion to be positive, the atom must have more protons than electrons. ![]() needs to have three more protons than electrons, because the charge is +3. With an atomic number of 13, the aluminum ion must have three fewer electrons than protons: 13 – 3 = 10 electrons.
needs to have three more protons than electrons, because the charge is +3. With an atomic number of 13, the aluminum ion must have three fewer electrons than protons: 13 – 3 = 10 electrons.
215. 15 protons and 18 electrons
Sometimes you need to look up the atomic number to determine the number of protons. The atomic number of phosphorus, P, is 15, so phosphorus has 15 protons. You can find the number of electrons by subtracting the charge from the number of protons: 15 – (–3) = 15 + 3 = 18 electrons.
216. ![]()
To write the isotope notation, start with the symbol for silver, Ag. To the left of the symbol and slightly above the base line, write 109, the mass number that follows the hyphen after the element’s name. Then write the atomic number, 47 (from the periodic table), below 109. Write a plus sign as the superscript after the Ag to indicate the charge; writing the 1 isn’t necessary because it’s understood.
217. ![]()
The symbol for sulfur is S. Write the mass number, 34, to the upper left of the S. Sulfur has an atomic number of 16, so write 16 under the 34. Represent the charge of negative 2 by writing the superscript 2– after the S.
218. 24 protons, 28 neutrons, 18 electrons
Chromium, Cr, has an atomic number of 24, which is equal to the number of protons. If the mass number is 52, then the atom contains 28 neutrons, because the mass number minus the atomic number (from the periodic table) gives you the number of neutrons: 52 – 24 = 28 neutrons.
To get the number of electrons, subtract the ion’s charge from the number of protons: 24 – 6 = 18 electrons. This answer makes sense because a +6 charge means that there are six more protons than electrons.
219. 28 protons, 34 neutrons, 25 electrons
Nickel, Ni, has an atomic number of 28, which is equal to the number of protons. Because the top number in the isotope notation is 62, the mass number is 62. To find the number of neutrons, subtract the atomic number (from the periodic table) from the mass number: 62 – 28 = 34 neutrons.
To get the number of electrons, subtract the ion’s charge from the number of protons: 28 – 3 = 25 electrons. This answer makes sense because a +3 charge means that there are three more protons than electrons.
220. Hund’s rule
“Electrons fill subshells singly before doubly” is a shortened restatement of Hund’s rule.
221. Aufbau principle
The Aufbau principle describes the order in which electrons fill orbitals.
222. 14
There are seven f orbitals. Each orbital can hold a maximum of two electrons, so a maximum of fourteen electrons are in the f orbitals.
223. 6
There are three p orbitals. Each orbital can hold a maximum of two electrons, so a maximum of six electrons are in the p orbitals.
224. 1s22s22p2
Carbon, C, has an atomic number of 6, which means that it has six protons and six electrons. Electrons fill orbitals from lowest energy to highest energy, so the first energy level gets filled before the second energy level. Remember that the s orbital can hold a maximum of two electrons and that the p orbitals can hold a maximum of six electrons.
To check the electron configuration, note that adding the superscripts on 1s22s22p2 gives you 2 + 2 + 2 = 6 electrons.
225. 1s22s22p63s2
Magnesium, Mg, has an atomic number of 12, so it has 12 protons and 12 electrons. Electrons fill orbitals from the first energy level up to the third in magnesium. The first energy level contains two electrons, the second energy level contains eight electrons (two in an s orbital and six in a p orbital), and the third energy level contains the last two electrons, for a total of 12 electrons.
226. 1s22s22p63s23p6
Argon, Ar, has an atomic number of 18, so it has 18 protons and 18 electrons. The electrons fill the first and second energy levels completely (two in each s orbital and six in the p orbital). The remaining electrons fill the 3s and 3p orbitals.
227. 1s22s22p63s23p64s23d104p5
An atom of bromine, Br, has 35 protons and 35 electrons. The first energy level can hold a maximum of two electrons, and the second energy level can hold a maximum of eight electrons (two in the s orbital and six in the p orbital). The third energy level can hold a maximum of 18 electrons (two in an s orbital, six in p orbitals, and 10 in d orbitals), and the fourth energy level contains the remainder of the electrons. Remember that the 4s orbital doesn’t require as much energy as the 3d orbital in order to fill with electrons, so the 4s orbital fills first.
228. 1s22s22p63s23p64s23d104p65s24d2
An atom of zirconium, Zr, has 40 protons and 40 electrons. The first energy level contains two electrons, the second energy level contains eight electrons (two in the s orbital and six in the p orbital), the third energy level contains 18 electrons, and the fourth energy level contains the remainder of the electrons. Remember that the 5s orbital doesn’t require as much energy as the 4d orbital in order to fill with electrons, so the 5s orbital fills first.
229. 1s22s22p63s23p64s23d104p65s24d105p66s24f145d106p67s25f6
Finding the expected electron configuration of plutonium, Pu, is hard because there are so many electrons. When you get down to the bottom of the periodic table, electrons are in every energy level. Just be sure to add them in the correct order based on the energy it takes to fill the orbitals.
Adding the superscripts in the expected electron configuration, 1s22s22p63s23p64s23d104p65s24d105p66s24f145d106p67s25f6, gives you 2 + 2 + 6 + 2 + 6 + 2 + 10 + 6 + 2 + 10 + 6 + 2 + 14 + 10 + 6 + 2 + 6 = 94 electrons.
230. the spin quantum number
The spin quantum number, s, describes the direction of the spin of the electron. According to this model, the electron spins either clockwise or counterclockwise.
231. the principal quantum number
The principal quantum number, n, describes the average distance between the nucleus and the orbital, with the first energy level at a distance of 1.
232. the magnetic quantum number
The magnetic quantum number, ml, describes how the orbitals are oriented in three-dimensional space.
233. the angular momentum quantum number
The angular momentum quantum number, l, describes the shape of the orbital.
234. ![]() ,
, ![]()
An electron can spin in only two directions: clockwise or counterclockwise. Because direction is relative, each has a numeric value of 1/2 assigned to the electron; one direction is considered positive, and the other is negative.
235. average atomic mass
The decimal number found in most blocks on the periodic table is the weighted average of the relative abundance of all known isotopes. The more common an isotope is, the more it affects the average atomic mass.
236. 6.940 amu
To find the average atomic mass, take the percent abundance and multiply it by the given mass of the element. Repeat for as many isotopes as are given. Then add the multiplied numbers together:
237. 35.45 amu
You have two isotopes, so take the percent of each isotope, multiply it by the given mass of the isotope, and then add the two numbers together:
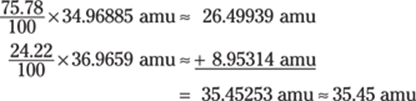
238. 24.31 amu
Multiply each atomic mass by the percent abundance of each isotope and add the three numbers together:
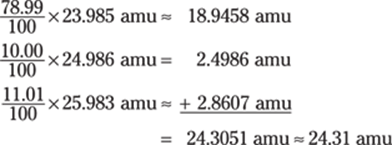
239. 39.098 amu
Multiply each atomic mass by the percent abundance of each isotope and add the three numbers together. Be careful in this question — remember to divide the percent that’s less than 1 by 100 just like the other percent abundances:
240. 55.845 amu
Multiply each atomic mass by the percent abundance of each isotope and add the four numbers together. Don’t forget to divide the percent that’s less than 1 by 100 just like the other percent abundances.
241. 83.80 amu
Multiply each atomic mass by the percent abundance of each isotope and add the six numbers together. What makes this problem challenging is the sheer number of numbers and decimal places that you have to keep up with. Having a calculator that can retain your display until you finish entering the numbers is a definite plus here.

242. 80.% boron-11
To solve this problem, you need to rely on your algebra skills. You have two isotopes of boron, and you have to solve for the percent abundance of one of the isotopes. So here’s your initial equation:
(% B-11)(mass B-11) + (% B-10)(mass B-10) = average atomic mass
Next, substitute in the given numbers from the problem:
(% B-11)(11.009306 amu) + (% B-10)(10.012397 amu) = 10.81 amu
You have two unknown percentages, and you need to figure out how they’re related. The two percentages have to add up to 100%: (% B-11) + (% B-10) = 100%. In decimal form, 100% equals 1, so if you consider the amount of boron-11 to be x, then the amount of boron-10 has to be 1 – x.
By substituting these values into the initial equation and solving for x, you can find the percent of boron-11:
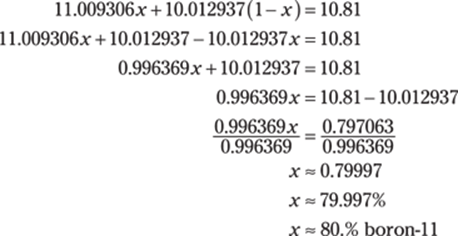
The subtraction in the fourth line (10.81 – 10.012937) limits the answer to two decimal places (two significant figures), which in turn limits the final answer to two significant figures.
243. nuclear fusion
Nuclear fusion is the process that occurs in the sun to give off energy. During nuclear fusion, two atomic nuclei join (fuse) to form a heavier nucleus.
244. nuclear fission
Nuclear fission is the process by which large atoms are broken into smaller atoms, producing large amounts of energy as well as additional neutrons as byproducts.
245. alpha decay
An alpha particle is a helium nucleus (two protons and two neutrons), so when a helium nucleus is ejected from the nucleus of an atom, the nucleus is said to have undergone alpha decay.
246. gamma decay
A ![]() ray, or gamma ray, is evidence of gamma decay. The superscript 0 is the mass number, the subscript 0 is the atomic number, and γ is the Greek letter gamma. A gamma ray is a high-energy form of electromagnetic radiation (light).
ray, or gamma ray, is evidence of gamma decay. The superscript 0 is the mass number, the subscript 0 is the atomic number, and γ is the Greek letter gamma. A gamma ray is a high-energy form of electromagnetic radiation (light).
247. ![]()
Beta decay produces a beta particle, which is essentially an electron. The superscript 0 is the mass number, the subscript –1 is the atomic number, and e is the symbol for an electron or positron. You can also write beta decay as ![]() .
.
248. positron emission
A ![]() particle is a positron. The superscript 0 is the mass number, the subscript +1 is the atomic number, and e is the symbol for a positron or electron. When a positron is produced in a nuclear reaction, the atom is said to have undergone positron emission.
particle is a positron. The superscript 0 is the mass number, the subscript +1 is the atomic number, and e is the symbol for a positron or electron. When a positron is produced in a nuclear reaction, the atom is said to have undergone positron emission.
249. electron capture
An easy way to distinguish between electron capture and beta decay is to determine where the electron is in the equation. In beta decay, the electron is on the product side of the reaction, but with electron capture, the electron is on the reactants side. In this reaction, the beryllium atom has captured the beta particle, making a lithium atom.
For the beryllium atom (Be), electron (e), and lithium atom (Li), the superscripts are the mass numbers, and the subscripts are the atomic numbers. In a nuclear equation, the sum of the superscripts on each side must be equal, and the sum of the subscripts on each side must be equal.
250. alpha decay
The emission of an alpha particle — two protons and two neutrons — from a nucleus is evidence of alpha decay. Remember that an alpha particle is the same thing as a helium nucleus.
For the uranium atom (U), thorium atom (Th), and helium (He), the superscripts are the mass numbers and the subscripts are the atomic numbers. In a nuclear equation, the sum of the superscripts on each side must be equal, and the sum of the subscripts on each side must be equal.
251. gamma decay
The emission of a gamma ray ![]() is evidence of gamma decay.
is evidence of gamma decay.
252. beta decay
Beta decay occurs when a beta particle, or electron, is emitted from a nucleus. For the phosphorus atom (P), sulfur atom (S), and electron (e), the superscripts are the mass numbers, and the subscripts are the atomic numbers. In a nuclear equation, the sum of the superscripts on each side must be equal, and the sum of the subscripts on each side must be equal.
253. ![]()
The missing particle in this reaction is the only reactant. In nuclear reactions, the mass numbers on the left side of the arrow have to add up to the sum of the mass numbers on the right side of the arrow. That means the mass number of the missing particle on the left has to be 220 + 4 = 224.
Similarly, the atomic numbers on the left of the arrow have to add up to the sum of the atomic numbers on the right side of the arrow. Therefore, the atomic number of the missing particle on the left has to be 86 + 2 = 88.
Use the atomic number to identify the element symbol to finish the isotope notation. The element with atomic number 88 is radium, Ra.
254. ![]()
Because the total mass numbers and atomic numbers on the left and right sides of the arrow have to be equal, use a little algebra and solve for the missing piece. Subtract the numbers for helium from the numbers for curium: The mass number of the missing particle is 247 – 4 = 243, and the atomic number of the missing particle is 96 – 2 = 94.
Then look up the identity of the element whose atomic number is 94 to get the symbol, Pu, for plutonium.
255. ![]()
By subtracting the mass numbers and atomic numbers of the elements on the left side of the arrow from the mass numbers and atomic numbers of the isotopes on the right side of the arrow, you can find the missing piece of this equation. Take the plutonium numbers minus the americium numbers. The mass number of the missing particle is 241 – 241 = 0, and the atomic number of the missing particle is 94 – 95 = –1.
No element has an atomic number with a charge, so this must be something that basically has no mass and a charge of –1, which is a beta particle (electron).
256. ![]()
When gamma ray emission occurs, the same element with the same mass and atomic numbers shows up on both sides of the equation. No mass was lost, so the emitted particle must be a gamma ray, ![]() .
.
257. ![]()
To find the missing part of this equation, subtract the mass numbers and atomic numbers of the isotopes on the left side of the arrow from the mass numbers and atomic numbers of the isotope on the right side of the arrow. Take the niobium numbers minus the beta particle numbers. The mass number of the missing particle is 93 – 0 = 93, and the atomic number of the missing particle is 41 – (–1) = 42.
Next, use the periodic table to look up atomic number 42 to get the symbol, Mo, for molybdenum.
258. ![]()
This question involves two isotopes on the right side of the arrow, so add together their mass numbers and then their atomic numbers. The sum of the mass numbers is 4 + 1 = 5, and the sum of the atomic numbers is 2 + 0 = 2.
Next, subtract the mass number and the atomic number of the hydrogen isotope on the left side of the arrow: The difference in mass numbers is 5 – 2 = 3, and the difference in atomic numbers is 2 – 1 = 1.
The resulting atomic number is 1, which is the atomic number of hydrogen. This form of hydrogen, with a mass number of 3, is a very special isotope known as tritium.
259. ![]()
This question involves two isotopes on the left side of the arrow, so add together their mass numbers and then their atomic numbers. The sum of the mass numbers is 4 + 14 = 18, and the sum of the atomic numbers is 2 + 7 = 9.
Next, subtract the mass number and the atomic number of the hydrogen isotope on the right side of the arrow. The difference in mass numbers is 18 – 1 = 17, and the difference in atomic numbers is 9 – 1 = 8.
The resulting atomic number is 8, which is the atomic number of oxygen, O.
260. ![]()
To solve for the missing piece of this equation, not only do you have to add together the two parts on the left side of the arrow, but you also have to combine the two parts on the right side of the arrow. On the left, the sum of the mass numbers is 235 + 1 = 236, and the sum of the atomic numbers is 92 + 0 = 92. On the right, the sum of the mass numbers is 134 + 100 = 134, and the sum of the atomic numbers is 54 + 38 = 92.
Next, subtract the sums on the right side of the arrow from the sums on the left. The difference in mass numbers is 236 – 234 = 2, and the difference in atomic numbers is 92 – 92 = 0.
The missing part has an atomic mass of 2 and no protons or electrons. A neutron has a mass of 1 and has no charge, so two neutrons must be missing from the right side of this equation.
261. 12.5 g
To find the amount of undecayed atoms that remain after a certain number of half-lives, you can take the original amount and divide it by 2 as many times as there are half-lives. Visually, the process could look like this:
![]()
Or mathematically, you can write the mass of undecayed atoms as
![]()
which simplifies to the following:
![]()
where n is the number of half-lives that have occurred. Here are the calculations for this problem:
![]()
262. 43.8 g
To find the amount of decayed atoms, first calculate the mass of atoms that remain undecayed after a certain number of half-lives. You can take the original amount and divide it by 2 as many times as there are half-lives. Visually, the process could look like this:
![]()
Or mathematically, you can write the mass of undecayed atoms as
![]()
where n is the number of half-lives that have occurred. Enter the numbers and solve:
![]()
Then subtract the mass of undecayed atoms from the original sample to find out how much of the sample has decayed: 50.0 g – 6.25 g = 43.75 g, which is 43.8 g when you round the answer to the tenths place.
263. 330. g
To find the size of the original sample, simply multiply the mass of the final sample by 2 six times, because it went through six half-lives:
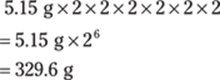
264. 4
The amount of undecayed material is the original amount of material divided by 2 raised to n, a power equal to the number of half-lives:
![]()
Enter the numbers and start to solve for the number of half-lives:
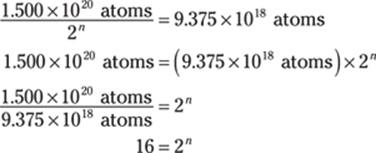
Take the log of both sides of the equation and finish solving for n:
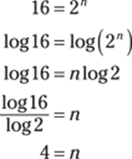
265. ![]()
First, determine how many half-lives have passed by dividing the total time elapsed by the duration of one half-life:
![]()
Next, divide 1 by 2 the number of times equal to the number of half-lives:
![]()
266. 24.8 g
First, determine how many half-lives have passed by dividing the total time elapsed by the length of one half-life:
![]()
Next, determine how many grams remain undecayed by dividing 25.0 g by 2 raised to the number of half-lives:
![]()
Finally, subtract the grams of undecayed iodine-131 from the original number of grams: 25.0 g – 0.195 g = 24.8 g decayed.
267. 176 g
First, determine how many half-lives have passed. To do this, divide the total number of years by the length of one half-life:
![]()
After you know how many half-lives have passed, you can get the size of the original sample. Multiply the 11 grams that remain undecayed by 2 multiplied by itself four times (the number of half-lives):
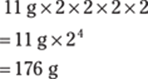
268. 1 minute
On this question, you first need to determine how many half-lives have passed. The amount of undecayed material is the original amount of material divided by 2 raised to n, a power equal to the number of half-lives:
![]()
Plug in the numbers and start to solve for the number of half-lives:

Take the log of both sides of the equation and solve for n:
Now divide the length of time that has passed, 5 minutes, by the number of half-lives to get the length of one half-life:
![]()
269. ![]()
First, determine how many half-lives have passed by dividing the total time elapsed by the length of one half-life:
![]()
Now you can pick any sample size and think about the decay process. After one half-life, 50% of the sample remains. After a second half-life, 1/2 of the 50%, or 25%, of the sample remains. And 25% is 1/4 of the original amount.
If 1/4 of the sample remains, then 3/4 of the sample has decayed.
270. 110 hours
You first need to determine how many half-lives have passed. The amount of undecayed material is the original amount of material divided by 2 raised to n, a power equal to the number of half-lives:
The units of mass don’t match, so convert the kilograms to grams (1,000 g = 1 kg). Then start to solve for the number of half-lives:
Take the log of both sides of the equation and solve for n:
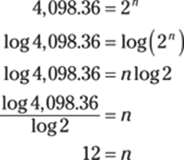
After you know how many half-lives have passed, you can determine the total amount of time by multiplying the length of one half-life by the number of half-lives:
![]()
271. C
The symbol for carbon is C.
272. Cl
The symbol for chlorine is Cl.
273. Al
The symbol for aluminum is Al.
274. Cd
The symbol for cadmium is Cd.
275. Cu
The symbol for copper is Cu. The symbol originates from the Latin word cuprum.
276. As
The symbol for arsenic is As. Ar is the symbol for argon, not arsenic.
277. Na
The symbol for sodium is Na. The symbol originates from the Latin word natrium.
278. K
The symbol for potassium is K. The symbol originates from the Latin word kalium.
279. Fe
The symbol for iron is Fe. The symbol’s origins are in the Latin word ferrum, which means iron.
280. Ag
The symbol for silver is Ag. The symbol originates from the Latin word argentum, which means silver.
281. nitrogen
The element with the symbol N is nitrogen.
282. sulfur
The element with the symbol S is sulfur.
283. bromine
The element with the symbol Br is bromine.
284. phosphorus
The element with the symbol P is phosphorus. The symbol for potassium is not P but K.
285. manganese
The element with the symbol Mn is manganese. The symbol for magnesium is Mg, not Mn.
286. astatine
The element with the symbol At is astatine.
287. radium
The element with the symbol Ra is radium. The symbol for radon is Rn.
288. mercury
The element with the symbol Hg is mercury. The symbol originates from the Latin word hydrargyrum, which means liquid silver.
289. tin
The element with the symbol Sn is tin. The symbol comes from the Latin word stannum, which means tin. The symbol for silicon is Si.
290. protactinium
The element with the symbol Pa is protactinium.
291. horizontally
A period goes horizontally — from left to right or right to left — on the periodic table.
292. touching the top and bottom of the stair-step line that starts between B and Al and goes down to Te and Po
The metalloids are elements that have characteristics of both metals and nonmetals; they form a border between the metals and nonmetals on the periodic table and include Si, Ge, As, Sb, Te, and Po. Elements (except for hydrogen) that are to the left of the stair-step line are metals, and elements to the right of the stair-step line are nonmetals.
293. in the second column of the periodic table
The alkaline earth metals include Be, Mg, Ca, Ba, and Ra, which are in the second column (Group 2 or IIA) of the periodic table.
294. in the middle of the periodic table
The transition metals are in the middle of the periodic table (from Sc to Zn and the rows directly below those ten elements). These groups are usually numbered 3 to 12 or IIIB to IIB.
295. in the rightmost column of the periodic table
The noble gases, He, Ne, Ar, Kr, Xe, and Rn, are in the rightmost column of the periodic table. This column is usually labeled Group 18, 0, or VIIIA.
296. in the bottom two rows of the periodic table
The inner transition metals (elements 58 to 71 and 90 to 103) are located in the bottom two rows of the periodic table, or between La and Hf and Ac and Rf in an expanded view of the periodic table. Elements 58 to 71 comprise the lanthanide series, and elements 90 to 103 comprise the actinide series. In both of these series, the f subshells are being filled.
297. in the leftmost column of the periodic table
Li, Na, K, Rb, Cs, and Fr are all members of the alkali metal family and are located in the leftmost column (Group 1 or IA) of the periodic table.
298. Dmitri Mendeleev
Dmitri Mendeleev arranged all the known elements of his time based on the physical properties of the elements, focusing on atomic mass.
German chemist Lothar Meyer did the same thing independently at the same time as Mendeleev, but Mendeleev receives more credit because he went further in his predictions.
299. Henry Moseley
Henry Moseley, an English physicist, is credited with arranging the modern periodic table with the elements in order of increasing atomic number.
300. above and to the right of the stair-step line that starts between B and Al and goes down to Te and Po
Most of the elements that exist as gases at room temperature (N, O, F, Cl, He, Ne, Ar, Kr, Xe, and Rn) are above and to the right of the stair-step line. The only exception is H, hydrogen, although some versions of the periodic table do place hydrogen above fluorine, in the halogen family.
301. the alkali metals
Potassium, K, is in the leftmost column of the periodic table, the alkali metal family.
302. the transition metals
Silver, Ag, is under copper, Cu, in the transition metals.
303. the oxygen family
Selenium, Se, is in the same column as oxygen, O, so selenium is a member of the oxygen family.
304. the carbon family
Tin, Sn, is two blocks below carbon on the periodic table, making tin a member of the carbon family.
305. the halogen family
Iodine, I, is in the same family as F, Cl, Br, and At, the halogen family.
306. the alkaline earth metals
Calcium, Ca, is in the same family as Be, Mg, Sr, Ba, and Ra, making calcium a member of the alkaline earth metals.
307. the boron family
Aluminum, Al, is under boron, B, on the periodic table, so aluminum is in the boron family.
308. the inner transition metals
The heaviest naturally occurring element is uranium, U. It’s in the inner transition metals on the periodic table.
309. the transition metals
Mercury, Hg, is the only metal that’s liquid at room temperature. Mercury is in the transition metals on the periodic table.
310. the halogen family
Bromine, Br, is the only nonmetal that’s a liquid at room temperature. Bromine is in the halogen family (Group 17 or VIIA).
311. the boron family
Members of the boron family (Group 13 or IIIA) have three valence electrons. Their electron configurations have s2p1 in their outermost energy level.
312. the nitrogen family
The members of the nitrogen family (Group 15 or VA) have five valence electrons. Their electron configurations have s2p3 in their outermost energy level.
313. the alkaline earth metals
The alkaline earth metals (Group 2 or IIA) have two valence electrons in their s orbital.
314. increase; increase
As you go across a period (row), the masses of the elements generally increase — with just a few exceptions. As you go down a period (column), the masses of the elements always increase.
315. decrease; increase
The radii of the elements generally decrease going across a period from left to right, and the radii increase going down a family.
316. ionization energy
Ionization energy is the amount of energy needed to remove an electron from a gaseous atom. The ion that forms has fewer electrons than protons, giving the ion a net positive charge.
317. electron affinity
Affinity means attraction, so if you have a chocolate affinity, you’re attracted to chocolate and would gladly add some to your collection. Similarly, if an atom has a high electron affinity, it has an attraction for electrons and would want to add them to its collection.
318. larger than; more
An anion is a negatively charged ion. This means that an anion has more negatively charged particles (electrons) than positively charged particles (protons). The ionic radius of an anion is larger than the radius of the atom because the ion has more electrons (and less of an effective nuclear charge) than the atom.
319. decrease; increases
The atomic radii of the elements decrease in size going from left to right across a period because with each successive element, an additional proton is added to the nucleus. These additional protons pull the electrons in closer to the nucleus, making the distance across the atom smaller.
320. Be < Ca < Ba
Ba, Be, and Ca (barium, beryllium, and calcium) are all in the same family. The atomic radius increases as you go down a family because more energy levels are being added, so Be < Ca < Ba.
321. Cl < S < P
Cl, P, and S (chlorine, phosphorus, and sulfur) are all in the same period on the periodic table. Within a period, the effective nuclear charge increases along with the atomic number. The greater positive charge in the nucleus pulls on the electrons more strongly, leading to a smaller atomic radius.
322. Li < B < C
B, C, and Li (boron, carbon, and lithium) are in the same period on the periodic table. Ionization energy increases going from left to right in a period because the additional protons pull electrons closer to the nucleus, so removing an electron takes more energy.
323. I < Br < Cl
Br, Cl, and I (bromine, chlorine, and iodine) are in the same family. In a family, the ionization energy decreases from top to bottom because the larger the atom is, the less energy it takes to remove an electron. All the extra electrons create shielding between the nucleus and the valence (outermost) electrons.
324. N < O < F
F, O, and N (fluorine, oxygen, and nitrogen) are in the same period on the periodic table. Within a period, the nucleus with the most protons attracts the electrons most strongly. N has the fewest protons of the given elements, so it has the lowest electron affinity; F has the most protons, so it has the highest electron affinity.
325. Te < Se < S
S, Se, and Te (sulfur, selenium, and tellurium) are in the same family on the periodic table. Within a family, the atomic radius increases with increasing atomic number.
The atom with the smallest radius has the highest electron affinity. The atom with the largest atomic radius has a hard time keeping the electrons that it has, so it has low ionization energy. Te has the largest radius and S has the smallest, so Te < Se < S.
326. Sn > Si > Cl
Chlorine (Cl) is a nonmetal, silicon (Si) is a metalloid, and tin (Sn) is a metal. When putting these elements in order from most to least metallic, place Sn before Si and Si before Cl.
327. Mg < Na < K
For any period on the periodic table, the effective nuclear charge increases with increasing atomic number. And for any column on the periodic table, the number of occupied energy levels increases with increasing atomic number.
Mg (magnesium) has a smaller radius than Na (sodium) because Mg has a larger effective nuclear charge. Na has a smaller radius than K (potassium) because Na has one fewer energy level.
328. S2– > O2– > F–
Adding electrons increases the radius. The greater the number of electrons added, the greater the increase in the radius, because the electrons spread out as they repel each other. This is why both O2– and S2– are larger than F–. S, which is lower on the periodic table than O, has an additional energy level, so it’s larger.
329. F > Li > Cs
The ionization energy of F (fluorine) is greater than the ionization energy of Li (lithium) because F has the greater effective nuclear charge. Cs (cesium) has a lower ionization energy than Li because the Cs atom has more shielding from the large number of electrons that exist between the nucleus and the valence electrons.
330. Ba < Bi < N
Within a period, effective nuclear charge increases with increasing atomic number, and within a column, atomic radius increases with increasing atomic number.
Ba (barium) has a lower ionization energy than Bi (bismuth) because Bi has a greater effective nuclear charge. However, Bi has a lower ionization energy than N (nitrogen) because removing an electron from Bi requires less energy; the valence electrons in Bi are farther away from their nucleus than the valence electrons in N are from their nucleus.
331. sodium chloride
A binary ionic compound contains two elements, a metal and a nonmetal. The metal is present as a cation (positive ion), and the nonmetal is present as an anion (negative ion).
The cation name, which matches the original metal name (sometimes with info on the oxidation state), always goes before the anion name. The name of the nonmetal is altered to have an -ide suffix (sometimes requiring minor spelling changes). Unlike molecular compounds, ionic compounds do not use prefixes.
In this case, NaCl is composed of Na+ and Cl–. Na+ is the sodium ion, and Cl– is the chloride ion. Therefore, NaCl is called sodium chloride.
332. calcium oxide
CaO is a compound made of calcium ions (Ca2+) and oxide ions (O2–), so CaO is calcium oxide.
333. aluminum bromide
To name AlBr3, first name the metal: Al is aluminum. Then name the nonmetal, changing the ending to -ide. Br is bromine, which becomes bromide, so AlBr3 is aluminum bromide.
334. potassium sulfide
When naming a binary ionic compound, don’t use prefixes; just name the metal and then the nonmetal with the suffix -ide. K2S is composed of potassium ions (K+) and sulfide ions (S2–), so K2S is potassium sulfide.
335. aluminum oxide
Al2O3 is composed of aluminum ions (Al3+) and oxide ions (O2–), so Al2O3 is aluminum oxide.
336. lithium nitride
When naming a binary ionic compound, don’t use prefixes; just name the metal and then the nonmetal with the suffix -ide. Li3N is lithium nitride.
337. magnesium iodide
To name MgI2, first name the metal: Mg is magnesium. Then name the nonmetal, changing the ending to -ide. I is iodine, which becomes iodide, so MgI2 is magnesium iodide.
338. strontium selenide
SrSe is a compound made of strontium and selenium. The nonmetal selenium becomes selenide, so SrSe is strontium selenide.
339. barium fluoride
To name BaF2, first name the metal: Ba is barium. Then name the nonmetal, changing the ending to -ide. F is fluorine, which becomes fluoride, so BaF2 is barium fluoride.
340. sodium hydride
In this case, hydrogen bonds to Na, sodium. The -ogen ending on hydrogen changes to -ide, so NaH is sodium hydride.
Note: Hydrogen is an odd nonmetal because it’s on the left side of the periodic table. Hydrogen is also unusual because it forms both positive and negative ions.
341. zinc phosphide
Even though zinc (Zn) is a transition metal, it has only one oxidation state. Because only one oxidation state (+2) is possible, you don’t need to specify the charge. Phosphorus forms the phosphide ion (P3–), so Zn3P2 is zinc phosphide.
342. copper(II) bromide
Cu, copper, is a transition metal that has multiple oxidation states, so you need to use a roman numeral to designate which form of copper is in the compound.
The total charge of ions and cations has to equal zero. Because CuBr2 contains two Br ions, each with a charge of –1, the copper has to have an oxidation number of +2 to cancel out the two negative charges. The first part of the name is copper(II) to take this charge into account. The second part of the name is bromide, as bromine changes to the bromide ion.
343. gold(III) chloride
Au, gold, is a transition metal that has multiple oxidation states, so you need to use a roman numeral to designate which form of gold is in the compound.
The total charge of ions and cations has to equal zero. Because AuCl3 contains three Cl ions, each with a charge of –1, the gold has to have an oxidation number of +3 to cancel out the three negative charges. The first part of the name is gold(III) to take this charge into account. The second part of the name is chloride, as chlorine changes to the chloride ion.
344. cobalt(II) sulfide
Co, cobalt, is a transition metal that has multiple oxidation states, so you need to use a roman numeral to designate which form of cobalt is in the compound.
The total charge of ions and cations has to equal zero. Because the sulfide ion has a –2 charge, the cobalt must have a +2 charge. The first part of the name is cobalt(II) to take this charge into account. The second part of the name is sulfide, as sulfur changes to the sulfide ion. CoS is cobalt(II) sulfide.
345. manganese(III) fluoride
Mn, manganese, is a transition metal that has multiple oxidation states, so you need to use a roman numeral to designate which form of manganese is in the compound.
The total charge of ions and cations has to equal zero. MnF3 contains three fluoride ions (F–), each with a charge of –1, so the manganese has to have an oxidation number of +3 to cancel out the three negative charges. The first part of the name is manganese(III) to take this charge into account. Thus, MnF3 is manganese (III) fluoride.
346. mercury(I) iodide
Hg, mercury, has two oxidation states, Hg22+ and Hg2+, so you need to use a roman numeral to designate which form of mercury is in the compound.
The total charge of ions and cations has to equal zero. Because Hg2I2 contains two iodide ions (I–), each with a charge of –1, the charge on each mercury ion must be +1. Therefore, Hg2I2 is mercury(I) iodide.
Note: Mercury(I) is Hg22+, a polyatomic ion consisting of two Hg atoms with a total charge of +2. The charge (+2) divided by the number of atoms (2) gives a +1 charge for each mercury atom. It’s improper to split this — or any other polyatomic ion — into smaller pieces.
347. tin(IV) oxide
Sn isn’t a transition metal, but it does have two common oxidation states, +2 and +4, so you need to use a roman numeral to designate which form of tin is in the compound.
The total charge of ions and cations has to equal zero. Oxide ions have a charge of –2. SnO2 contains two oxide ions, so the oxygen ions produce a charge of –4. To balance this –4 charge, the tin ion must have a charge of +4. SnO2 is tin(IV) oxide.
Note: Because tin is a metal, it can lose all four valence electrons to give it a +4 oxidation state. Such high charges rarely occur in stable ions.
348. sodium hypochlorite
Ternary ionic compounds (compounds containing three elements) and higher compounds usually contain polyatomic ions. The name of a polyatomic cation simply replaces the name of the cation (metal) in a binary compound, and the name of a polyatomic anion replaces the name of the anion (nonmetal).
Most polyatomic ions have either an -ate or an -ite suffix. A few, such as hydroxide, have an -ide suffix. Ions with -ate suffixes have more oxygen than those with an -ite suffix.
In this case, the polyatomic ion is ClO–, the hypochlorite ion. So name the metal ion first (Na is the sodium ion) and then add the name of the polyatomic ion ClO– to get sodium hypochlorite.
349. potassium hydroxide
In this case, the polyatomic ion is OH–, the hydroxide ion. Name the metal ion and list it first (K is the potassium ion) and then add the name of the polyatomic ion OH– to get potassium hydroxide.
350. strontium sulfite
The polyatomic ion in SrSO3 is SO32–, the sulfite ion. Sr2+ is the strontium ion, so SrSO3 is strontium sulfite.
351. calcium carbonate
The polyatomic ion in CaCO3 is CO32–, the carbonate ion. Ca2+ is the calcium ion, so CaCO3 is calcium carbonate.
352. aluminum phosphate
The polyatomic ion in AlPO4 is PO43–, the phosphate ion. Al is aluminum, so AlPO4 is aluminum phosphate.
353. sodium chlorate
The polyatomic ion in NaClO3 is ![]() , the chlorate ion. Na is sodium, so NaClO3 is sodium chlorate.
, the chlorate ion. Na is sodium, so NaClO3 is sodium chlorate.
354. gallium phosphite
The polyatomic ion in GaPO3 is PO33–, the phosphite ion. Ga3+ is the gallium ion, so GaPO3 is gallium phosphite.
355. ammonium chloride
The polyatomic ion in NH4Cl is ![]() , the ammonium ion. When you combine ammonium with the chloride ion (Cl–), you get ammonium chloride.
, the ammonium ion. When you combine ammonium with the chloride ion (Cl–), you get ammonium chloride.
356. zinc sulfate
The polyatomic ion in ZnSO4 is SO42–, the sulfate ion. Zn2+ is the zinc ion, a transition metal that has only one oxidation state (and therefore doesn’t require a roman numeral), so ZnSO4 is zinc sulfate.
357. ammonium oxalate
(NH4)2C2O4 contains two polyatomic ions, ![]() (ammonium) and C2O42– (oxalate). All you need to do is put both names together: ammonium oxalate.
(ammonium) and C2O42– (oxalate). All you need to do is put both names together: ammonium oxalate.
358. potassium permanganate
In KMnO4, the ![]() is the polyatomic ion. K+ is the potassium ion, and
is the polyatomic ion. K+ is the potassium ion, and ![]() is permanganate, so KMnO4 is potassium permanganate.
is permanganate, so KMnO4 is potassium permanganate.
359. beryllium nitrite
When a set of parentheses appears in a chemical formula, the contents are often a polyatomic ion. ![]() is the nitrite ion, so Be(NO2)2 is beryllium nitrite.
is the nitrite ion, so Be(NO2)2 is beryllium nitrite.
360. copper(II) cyanide
Cu(CN)2 begins with copper, a transition metal that has more than one oxidation state. To determine the oxidation state, look at how many polyatomic ion units are with Cu. The molecule contains two CN– ions, each with a charge of –1. To balance out this –2 charge, the copper must have a +2 charge.
Name the metal ion using roman numerals to represent its oxidation number, and then name the polyatomic ion (which happens to be inside the parentheses). Cu2+ is the copper(II) ion, and CN– is cyanide, so Cu(CN)2 is copper(II) cyanide. Note that this compound doesn’t actually exist, but you can still give it a name.
361. silver hypobromite
Even though Ag is a transition metal, it has only the oxidation state of +1, so you don’t need to use a roman numeral when naming this compound. Ag+ is the silver ion, and BrO– is hypobromite, so AgBrO is silver hypobromite.
362. ammonium phosphate
(NH4)3PO4 is made up of two polyatomic ions, so name the cation first and then name the anion. ![]() is the ammonium ion, and PO43– is the phosphate ion, so (NH4)3PO4 is ammonium phosphate.
is the ammonium ion, and PO43– is the phosphate ion, so (NH4)3PO4 is ammonium phosphate.
363. nickel(III) sulfate
Ni, nickel, is a transition metal with more than one oxidation state, so you have to look at the number of polyatomic ion units and their charge in order to determine which oxidation state to assign to the nickel. The sum of the charges of the ions in the compound has to equal zero, so you can set up an equation to solve for nickel’s oxidation state:
Therefore, Ni2(SO4)3 is nickel(III) sulfate.
364. lead(II) acetate
Pb, lead, is a metal with more than one oxidation state, so you have to look at the number of polyatomic ion units and their charge in order to determine which oxidation state to assign to the lead. The sum of the charges of the ions in the compound has to equal zero, so you can set up an equation to solve for lead’s oxidation state:
Therefore, Pb(C2H3O2)2 is lead(II) acetate.
365. CsCl
A binary ionic compound contains two elements, a metal and a nonmetal. The metal is present as a cation (positive ion), and the nonmetal is present as an anion (negative ion).
The cation name always comes before the anion name. If the cation (metal) is able to form different ions, a roman numeral after the name specifies the oxidation state. The total charge of the cations and anions must equal zero.
In this case, the cesium ion is Cs+, and chloride, Cl–, is the ionic form of the chlorine atom. The +1 charge of the Cs cancels out the –1 charge of the Cl, so the formula is CsCl.
366. InF3
The indium(III) ion is In3+, and fluoride, F–, is the ion formed by fluorine. The +3 charge of the In needs to be balanced by the negative charge of the F, so you need three –1 fluoride ions to cancel out the +3 charge of the In. The formula is InF3.
367. MgO
The magnesium ion is Mg2+, and oxide, O2–, is the ion formed by oxygen. The +2 charge of the Mg cancels out the –2 charge of the O, so the formula is MgO.
368. BaBr2
The barium ion is Ba2+, and bromide, Br–, is the ionic form of the bromine atom. You need two negatively charged bromide ions to cancel out the +2 charge on the barium ion, so the formula is BaBr2.
369. KI
The potassium ion is K+, and iodide, I–, is the ionic form of the iodine atom. The +1 charge of the K cancels out the –1 charge of the I, so the formula is KI.
370. AlCl3
The aluminum ion is Al3+, and chloride, Cl–, is the ionic form of the chlorine atom. You need three negatively charged chloride ions to cancel out the +3 charge of the aluminum, so the formula is AlCl3.
371. CrF3
Writing formulas of compounds that contain transition metals with variable charges is relatively easy because the roman numeral tells you the charge of the metal ion. Chromium(III) is Cr3+, and fluoride, F–, is the ionic form of the fluorine atom. You need three fluoride ions to balance out the chromium’s +3 charge, so the formula is CrF3.
372. FeS
Iron(II) is the Fe2+ ion, and sulfide, S2–, is the ionic form of the sulfur atom. The +2 charge cancels out the –2 charge, so the formula is FeS.
373. Cu3N
Copper(I) is the Cu+ ion, and nitride, N3–, is the ionic form of the nitrogen atom. You need three positively charged copper ions to balance out the –3 charge of the nitride ion, so the formula is Cu3N.
374. PbO
Lead(II) is the Pb2+ ion, and oxide, O2–, is the ionic form of the oxygen atom. The +2 charge of the Pb cancels out the –2 charge of the O, so the formula is PbO.
375. Ni2Se
Nickel(I) is the Ni+ ion, and selenide, Se2–, is the ionic form of the selenium atom. You need two Ni+ ions to balance out the –2 charge of the selenium, so the formula is Ni2Se.
376. Ag2O
The silver ion is Ag+, and oxide, O2–, is the ionic form of the oxygen atom. You need two Ag+ ions to balance out the –2 charge of the oxide ion, so the formula is Ag2O.
377. SrBr2
The strontium ion is Sr2+, and the bromide ion is Br–. You need two Br– ions to balance out the +2 charge of the strontium ion, so the formula is SrBr2.
378. Mg3N2
The magnesium ion is Mg2+, and nitride, N3–, is the ionic form of the nitrogen atom. The lowest common multiple of the two charges is 6. For the charges to cancel out at +6 and –6, you need three Mg2+ ions and two N3–ions. Therefore, the formula for magnesium nitride is Mg3N2.
379. LiH
The lithium ion is Li+. Hydride, H–, is the anion (negative ion) form that hydrogen takes when bonded to a metal. The +1 and –1 charges cancel out, so the formula is LiH.
380. ZnCl2
Zinc is a transition metal. For the most part, its oxidation number is +2, making the zinc ion Zn2+. Chloride, Cl–, is the ionic form of the chlorine atom. You need two Cl– ions to balance out the +2 charge of the zinc ion, so the formula is ZnCl2.
381. CrS
Chromium(II) is Cr2+, and sulfide, S2–, is the ionic form of the sulfur atom. The +2 and –2 charges cancel out, so the formula is CrS.
382. MnSe
Manganese(II) is Mn2+, and selenide, Se2–, is the ionic form of the selenium atom. The +2 and –2 charges cancel out, so the formula is MnSe.
383. SnF4
Tin(IV) is Sn4+, and fluoride, F–, is the ionic form of the fluorine atom. You need four times as many fluoride ions to balance out the +4 charge on Sn4+, so the formula for tin(IV) fluoride is SnF4.
384. CuI
The copper(I) ion is Cu+, and the iodide ion is I–. You need one –1 iodide ion to balance out the +1 charge of the copper ion, so the formula is CuI.
385. NiP
The nickel(III) ion is Ni3+, and phosphide, P3–, is the ionic form of the phosphorus atom. The +3 charge from the nickel cancels out the –3 charge of the phosphide ion, so the formula is NiP.
386. Al2Se3
The aluminum ion is Al3+, and selenide, Se2–, is the ionic form of selenium. The lowest common multiple of the two charges is 6. For the charges to cancel out at +6 and –6, you need two Al3+ ions and three Se2– ions. Therefore, the formula for aluminum selenide is Al2Se3.
387. SnO2
Tin(IV) is Sn4+, and oxide, O2–, is the ionic form of the oxygen atom. You need two –2 charged oxide ions to balance out the +4 charge of the tin ion, so the formula is SnO2.
388. Ca3P2
The calcium ion is Ca2+, and the phosphide ion is P3–. The lowest common multiple of the two charges is 6. For the charges to cancel out at +6 and –6, you need three Ca2+ ions and two P3– ions. Therefore, the formula for calcium phosphide is Ca3P2.
389. Fe2O3
The iron(III) ion is Fe3+, and the oxide ion, O2–, is the ionic form of the oxygen atom. The lowest common multiple of the two charges is 6. For the charges to cancel out at +6 and –6, you need two Fe3+ ions and three O2–ions. The formula for iron(III) oxide is Fe2O3.
390. MnS2
Manganese(IV) is the Mn4+ ion, and sulfide, S2–, is the ionic form of the sulfur atom. You need two –2 sulfide ions to balance out the +4 charge of the manganese(IV) ion, so the formula for manganese(IV) sulfide is MnS2.
391. RbClO
In compounds containing polyatomic ions, the name of a polyatomic cation replaces the name of the cation (metal) in a binary compound, or the name of a polyatomic anion replaces the name of the anion (nonmetal).
The cation name always comes before the anion name. If the cation (metal) is able to form different ions, a roman numeral after the name specifies the oxidation state. The total charge of the cations and anions must equal zero.
The rubidium ion is Rb+, and hypochlorite is ClO–. The +1 charge cancels out the –1 charge, so the formula for rubidium hypochlorite is RbClO.
392. BeCO3
The beryllium ion is Be2+, and carbonate is CO32–. The +2 charge cancels out the –2 charge, so the formula for beryllium carbonate is BeCO3.
393. AlPO3
The aluminum ion is Al3+, and phosphite is PO33–. The +3 charge cancels out the –3 charge, so the formula for aluminum phosphite is AlPO3.
394. NaHCO3
The sodium ion is Na+, and hydrogen carbonate is ![]() . The +1 charge cancels out the –1 charge, so the formula for sodium hydrogen carbonate is NaHCO3. Another name for this compound is sodium bicarbonate.
. The +1 charge cancels out the –1 charge, so the formula for sodium hydrogen carbonate is NaHCO3. Another name for this compound is sodium bicarbonate.
395. NaOH
The sodium ion is Na+, and hydroxide is OH–. The +1 charge cancels out the –1 charge, so the formula for sodium hydroxide is NaOH.
396. KHSO4
The potassium ion is K+, and hydrogen sulfate is ![]() . The +1 charge cancels out the –1 charge, so the formula for potassium hydrogen sulfate is KHSO4. Another name for this compound is potassium bisulfate.
. The +1 charge cancels out the –1 charge, so the formula for potassium hydrogen sulfate is KHSO4. Another name for this compound is potassium bisulfate.
397. LiClO4
The lithium ion is Li+, and perchlorate is ![]() . The +1 charge cancels out the –1 charge, so the formula for lithium perchlorate is LiClO4.
. The +1 charge cancels out the –1 charge, so the formula for lithium perchlorate is LiClO4.
398. BaC2O4
The barium ion is Ba2+, and oxalate is C2O42–. The +2 charge cancels out the –2 charge, so the formula for barium oxalate is BaC2O4.
399. CrAsO4
Chromium(III) is Cr3+, and arsenate is AsO43–. The +3 charge cancels out the –3 charge, so the formula for chromium(III) arsenate is CrAsO4.
400. AgNO3
The silver ion is Ag+, and nitrate is ![]() . The +1 charge cancels out the –1 charge, so the formula for silver nitrate is AgNO3.
. The +1 charge cancels out the –1 charge, so the formula for silver nitrate is AgNO3.
401. PbSO3
Lead(II) is Pb2+, and sulfite is SO32–. The +2 charge cancels out the –2 charge, so the formula for lead sulfite is PbSO3.
402. TlBrO2
Thallium(I) is Tl+, and bromite is ![]() . The +1 charge cancels out the –1 charge, so the formula for thallium(I) bromite is TlBrO2.
. The +1 charge cancels out the –1 charge, so the formula for thallium(I) bromite is TlBrO2.
403. AuPO4
Gold(III) is Au3+, and phosphate is PO43–. The +3 charge cancels out the –3 charge, so the formula for gold(III) phosphate is AuPO4.
404. FeSO4
Iron(II) is Fe2+, and sulfate is SO42–. The +2 charge cancels out the –2 charge, so the formula for iron(II) sulfate is FeSO4.
405. CaS2O3
The calcium ion is Ca2+, and thiosulfate is S2O32–. The +2 charge cancels out the –2 charge, so the formula for calcium thiosulfate is CaS2O3.
406. Na2O2
The sodium ion is Na+, and peroxide is O22–. You need two Na+ ions to balance out the –2 charge on the peroxide ion, so the formula for sodium peroxide is Na2O2.
407. NH4NO2
The ammonium ion is NH4+, and nitrite is ![]() . The ammonium’s +1 charge cancels out the nitrite’s –1 charge, so the formula for ammonium nitrite is NH4NO2. Note: Nitrogen appears twice in the formula because it’s in two separate polyatomic ions; combining parts of two separate ions isn’t appropriate.
. The ammonium’s +1 charge cancels out the nitrite’s –1 charge, so the formula for ammonium nitrite is NH4NO2. Note: Nitrogen appears twice in the formula because it’s in two separate polyatomic ions; combining parts of two separate ions isn’t appropriate.
408. Be(ClO2)2
The beryllium ion is Be2+, and the chlorite ion is ![]() . You need two –1 charged
. You need two –1 charged ![]() ions to balance out the +2 charge of the beryllium ion, so the formula for beryllium chlorite is Be(ClO2)2.
ions to balance out the +2 charge of the beryllium ion, so the formula for beryllium chlorite is Be(ClO2)2.
409. NaCN
The sodium ion is Na+, and cyanide is CN–. The +1 charge of the sodium cancels out the –1 charge of the cyanide, so the formula for sodium cyanide is NaCN.
410. Mg(MnO4)2
The magnesium ion is Mg2+, and the permanganate ion is ![]() . You need two –1 charged permanganate ions to balance out the +2 charge of the magnesium ion, so the formula for magnesium permanganate is Mg(MnO4)2.
. You need two –1 charged permanganate ions to balance out the +2 charge of the magnesium ion, so the formula for magnesium permanganate is Mg(MnO4)2.
411. (NH4)2Cr2O7
The ammonium ion is NH4+, and the dichromate ion is Cr2O72–. You need two +1 charged ammonium ions to balance out the –2 charge of the dichromate ion, so the formula for ammonium dichromate is (NH4)2Cr2O7.
412. Co(IO4)3
Cobalt(III) is Co3+, and periodate is ![]() . You need three –1 charged periodate ions to cancel out the +3 charged cobalt ion, so the formula for cobalt(III) periodate is Co(IO4)3.
. You need three –1 charged periodate ions to cancel out the +3 charged cobalt ion, so the formula for cobalt(III) periodate is Co(IO4)3.
413. Pb(C2O4)2
The lead(IV) ion is Pb4+, and the oxalate ion is C2O42–. You need two –2 charged oxalate ions to balance out the +4 charge of the lead(IV) ion, so the formula for lead(IV) oxalate is Pb(C2O4)2.
414. CaHPO4
The calcium ion is Ca2+, and the hydrogen phosphate ion is HPO42–. The +2 charge of the calcium ion cancels out the –2 charge of the hydrogen phosphate ion, so the formula for calcium hydrogen phosphate is CaHPO4.
415. Fe(C2H3O2)3
Iron(III) is Fe3+, and the acetate ion is ![]() . You need three –1 charged acetate ions to cancel out the +3 charged iron(III) ion, so the formula for iron(III) acetate is Fe(C2H3O2)3.
. You need three –1 charged acetate ions to cancel out the +3 charged iron(III) ion, so the formula for iron(III) acetate is Fe(C2H3O2)3.
Note: You can also write the acetate ion as CH3COO–, so Fe(CH3COO)3 is another way to write the chemical formula for iron(III) acetate.
416. KSCN
The potassium ion is K+, and the thiocyanate ion is SCN–. The +1 charge of the potassium ion cancels out the –1 charge of the thiocyanate ion, so the formula for potassium thiocyanate is KSCN.
417. Cu(HSO3)2
The copper(II) ion is Cu2+, and the hydrogen sulfite ion is ![]() . You need two
. You need two ![]() ions to balance out the +2 charge of the copper(II) ion, so the formula for copper(II) hydrogen sulfite is Cu(HSO3)2. This compound is also known as copper(II) bisulfite.
ions to balance out the +2 charge of the copper(II) ion, so the formula for copper(II) hydrogen sulfite is Cu(HSO3)2. This compound is also known as copper(II) bisulfite.
418. HgO2
The mercury(II) ion is Hg2+, and peroxide is O22–. The +2 charge of the Hg2+ ion cancels the –2 charge of O22–, so the formula for mercury(II) peroxide is HgO2.
419. AuOCN
Gold(I) is Au+, and the cyanate ion is OCN–. The +1 charge cancels out the –1 charge, so the formula for gold(I) cyanate is AuOCN.
420. Al(H2PO4)3
The aluminum ion is Al3+, and dihydrogen phosphate is ![]() . To cancel out the +3 charge of the Al3+, you need three
. To cancel out the +3 charge of the Al3+, you need three ![]() ions, so the formula for aluminum dihydrogen phosphate is Al(H2PO4)3.
ions, so the formula for aluminum dihydrogen phosphate is Al(H2PO4)3.
421. 2
The prefix di- represents two atoms.
422. 6
The prefix hexa- represents six atoms.
423. 7
The prefix hepta- represents seven atoms.
424. 4
The prefix tetra- represents four atoms.
425. 9
The prefix nona- represents nine atoms.
426. 3
The prefix tri- represents three atoms.
427. 1
The prefix mono- represents one atom. In more recent naming systems, this prefix is rarely used. Carbon monoxide is one of the few cases where you still see mono-.
428. 5
The prefix penta- represents five atoms.
429. 8
The prefix octa- represents eight atoms.
430. 10
The prefix deca- represents ten atoms.
431. carbon monoxide
CO contains two nonmetals: carbon and oxygen. The molecule has only one atom of carbon, so just use the name of the first element as-is. The second nonmetal, oxygen, gets a prefix (which describes how many oxygen atoms are in the compound) and a suffix of -ide. The prefix for one is mon(o)-, so CO is carbon monoxide. This is one of the few cases where mono- is still in use.
432. sulfur dibromide
SBr2 contains one sulfur atom and two bromine atoms. The molecule has only one sulfur atom, so use the name of the first element as-is. The second element, bromine, gets a suffix of -ide and the prefix di- (two) to indicate the number of atoms. Therefore, SBr2 is sulfur dibromide.
433. iodine chloride
ICl contains one iodine atom and one chlorine atom. The molecule has only one iodine atom, so use the name of the first element as-is. The second element, chlorine, gets a suffix of -ide; the prefix mono- (one) is usually omitted in modern naming systems, so you don’t need a prefix to indicate the number of chlorine atoms. Therefore, ICl is iodine chloride. The name iodine monochloride is also acceptable.
434. sulfur dioxide
SO2 contains one sulfur atom and two oxygen atoms. The molecule has only one sulfur atom, so use the name of the first element as-is. The second element, oxygen, gets a suffix of -ide and the prefix di- (two) to indicate the number of atoms. Therefore, SO2 is sulfur dioxide, a compound that contributes to the odor of a freshly struck match.
435. phosphorus pentachloride
PCl5 contains one phosphorus atom and five chlorine atoms. The molecule has only one phosphorus atom, so phosphorus doesn’t need a prefix. The second element, chlorine, gets a suffix of -ide and the prefix penta- (five) to indicate the number of atoms. Therefore, PCl5 is phosphorus pentachloride.
436. xenon difluoride
XeF2 contains one xenon atom and two fluorine atoms. The molecule has only one xenon atom, so use the name of the first element as-is. The second element, fluorine, gets a suffix of -ide and the prefix di- (two) to indicate the number of atoms. Therefore, XeF2 is xenon difluoride.
437. sulfur hexafluoride
SF6 contains one sulfur atom and six fluorine atoms. The molecule has only one sulfur atom, so use the name of the first element as-is. The second element, fluorine, gets a suffix of -ide and the prefix hexa- (six) to indicate the number of atoms. Therefore, SF6 is sulfur hexafluoride.
438. carbon tetrabromide
CBr4 contains one carbon atom and four bromine atoms. The molecule has only one carbon atom, so carbon doesn’t need a prefix. The second element, bromine, gets a suffix of -ide and the prefix tetra- (four) to indicate the number of atoms. Therefore, CBr4 is carbon tetrabromide.
439. boron trichloride
BCl3 contains one boron atom and three chlorine atoms. The molecule has only one boron atom, so you don’t need a prefix on boron. The second element, chlorine, gets a suffix of -ide and the prefix tri- (three) to indicate the number of atoms. Therefore, BCl3 is boron trichloride.
440. silicon dioxide
SiO2 contains one silicon atom and two oxygen atoms. The molecule has only one silicon atom, so silicon doesn’t need a prefix. The second element, oxygen, gets a suffix of -ide and the prefix di- (two) to indicate the number of atoms. Therefore, SiO2 is silicon dioxide, which you may recognize as the mineral quartz. This compound is a major component of sand and glass.
441. arsenic pentachloride
AsCl5 contains one arsenic atom and five chlorine atoms. The molecule has only one arsenic atom, so use the name of the first element as-is. The second element, chlorine, gets a suffix of -ide and the prefix penta- (five) to indicate the number of atoms. Therefore, AsCl5 is arsenic pentachloride.
442. antimony trichloride
SbCl3 contains one antimony atom and three chlorine atoms. The molecule has only one antimony atom, so antimony doesn’t need a prefix. The second element, chlorine, gets a suffix of -ide and the prefix tri- (three) to indicate the number of atoms. Therefore, SbCl3 is antimony trichloride.
443. silicon tetraiodide
SiI4 contains one silicon atom and four iodine atoms. The molecule has only one silicon atom, so use the name of the first element as-is. The second element, iodine, gets a suffix of -ide and the prefix tetra- (four) to indicate the number of atoms. Therefore, SiI4 is silicon tetraiodide.
444. nitrogen trifluoride
NF3 contains one nitrogen atom and three fluorine atoms. The molecule has only one nitrogen atom, so use the name of the first element as-is. The second element, fluorine, gets a suffix of -ide and the prefix tri- (three) to indicate the number of atoms. Therefore, NF3 is nitrogen trifluoride.
445. carbon disulfide
CS2 contains one carbon atom and two sulfur atoms. The molecule has only one carbon atom, so you don’t need a prefix on the carbon. The second element, sulfur, gets a suffix of -ide and the prefix di- (two) to indicate the number of atoms. Therefore, CS2 is carbon disulfide.
446. chlorine dioxide
ClO2 contains one chlorine atom and two oxygen atoms. The molecule has only one chlorine atom, so use the name of the first element as-is. The second element, oxygen, gets a suffix of -ide and the prefix di- (two) to indicate the number of atoms. Therefore, ClO2 is chlorine dioxide. This compound is useful as an industrial bleach.
447. xenon tetroxide
XeO4 contains one xenon atom and four oxygen atoms. The molecule has only one xenon atom, so xenon doesn’t need a prefix. The second element, oxygen, gets a suffix of -ide and the prefix tetr(a)- (four) to indicate the number of atoms. Therefore, XeO4 is xenon tetroxide. This compound is very unstable and will spontaneously detonate.
448. water
H2O contains two hydrogen atoms and one oxygen atom. The compound might appear to be dihydrogen oxide, but the current systematic (IUPAC) name is water.
449. selenium hexafluoride
SeF6 contains one selenium atom and six fluorine atoms. The molecule has only one selenium atom, so use the name of the first element as-is. The second element, fluorine, gets a suffix of -ide and the prefix hexa- (six) to indicate the number of atoms. Therefore, SeF6 is selenium hexafluoride.
450. disulfur dichloride
S2Cl2 contains two sulfur atoms and two chlorine atoms. Because you have more than one atom of the first element, it needs a prefix. The prefix for two is di-, giving you disulfur. The second element, chlorine, gets a suffix of -ide and also takes the prefix di-. Therefore, S2Cl2 is disulfur dichloride.
451. dinitrogen trioxide
N2O3 contains two nitrogen atoms and three oxygen atoms. The nitrogen needs the prefix di- to represent two atoms, and the oxygen (which becomes oxide) needs the prefix tri- to indicate three atoms. Therefore, N2O3 is dinitrogen trioxide.
452. tetraphosphorus hexoxide
P4O6 contains four phosphorus atoms and six oxygen atoms. The phosphorus needs the prefix tetra- to indicate four atoms, and the oxygen needs the prefix hex(a)- (six) and the suffix -ide. Therefore, P4O6 is tetraphosphorus hexoxide.
453. diboron tetrachloride
B2Cl4 contains two boron atoms and four chlorine atoms. The boron needs the prefix di- (two), and chlorine needs the prefix tetra- (four). Chlorine changes to chloride, so B2Cl4 is diboron tetrachloride.
454. bromine trifluoride
BrF3 contains one bromine atom and three fluorine atoms. The molecule has only one bromine atom, so use the name of the first element as-is. The second element, fluorine, gets a suffix of -ide and the prefix tri- (three) to indicate the number of atoms. Therefore, BrF3 is bromine trifluoride.
455. disulfur decafluoride
S2F10 contains two sulfur atoms and ten fluorine atoms. Use the prefix di- (two) to represent the number of sulfur atoms and the prefix deca- (ten) to represent the number of fluorine atoms. Fluorine changes to fluoride, so S2F10 is disulfur decafluoride.
456. SiBr4
The first element in the name of the compound doesn’t have a prefix, and by convention, this means that there’s only one atom of that element. The second element in the compound has a prefix to tell you how many atoms of that element are present. In silicon tetrabromide, the prefix on the bromide is tetra-, which represents four. You have one silicon (Si) atom and four bromine (Br) atoms, so silicon tetrabromide is SiBr4.
457. NI3
In nitrogen triiodide, the absence of a prefix on the nitrogen tells you that the compound has just one nitrogen (N) atom, and the tri- prefix on the iodide indicates three iodine (I) atoms. The formula for nitrogen triiodide is NI3. This compound is very unstable and explodes at the lightest touch.
458. CO2
You’re probably familiar with carbon dioxide and didn’t need to think about the formula too much. The absence of a prefix on the carbon and the di- (two) prefix on the oxide lead to the formula CO2. Carbon dioxide is one of the products of respiration and the combustion of most fuels.
459. AsF5
In arsenic pentafluoride, the absence of a prefix on the arsenic means you have just one arsenic (As) atom. The penta- prefix on the fluoride tells you that there are five fluorine (F) atoms, so the formula for arsenic pentafluoride is AsF5.
460. NO
In nitrogen monoxide, nitrogen doesn’t have a prefix, and the prefix on the oxide is mon(o)-, which means one, so this compound has one atom of nitrogen (N) and one atom of oxygen (O). The formula for nitrogen monoxide is NO. This compound is also known as nitrogen oxide.
461. SO3
In sulfur trioxide, the sulfur has no prefix, so the formula contains just one atom of sulfur (S). The oxide has a prefix of tri-, which means three, so you have three oxygen (O) atoms. This makes sulfur trioxide SO3.
462. ClF
In chlorine monofluoride, chlorine doesn’t have a prefix, and the prefix on the fluoride is mono-, which means one. So chlorine monofluoride has one atom of chlorine (Cl) and one atom of fluorine (F), making the formula ClF. This compound is also known as chlorine fluoride.
463. XeF4
In xenon tetrafluoride, xenon doesn’t have a prefix, and the prefix on the fluoride is tetra-, which means four. So xenon tetrafluoride contains one atom of xenon (Xe) and four atoms of fluorine (F), making the formula XeF4. This compound was the first binary compound of xenon to be prepared in a laboratory.
464. NO2
In nitrogen dioxide, no prefix on the nitrogen tells you that the formula includes one nitrogen (N) atom, and the di- prefix on the oxide indicates two oxygen (O) atoms. The formula for nitrogen dioxide is NO2. Nitrogen dioxide is one of the contributors to photochemical smog.
465. CCl4
In carbon tetrachloride, no prefix on the carbon means that the formula includes only one carbon (C) atom, and the tetra- prefix on the chloride indicates four chlorine (Cl) atoms, so carbon tetrachloride is CCl4.
466. IF7
In iodine heptafluoride, no prefix on the iodine means that the formula includes only one iodine (I) atom. The hepta- prefix represents seven, so there are seven fluorine (F) atoms in the formula. This makes the formula for iodine heptafluoride IF7. Iodine heptafluoride is one of the few compounds where one atom is surrounded by seven others.
467. PBr3
In phosphorus tribromide, no prefix on the phosphorus tells you that the formula includes just one atom of phosphorus (P), and the tri- prefix on the bromide indicates three bromine (Br) atoms. Therefore, the formula for phosphorus tribromide is PBr3.
468. SeF4
In selenium tetrafluoride, no prefix on the selenium means you have just one atom of selenium (Se), and the prefix on the fluoride is tetra-, which means four, so you have four fluorine atoms (F). Therefore, selenium tetrafluoride is SeF4.
469. ClO2
In chlorine dioxide, the absence of a prefix on the chlorine tells you that there’s only one chlorine (Cl) atom, and the di- prefix on the oxide tells you that there are two oxygen (O) atoms in the compound. The formula for chlorine dioxide is ClO2.
470. BF3
In boron trifluoride, the absence of a prefix on the boron reflects that there’s only one boron (B) atom, and the tri- prefix on the fluoride indicates three fluorine (F) atoms. This makes the formula for boron trifluoride BF3.
471. N2O5
Dinitrogen pentoxide has a prefix on both the nitrogen and the oxide. The di- represents two, and the pent(a)- represents five, so there are two nitrogen (N) atoms and five oxygen (O) atoms in dinitrogen pentoxide. The formula is N2O5.
472. P2O3
Diphosphorus trioxide has a prefix on both the phosphorus and the oxide. The di- represents two, and the tri- represents three, so there are two phosphorus (P) atoms and three oxygen (O) atoms in diphosphorus trioxide. The formula is P2O3.
Note that the formula P2O3 is the empirical formula. The molecular formula is P4O6, which has the name tetraphosphorus hexoxide (or hexaoxide).
473. N2Cl2
Dinitrogen dichloride has two nitrogen (N) atoms and two chlorine (Cl) atoms because both elements have the prefix di-, which means two. Therefore, the formula for dinitrogen dichloride is N2Cl2.
474. P4O10
Tetraphosphorus decoxide has the prefix tetra-, meaning four, and the prefix dec(a)-, meaning ten, so you need four phosphorus (P) atoms and ten oxygen (O) atoms. This makes the formula for tetraphosphorus decoxide P4O10.
475. AsF3
In arsenic trifluoride, the absence of a prefix on arsenic reflects that there’s only one arsenic (As) atom, and the tri- prefix on the fluoride indicates three fluorine (F) atoms. This makes the formula for arsenic trifluoride AsF3.
476. N2O
In dinitrogen oxide, the prefix di- on the nitrogen tells you that there are two nitrogen (N) atoms, and the lack of a prefix on the oxide tells you that there’s just one oxygen (O) atom. The formula for dinitrogen oxide is N2O.
This compound is also called dinitrogen monoxide. Its common names include nitrous oxide and laughing gas, which you may recognize as an anesthetic used by dentists.
477. P4O3
Tetraphosphorus trioxide has the prefix tetra-, meaning four, and the prefix tri-, meaning three, so the compound contains four phosphorus (P) atoms and three oxygen (O) atoms. This makes the formula for tetraphosphorus trioxide P4O3.
478. XeO3
In xenon trioxide, the absence of a prefix on xenon reflects that there’s only one xenon (Xe) atom, and the tri- prefix on the oxide indicates three oxygen (O) atoms. This makes the formula for xenon trioxide XeO3.
479. SbCl5
In antimony pentachloride, the absence of a prefix on antimony indicates one antimony (Sb) atom, and the penta- prefix on the chloride indicates five chlorine (Cl) atoms, so the formula for antimony pentachloride is SbCl5.
480. Cl2O7
The name dichlorine heptoxide has the prefix di-, meaning two, and the prefix hept(a)-, meaning seven, so the formula includes two chlorine (Cl) atoms and seven oxygen (O) atoms. This means that the formula for dichlorine heptoxide is Cl2O7.
481. 3
A Lewis dot diagram shows only the valence electrons. Boron has an electron configuration of 1s22s22p1. The valence electrons are the ones in the second energy level, 2s22p1, so the diagram contains three electrons.
482. 2
Barium has a noble gas electron configuration [Xe]6s2 with two valence electrons. (The electron configuration of a noble gas like xenon is very stable, and any electrons beyond that configuration are an atom’s valence electrons.) The Lewis dot diagram shows only valence electrons, so the diagram for barium has two electrons.
483. 7
The noble gas electron configuration of chlorine is [Ne]3s23p5. The Lewis dot diagram shows valence electrons, so the diagram for chlorine has seven electrons.
484. 8
An oxygen atom has eight electrons (1s22s22p4), and the oxygen ion, with a charge of –2, has two more electrons (1s22s22p6). This means that there are eight valence electrons in the oxygen ion (2s22p6), so eight electrons go in the Lewis dot diagram.
485. 0
The sodium ion, Na+, is a sodium atom that has lost its one valence electron, going from an electron configuration of 1s22s22p63s1 to 1s22s22p6. The orbitals in the third energy level are still there, but they don’t contain any electrons. The Lewis dot diagram, which shows only the valence electrons, has zero electrons for the sodium ion.
486. the nitrogen group
Members of the nitrogen group (Group VA or 15) have five valence electrons because their highest energy level has electrons filling s2p3 orbitals.
487. the oxygen group
Members of the oxygen group (Group VIA or 16) have six valence electrons because their highest energy level has electrons filling s2p4 orbitals.
488. the boron group
Members of the boron group (Group IIIA or 13) have three valence electrons because their highest energy level has electrons filling s2p1 orbitals.
489. the alkaline earth metals
Members of the alkaline earth metals have two valence electrons because their highest energy level has electrons filling s2 orbitals. Elements with two valence electrons are in Group 2 (or 2A, IIA, or IIB) of the periodic table.
490. the noble gases
The noble gases, except helium, have eight valence electrons because their highest energy level has electrons filling s2p6 orbitals (helium, however, has only two valence electrons). The noble gases are in Group 18 (or 8, VIIIA, VIIIB, or 0) of the periodic table.
491. electronegativity
Electronegativity is how strongly an atom attracts a bonding pair of electrons to itself. You can look up electronegativity values in a table.
Tip: In most cases, you need to know only relative electronegativity values. Fluorine has the highest value, so you can assume that the closer an element is to fluorine, the higher its electronegativity. If two elements are the same distance away from fluorine, the one that’s closer to the top of the periodic table has the higher value. This tip works for all representative elements except hydrogen.
492. nonpolar covalent
Nonpolar covalent bonds tend to form between elements that have an electronegativity difference between 0.0 and 0.3 (or between 0.0 and 0.4; the cutoff depends on which electronegativity values are used).
Note that in terms of electronegativity, the division between bond types is not a precise division. Additional factors, such as the elements present, neighboring elements, and hybridization, affect the type of bond.
493. polar covalent
Polar covalent bonds form between elements that have a moderate electronegativity difference, typically between 0.3 and 1.7. In general, differences less than 0.3 (or 0.4, depending on the electronegativity values used) are considered nonpolar; differences greater than 1.7 are generally considered ionic.
494. lower; higher
Electronegativity is the relative attraction that an atom in a compound has for electrons. Metals are more likely to give away electrons than accept them, so metals tend to have lower electronegativity values. Nonmetals are more likely to accept electrons, so nonmetals tend to have higher electronegativity values. Of course, there are exceptions. For example, gold, a metal, has an unusually high electronegativity. The electronegativity of gold (2.4) is higher than that of the nonmetal phosphorus (2.1).
Note: The representative elements are the elements in Groups 1, 2, and 13 through 18 on the periodic table. In the Chemical Abstract Services (CAS) version of the periodic table, which uses roman numerals, the representative elements are the A elements (as opposed to the transition elements, which are the B elements). The A elements always have an A in their group number, as in IA and IIA.
495. polar covalent
Elements that have a moderate difference in their electronegativity values normally form polar covalent bonds. Nonmetals that aren’t adjacent on the periodic table, such as carbon and bromine, usually have moderate differences.
496. polar covalent
Elements that have a moderate difference in their electronegativity values normally form polar covalent bonds. Nonmetals that aren’t adjacent on the periodic table, such as carbon and fluorine, usually have moderate differences.
497. polar covalent
Elements that have a moderate difference in their electronegativity values normally form polar covalent bonds. Nonmetals that aren’t adjacent on the periodic table usually have moderate differences.
Elements that are diagonal to each other, such as chlorine and fluorine, are 1.4 times as far away from each other as side-by-side elements. If you go over one and down one, you can think of the elements as though they’re nonadjacent; they’re twice as far away.
498. nonpolar covalent
Elements with similar electronegativity values normally form nonpolar covalent bonds. Similar values occur for a nonmetal bonding with itself or with an adjacent nonmetal on the periodic table. Remember that hydrogen is an exception; its electronegativity places it between boron and carbon. The electronegativity difference between boron and hydrogen is small, so the bond is nonpolar covalent.
499. nonpolar covalent
Elements with similar electronegativity values normally form nonpolar covalent bonds. Similar values occur for a nonmetal bonding with itself or with an adjacent nonmetal on the periodic table. Remember that hydrogen is an exception; its electronegativity places it between boron and carbon. The electronegativity difference between carbon and hydrogen is small, so the bond is nonpolar covalent.
500. polar covalent
Elements that have a moderate difference in their electronegativity values normally form polar covalent bonds. Nonmetals that aren’t adjacent on the periodic table, such as phosphorus and fluorine, usually have moderate differences.
501. S–Cl < O–S < Si–Cl
In general, the farther two nonmetals are from each other on the periodic table, the more polar the bond. Remember that hydrogen is an exception in that in terms of electronegativity, it fits between boron and carbon. If two pairs have the same separation, the one closer to the top (or the right) of the periodic table is more polar.
Sulfur and chlorine are adjacent on the periodic table, so the S–Cl bond is slightly polar. Oxygen is higher on the periodic table than chlorine, so its electronegativity is higher. For this reason, even though oxygen is also adjacent to sulfur, the S–O bond is more polar than the S–Cl bond. In this problem, the greatest separation on the periodic table is between silicon and chlorine, so Si–Cl is the most polar bond.
502. trigonal pyramidal
This molecule has three bonding pairs of electrons (indicated by the lines between the atoms) and one nonbonding pair of electrons (indicated by the two dots in the region above the central atom). This combination yields a trigonal pyramid shape — a pyramid with a three-sided base. The three black spheres form the triangular base of the pyramid, and the gray sphere — the central atom — represents the apex.
503. linear
This molecule has two bonding pairs of electrons (indicated by the lines between the atoms) and no nonbonding pairs of electrons. The bonding pairs orient themselves on opposite sides of the central atom, giving the molecule a linear shape.
504. tetrahedral
This molecule has four bonding pairs of electrons (indicated by the four lines between the atoms) and no lone pairs of electrons. The bonding pairs arrange themselves to maximize the distance from each other, so the angle measures are 109.5° at the central atom. A tetrahedron is a pyramid with four faces that are equilateral triangles, so this shape is tetrahedral.
505. trigonal planar
This molecule has three bonding pairs of electrons (indicated by the lines between the atoms) and no lone pairs of electrons. The three pairs arrange themselves to maximize their distance from each other (120° apart). This arrangement is known as trigonal planar, because the three pairs lie in a plane.
506. linear
This molecule has one bonding pair (indicated by the line between the atoms) and no nonbonding pairs of electrons. There are only two atoms, so the molecular geometry must be linear.
507. bent
This molecule has two bonding areas (indicated by the lines between the atoms) and two nonbonding pairs (indicated by the two pairs of dots). The four total pairs arrange themselves to maximize the distance between pairs. Bonding atoms to two of the pairs results in a bent shape.
508. linear
A bond angle of 180° — a straight line — corresponds to the linear shape.
509. trigonal planar
A bond angle of 120° is in trigonal planar molecules. In this shape, the three bonding pairs of electrons lie in a plane, maximizing their distance from each other.
510. tetrahedral
The tetrahedral molecular shape has a bond angle of approximately 109.5°.

511. trigonal planar
Start with the Lewis structure of BCl3:
The lines between the boron and each of the chlorine atoms represent bonding pairs of electrons. There are no other pairs on the boron, and the additional pairs on the noncentral atoms aren’t important in determining molecular geometry. Because boron has only three pairs, the bonds will be at 120° angles, yielding a trigonal planar structure.
512. linear
Bromine, Br2, is diatomic, so it has only one bonding pair area. Therefore, it’s linear in shape.
513. tetrahedral
Start by drawing the Lewis structure of CH4:
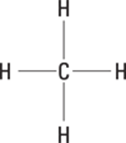
The lines between the carbon and each of the hydrogen atoms represent bonding pairs of electrons. There are no other pairs on the carbon. Because carbon has only four electron pairs, the bonds will be at 109.5° angles, yielding a tetrahedral structure.
514. bent
Start by drawing the Lewis structure of H2O:
The lines between the oxygen and each of the hydrogen atoms represent bonding pairs of electrons. The oxygen also has two lone pairs. Oxygen has four pairs, so the pairs will be at 109.5° angles, yielding a tetrahedral electron-pair structure. But because two of the pairs are lone pairs, they aren’t part of the molecular geometry. This leaves a bent geometry. The lone pairs cause the angle between the bonding pairs to be a little less than expected because the electron pairs repel each other, spreading farther apart.
515. trigonal pyramidal
Start by drawing the Lewis structure of NH3:
The lines between the nitrogen and each of the hydrogen atoms represent bonding pairs of electrons. The nitrogen also has one lone pair. Nitrogen has four pairs, so they’ll be at 109.5° angles, yielding a tetrahedral electron-pair structure. But because one of the pairs is a lone pair, it isn’t part of the molecular geometry. This leaves a trigonal pyramidal geometry. The lone pair causes the angle between the bonding pairs to be a little less than expected.
516. linear
Start by drawing the Lewis structure of CO2:
![]()
The lines between the carbon and each of the oxygen atoms represent bonding pairs of electrons; however, when dealing with double bonds, only one of the two bonding pairs counts. Because the carbon has two bonding pair areas, not four, they’ll be at 180° angles, yielding a linear geometry. The electron pairs not on the central atom don’t affect the molecular geometry.
517. Its central atom has four electron pairs, two of which are nonbonding.
Look at the Lewis structure of H2O:
The lines between the oxygen and each of the hydrogen atoms represent bonding pairs of electrons. The oxygen also has two lone pairs. Oxygen has four pairs, so the pairs will be at 109.5° angles, yielding a tetrahedral electron-pair structure. But because two of the pairs are lone pairs, they aren’t part of the molecular geometry. This leaves a bent geometry. The lone pairs cause the angle between the bonding pairs to be a little less than expected.
518. Its central atom has three bonding pair areas and one nonbonding pair area.
Look at the Lewis structure of NF3:

The lines between the nitrogen and each of the fluorine atoms represent bonding pairs of electrons. The nitrogen also has one lone pair. Nitrogen has four pairs, so the pairs will be at 109.5° angles, yielding a tetrahedral electron-pair structure. But because one of the pairs is a lone pair, it isn’t part of the molecular geometry. This leaves a trigonal pyramidal geometry. The lone pair causes the angle between the bonding pairs to be a little less than expected.
519. Its central atom has two bonding pairs and no lone pairs of electrons.
Look at the Lewis structure of CO2:
![]()
The lines between the carbon and each of the oxygen atoms represent bonding pairs of electrons; however, when dealing with double bonds, only one of the two bonding pairs counts. Because the carbon has two pairs, not four, they’ll be at 180° angles, yielding a linear molecular geometry. The pairs not on the central atom don’t affect the molecular geometry.
520. It has four bonding pairs and no lone pairs of electrons.
Look at the Lewis structure of CCl4:
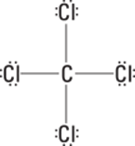
The lines between the carbon and each of the chlorine atoms represent bonding pairs of electrons. There are no other electron pairs on the carbon. Because carbon has only four pairs, they’ll be at 109.5° angles, yielding a tetrahedral structure.
521. Its central atom has three bonding pair areas and zero nonbonding pair areas.
Look at the Lewis structure of BF3:
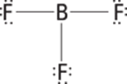
The lines between the boron and each of the fluorine atoms represent bonding pairs of electrons. There are no other pairs on the boron, and the additional pairs on the noncentral atoms aren’t important in determining the molecular geometry. Because boron has only three pairs, they’ll be at 120° angles, yielding a trigonal planar structure.
522. octahedral
This molecule has six bonding pairs of electrons (indicated by the lines between the atoms) and no nonbonding pairs of electrons. The six pairs arrange themselves 90° apart to maximize their separation. The result is an octahedral shape (see the following figure). An octahedron has eight faces that are equilateral triangles.
523. T-shaped
The drawing shows a molecule with three bonding pairs and two lone pairs of electrons. This combination indicates a T-shaped molecule.
524. seesaw
This molecule has four bonding pairs of electrons (indicated by the lines between the atoms) and one nonbonding pair of electrons (indicated by the pair of dots). The five electron pairs arrange themselves in a trigonal bipyramidal structure — a double pyramid with a triangular base. The lone pair occupies one of the central positions, leaving a seesaw arrangement.
525. square pyramidal
This molecule has five bonding pairs of electrons (indicated by the lines between the atoms) and one nonbonding pair of electrons (indicated by the pair of dots). The six pairs arrange themselves pointing toward the center of an octahedron, with the lone pair occupying one corner. Four of the atoms surrounding the central atom form the base of a square pyramid, and the fifth atom forms the apex, creating a structure known as square pyramidal:

526. trigonal bipyramidal
The drawing depicts a molecule that has five bonding pairs of electrons (indicated by the lines between the atoms) and no lone pairs of electrons. The five pairs arrange themselves as far apart as possible to minimize repulsion. This results in the only basic structure that has two types of bond angles: 90° and 120°. The three central bonding pairs form a trigonal (three-sided) plane, with the pairs at 120° angles to each other. The upper bonding pair (at 90° to the three in the plane) forms the apex to a trigonal pyramid (a pyramid with a three-sided base), and the lower bonding pair (also at 90° to the three in the plane) forms the apex of a second trigonal pyramid.
527. square planar
The drawing depicts a molecule with four bonding pairs and two lone pairs of electrons. The bonding pairs form a square around the central atom, with nonbonding pairs above and below the plane.

528. trigonal bipyramidal
The trigonal bipyramidal shape has atoms that are 90° apart (the three atoms that are in same the plane are 90° from the atoms above and below the plane), 120° apart (the three atoms in the plane are 120° away from each other), and 180° apart (the atoms above and below the plane are in a line 180° apart).

529. tetrahedral
A tetrahedral geometry has bond angles of 109.5°. The bond angles are slightly less than 109.5° in the trigonal pyramidal and bent structures because repulsion between electrons causes the nonbonding electron pairs to take up more room in the compound.
530. seesaw
Start with the Lewis structure of SF4:
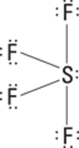
The diagram shows four bonding pairs of electrons (indicated by the lines) between the sulfur and the fluorine atoms. The sulfur also has a lone pair. You can disregard the lone pairs on the fluorine because they don’t determine the structure. The five pairs around the sulfur adopt a trigonal bipyramidal electron-pair geometry (a double pyramid with a triangular base).
The lone pair is one of the trigonal pairs (separated from each other by 120°), leaving atoms at the other two trigonal pairs and at the two pairs at the apexes of the pyramids. This shape is a seesaw molecular geometry.
531. trigonal bipyramidal
Look at the Lewis structure of PCl5:
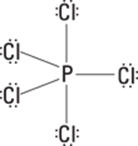
The diagram shows five bonding pairs of electrons (indicated by the lines) between the phosphorus and the chlorine atoms. The five pairs around the phosphorus adopt a trigonal bipyramidal electron pair geometry (a double pyramid with a triangular base). Three of the pairs form a trigonal planar arrangement (the bonds are 120° apart).
532. square pyramidal
Start with the Lewis structure of BrF5:
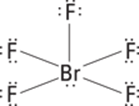
The diagram shows five bonding pairs of electrons (indicated by the lines) between the bromine and the fluorine atoms. The bromine also has a lone pair of electrons. The six pairs around the bromine adopt an octahedral electron-pair geometry (all pairs are at 90° angles, forming a double pyramid with a square base). One of the bonding pairs, at 90° to the square plane, forms the apex of a pyramid.
533. octahedral
Look at the Lewis structure of SF6:
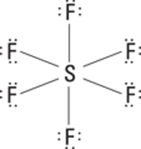
The diagram shows six bonding pairs of electrons (indicated by the lines) between the sulfur and the fluorine atoms. The six pairs around the sulfur adopt an octahedral electron pair geometry (all pairs are at 90° angles, forming a double pyramid with a square base).
534. square planar
Start with the Lewis structure of XeF4:

The diagram shows four bonding pairs of electrons (indicated by the lines) between the xenon and the fluorine atoms. The xenon also has two lone pairs of electrons. The six pairs around the xenon adopt an octahedral electron-pair geometry (all pairs are at 90° angles, forming a double pyramid with a square base). The two lone pairs are on opposite sides of the xenon, leaving all the atoms in a plane. This is a square planar geometry.
535. T-shaped
Start with the Lewis structure of ClF3:
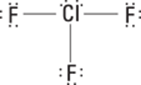
The diagram shows three bonding pairs of electrons (indicated by the lines) between the chlorine and the fluorine atoms. The chlorine also has two lone pairs. You can disregard the lone pairs on the fluorine, because they don’t determine the structure. The five pairs around the sulfur adopt a trigonal bipyramidal electron-pair geometry (a double pyramid with a triangular base).
The lone pairs are two of the trigonal pairs (separated by 120°), leaving atoms at the other trigonal pair and at the two pairs at the apexes of the pyramids. This is a T-shaped molecular geometry.
536. trigonal bipyramidal; linear
Start with the Lewis structure of xenon difluoride, XeF2:
![]()
The diagram shows two bonding pairs of electrons (indicated by the lines) between the xenon and the fluorine atoms. The xenon also has three lone pairs. You can disregard the lone pairs on the fluorine, because they don’t determine the structure. The five pairs around the xenon adopt a trigonal bipyramidal electron-pair geometry (a double pyramid with a triangular base).
The lone pairs are the trigonal pairs (separated by 120°), leaving atoms at both pairs at the apexes of the pyramids. This is a linear molecular geometry.
537. the polarity of the bonds, shape of the molecule, and distribution of the electron pairs on the atoms
To determine the polarity of a molecule, you need to know the polarity of the bonds in the molecule, the shape of the molecule, and the distribution of electron pairs on all the atoms in the molecule.
538. nonpolar; nonpolar
Elements with similar electronegativity values normally form nonpolar covalent bonds. Hydrogen is bonding to itself in H2, so the electronegativity difference is zero. If a molecule has only nonpolar bonds, it’s nonpolar.
539. polar; polar
Elements that have a moderate difference in their electronegativity values normally form polar covalent bonds. Nonmetals that aren’t adjacent on the periodic table, such as hydrogen and chlorine, usually have moderate differences (note that hydrogen is special in that its electronegativity places it between boron and carbon). For diatomic molecules like HCl, if the bond is polar covalent, so is the molecule.
540. polar; nonpolar
Elements that have a moderate difference in their electronegativity values normally form polar covalent bonds. Nonmetals that aren’t adjacent on the periodic table, such as carbon and chlorine, usually have moderate differences.
In CCl4, the symmetrical arrangement of the four polar bonds (tetrahedral) results in their polarities canceling, leaving a nonpolar molecule:
![]()
541. polar; polar
Elements that have a moderate difference in their electronegativity values normally form polar covalent bonds. Nonmetals that aren’t adjacent on the periodic table usually have moderate differences (note that hydrogen is special in that its electronegativity places it between boron and the carbon).
In NH3, the three bonding pairs are on one side of the molecule (the base of the trigonal pyramid), making the base of the pyramid positive and the top negative. This makes the molecule polar.
![]()
542. polar; polar
Elements that have a moderate difference in their electronegativity values normally form polar covalent bonds. Nonmetals that aren’t adjacent on the periodic table usually have moderate differences. (Note: Diagonal elements are 1.4 times as far away from each other as side-by-side elements; if you go over one and down one, you can think of the elements as though they’re nonadjacent; they’re twice as far away.)
The SeCl4 molecule has a seesaw shape, so the polarities of the end atoms (180° apart) cancel. The other two polar bonds pull in the same general direction to make the molecule polar.
543. polar; nonpolar
Elements that have a moderate difference in their electronegativity values normally form polar covalent bonds. Nonmetals that aren’t adjacent on the period table, such as xenon and oxygen, usually have moderate differences.
In XeO4, the symmetrical arrangement of the four polar bonds (tetrahedral) results in their polarities canceling, leaving a nonpolar molecule.
544. polar; polar
Elements that have a moderate difference in their electronegativity values normally form polar covalent bonds. Nonmetals that aren’t adjacent on the periodic table, such as iodine and chlorine, usually have moderate differences.
ICl3 is a T-shaped molecule. The two polar covalent bonds forming the top of the T cancel, and the remaining polar covalent bond makes the molecule polar.
545. OF2, NBr3, PCl3, and IF5
OF2, PCl3, and IF5 are all polar because they contain polar bonds and have uneven distributions of electrons on asymmetric molecules (their respective molecular shapes are bent, trigonal pyramidal, and square pyramidal). NBr3 has nonpolar bonds and an even distribution of electrons all the way around the molecule, but because the molecule is trigonal pyramidal (because it has three bonding pairs of electrons and one nonbonding pair), the molecule is polar.
546. decomposition
When one reactant forms two or more products, you have a decomposition reaction. In this case, NaCl breaks down into Na and Cl2.
547. single displacement
In this reaction, the sodium displaces the hydrogen from the HCl. When an element replaces an element with similar charge in a compound, forming a new compound and leaving the replaced element by itself, the reaction is a single displacement reaction.
548. combination
In a combination reaction, also known as a synthesis reaction, two elements react to form a single compound.
549. combustion
When oxygen in the air reacts with a compound containing carbon (with an ignition source) to form carbon dioxide and water, you have a combustion reaction.
If insufficient oxygen is present, incomplete combustion may occur. In that case, CO or even C(s) may form in place of the CO2.
550. double displacement
A reaction that occurs when two compounds exchange ions is a double displacement reaction. To make the swap easier to see, note that you can write H2O as HOH (or H–OH).
This specific type of double displacement reaction (an acid reacting with a base) is also known as a neutralization reaction.
551. combination
The two elements hydrogen and oxygen react to form a single compound, so this is a combination reaction.
552. combustion
When oxygen in the air reacts with a compound containing carbon (with an ignition source) to form carbon dioxide and water, you have a combustion reaction. Incomplete combustion may also have carbon monoxide as a product.
553. combination
The two elements nitrogen and hydrogen react to form a single compound, so this is a combination reaction.
554. decomposition
When one reactant forms two or more products, the reaction is a decomposition reaction.
555. single displacement
In a single displacement reaction, an element replaces an element of similar charge in a compound, forming a new compound and leaving the replaced element by itself. In this case, potassium displaces silver.
556. combination
![]()
When two elements react to form a single compound, you have a combination reaction.
557. double displacement
![]()
A reaction that occurs when two compounds exchange ions is a double displacement reaction. In this case, the sodium trades places with the lead (or equivalently, the hydroxide trades places with the nitrate).
558. single displacement
![]()
When an element replaces an element of similar charge in a compound, forming a new compound and leaving the replaced element by itself, the reaction is a single displacement reaction. In this case, magnesium displaces hydrogen.
559. decomposition
![]()
When one reactant forms two or more products, the reaction is a decomposition reaction.
560. combustion
![]()
When oxygen in the air reacts with a compound containing carbon (with an ignition source) to form carbon dioxide and water, the reaction is a combustion reaction. Incomplete combustion may also have carbon monoxide as a product.
561. single displacement
![]()
In a single displacement reaction, an element replaces an element of similar charge in a compound, forming a new compound and leaving the replaced element by itself. Here, iron takes the place of copper.
562. decomposition
![]()
When one reactant forms two or more products, the reaction is a decomposition reaction.
563. combustion
![]()
In a combustion reaction, oxygen in the air reacts with a compound containing carbon (with an ignition source) to form carbon dioxide and water. Incomplete combustion may also have carbon monoxide as a product.
564. combination
![]()
When two elements react to form a single compound, it’s a combination reaction.
565. combination
![]()
Two elements react to form a single compound, so this is a combination reaction. You can also classify this reaction as a combustion reaction.
566. single displacement
![]()
When an element replaces an element with similar charge in a compound, forming a new compound and leaving the replaced element by itself, you have a single displacement reaction.
567. decomposition
![]()
When one reactant forms two or more products, you have a decomposition reaction.
568. double displacement
![]()
A reaction that occurs when two compounds exchange ions is a double displacement reaction. Here, the calcium trades places with the hydrogen (or equivalently, the hydroxide trades places with the sulfate).
This type of double displacement reaction (an acid plus a base) is also known as a neutralization reaction.
569. combination
![]()
When two elements react to form a single compound, you have a combination reaction.
570. combustion
![]()
When oxygen in the air reacts with a compound containing carbon (with an ignition source) to form carbon dioxide and water, you have a combustion reaction. Incomplete combustion may also have carbon monoxide as a product.
571. double displacement
![]()
A reaction that occurs when two compounds exchange ions of the same charge is a double displacement reaction. In this case, the sodium trades places with the silver (or equivalently, the nitrate trades places with the chloride).
572. combination
![]()
When two elements react to form a single compound, the reaction is a combination reaction.
573. single displacement
![]()
In a single displacement reaction, an element replaces an element of similar charge in a compound, forming a new compound and leaving the replaced element by itself. In this case, chlorine displaces bromine.
574. decomposition
![]()
When one reactant forms two or more products, you have a decomposition reaction.
575. double displacement
![]()
A reaction that occurs when two compounds exchange ions is a double displacement reaction. In this case, the aluminum trades places with the calcium (or equivalently, the sulfate trades places with the phosphate).
576. 1, 2
For every N2O4 molecule that decomposes, you need two NO2 molecules, making the coefficients of the balanced equation 1 and 2:
![]()
577. 1, 3, 1, 1
The only element out of balance in this reaction is the chlorine. Two atoms of chlorine are on the reactants side of the equation, and six atoms of chlorine are on the products side of the equation. Multiplying the Cl2 by 3 makes the coefficients 1, 3, 1, and 1:
![]()
578. 1, 1, 1, 2
In this equation, the Na atoms and the ![]() ions are not in balance. Two sodium atoms and two nitrate ions are on the reactants side, and only one sodium atom and one nitrate ion are on the products side. Multiplying the NaNO3 by 2 balances the equation and makes the coefficients 1, 1, 1, and 2:
ions are not in balance. Two sodium atoms and two nitrate ions are on the reactants side, and only one sodium atom and one nitrate ion are on the products side. Multiplying the NaNO3 by 2 balances the equation and makes the coefficients 1, 1, 1, and 2:
![]()
579. 1, 1, 2
In this equation, neither the iodine nor the chlorine is balanced. You see two atoms of iodine and two atoms of chlorine on the reactants side but only one atom of iodine and one atom of chlorine on the product side. Multiplying ICl by 2 balances the iodine and the chlorine atoms, making the coefficients 1, 1, and 2:
![]()
580. 2, 1, 2, 1
In this equation, neither the chlorine nor the fluorine is balanced. You have one atom of chlorine and two atoms of fluorine on the reactants side but only one atom of fluorine and two atoms of chlorine on the products side. Multiplying the KCl by 2 on the reactants side balances the chlorine atoms, but that gives you two atoms of potassium on the reactants side. Multiplying the KF on the products side then balances the potassium and fluorine atoms, making the coefficients 2, 1, 2, and 1:
![]()
581. 1, 2, 1, 2
In this equation, only the carbon atoms are balanced. Multiplying the water on the products side by 2 balances the hydrogen atoms, giving you four on each side; it also increases the number of oxygen atoms on the products side to four. To balance the oxygen atoms, you need to multiply the O2 on the reactants side by 2, making the coefficients 1, 2, 1, and 2:
![]()
582. 2, 1, 2
To balance this equation, first balance the oxygen by multiplying CaO by 2. Next, balance the calcium by multiplying the Ca on the reactants side by 2. The coefficients are 2, 1, and 2:
![]()
583. 1, 3, 2
Neither the nitrogen nor the hydrogen is balanced in this equation. First, balance the nitrogen by multiplying the NH3 by 2, giving you two nitrogen atoms on each side. You now have six hydrogen atoms on the product side. To balance the hydrogen atoms, multiply the H2 by 3. This final coefficients are 1, 3, and 2:
![]()
584. 2, 1, 2
Initially, only the sulfur is balanced in this equation. You need more oxygen atoms on the product side, so begin by multiplying the SO3 by 2; this gives you six oxygen atoms and two sulfur atoms on the product side.
To balance the sulfur atoms, multiply the SO2 by 2 on the reactants side; this gives you two sulfur atoms and four oxygen atoms. When you add the four oxygen atoms from the 2SO2 to the two oxygen atoms from the O2, you see that you have six oxygen atoms on the reactants side, so the oxygen is balanced. The coefficients are 2, 1, and 2:
![]()
585. 3, 2, 1, 6
Nothing is balanced in this equation. Start with the Mg atoms. You have one Mg atom on the reactants side and three on the products side, so multiply the Mg(NO3)2 by 3; not only does this give you three Mg atoms on both sides, but it also gives you six NO3 ions on the reactants side, compared to the one NO3 ion on the products side.
To balance the NO3 ions, multiply KNO3 by 6 on the products side; this also gives you six K atoms. Last, multiply the K3PO4 by 2 to balance the K and PO4 ions. The final coefficients 3, 2, 1, and 6:
![]()
586. 1, 1, 1
Here’s the balanced equation:
![]()
The coefficients are 1, 1, and 1.
587. 1, 2, 1, 1
Write the unbalanced equation:
![]()
Only the chlorine atoms are unbalanced. To balance them, multiply the Cl2 by 2:
![]()
The coefficients are 1, 2, 1, and 1.
588. 1, 1, 2, 1
Write the unbalanced equation:
![]()
The silver atoms and the chlorate ions aren’t balanced. Multiplying the AgClO3 on the products side by 2 balances the equation:
![]()
The coefficients are 1, 1, 2, and 1.
589. 1, 2, 1, 1, 1
Write the unbalanced equation:
![]()
Only the hydrogen and chlorine atoms are unbalanced. Multiplying the HCl on the reactants side by 2 balances the equation:
![]()
The resulting coefficients are 1, 2, 1, 1, and 1.
590. 4, 3, 2
Write the unbalanced equation:
![]()
To balance the oxygen atoms on both sides of the equation, you look for the lowest common multiple of 2 and 3, which is 6. Multiply the O2 by 3 and the B2O3 by 2 to balance the oxygen atoms, giving you six on each side.
You end up with four boron atoms on the product side, so multiply the B on the reactants side by 4. The coefficients of the balanced reaction are 4, 3, and 2:
![]()
591. 5
Write the unbalanced equation:
![]()
To balance the iodine, multiply the NaI by 2. That gives you two sodium atoms on the product side, so multiply Na by 2 on the reactants side:
![]()
The coefficients are 2, 1, and 2, and they add up to 5.
592. 7
Write the unbalanced equation, remembering that oxygen gas is diatomic:
![]()
To balance this equation, you need to balance the oxygen atoms. The lowest common multiple of 3 and 2 is 6, so multiply the KClO3 by 2 and the O2 by 3 to get six oxygen atoms on each side.
Now you have two potassium and two chlorine atoms on the reactant side. To balance those atoms, multiply the KCl on the products side by 2, which gives you
![]()
The coefficients are 2, 2, and 3, and their sum is 7.
593. 9
Write the unbalanced equation, remembering that oxygen gas is diatomic:
![]()
First, balance the oxygen atoms. The lowest common multiple of 2 and 3 is 6, so multiply the Fe2O3 by 2 (which also gives you four Fe atoms) and the O2 by 3, giving you six oxygen atoms on each side.
Multiply the Fe on the reactants side by 4 to finish balancing the equation:
![]()
The coefficients are 4, 3, and 2, and they add up to 9.
594. 12
Write the unbalanced equation, remembering that Ca2+ plus N3– gives you Ca3N2 (calcium nitride):
![]()
Tip: As you balance this equation, thinking of H2O as HOH (or H–OH) may be helpful. That way, you can easily see where the OH in Ca(OH)2 and the H in NH3 come from.
Balance the calcium first by multiplying the Ca(OH)2 on the products side by 3. Then balance the nitrogen by multiplying the NH3 by 2.
Now you need to balance the hydrogens and hydroxides. You can do this by multiplying the H2O by 6. Here’s the final, balanced equation:
![]()
The coefficients are 1, 6, 3, and 2, and they add up to 12.
595. 2, 25, 16, 18
Write the unbalanced equation:
![]()
Tip: When balancing a combustion reaction with a hydrocarbon, balance the carbon first, the hydrogen second, and the oxygen last.
Balance the carbon and hydrogen by multiplying the carbon dioxide on the products side by 8 and multiplying the water on the products side by 9:
![]()
Sometimes when balancing the C and H, you end up with an odd number of O atoms on the products side. In this case, 8CO2 + 9H2O gives you 16 + 9 = 25 oxygen atoms. To remedy this, multiply the C8H18 by 2. Then continue to balance, doubling your carbon and hydrogen atoms on the products side:
![]()
That gives you an even number of oxygen atoms on the products side (32 + 18 = 50), so multiply the O2 by 25. The final coefficients are 2, 25, 16, and 18:
![]()
596. 3
Iron(III) bromide and barium hydroxide are two ionic compounds, so first determine what kind of reaction takes place. You may be able to do that from just the words, or you may need to write the formulas for the reactants first:
![]()
You should recognize that a double displacement reaction will occur. The anions or cations will change places in the two compounds, and the products will be iron(III) hydroxide and barium bromide. Here’s the unbalanced equation:
![]()
The bromine atoms and the hydroxide ions aren’t balanced. You can remedy the bromine imbalance by multiplying the FeBr3 by 2 and the BaBr2 by 3, giving you six bromine atoms on each side.
Now you need to balance the Ba2+, Fe3+, and OH–, which you can do by multiplying the Ba(OH)2 by 3 and the Fe(OH)3 by 2. The balanced equation is
![]()
The coefficient on the barium hydroxide is 3.
597. 1
In this reaction, an element (potassium) reacts with a compound (nickel(II) chloride), and that pattern characterizes this reaction as a single displacement reaction. The positively charged ions trade places, so the potassium replaces the nickel to make potassium chloride. Here’s the unbalanced equation:
![]()
To balance the chlorine, multiply the potassium chloride on the products side by 2. Then multiply the potassium on the reactants side by 2 to balance the potassium. Here’s the balanced equation:
![]()
The coefficient on the nickel(II) chloride is 1.
598. 5
When only one reactant shows up in an equation, you can be pretty sure that the reaction is a decomposition reaction. Metallic oxides with high metal charges decompose into metallic oxides with lower metal charges. Lead(IV) oxide decomposes into lead(II) oxide and oxygen gas, so here’s the unbalanced equation:
![]()
To balance this equation, simply multiply the PbO by 2 to get an even number of oxygen atoms; then multiply the PbO2 by 2 to balance the lead atoms:
![]()
The coefficients are 2, 2, and 1, which add up to 5.
Here’s another possible reaction (note that the coefficients still sum to 5):
![]()
599. 3
This reaction involves two compounds, each with a positive and a negative ion, so this must be a double displacement reaction. The silver will change places with the aluminum (or equivalently, the nitrate will change places with the chlorine) to form silver chloride and aluminum nitrate. Here’s the unbalanced equation:
![]()
The chlorine atoms and the nitrate ions aren’t balanced. To remedy this situation, multiply the AgCl by 3 to balance the chlorine; then multiply the AgNO3 by 3 to balance the silver and nitrate:
![]()
The coefficient on the silver nitrate is 3.
600. 6
When only one reactant shows up in an equation, you can be pretty sure you’re dealing with a decomposition reaction. With a nonmetallic halide, the products are two elements (the nonmetal and the halogen), so here’s the unbalanced equation:
![]()
First, multiply the NCl3 by 2 to even out the nitrogen atoms on each side of the equation. Next, balance the chlorine atoms by multiplying the Cl2 by 3:
![]()
The sum of the coefficients is 2 + 1 + 3 = 6.
601. ![]()
Here, two elements are reacting with each other to form a compound, making this a combination reaction. Write the unbalanced equation, remembering that oxygen gas is diatomic and that zinc, as a metal, is monatomic:
![]()
Multiply the ZnO by 2 to balance the oxygen atoms and multiply the Zn by 2 to balance the zinc atoms:
![]()
602. 9
In this reaction, a compound is reacting with an element — the pattern of a single displacement reaction. The positive hydrogen ion will displace the positive iron ion in iron(III) oxide to make water, leaving Fe as the metal. Here’s the unbalanced equation:
![]()
First balance the oxygen atoms by multiplying the H2O by 3. Then multiply the H2 by 3 to balance the hydrogen atoms. Next, you need to balance the iron atoms, so multiply the Fe on the products side by 2. Here’s the balanced equation:
![]()
The sum of the coefficients of this reaction is 1 + 3 + 2 + 3 = 9.
603. 2
Only one reactant is given, so this reaction is a decomposition reaction. Nonmetallic oxides decompose to give you a nonmetal and oxygen gas. Write the unbalanced equation, remembering that elemental gases are diatomic (except for the noble gases, which are monatomic):
![]()
To balance the nitrogen, multiply the NO2 on the reactant side by 2. This gives you four oxygen atoms on the reactant side, so also multiply the O2 by 2. Here’s the balanced equation:
![]()
The coefficient on the nitrogen dioxide is 2.
604. ![]()
The reactants are two compounds, each with a positive ion and a negative ion, so this must be a double displacement reaction. The silver will change places with the potassium (or equivalently, the nitrate will change places with the dichromate) to form silver dichromate and potassium nitrate. Here’s the unbalanced equation:
![]()
The potassium atoms and the silver ions aren’t balanced. To balance the potassium, multiply the KNO3 by 2. Then multiply the AgNO3 by 2 to balance the silver and the nitrate:
![]()
605. 3
In this case, an element (aluminum) reacts with a compound (copper(II) sulfate), which is the pattern of a single displacement reaction. The ions with the positive charges trade places, so the aluminum replaces the copper to make aluminum sulfate:
![]()
To balance this equation, you need to multiply the CuSO4 by 3, because you have three SO42– ions in aluminum sulfate. Next, multiply the Cu on the products side by 3 to balance the copper atoms. Last, multiply the Al on the reactants side by 2, giving you the balanced equation:
![]()
The coefficient on the copper(II) sulfate is now 3.
606. +7
To find the oxidation number of an element in a compound, set up an algebraic equation. Because KMnO4 is a neutral compound, the sum of the charges of all the atoms in the compound equals zero:
K + Mn + 4O = 0
Substitute in the oxidation numbers of the elements other than manganese. Potassium is in Group 1 (IA) on the periodic table, so its expected oxidation state is +1. Oxygen is in Group 16 (VIA), so its expected oxidation state is –2.
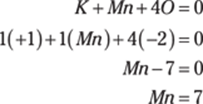
The oxidation number of manganese is +7.
607. +5
To find the oxidation number of an element in a compound, set up an algebraic equation. Because NaBrO3 is a neutral compound, the sum of the charges of all the atoms in the compound equals zero:
Na + Br + 3O = 0
Substitute in the oxidation numbers of the elements other than bromine. Sodium is in Group 1 (IA) on the periodic table, so its expected oxidation state is +1. Oxygen is in Group 16 (VIA), so its expected oxidation state is –2. Enter the values and then solve:
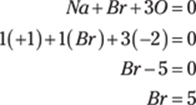
The oxidation number of bromine is +5.
608. +6
To find the oxidation number of an element in a compound, set up an algebraic equation. Because Cr2O72– is a polyatomic ion, the sum of the charges of all the atoms in the ion equals the charge on the ion:
2Cr + 7O = –2
Next, substitute in the oxidation numbers of any elements other than chromium. Oxygen is in Group 16 (VIA), so its most common oxidation state is –2. Then solve:
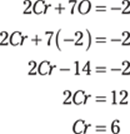
The oxidation number of chromium is +6.
609. +2
To find the oxidation number of an element in a compound, set up an algebraic equation. Because S2O32– is a polyatomic ion, the sum of the charges of all the atoms in the ion equals the charge on the ion:
2S + 3O = –2
Substitute in the oxidation number of the oxygen, which is more electronegative than sulfur. Oxygen is in Group 16 (VIA) on the periodic table, so its expected oxidation state is –2. Then solve:
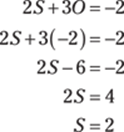
The oxidation number of sulfur is +2.
610. oxidation
Oxidation occurs when a substance loses electrons.
611. reduction
Reduction occurs when a substance gains electrons.
612. Fe
Fe loses two electrons to become Fe2+, so Fe is oxidized.
613. Fe2+
Fe2+ gains two electrons to become Fe, so it’s reduced.
614. Cu
The reducing agent is the substance that’s been oxidized; that is, it’s the one that loses electrons. In this equation, copper loses electrons:
![]()
615. Br2
The oxidizing agent is the substance that’s been reduced; that is, it’s the one that gains electrons. In this equation, Br2 gains electrons:
![]()
Notice that Br2 is also the reducing agent:
![]()
616. 3e–
To balance this half-reaction, first balance the oxygen atoms:
![]()
The water comes from the oxygen and hydrogen present in the acidic environment.
Then balance the hydrogen atoms:
![]()
Finally, balance the charge, giving you three electrons:
![]()
617. 10e–
To balance this half-reaction, first balance the bromine atoms:
![]()
Then balance the oxygen atoms, followed by the hydrogen atoms. Add twice as many OH– as you need to the oxygen-deficient side. Then add half as many H2O molecules to the opposite side to balance the H atoms:
![]()
Finally, balance the charge, giving you 10 electrons:
![]()
618. 8
The two half-reactions for this equation are as follows:
![]()
You need to balance the first half-reaction only for charge. To do this, add two electrons to the products side:
![]()
The second half-reaction needs oxygen atoms on the products side, so add water to the products side and then add hydrogen ions to the reactants side:
![]()
Next, add eight electrons to the reactants side to balance the charge:
![]()
The number of electrons on each side of the half-reactions needs to match, so finish balancing the equation by multiplying the zinc half-reaction ![]() by 4 and then adding the two half-reactions back together:
by 4 and then adding the two half-reactions back together:
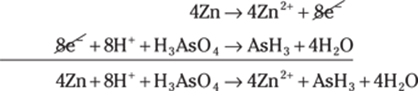
The equation is now balanced, with eight hydrogen ions on the reactants side.
619. 6
The two half-reactions for this equation are as follows:
![]()
You need to balance the first half-reaction only for charge. To do this, add three electrons to the reactant side:
![]()
For the second half-reaction, you need to balance the oxygen atoms. To do this in the presence of a base (OH–), note how many oxygen atoms you need to add to the oxygen-deficient side and add twice as many hydroxide ions; then balance out the hydrogen atoms by adding half as many water molecules to the opposite side. In this case, you add two hydroxide ions to the reactants and one water molecule to the products side, for a net gain of one oxygen atom:
![]()
Next, balance the charge by adding two electrons to the products side:
![]()
When you add the two half-reactions together, the number of electrons needs to cancel. So multiply the first half-reaction ![]() by 2 and multiply the second half-reaction
by 2 and multiply the second half-reaction ![]() by 3. Here’s what you get when you add the half-reactions:
by 3. Here’s what you get when you add the half-reactions:
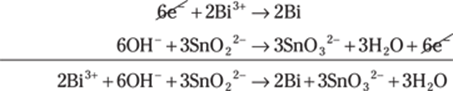
Both the atoms and charge are in balance. The question asks for the coefficient on the hydroxide ion in the balanced equation, so the answer is 6.
620. 1, 5, 3, 3, 3, 3
First, separate the substances that would dissolve in water and eliminate the spectator ions:
![]()
Then write your two half-reactions:
![]()
Next, balance the iodine atoms, then the oxygen atoms, and then the hydrogen atoms in the first half-reaction. Note that in an acidic solution, excess oxygen combines with hydrogen atoms from the acidic environment to produce water. Here’s the first half-reaction:
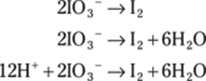
For the second half-reaction, you just balance the iodine atoms:
![]()
Now balance the charge in each half-reaction by adding electrons where needed:
![]()
Next, balance the charge — that is, make the number of electrons in each half-reaction equal. In this case, the lowest common multiple of 10 and 2 is 10, so you multiply the second half-reaction ![]() by 5 to get 10 electrons. Add the half-reactions and cancel the electrons:
by 5 to get 10 electrons. Add the half-reactions and cancel the electrons:
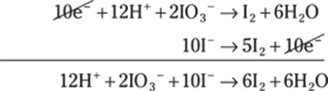
Now substitute this equation into the original molecular equation, reinserting the spectator ions (K+ and ![]() ):
):
![]()
You have 12 hydrogen ions on the reactants side, so multiply the H2SO4 by 6; then multiply K2SO4 by 6 to balance the potassium and sulfate ions with the ones on the reactants side of the equation:
![]()
You can divide everything by 2, so here’s the final equation:
![]()
The resulting coefficients are 1, 5, 3, 3, 3, and 3.
621. 3
At first, this equation may appear to be balanced. In fact, the atoms are balanced, but the charge isn’t. The two half-reactions for this equation are as follows:
![]()
You need to balance the oxygen atoms in each half-reaction. To do this in a basic solution, think of how many oxygen atoms you need on the oxygen-deficient side and add twice as many hydroxide ions; then balance out the hydrogen atoms by adding half as many water molecules to the opposite side. In this case, you add four hydroxide ions to the oxygen-deficient side and two water molecules to the opposite side, for a net gain of two oxygen atoms:
![]()
Next, balance the charge in each half-reaction by adding electrons. In this case, add three electrons to the reactants in the first half-reaction:
![]()
Then add four electrons to the products in the second half-reaction:
![]()
The number of electrons in each half-reaction must be equal. The lowest common multiple of 3 and 4 is 12, so multiply the first half-reaction ![]() by 4 and the second half-reaction
by 4 and the second half-reaction ![]() by 3. Then add the half-reactions together and simplify:
by 3. Then add the half-reactions together and simplify:
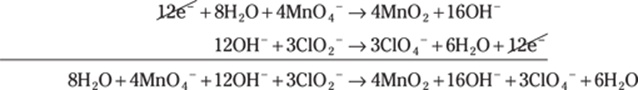
Last, remove the terms that appear on both sides of the equation. Six water molecules cancel out, reducing the eight water molecules on the reactants side to two. Twelve hydroxide ions cancel out, reducing the number of hydroxide ions on the products side from sixteen to four. Here’s the reaction:
![]()
Double-check to make sure all the atoms and the charge are in balance. They’re balanced, so the coefficient of ![]() is 3.
is 3.
622. 74.56 g/mol
Find the molar masses of the elements in KCl by looking up their atomic masses on the periodic table. The molar mass of potassium is 39.102 g/mol, and the molar mass of chlorine is 35.453 g/mol. Adding the masses gives you 39.102 + 35.453 = 74.555 g/mol, which rounds to 74.56 g/mol.
623. 56.08 g/mol
Find the molar masses of the elements in CaO by looking up their atomic masses on the periodic table. The molar mass of calcium is 40.08 g/mol, and the molar mass of oxygen is 15.9994 g/mol. The sum is 40.08 + 15.9994 = 56.0794 g/mol, which rounds to 56.08 g/mol.
624. 83.98 g/mol
Find the molar masses of the elements in AlF3 by looking up their atomic masses on the periodic table. Based on the formula, you need to multiply the molar mass of fluorine (18.9984 g/mol) by 3, the number of fluorine atoms. You can use the molar mass of aluminum (26.9815 g/mol) directly.
Add the masses of the elements together and then round to the hundredths place. The sum is 26.9815 + (18.9984 × 3) = 83.9767 g/mol, which rounds to 83.98 g/mol.
625. 69.62 g/mol
Find the molar masses of the elements in B2O3 by looking up their atomic masses on the periodic table. The formula includes two boron atoms and three oxygen atoms, so multiply the molar mass of boron (10.811 g/mol) by 2 and multiply the molar mass of oxygen (15.9994 g/mol) by 3.
Add the masses of the elements and round to the hundredths place. The sum is (10.811 × 2) + (15.9994 × 3) = 69.6202 g/mol, which rounds to 69.62 g/mol.
626. 331.63 g/mol
Find the molar masses of the elements in CBr4 by looking up their atomic masses on the periodic table. Carbon has a molar mass of 12.01115 g/mol, and bromine has a molar mass of 79.904 g/mol. The formula includes four bromine atoms, so multiply the molar mass of bromine by 4.
Add the masses of the elements and round to the hundredths place. The sum is 12.01115 + (79.904 × 4) = 331.62715 g/mol, which rounds to 331.63 g/mol.
627. 53.49 g/mol
Find the molar masses of the elements in NH4Cl by looking up their atomic masses on the periodic table. Nitrogen has a molar mass of 14.0067 g/mol, hydrogen has a molar mass of 1.00797 g/mol, and chlorine has a molar mass of 35.453 g/mol. The formula includes four hydrogen atoms, so multiply the molar mass of hydrogen by 4.
Add the masses of the elements and round to the hundredths place. The sum is 14.0067 + (1.00797 × 4) + 35.453 = 53.49158 g/mol, which rounds to 53.49 g/mol.
628. 261.35 g/mol
Find the molar masses of the elements in Ba(NO3)2 by looking up their atomic masses on the periodic table. Based on the formula for barium nitrate, you need to multiply the molar mass of nitrogen (14.0067 g/mol) by 2, the number of nitrogen atoms present; you also need to multiply the molar mass of oxygen (15.9994 g/mol) by 6, the number of oxygen atoms present. You can use the molar mass of barium (137.34 g/mol) directly.
Add the masses of the elements and round to the hundredths place. The sum is 137.34 + (14.0067 × 2) + (15.9994 × 3 × 2) = 261.3498 g/mol, which rounds to 261.35 g/mol.
629. 132.14 g/mol
Find the molar masses of the elements in (NH4)2SO4 by looking up their atomic masses on the periodic table. Ammonium sulfate contains two ammonium ions for every sulfate ion, giving you two nitrogen atoms, eight hydrogen atoms, one sulfur atom, and four oxygen atoms. Multiply the molar mass of each element by the proper number of atoms, add the results, and round to the hundredths place. The sum is (14.0067 × 2) + (1.00797 × 4 × 2) + 32.064 + (15.9994 × 4) = 132.13876 g/mol, which rounds to 132.14 g/mol.
630. 247.80 g/mol
First, write the formula for silver sulfide. Silver is Ag+ and sulfide is S2–, so the formula is Ag2S. Next, find the molar masses of the elements by looking up their atomic masses on the periodic table.
Based on the formula, you need to multiply the molar mass of silver (107.8682 g/mol) by 2, the number of silver atoms. You can use the molar mass of sulfur (32.064 g/mol) directly. Add the masses together and round to the hundredths place. The sum is (107.8682 × 2) + 32.064 = 247.8004 g/mol, which rounds to 247.80 g/mol.
631. 105.99 g/mol
First, write the formula for sodium carbonate. The sodium ion is Na+ and carbonate is ![]() , so the formula is Na2CO3. Next, find the molar masses of the elements by looking up their atomic masses on the periodic table.
, so the formula is Na2CO3. Next, find the molar masses of the elements by looking up their atomic masses on the periodic table.
Based on the formula, you need to multiply the molar mass of sodium (28.9898 g/mol) by 2, the number of sodium atoms, and multiply the molar mass of oxygen (15.9994 g/mol) by 3, the number of oxygen atoms. You can use the molar mass of carbon (12.0115 g/mol) directly. Add the masses together and round to the hundredths place. The sum is (22.9898 × 2) + 12.0115 + (15.9994 × 3) = 105.9893 g/mol, which rounds to 105.99 g/mol.
632. 318.02 g/mol
Find the molar masses of the elements in Al2(C2O4)3 by looking up their atomic masses on the periodic table. In aluminum oxalate, you have two aluminum ions and three oxalate ions. Each oxalate ion (C2O42–) contains two carbon atoms and four oxygen atoms. When you multiply the atoms in the oxalate ion by 3, you get a total of six carbon atoms (2 × 3) and twelve oxygen atoms (4 × 3) in the formula. The sum of the molar masses is (26.9815 × 2) + (12.0115 × 2 × 3) + (15.9994 × 4 × 3) = 318.0248 g/mol, which rounds to 318.02 g/mol.
633. 204.40 g/mol
Find the molar masses of the elements in Zn(NH3)4Cl2 by looking up their atomic masses on the periodic table. This compound, which contains a complexion, includes four ammonia molecules combined with a zinc ion and two chlorine ions. Based on the formula, you need to multiply the molar mass of nitrogen (14.0067 g/mol) by 4, the number of nitrogen atoms; multiply the molar mass of hydrogen (1.00979 g/mol) by 12, the number of hydrogen atoms; multiply the molar mass of chlorine (35.453 g/mol) by 2, the number of chlorine atoms; and use the molar mass of zinc (65.37 g/mol) directly. The sum of the molar masses is 65.37 + (14.0067 × 4) + (1.00797 × 3 × 4) + (35.453 × 2) = 204.39844 g/mol, which rounds to 204.40 g/mol.
634. 249.68 g/mol
This formula is for copper(II) sulfate pentahydrate. To calculate its molar mass, think of the molecule as copper sulfate plus five water molecules. So not only do you have one copper atom, one sulfur atom, and four oxygen atoms, but you also have ten hydrogen atoms and five more oxygen atoms to account for when calculating the molar mass.
Find the molar masses of the elements in CuSO4·5H2O by looking up their atomic masses on the periodic table, multiply the molar mass of each element by the proper number of atoms, and add the results. The sum is [63.546 + 32.064 + (15.9994 × 4)] + [(5 × 1.00797 × 2) + (5 × 15.9994)] = 249.6843 g/mol, which rounds to 249.68 g/mol.
635. 287.04 g/mol
The formula for manganese(II) nitrate hexahydrate begins with the manganese(II) (Mn2+) ion, followed by the nitrate ion ![]() . You need two nitrate ions to balance the +2 charge on the manganese. After the Mn(NO3)2, you need to account for the hexahydrate, which is six water molecules. The formula becomes Mn(NO3)2·6H2O.
. You need two nitrate ions to balance the +2 charge on the manganese. After the Mn(NO3)2, you need to account for the hexahydrate, which is six water molecules. The formula becomes Mn(NO3)2·6H2O.
The formula shows one manganese atom, two nitrogen atoms, six oxygen atoms, twelve hydrogen atoms, and six more oxygen atoms. Find the molar masses of the elements in Mn(NO2)3·6H2O by looking up their atomic masses on the periodic table, multiply the molar mass of each element by the proper number of atoms, and add the results. The sum is 54.9380 + (14.0067 × 2) + (15.9994 × 3 × 2) + (6 × 1.00797 × 2) + (6 × 15.9994) = 287.0384 g/mol, which rounds to 287.04 g/mol.
636. 501.61 g/mol
The formula for iron(II) phosphate octahydrate begins with the iron(II) ion (Fe2+), followed by the phosphate ion (PO43–), giving you the formula Fe3(PO4)2. Add the octahydrate (eight water molecules) to the Fe3(PO4)2, and you get Fe3(PO4)2·8H2O.
The formula shows three iron atoms, two phosphorus atoms, eight oxygen atoms, sixteen hydrogen atoms, and eight more oxygen atoms. Find the molar masses of the elements in Fe3(PO4)2·8H2O by looking up their atomic masses on the periodic table, multiply the molar mass of each element by the proper number of atoms, and add the results. The sum is [(55.847 × 3) + (30.9738 × 2) + (15.9994 × 4 × 2)] + [(8 × 1.00797 × 2) + (8 ×15.9994)] = 501.60652 g/mol, which rounds to 501.61 g/mol.
637. 22.34% Na
Divide the mass of 1 mol Na by the mass of 1 mol NaBr. The mass of NaBr is the sum of the masses of the atoms present. Then multiply by 100 to get the percent.
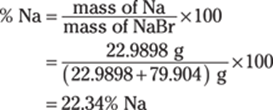
638. 73.21% Sr
Divide the mass of 1 mol Sr by the mass of 1 mol SrS. The mass of SrS is the sum of the masses of the atoms present. Then multiply by 100 to get the percent.
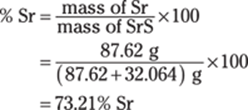
639. 28.93% Cl
Divide the mass of 1 mol Cl by the mass of 1 mol KClO3. In finding the mass of KClO3, multiply the mass of the oxygen by 3, because three oxygen atoms are present in the compound. Then multiply by 100 to get the percent.
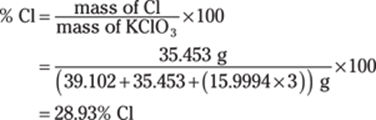
640. 49.96% O
Divide the mass of 4 mol O by the mass of 1 mol CaC2O4. Remember to multiply the molar mass of oxygen by 4, because four oxygen atoms are present in the compound. The mass of CaC2O4 is the sum of the masses of the atoms present, written in grams.
After dividing the masses, multiply by 100 to get the percent:
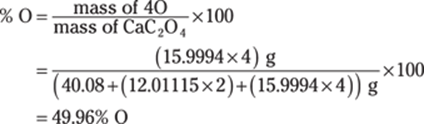
641. 40.56% S
Divide the mass of 2 mol S by the mass of 1 mol Na2S2O3. Remember to multiply the molar mass of sulfur by 2, because two sulfur atoms are present in the compound. The mass of Na2S2O3 is the sum of the atomic masses of the atoms present, written in grams.
After dividing the masses, multiply by 100 to get the percent:
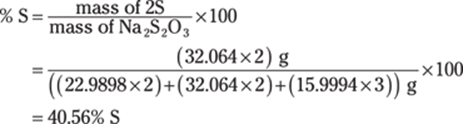
642. 35.00% N
Divide the mass of 2 mol N by the mass of 1 mol NH4NO3. Make sure you include both the nitrogen from the ammonium and the nitrogen from the nitrate in the mass of the nitrogen. The mass of NH4NO3 is the sum of the atomic masses of the atoms present, written in grams.
After dividing the masses, multiply by 100 to get the percent:
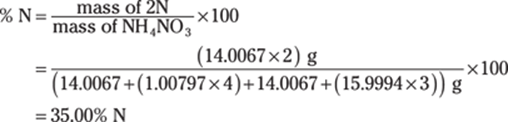
643. 10.21% Li
The first step in finding the mass percent of lithium is to write the formula of lithium hydrogen carbonate. The lithium ion is Li+ and the hydrogen carbonate ion is ![]() , so the formula is LiHCO3.
, so the formula is LiHCO3.
Next, divide the mass of 1 mol Li by the mass of 1 mol LiHCO3. The mass of LiHCO3 is the sum of the atomic masses of the atoms present, written in grams. Then multiply by 100 to get the percent:
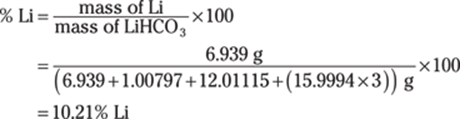
644. 87.06% Ag
First, write the formula for silver sulfide. The silver ion is Ag+ and the sulfide ion is S2–, so you need two positive silver ions to balance out the –2 charge of the sulfide ion; the formula is Ag2S.
Next, divide the mass of 2 mol Ag by the mass of 1 mol Ag2S. The mass of Ag2S is the sum of the atomic masses of the atoms present, written in grams. Multiply the result by 100 to get the percent silver:
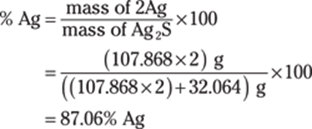
645. 34.59% Al, 61.53% O, and 3.88% H
To find the mass percent of each element in aluminum hydroxide, first write the formula for the compound. The aluminum ion is Al3+ and the hydroxide ion is OH–, so you need three hydroxide ions to balance the +3 charge of the aluminum; therefore, the formula is Al(OH)3.
Next, find the mass percent of each element by taking the molar mass of each element (based on the number of atoms in the formula), dividing by the mass of the compound, and then multiplying by 100 to get the percent:
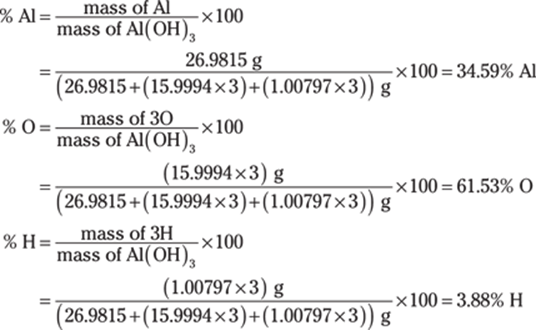
646. 15.75% Zn, 61.13% I, and 23.12% O
To find the mass percent of each element in zinc iodate, first write the formula for the compound. The formula comes from the Zn2+ ion and the ![]() ion. You need two negatively charged iodate ions to balance the +2 charge of the zinc ion, so the formula is Zn(IO3)2.
ion. You need two negatively charged iodate ions to balance the +2 charge of the zinc ion, so the formula is Zn(IO3)2.
To find the mass percent of each element, take the molar mass of each element (based on the number of atoms in the formula), divide by the molar mass of the compound, and then multiply by 100 to get the percent.
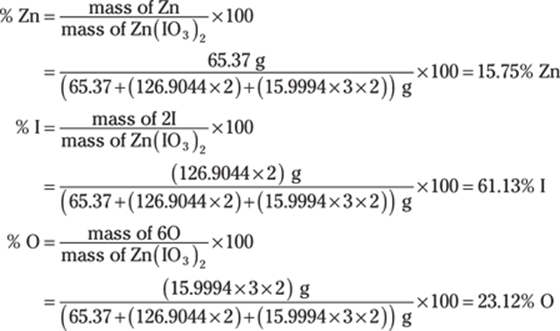
647. N2O5, NO2, N2O3, NO, N2O
In this problem, calculating the actual mass percent for each compound isn’t necessary because the compounds contain only nitrogen and oxygen (although you may still choose to calculate the values). Because only approximate values are necessary, you can simplify the calculations by rounding the molar masses to whole numbers, with 14 g/mol for nitrogen and 16 g/mol for oxygen.
For each compound, set up a ratio of the mass of nitrogen to the mass of the compound; then compare the ratios with a common numerator:
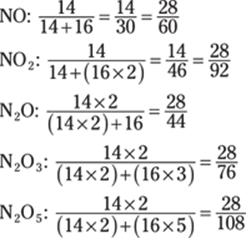
Arranging the ratios from smallest to largest gives you ![]() , so the correct order is N2O5, NO2, N2O3, NO, N2O.
, so the correct order is N2O5, NO2, N2O3, NO, N2O.
648. C2H6, CH4, CaC2O4, K2CO3, CCl4
Find the mass percent of carbon by taking the molar mass of carbon in each compound (based on the number of atoms in the formula), dividing that mass by the mass of the compound, and then multiplying the result by 100 to get the percent:
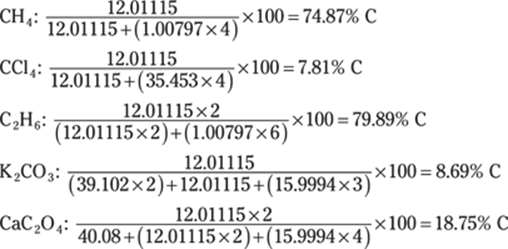
Arranging the percentages from largest to smallest gives you 79.89% C > 74.87% C > 18.75% C > 8.69% C > 7.81% C, so the correct order is C2H6, CH4, CaC2O4, K2CO3, CCl4.
649. K2SO4, K2SO3, K2S, KSCN, K2S2O3
To find the mass percent of sulfur, take the molar mass of sulfur in each compound (based on the number of atoms in the formula), divide by the mass of the compound, and then multiply by 100 to get the percent:
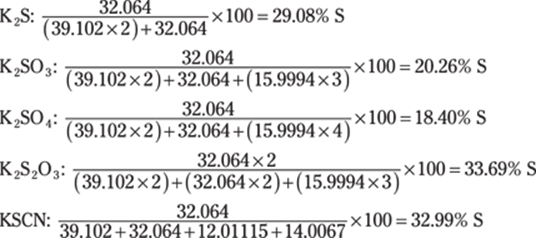
Arranging the percentages from smallest to largest gives you 18.40% S < 20.26% S < 29.08% S < 32.99% S < 33.69% S, so the correct order is K2SO4, K2SO3, K2S, KSCN, K2S2O3.
650. 36.08% H2O
To find the mass percent of water in the hydrate, take the mass of 1 mol of water (H2O), divide it by the mass 1 mol of the hydrated crystal (CuSO4·5H2O), and multiply by 100 to get the percent:
![]()
651. 67.34% O
When given the name of the compound, first write the formula. Sodium phosphate contains the sodium ion Na+ and the phosphate ion PO43–. To balance out the –3 charge on the phosphate ion, you need three sodium ions, so the first part of the formula is Na3PO4.
The dodecahydrate part of the name represents the 12 water molecules in the hydrated crystal. Remember to insert the dot between the sodium phosphate part of the formula and the water part (Na3PO4·12H2O), but don’t treat it as a multiplication sign when calculating the molar mass of the hydrated crystal. Think of it more like sodium phosphate plus 12 water molecules.
Take the mass of the oxygen (accounting for the four oxygen atoms in the phosphate and the twelve oxygen atoms in the water), divide it by the mass of 1 mol of the compound, and multiply by 100 to get the percent:
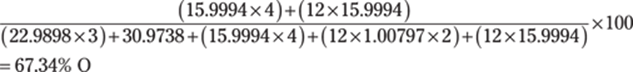
652. C2H3O2
The empirical formula is the simplest whole-number ratio of atoms in a compound. Out of the formulas listed, only C2H3O2 can’t be simplified. In each of the other compounds, the number of atoms in the formula is divisible by 2.
653. CH2
To find the empirical formula of a compound, you need to have counting units (or groupings) of each element, not the mass. If the question gives you the mass percent of each element, suppose that you have a 100.00-g sample such that 85.62% = 85.62 g. The first step for each element is to change the percent to grams. The second step is to convert grams to moles by dividing by the molar mass:
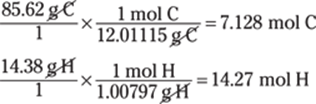
Next, compare the number of moles of each element: Divide the moles of each element by the smaller number of moles to get a ratio. Here, divide by 7.128 mol:
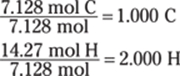
You have two hydrogen atoms for every atom of carbon, so the empirical formula is CH2.
654. CHCl3
To find the empirical formula of a compound, you need to have counting units (or groupings) of each element, not the mass. This question gives you the mass in grams, so convert the grams to moles by dividing by the molar mass:
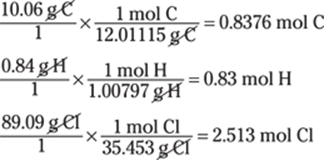
Next, compare the number of moles of each element: Divide the moles of each element by the smallest number of moles to get a ratio. Here, divide by 0.83 mol:
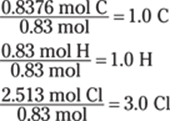
You have three chlorine atoms for every carbon and hydrogen atom, so the empirical formula for the compound is CHCl3.
655. FeCrO4
To find the empirical formula of a compound, you need to have counting units (or groupings) of each element, not the mass. This question gives you the mass in grams, so convert the grams to moles by dividing by the molar masses:
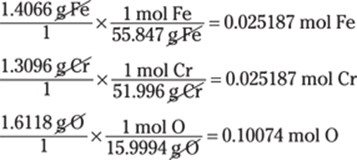
Next, compare the number of moles of each element: Divide the moles of each element by the smallest number of moles to get a ratio. Here, divide by 0.025187 mol:
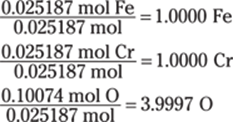
You have four oxygen atoms for every atom of iron and chromium, so the empirical formula for the compound is FeCrO4.
656. CdSO4
First, determine the mass of the anhydrous salt. If the 100.00-g sample includes 18.73 g of water, then the anhydrous salt has a mass of 100.00 g – 18.73 g = 81.27 g.
To find the number of grams of oxygen, subtract the mass of the cadmium and sulfur from the mass of the anhydrous salt: 81.27 g – (43.82 g + 12.50 g) = 24.95 g O. Use these numbers to find the empirical formula. First, change the masses to moles by dividing by the molar mass of each element:
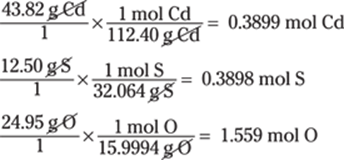
Then compare the number of moles of each element: Divide the moles of each element by the smallest number of moles to get a ratio. Here, divide by 0.3898 mol:
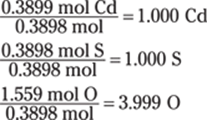
You have four oxygen atoms for every atom of cadmium and sulfur, so the anhydrous salt is CdSO4.
657. II only
The empirical formula is the lowest whole-number ratio of the each kind of atom in the compound. CH4 and C3H8 can’t be reduced, but you can reduce C2H2 to CH.
658. II and III
The empirical formula is the lowest whole-number ratio of the each kind of atom in the compound. CO2 can’t be reduced, but you can reduce C6H12 to CH2 and reduce C6H12O6 to CH2O.
659. C6H6
To find the molecular formula given the empirical formula and the molar mass of the molecular formula, find the ratio of the molecular formula to the empirical formula. Divide the molar mass of the molecular formula by the molar mass of the empirical formula:
![]()
Use that number to scale the empirical formula: 6 × CH = C6H6.
660. C6H14O2N4
To find the empirical formula of a compound, you need to have counting units (or groupings) of each element, not the mass. If the question gives you the mass percent of each element as this question does, suppose you have a 100.00-g sample, such that 41.37% = 41.37 g. The first step for each element is to change the percent to grams. The second step for each element is to convert grams to moles by dividing by the molar mass:
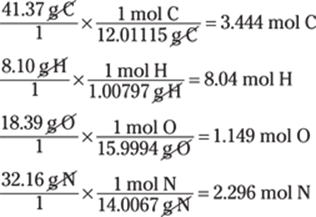
Next, compare the number of moles of each element. Divide the moles of each element by the smallest number of moles to get a ratio. Here, divide by 1.149 mol:
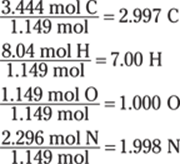
The ratios tell you that the empirical formula of the compound is C3H7ON2. To find the molecular formula, take the given molar mass and divide it by the molecular mass of the empirical formula:
![]()
The molecular formula is twice as big as the empirical formula, so the molecular formula for the compound is C6H14O2N4.
661. C9H9O5NI2 and C9H9O5NI2
To find the empirical formula of a compound, you need to have counting units (or groupings) of each element, not the mass. When a question gives you the mass percent of each element, suppose you have a 100.00-g sample, such that 23.25% = 23.25 g. The first step for each element is the change the percent to grams. The second step for each element is to convert grams to moles by dividing by the molar mass. To find the amount of iodine, subtract the masses of the other elements from 100.0 g before converting to moles.
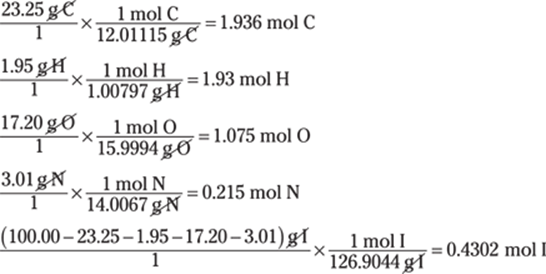
Next, compare the number of moles of each element. Divide the moles of each element by the smallest number of moles to get a ratio. Here, divide by 0.215 mol:
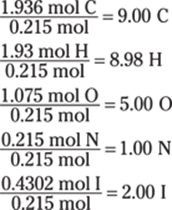
Therefore, the empirical formula of the compound is C9H9O5NI2. To find the molecular formula, take the given molar mass and divide it by the molar mass of the empirical formula:
![]()
This means that the molecular formula is the same as the empirical formula, C9H9O5NI2.
662. 0.171 mol NaCl
To find the number of moles when given the number of grams, divide the given mass by the molar mass of the compound and then round to the correct number of significant figures:
![]()
663. 0.361 mol BH3
To find the number of moles when given the number of grams, divide the given mass by the molar mass of the compound and then round to the correct number of significant figures:
![]()
664. 2.59 mol Na2CO3
To find the number of moles when given the number of grams, divide the given mass by the molar mass of the compound and round to the correct number of significant figures:
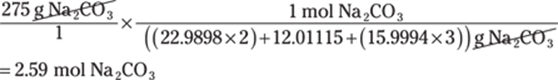
665. 11.4 mol NH4OH
To find the number of moles when given the number of grams, divide the given mass by the molar mass of the compound and round to the correct number of significant figures:
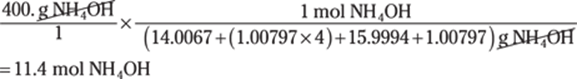
666. 0.63 mol KMnO4
To find the number of moles when given the number of grams, divide the given mass by the molar mass of the compound and round to the correct number of significant figures:
![]()
667. 3.65 mol BeS
First, write the formula for beryllium sulfide. The beryllium ion is Be2+, and the sulfide ion is S2–. The +2 charge on the beryllium ion cancels out the –2 charge on the sulfide ion, so the formula is BeS.
To find the number of moles when given the number of grams, divide the given mass by the molar mass of the compound and round to the correct number of significant figures:
![]()
668. 0.245 mol Al2O3
First, write the formula for aluminum oxide. The aluminum ion is Al3+, and the oxide ion is O2–. You need two +3 charged aluminum ions to balance out the three –2 charged oxygen ions, so the formula is Al2O3.
To find the number of moles when given the number of grams, divide the given mass by the molar mass of the compound and round to the correct number of significant figures:
![]()
669. 1.17 mol CCl4
First, write the formula for carbon tetrachloride. The prefix tetra- indicates that there are four chlorine atoms in the formula, and the lack of a prefix on the carbon means that there’s only one carbon atom, so the formula is CCl4.
Next, to find the number of moles when given the number of grams, divide the given mass by the molar mass of the compound and round to the correct number of significant figures:
![]()
670. 1.76 mol Ca3P2
First, write the formula for calcium phosphide. The calcium ion is Ca2+, and the phosphide ion is P3–. You need three +2 charged calcium ions to cancel out the two –3 charged phosphorus ions, so the formula is Ca3P2.
Next, to find the number of moles when given the number of grams, divide the given mass by the molar mass of the compound and round to the correct number of significant figures:
![]()
671. 0.00648 mol Mg(MnO4)2
First, write the formula for magnesium permanganate. The magnesium ion is Mg2+, and the permanganate ion is ![]() . You need two –1 charged permanganate ions to cancel out the +2 charged magnesium ion, so the formula is Mg(MnO4)2.
. You need two –1 charged permanganate ions to cancel out the +2 charged magnesium ion, so the formula is Mg(MnO4)2.
Next, to find the number of moles when given the number of grams, divide the given mass by the molar mass of the compound and round to the correct number of significant figures:
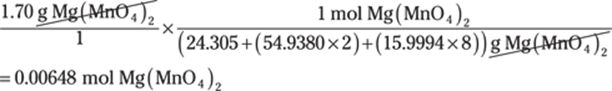
672. 377 g FeO
To find the number of grams when given the number of moles, multiply the given number of moles by the molar mass of the compound and round to the correct number of significant figures:
![]()
673. 101 g S2Cl2
To find the number of grams when given the number of moles, multiply the given number of moles by the molar mass of the compound and round to the correct number of significant figures:
![]()
674. 16,600 g KI
To find the number of grams when given the number of moles, multiply the given number of moles by the molar mass of the compound and round to the correct number of significant figures:
![]()
675. 4,230 g CaCO3
To find the number of grams when given the number of moles, multiply the given number of moles by the molar mass of the compound and round to the correct number of significant figures:
![]()
676. 1,700 g Al(NO3)3
To find the mass of given substance when given the number of moles, multiply the given number of moles by the molar mass of the compound and round to the correct number of significant figures:
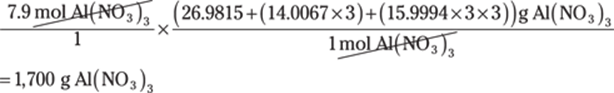
677. 50. g Pb(NO3)2
First, write the formula for lead(II) nitrate. The lead(II) ion is Pb2+, and the nitrate ion is ![]() . You need two –1 charged nitrate ions to cancel out the +2 charged lead ion, so the formula is Pb(NO3)2.
. You need two –1 charged nitrate ions to cancel out the +2 charged lead ion, so the formula is Pb(NO3)2.
Next, to find the number of grams when given the number of moles, multiply the given number of moles by the molar mass of the compound and round to the correct number of significant figures:
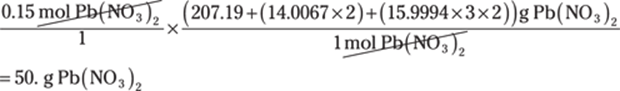
678. 3,400 g P4O10
First, write the formula for tetraphosphorus decoxide. The prefix tetra- indicates that there are four phosphorus atoms in the formula, and the prefix dec(a)- on the oxide means that there are ten oxygen atoms, so the formula is P4O10.
Next, to find the number of grams when given the number of moles, multiply the given number of moles by the molar mass of the compound and round to the correct number of significant figures:
![]()
679. 36 g Cu2O
First, write the formula for copper(I) oxide. The copper(I) ion is Cu+, and the oxide ion is O2–. You need two +1 charged copper ions to cancel out the –2 charged oxygen ion, so the formula is Cu2O.
Next, to find the number of grams when given the number of moles, multiply the given number of moles by the molar mass of the compound and round to the correct number of significant figures.
![]()
680. 24.6 kg Zn(OH)2
First, write the formula for zinc hydroxide. The zinc ion is Zn2+, and the hydroxide ion is OH–. You need two –1 charged hydroxide ions to cancel out the +2 charged zinc ion, so the formula is Zn(OH)2.
Next, to find the number of kilograms when given the number of moles, multiply the given number of moles by the molar mass of the compound, convert to kilograms, and round to the correct number of significant figures:
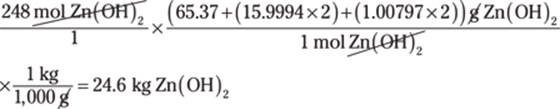
681. 48.5 kg (NH4)2SO4
First, write the formula for ammonium sulfate. The ammonium ion is ![]() , and the sulfate ion is SO42–. You need two +1 charged ammonium ions to cancel out the –2 charged sulfate ion, so the formula is (NH4)2SO4.
, and the sulfate ion is SO42–. You need two +1 charged ammonium ions to cancel out the –2 charged sulfate ion, so the formula is (NH4)2SO4.
Next, to find the number of kilograms when given the number of moles, multiply the given number of moles by the molar mass of the compound, convert to kilograms, and round to the correct number of significant figures:
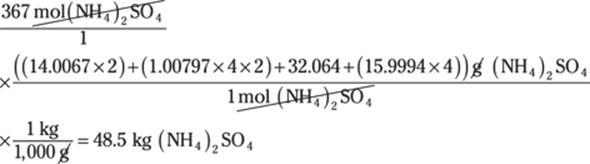
682. 3.76 × 1022 atoms of He
To find the number of atoms in a given the mass of a substance, first divide the given mass by the molar mass to get moles. Then multiply the result by Avogadro’s number of atoms in a mole. Finally, round the answer to the correct number of significant figures.
![]()
683. 6.7 × 1023 atoms of Al
To find the number of atoms in a given the mass of a substance, first divide the given mass by the molar mass to get moles. Then multiply the result by Avogadro’s number of atoms in a mole. Finally, round the answer to the correct number of significant figures.
![]()
684. 2.74 × 1024 molecules of CO2
Before starting the math on this question, be sure to write the formula for carbon dioxide: CO2.
To find the number of molecules in a given the mass of a substance, divide the given mass by the molar mass to get moles. Then multiply the result by Avogadro’s number of molecules in a mole. Finally, round the answer to the correct number of significant figures.
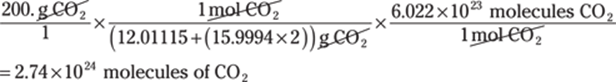
685. 3.3 × 1020 molecules of N2O4
First, write the formula for dinitrogen trioxide. The di- prefix on the nitrogen indicates that there are two nitrogen atoms in the formula, and the prefix tetr(a)- on the oxide tells you that there are four atoms of oxygen, so the formula is N2O4.
To find the number of molecules in a given the mass of a substance, first divide the given mass by the molar mass to get moles. Then multiply the result by Avogadro’s number of molecules in a mole. Finally, round answer to the correct number of significant figures.
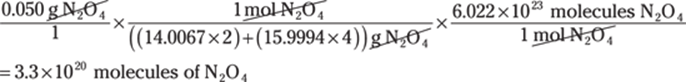
686. 4.70 × 1024 atoms of S
In this question, you need to take into account that there are eight atoms of sulfur in a molecule of S8, which will add one step to the end of the problem. First divide the given mass of S8 by the molar mass of S8 to get the moles of S8. Then multiply the result by Avogadro’s number of molecules in a mole. Finally, use the ratio of atoms of S in a molecule of S8 to get your answer, rounding to the correct number of significant figures.
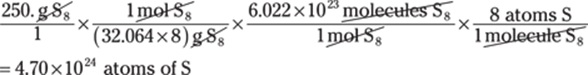
687. 2.5 × 105 g Br2
To find the mass in grams of a given number of molecules of a substance, first divide the given number of molecules by Avogadro’s number of molecules in a mole. Then multiply the result by the molar mass of the substance. Finally, round the answer to the correct number of significant figures.
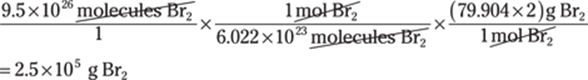
688. 8.095 × 10–1 g Ni
To find the mass in grams of a given number of atoms of a substance, first divide the given number of atoms by Avogadro’s number of atoms in a mole. Then multiply the result by the molar mass of the substance. Finally, round your answer to the correct number of significant figures.
![]()
689. 8.98 × 101 g C6H12O6
To find the mass in grams of a given number of molecules of a substance, first divide the given number of molecules by Avogadro’s number of molecules in a mole. Then multiply the result by the molar mass of the substance. Finally, round your answer to the correct number of significant figures.
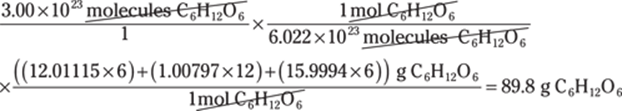
690. 9.0 × 10–15 g Kr
Krypton is a noble gas, so it’s made up of single atoms, not diatomic molecules. To find the mass in grams of a given number of atoms of a substance, first divide the given number of atoms by Avogadro’s number of atoms in a mole. Then multiply the result by the molar mass of the substance. Finally, round your answer to the correct number of significant figures.
![]()
691. 1.7 × 101 g AuF3
Before starting the math on this question, write the formula for gold(III) fluoride. The gold(III) ion is Au3+, and the fluoride ion is F–, so the formula is AuF3.
To find the number of grams in a given number of formula units of a substance, first divide the given number of formula units by Avogadro’s number of formula units in a mole. Then multiply the result by the molar mass of the substance. Finally, round the answer to the correct number of significant figures.
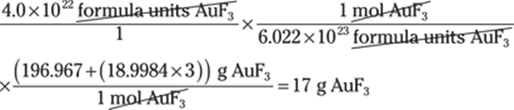
692. 10.0 mol NH3
To find the moles of ammonia produced, use the mole ratio from the balanced equation. For every mole of nitrogen gas consumed, 2 mol of ammonia are made.
![]()
693. 8.00 mol NH3
To find the moles of ammonia produced, use the mole ratio from the balanced equation. For every 3 mol of hydrogen gas consumed, 2 mol of ammonia are made.
![]()
694. 12.0 mol N2
To find the moles of nitrogen gas that would be necessary to make 24.0 mol of ammonia, use the mole ratio from the balanced equation. For every 2 mol of ammonia produced, 1 mol of nitrogen gas is consumed.
![]()
695. 54.0 mol H2
To find the moles of hydrogen gas that would be necessary to make 36.0 mol of ammonia, use the mole ratio from the balanced equation. For every 2 mole of ammonia produced, 3 mol of hydrogen gas are consumed:
![]()
696. 9.00 mol CO2 and 12.0 mol H2O
To find the moles of each product produced, you need to complete two separate calculations using the mole ratio from the balanced equation. To find the moles of CO2, note that there are 3 mol of CO2 produced for every mole of C3H8 combusted:
![]()
To find the moles of H2O, note that there are 4 mol of H2O produced for every mole of C3H8 combusted:
![]()
697. 3.0 × 100 g KCl
To find the mass of a substance when given the mass of a different substance in the same reaction, complete the following conversions:
g given → mol given → mol desired substance → g desired substance
To change from grams to moles of the given substance, divide by the molar mass. To get from moles of the given substance to moles of the desired substance, use the coefficients from the balanced reaction to form a mole ratio. And to get the mass of the desired substance, multiply the moles of the desired substance by the molar mass of the desired substance. Tip: To avoid rounding errors, complete all the calculations on your calculator and round the final answer to the correct number of significant figures.
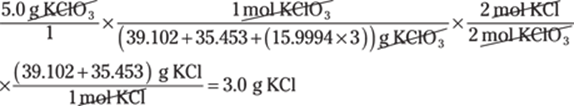
698. 2.0 g O2
To find the mass of a substance when given the mass of a different substance in the same reaction, complete the following conversions:
g given → mol given → mol desired substance → g desired substance
To change from grams of the given substance to moles, divide by the molar mass. To get from moles of the given substance to moles of the desired substance, use the coefficients from the balanced reaction to form a mole ratio. To get the mass of the desired substance, multiply the moles of the desired substance by the molar mass of the desired substance.
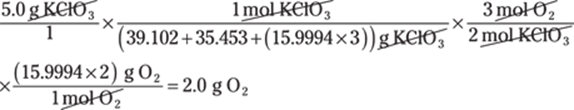
699. 27.0 g CaCO3
To find the mass of a substance when given the mass of a different substance in the same reaction, complete the following conversions:
g given → mol given → mol desired substance → g desired substance
To change from grams of the given substance to moles, divide by the molar mass. To get from moles of the given substance to moles of the desired substance, use the coefficients from the balanced reaction to form a mole ratio. To get the mass of the desired substance, multiply the moles of the desired substance by the molar mass of the desired substance.
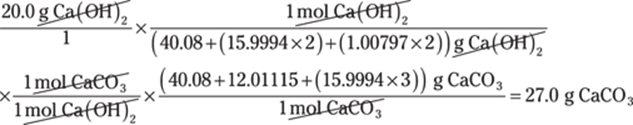
700. 117 g NaCl
To find the mass of a substance when given the mass of a different substance in the same reaction, complete the following conversions:
g given → mol given → mol desired substance → g desired substance
To change from grams of the given substance to moles, divide by the molar mass. To get from moles of the given substance to moles of the desired substance, use the coefficients from the balanced reaction to form a mole ratio. To get the mass of the desired substance, multiply the moles of the desired substance by the molar mass of the desired substance.
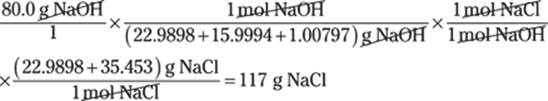
701. 4.6 g Ag3PO4
First, you need a balanced chemical equation from the formulas. Potassium phosphate consists of three K+ and one PO43–. Silver nitrate consists of one Ag+ and one ![]() . Potassium nitrate consists of one K+ and one
. Potassium nitrate consists of one K+ and one ![]() . Silver phosphate consists of three Ag+ and one PO43–. Putting all these together gives you the following reaction:
. Silver phosphate consists of three Ag+ and one PO43–. Putting all these together gives you the following reaction:
![]()
To balance this reaction, the potassium and silver atoms need to be evened out, so multiply the silver nitrate and the potassium nitrate by 3:
![]()
To find the mass of silver phosphate, first divide the given mass of silver nitrate by its molar mass. To get from moles of silver nitrate to the moles of silver phosphate, use the coefficients from the balanced reaction to form a mole ratio. To get the mass of silver phosphate, multiply the moles of the silver phosphate by its molar mass.
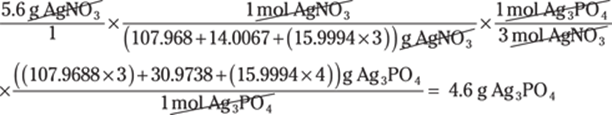
702. 8.20 g NaOH
First, you need a balanced chemical equation from the formulas. Sodium hydroxide consists of one Na+ and one OH–. Copper(II) sulfate consists of one Cu2+ and one SO42–. Copper(II) hydroxide consists of one Cu2+ and two OH–. Sodium sulfate consists of two Na+ and one SO42–. Putting all these together gives you the following equation:
![]()
To balance this equation, you need two sodium ions and two hydroxide ions on the reactants side, so multiply the NaOH by 2:
![]()
Now that you have the formula, you can find the number of grams of NaOH that would be necessary to make 10.0 g of Cu(OH)2. Start by dividing the given mass of Cu(OH)2 by its molar mass. Then use the mole ratio from the balanced equation to get the number of moles of NaOH. Last, convert the moles of NaOH to grams of NaOH by multiplying by the molar mass of NaOH.
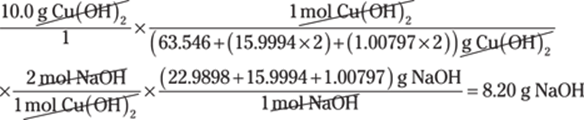
703. 66.8 g Zn
This equation is incomplete, so the first thing you need to do is determine what type of reaction it is. A single element is reacting with a compound, so this is a single displacement reaction: The Zn will displace the H in the HCl. Remember that hydrogen gas is diatomic. Zinc as an ion is Zn2+, so it will take two negatively charged chlorine ions to make the compound neutral:
![]()
To balance the equation, multiply the HCl on the reactants side by 2:
![]()
To find the number of grams of Zn from the HCl, first take the number of grams of HCl and divide by HCl’s molar mass. Then use the mole ratio from the balanced equation to find the number of moles of Zn. Next, convert the moles of Zn to grams of Zn by multiplying by the molar mass of Zn.
![]()
704. 430 g KNO3
The identities of the products are given in the question, so complete the equation with the formulas. You need three +2 charged calcium ions to balance out two –3 charged phosphate ions, and you need one positively charged potassium ion to balance out the one negatively charged nitrate ion:
![]()
This equation is unbalanced, so start with balancing the calcium ions by multiplying the Ca(NO3)2 by 3:
![]()
Next, balance the nitrate ions by multiplying the KNO3 by 6:
![]()
Now you can finish balancing the potassium by multiplying the K3PO4 by 2:
![]()
To find the number of grams of potassium nitrate (KNO3) that would be produced along with 220 g of calcium phosphate (Ca3(PO4)2), start by dividing 220 g of Ca3(PO4)2 by its molar mass. Next, use the mole ratio from the balanced equation to find the moles of KNO3 needed. Then multiply the number of moles of KNO3 by the molar mass of KNO3.
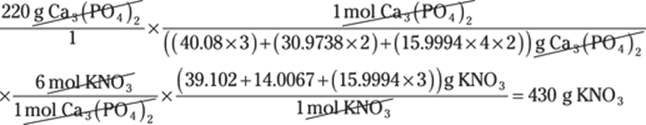
705. 0.15 g BaCl2 and 0.21 g K2Cr2O7
To work this problem, the first thing you need is a complete, balanced equation. Determine the formulas from the names, determine the type of reaction so you can predict the products, and then balance the equation.
Barium chloride comes from the combination of barium ions, Ba2+, and chloride ions, Cl–. Potassium dichromate comes from the combination of potassium ions, K+, and dichromate ions, Cr2O72–. This gives you
![]()
The reactants are a pair of ionic compounds that can switch partners, so this is a double displacement reaction. Therefore, the products are barium dichromate and potassium chloride:
![]()
Balancing this equation requires making the number of chlorine and potassium atoms equal on each side. Simply multiply the KCl on the products side of the reaction by 2:
![]()
Now calculate the number of grams of BaCl2 and K2Cr2O7 needed. You need to do the following conversions:
g given → mol given → mol desired substance → g desired substance
To change from grams to moles of barium dichromate, divide by the molar mass. To get from moles of barium dichromate to moles of the reactants, use the coefficients from the balanced reaction to form a mole ratio. To get the mass of the desired substance, multiply the moles of the desired substance by the molar mass of the desired substance.
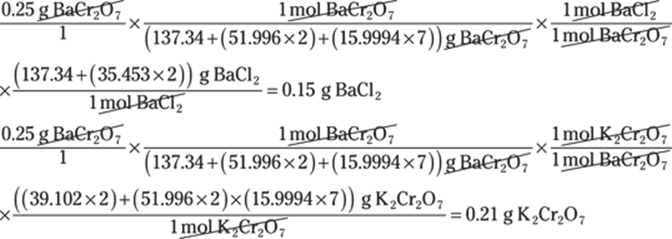
706. 167 g AlBr3
First, complete and balance the equation. There’s only one product, so this is a synthesis (combination) reaction. Aluminum as an ion has a charge of +3, and the bromine ion has a charge of –1, so you need three bromine ions to balance out the +3 charge of the aluminum ion. The formula is AlBr3. Here’s the unbalanced equation:
![]()
To balance this equation, you need the lowest common multiple of 2 and 3, which is 6, to balance the bromine atoms. Multiply the Br2 on the reactants side by 3, and multiply the AlBr3 on the product side by 2:
![]()
Then you can balance the aluminum atoms by multiplying the Al on the left by 2:
![]()
Now find the maximum amount of aluminum bromide that can form if all of each reactant is used up. You need to do the following conversions:
g given → mol given → mol desired substance → g desired substance
To change from grams of a given substance to moles of that substance, divide by the molar mass. To get from moles of the given substance to moles of the desired substance, use the coefficients from the balanced reaction to form a mole ratio. To get the mass of the desired substance, multiply the moles of the desired substance by the molar mass of the desired substance.
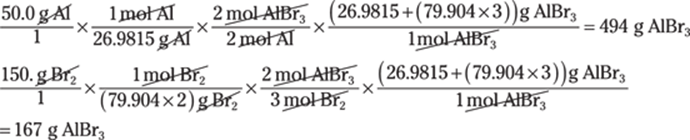
If 50.0 g Al can make 494 g AlBr3 but 150. g Br2 can make only 167 g AlBr3, then the maximum amount that can be made with the reactants provided is the smaller number, 167 g.
707. 2.01 × 1025 molecules of O2
First, balance the equation. To balance the oxygen atoms, you need the lowest common multiple of 2 and 5, which is 10. So multiply the P2O5 by 2 and multiply the O2 by 5:
![]()
Next, balance the atoms of phosphorus by multiplying the P on the reactants side by 4:
![]()
To find how many molecules of O2 you need, start with the given number of P2O5 and divide by Avogadro’s number of molecules in a mole to get moles of P2O5. Next, use the mole ratio to get moles of O2. Then multiply by Avogadro’s number of molecules in a mole to get the molecules of O2.
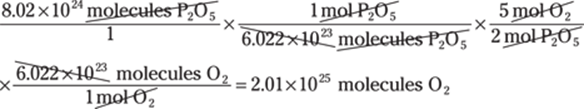
708. 8.45 × 1021 molecules of H2O
This equation is already balanced, so you can start the math part of the problem. To find the number of molecules of H2O needed, start with the given number of HNO3 molecules and divide by Avogadro’s number of molecules in a mole to get moles of HNO3. Then use the mole ratio to get moles of H2O. Multiply by Avogadro’s number of molecules in a mole to get the molecules of H2O.
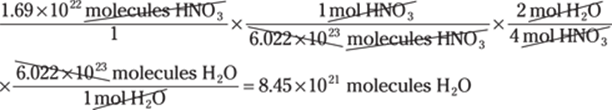
709. 3.16 × 1021 molecules of BaSO3
First, write and balance the equation for this reaction. Barium sulfite contains barium ions, Ba2+, and sulfite ions, SO32–, which gives you a formula of BaSO3. Barium sulfide contains barium ions, Ba2+, and sulfide ions, S2–, which gives you a formula of BaS. Sulfur dioxide contains one sulfur atom and two oxygen atoms, so the formula is SO2. Here’s the reaction:
![]()
This just happens to be balanced, so on to the calculations! To find the number of molecules of BaSO3 needed, start with the given number of SO2 molecules and divide by Avogadro’s number of molecules in a mole to get moles of SO2. Then use the mole ratio to get moles of BaSO3. Multiply by Avogadro’s number of molecules in a mole to get the molecules of BaSO3.
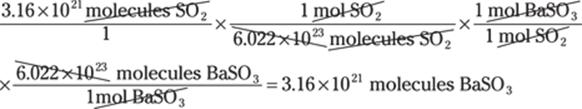
710. 2.64 × 1024 molecules of I2
First, write and balance the equation for this reaction. Iodine and chlorine gases are diatomic, so the reactants are I2 and Cl2. Iodine monochloride has one iodine atom and one chlorine atom, for the formula ICl. Here’s the equation:
![]()
This equation isn’t balanced, but to balance it, you only need to put a 2 in front of the ICl:
![]()
To find the number of molecules of iodine needed, start with the given number of ICl molecules and divide by Avogadro’s number of molecules in a mole to get moles of ICl. Then use the mole ratio to get moles of iodine. Multiply by Avogadro’s number of molecules in a mole to get the molecules of iodine.
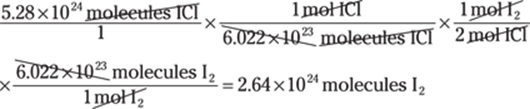
711. 5.65 × 1023 molecules C8H18
This combustion reaction is not balanced. Start by balancing the carbon:
![]()
Then balance the hydrogen:
![]()
Then balance the oxygen:
![]()
Fractions aren’t appropriate for balanced equations, so multiply everything by 2:
![]()
Now calculate the number of molecules of octane needed. Here’s the plan for the conversions:
g given → mol given → mol desired substance → g desired substance
To change from molecules to moles of a given substance, divide by Avogadro’s number of molecules in a mole. To get from moles of the given substance to moles of the desired substance, use the coefficients from the balanced reaction to form a mole ratio. To get the molecules of the desired substance, multiply the moles of the desired substance by Avogadro’s number of molecules in a mole of the desired substance.
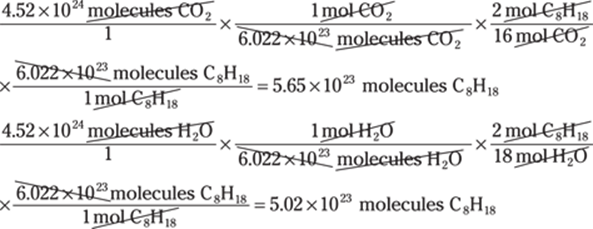
Compare the two numbers. You need at least 5.65 × 1023 molecules of C8H18, because 5.02 × 1023 molecules C8H18 would produce less than the required amount of carbon dioxide.
712. 84.39%
To find the percent yield, divide the actual yield (the amount produced in the experiment) by the theoretical yield (the maximum amount possible based on calculations) and multiply by 100:
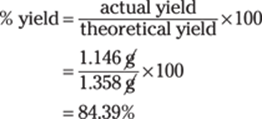
713. 104%
To find the percent yield, divide the actual yield (the amount produced in the experiment) by the theoretical yield (the maximum amount possible based on calculations) and multiply by 100:
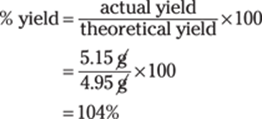
The student in this lab hasn’t violated the law of conservation of mass by creating matter. Most likely, there was an error in finding the mass of product synthesized, or some contaminants got in the sample.
714. 67.4%
To solve for the percent yield, you need both the actual yield and the theoretical yield. The actual yield and the mass of a reactant are given, so you need to calculate the theoretical yield. To do this, convert the grams of NH3 to moles by dividing by the molar mass of NH3; then use the mole ratio to get to moles of NH4Br and multiply the moles of NH4Br by the molar mass:
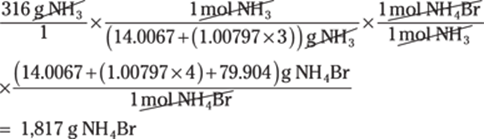
To find the percent yield, divide the actual yield by the theoretical yield and multiply by 100:
![]()
715. 87.1%
To solve for percent yield, you need both the actual yield and the theoretical yield. The problem gives only the actual yield and the mass of a reactant, so you need to calculate the theoretical yield. To do this, convert the grams of NH3 to moles by dividing by the molar mass of NH3; then use the mole ratio to get to moles of NH4Br and multiply the moles of NH4Br by the molar mass:
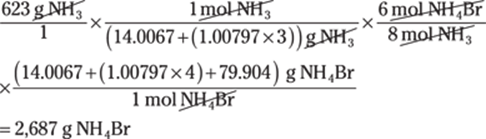
To find the percent yield, divide the actual yield by the theoretical yield and multiply by 100:
![]()
716. 93.2% BaSO4
First, having a balanced reaction would be good. Both reactants and one of the products are given in the question, so here’s your initial equation:
![]()
This reaction appears to be a double displacement, so the other product is sodium chloride:
![]()
To balance this equation, just multiply the NaCl by 2:
![]()
To find the percent yield of the reaction, you first need to calculate the theoretical yield. To do this, convert the grams of BaCl2 to moles by dividing by the molar mass of BaCl2; then use the mole ratio to get to moles of BaSO4 and multiply the moles of BaSO4 by the molar mass:
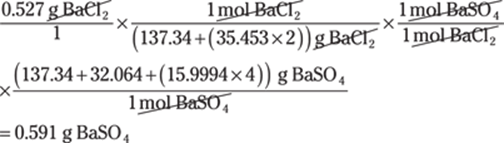
The actual yield divided by the theoretical yield times 100 gives you the percent yield:
![]()
717. 3.60 × 103 g SO3
The equation is already balanced, so the first step is to determine which reactant is the limiting reactant. Divide the moles of each substance by the coefficient of that substance from the balanced chemical equation. The lowest value from this division is the limiting reactant. The limiting reactant is the key to all subsequent calculations.
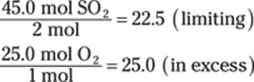
The limiting reactant controls the amount of product formed, so start with 45.0 mol of SO2 and perform a normal stoichiometry calculation to find the maximum amount of SO3 that can be produced:
![]()
718. 99.5 g NaCl
The chlorine is out of balance in the equation: You have three chlorine atoms on the reactants side and two chlorine atoms on the products side. An easy way to try to balance the odd number of chlorine atoms is to multiply each appearance of chlorine that appears singly in the formula (the NaClO2, the ClO2, and the NaCl) by 2, which gives you
![]()
Now the equation is balanced. To find the maximum amount of a substance that can be produced, you need to figure out which reactant will run out first. First, change the grams of each reactant to moles (so you have counting units to work with) by dividing by molar mass:
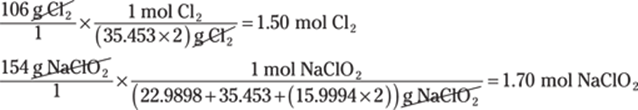
To determine the limiting reactant, divide the moles of each substance by the coefficient of that substance from the balanced chemical equation. The lowest value from this division is the limiting reactant. The limiting reactant is the key to all subsequent calculations.
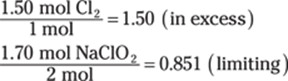
Now knowing the limiting reactant, you can find the mass of the product from the given mass. The limiting reactant controls the amount of product formed, so start with 154 g of NaClO2 and perform a normal stoichiometry calculation to find the grams of NaCl produced:
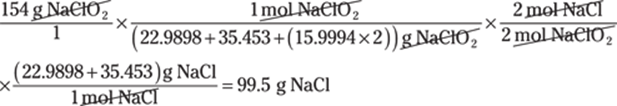
719. 188.9 g Al2O3, and the Al is limiting
First, you need a balanced equation. Aluminum is a metal that you write as Al; oxygen gas is diatomic, so it’s written O2; and aluminum oxide is given as Al2O3. With this info, you can write the unbalanced equation:
![]()
To balance the oxygen atoms, you need the lowest common multiple of 2 and 3, which is 6. Multiply the O2 by 3 and the Al2O3 by 2:
![]()
Then balance the aluminum by multiplying the Al on the reactants side by 4:
![]()
To find the limiting reactant, change the grams of each reactant to moles (so you have counting units to work with) by dividing by molar mass:
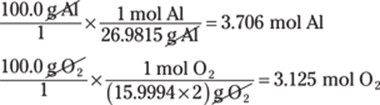
To determine the limiting reactant, divide the moles of each substance by the coefficient of that substance from the balanced chemical equation. The lowest value from this division is the limiting reactant. The limiting reactant is the key to all subsequent calculations.
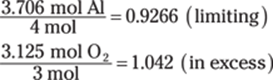
The Al is limiting because you have a smaller relative amount of it available to react. Now find the mass of Al2O3. Take the original mass of aluminum and perform a normal stoichiometry calculation to find the grams of Al2O3 produced:
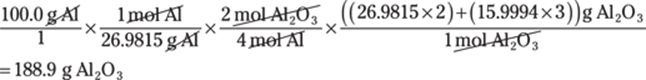
720. 9.34 g NO, and HNO3 is in excess
The equation is balanced, so the first step is to determine which reactant is limiting. First, change the grams of each reactant to moles (so you have counting units to work with) by dividing by molar mass:
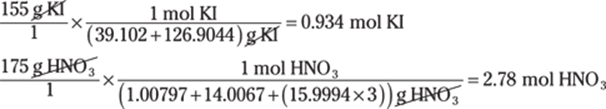
The mole ratio from the balanced equation shows that it takes 6 mol of KI for every 8 mol of HNO3.
To determine the limiting reactant, divide the moles of each substance by the coefficient of that substance from the balanced chemical equation. The lowest value from this division is the limiting reactant. The limiting reactant is the key to all subsequent calculations.
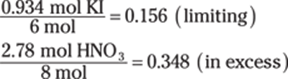
KI is the limiting reactant because you have a smaller amount of it available compared to the amount of HNO3.
Now knowing the limiting reactant, you can calculate the number of grams of NO. The limiting reactant controls the amount of product formed, so begin with 155 g of KI and perform a normal stoichiometry calculation:
![]()
721. O2 is the limiting reactant, 56.3 g of water is produced, and 43.7 g of H2 is in excess
First, you need an equation for the reaction. Hydrogen and oxygen both exist as diatomic gases, and with sufficient energy, they react to form water:
![]()
Balance the oxygen atoms by multiplying the water by 2, and then balance the hydrogen atoms by multiplying the H2 by 2:
![]()
Now you can determine which reactant is limiting and which is in excess. First, change the grams of each reactant to moles (so you have counting units to work with) by dividing by molar mass:
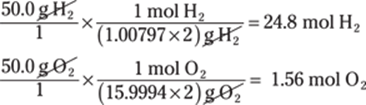
To determine the limiting reactant, divide the moles of each substance by the coefficient of that substance from the balanced chemical equation. The lowest value from this division is the limiting reactant. The limiting reactant is the key to all subsequent calculations.
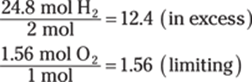
The O2 is the limiting reactant because a smaller relative amount of it is available. Now knowing the limiting reactant, you can find the mass of the water produced from the given mass of the oxygen. The limiting reactant controls the amount of product formed, so begin with 50.0 g of O2 and perform a normal stoichiometry calculation:

Using the law of conservation of mass, the mass of the hydrogen used plus the mass of the water used should equal the mass of the water produced (x g H2 + 50.0 g O2 = 56.3 g H2O), so the mass of the hydrogen used is 6.3 grams. The original mass of hydrogen available was 50.0 g, so subtract the hydrogen used from 50.0 g to find the H2 in excess:
50.0 g H2 – 6.3 g H2 = 43.7 g H2
722. 573 K
To find the Kelvin temperature when given degrees Celsius, add 273 to the Celsius temperature:
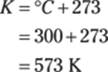
723. 423 K
To find the Kelvin temperature when given degrees Celsius, add 273 to the Celsius temperature:
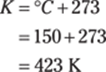
724. 73 K
To find the Kelvin temperature when given degrees Celsius, add 273 to the Celsius temperature:
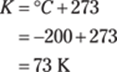
725. 195 K
To find the Kelvin temperature when given degrees Celsius, add 273 to the Celsius temperature:
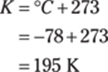
726. 310 K
To find the Kelvin temperature when given degrees Celsius, add 273 to the Celsius temperature:
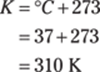
727. –173°C
To find the temperature in degrees Celsius when given kelvins, subtract 273 from the Kelvin temperature:
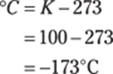
728. 27°C
To find the temperature in degrees Celsius when given kelvins, subtract 273 from the Kelvin temperature:
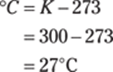
729. –273°C
To find the temperature in degrees Celsius when given kelvins, subtract 273 from the Kelvin temperature:
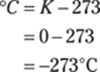
730. 40°C and 104°F
To find the temperature in degrees Celsius when given kelvins, subtract 273 from the Kelvin temperature:
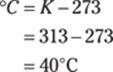
To find the temperature in degrees Fahrenheit, multiply the temperature in degrees Celsius by 9/5 and then add 32:
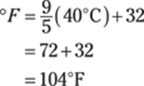
731. –40.00°C and –40.00°F
To find the temperature in degrees Celsius when given kelvins, subtract 273.15 from the Kelvin temperature. Use 273.15 (not 273) for this conversion to match the number of decimal places in the given temperature (233.15 K):
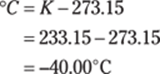
To find the temperature in degrees Fahrenheit, multiply the temperature in degrees Celsius by 9/5 and then add 32.00:
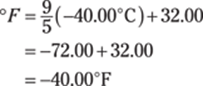
Note that answers of –40°C and –40°F have the wrong number of significant figures. You need two places after the decimal point to correspond to the original temperature.
732. ![]()
The lower segment that has no slope (from B to C) represents melting, turning from a solid into a liquid. All added heat energy goes toward the phase change, so no heat is available to change the temperature. The curve begins with a solid, at Point A, and heat raises the temperature of the solid to Point B, where the substance begins to melt. Melting is complete at Point C, and heating of the liquid begins.
733. ![]()
Condensation is the change from a gas to a liquid, and this occurs when the substance cools from Point B to C. All the heat energy removed comes from the phase change, so there’s no change in temperature; therefore, the slope in this region is zero.
Cooling is the removal of heat energy and may or may not involve a temperature change. The two line segments representing phase changes, ![]() and
and ![]() , have different lengths because the amount of energy released when a substance condenses is greater than the amount released when the same amount of material freezes.
, have different lengths because the amount of energy released when a substance condenses is greater than the amount released when the same amount of material freezes.
734. ![]()
Areas on the graph that have a slope are areas where the substance is being heated but not changing phase. The liquid phase exists between Points C and D. The curve begins with a solid, at Point A, and heating warms the solid to Point B, where the substance begins to melt. Melting is complete at Point C, and heating of the liquid begins. Heating of the liquid continues until the substance reaches the boiling point (Point D), where vaporization begins.
735. ![]()
Boiling is the change from a liquid to a gas. This occurs with no change in temperature (a line with no slope in the graph). The substance is a liquid at Point D and a gas at Point E. The curve begins with a solid, at Point A, and heating warms the solid to Point B, where the substance begins to melt. Melting is complete at Point C, and heating of the liquid begins. Heating of the liquid continues until the substance reaches the boiling point (Point D), where vaporization begins. Vaporization continues until all the liquid is converted to gas (Point E). The gas begins to heat at Point E and continues to heat until the end of the experiment at Point F.
736. ![]()
The solid is being heated at the bottom left-hand corner of the graph in the segment going from A to B. The curve begins with a solid, at Point A, and heating warms the solid to Point B, where the substance begins to melt.
737. ![]()
Freezing is the change from a liquid to a solid. This process takes place from Point D to Point E. The curve begins with a gas, at Point A, and removing heat cools the gas to Point B, where it begins to condense. Condensation is complete at Point C, and cooling of the liquid begins. Cooling of the liquid continues until it reaches the freezing point (Point D), where freezing begins. Freezing continues until all the liquid is converted to a solid (Point E). The substance cools from Point E to the end of the experiment (Point F).
738. ![]()
A gas is being heated starting at Point E and going to Point F and beyond. The curve begins with a solid, at Point A, and heating warms the solid to Point B, where the substance begins to melt. Melting is complete at Point C, and heating of the liquid begins. Heating of the liquid continues until the substance reaches the boiling point (Point D), where vaporization begins. Vaporization continues until all the liquid is converted to gas (Point E). The gas heats from Point E to the end of the experiment (Point F).
739. 16,700 J
To calculate the amount of energy needed to melt a substance, you can use the following formula:
Q = mΔHfus
where Q is the quantity of heat energy, m is the mass, and ΔHfus is the enthalpy of fusion. The enthalpy of fusion, sometimes called the heat of fusion, is the energy necessary to fuse (melt) a quantity of solid.
Enter the numbers in the formula and solve for Q:
![]()
You need 16,700 J of heat energy to melt the ice.
740. –2,500 calories
The temperature at the melting point and the freezing point are the same, so you can use the following formula:
Q = mΔHsol
where Q is the quantity of heat energy, m is the mass, and ΔHsol is the enthalpy of solidification. The enthalpy of solidification, sometimes called the heat of solidification, is the energy that has to be removed to freeze (solidify) a quantity of liquid; at a substance’s melting point, the heats of fusion and of solidification (freezing) are numerically the same; however, the signs are opposite. This means ΔHsol = –ΔHfus. Therefore, if the heat of fusion is 25cal/g, then the heat of solidification is –25 cal/g.
Enter the numbers in the formula and solve for Q:
![]()
When the ethanol freezes, it releases –2,500 calories of heat energy.
741. 27,000 calories
To calculate the amount of energy needed to evaporate a substance, use the following formula:
Q = mΔHvap
where Q is the quantity of heat energy, m is the mass, and ΔHvap is the enthalpy of vaporization. The enthalpy of vaporization, sometimes called the heat of vaporization, is the energy necessary to vaporize a quantity of liquid.
Enter the numbers in the formula and solve for Q:
![]()
You need 27,000 calories to evaporate the water.
742. –4,020 J
To calculate the amount of energy released when a substance is condensed, use the following formula:
Q = mΔHcond
where Q is the quantity of heat energy, m is the mass, and ΔHcond is the enthalpy of condensation. The enthalpy of condensation, sometimes called the heat of condensation, is the energy removed to condense a quantity of gas; at a substance’s boiling point, the heats of vaporization and of condensation are numerically the same; however, the signs are opposite. This means ΔHvap = –ΔHcond. Therefore, if the heat of vaporization is 201 J/g, then the heat of condensation is –201 J/g.
Enter the numbers in the formula and solve for Q:
![]()
When the nitrogen condenses, it releases –4,020 J of heat energy.
743. 50. calories
If the substance doesn’t go through any phase changes, you can use the following formula to calculate the amount of energy needed to raise the substance’s temperature:
Q = mCpΔT
In this equation, Q is the quantity of heat energy, m is the mass, Cp is the specific heat of the material, and ΔT is the change in temperature (Tfinal – Tinitial). The specific heat is the quantity of heat energy necessary to increase the temperature of 1 g of a substance by 1°C.
Enter the numbers in the formula and solve for Q:
![]()
You need 50. calories to raise the temperature of the steam to 112°C.
744. 19,000 J
If the substance doesn’t go through any phase changes, you can use the following formula to calculate the amount of energy needed to raise the substance’s temperature:
Q = mCpΔT
In this equation, Q is the quantity of heat energy, m is the mass, Cp is the specific heat of the material, and ΔT is the change in temperature (Tfinal – Tinitial). The specific heat is the quantity of heat energy necessary to increase the temperature of 1 g of a substance by 1°C.
Enter the numbers in the formula and solve for Q:
![]()
Round the intermediate answer (18,810 J) to 19,000 J so that the answer has the same number of significant figures as the temperatures given in the problem. (Remember: In multiplication problems, the answer should have the same number of significant figures as the measurement with the fewest significant figures.) You need 19,000 J to raise the temperature of the water to 80.°C.
745. 2,130 calories
You need heat to raise the temperature of the ice to the melting point, 0.0°C, and additional heat to melt the ice. The first step, raising the temperature, requires the formula Q = mCpΔT, where Q is the heat energy, m is the mass, Cp is the specific heat of the material, and ΔT is the change in temperature (Tfinal – Tinitial). The second step, melting the ice, requires the formula Q = mΔHfus, where Q is the quantity of heat energy, m is the mass, and ΔHfus is the enthalpy (heat) of fusion. Combining the formulas gives you the following:
746. –5.48 × 104 J
You first need to remove enough energy to lower the temperature to the freezing point of water, 0.0°C; then remove enough energy from the water for it to freeze. The first step, bringing the water to its freezing point, removes Q = mCpΔT energy, where Q is the energy, m is the mass, Cp is the specific heat of the material, and ΔT is the change in temperature (Tfinal – Tinitial). The second step, freezing the water, removes Q = mΔHsol energy, where Q is the quantity of heat energy, m is the mass, and ΔHsol is the enthalpy (heat) of solidification, which is the opposite of the enthalpy of fusion: ΔHsol = –ΔHfus.
This combination of formulas gives you the following:
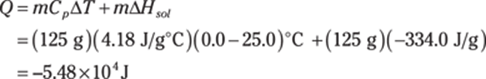
747. –2.50 × 104 cal
Solving this problem is a three-step process. The first step is to reduce the temperature of the steam to the boiling point of water, 100.0°C, using the specific heat of steam: Q = (mCpΔT)g, where Q is the heat energy, m is the mass, Cp is the specific heat of the material, and ΔT is the change in temperature (Tfinal – Tinitial). The second step is to condense the steam to liquid water using the heat of condensation (ΔHcond): Q = mΔHcond, where m is the mass and ΔHcond = –ΔHvap, the opposite of the heat of vaporization. The final step is to remove enough heat from the hot water to cool it to the final temperature using the specific heat of water: Q = (mCpΔT)l.
This combination gives you the following equation, with g referring to gas and l referring to liquid:
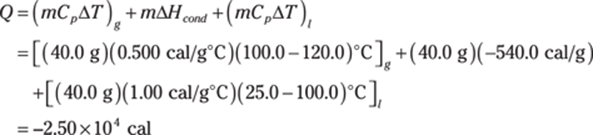
748. 5.043 × 105 J
The first thing to do is to raise the temperature of the water to the boiling point, 100.0°C, using the specific heat of water: Q = (mCpΔT)l, where Q is the energy, m is the mass, Cp is the specific heat of the material, and ΔTis the change in temperature (Tfinal – Tinitial). Then boil (vaporize) the water using the heat of vaporization (ΔHvap): Q = mΔHvap. Last, heat the steam from the boiling point of water to the final temperature using the specific heat of steam: Q = (mCpΔT)g.
This combination gives you the following equation, with g referring to gas and l referring to liquid:
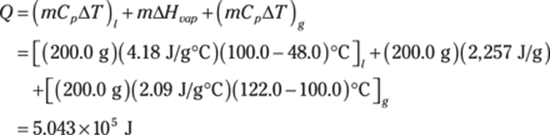
749. 2.66 × 104 cal
This problem involves a four-step process. First, raise the temperature of the ice to the melting point, 0.0°C, using the specific heat of ice: Q = (mCpΔT)s, where Q is the energy, m is the mass, Cp is the specific heat of the material, and ΔT is the change in temperature (Tfinal – Tinitial). Then melt the ice using the heat of fusion (ΔHfus): Q = mΔHfus. Next, raise the temperature of the liquid to the boiling point, 100.0° C, using the specific heat of water: Q = (mCpΔT)l. Finally, use the heat of vaporization, ΔHvap, to convert the liquid water to steam: Q = mΔHvap.
This combination, with s referring to solid and l referring to liquid, gives you the following equation:
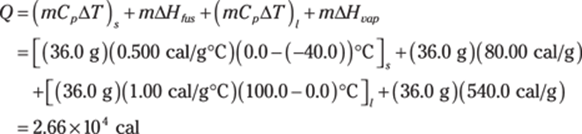
750. –2.20 × 105 J
Removing energy involves a series of four steps in this case. First, use the specific heat of steam to lower the temperature to the boiling point of water, 100.0°C: Q = (mCpΔT)g, where Q is the energy, m is the mass, Cp is the specific heat of the material, and ΔT is the change in temperature (Tfinal – Tinitial). At the boiling (condensation) point, remove the heat of condensation from the steam: Q = mΔHcond, where ΔHcond = –ΔHvap, the opposite of the heat of vaporization. The temperature of the liquid water formed from the condensation must be lowered to the freezing point, 0.0°C, using the specific heat of water: Q = (mCpΔT)l. Finally, freeze the water using the heat of solidification (ΔHsol = –ΔHfus): Q = mΔHsol.
This combination, with g referring to gas and l referring to liquid, gives you the following equation:
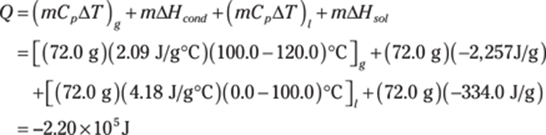
751. 1.444 × 107 J
You need five separate steps to solve this problem. In each step, you add a given amount of energy. The amount of energy in the first step, raising the temperature of the ice to the melting point, 0.0°C, depends on the specific heat of ice: Q = (mCpΔT)s, where Q is the energy, m is the mass, Cp is the specific heat of the material, and ΔT is the change in temperature (Tfinal – Tinitial). The second step, melting the ice, uses the heat of fusion of ice: Q = mΔHfus. Next, to raise the temperature of the water to the boiling point, 100.0°C, use the specific heat of water: Q = (mCpΔT)l. The fourth step, changing the water to steam, involves the heat of vaporization: Q = mΔHvap. The final step, raising the temperature of the steam, uses the specific heat of steam: Q = (mCpΔT)g.
This combination, with g referring to gas, l referring to liquid, and s referring to solid, gives you the following equation:
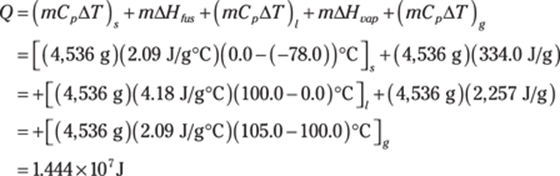
752. –385 cal
This problem involves five steps. In each step, you remove a given amount of energy. The amount of energy in the first step, cooling the steam to the condensation point, 100.0°C, depends on the specific heat of steam: Q = (mCpΔT)g, where Q is the energy, m is the mass, Cp is the specific heat of the material, and ΔT is the change in temperature (Tfinal – Tinitial). The second step, condensing the steam to water, utilizes the heat of condensation of steam (ΔHcond = –ΔHvap): mΔHcond. Next, use the specific heat of water to lower the water temperature to the freezing point, 0.0°C: Q = (mCpΔT)l. The fourth step, freezing the water, involves the heat of solidification (ΔHsol= –ΔHfus): mΔHsol. The final step, lowering the temperature of the ice, uses the specific heat of ice: Q = (mCpΔT)s.
This combination, with g referring to gas, l referring to liquid, and s referring to solid, gives you the following equation:
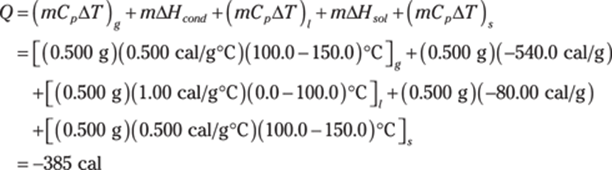
753. 60.0 g
The basic equation relating energy to mass, specific heat, and temperature is
Q = mCpΔT
where Q is the energy, m is the mass, Cp is the specific heat, and ΔT is the change in temperature. You want to know the mass, so rearrange the equation:
![]()
Entering the appropriate values and doing the math gives you
![]()
The sample has a mass of 60.0 g.
754. 4.16 J/g°C
The basic equation relating energy to mass, specific heat, and temperature is
Q = mCpΔT
where Q is the energy, m is the mass, Cp is the specific heat, and ΔT is the change in temperature. You want to know the specific heat, so solve the equation for Cp:
![]()
Now just enter the numbers and do the math:
![]()
The specific heat of the substance is 4.16 J/g°C.
755. 18.5°C
The basic equation relating energy to mass, specific heat, and temperature is
Q = mCpΔT
where Q is the energy, m is the mass, Cp is the specific heat, and ΔT is the change in temperature. You want to find the change in temperature, so solve the equation for ΔT:
![]()
Entering the appropriate values gives you
![]()
The temperature increased by 18.5°C.
756. 46.0°C
The basic equation relating energy to mass, specific heat, and temperature is
![]()
where Q is the energy, m is the mass, Cp is the specific heat, and ΔT is the change in temperature (final temperature minus initial temperature). You want to know the final temperature, so solve the equation for Tf:
![]()
Enter the numbers and complete the calculations:
![]()
The final temperature is 46.0°C.
757. 32.0°C
The basic equation relating energy to mass, specific heat, and temperature is
![]()
where Q is the energy, m is the mass, Cp is the specific heat, and ΔT is the change in temperature (final temperature minus initial temperature). You want to know the initial temperature. Solving the equation for Ti gives you
![]()
Enter the numbers and do the math:
![]()
The sample’s initial temperature was 32.0°C.
758. 9.10 cal
The basic equation relating energy to mass, specific heat, and temperature is
Q = mCpΔT
where Q is the energy, m is the mass, Cp is the specific heat, and ΔT is the change in temperature. You want to find the heat (energy), so simply enter the numbers and solve:
Q = (3.75 g)(0.986 cal/g°C)(2.46°C) = 9.10 cal
The answer is positive, so the sample absorbed 9.10 calories.
759. –0.400 J
The basic equation relating energy to mass, specific heat, and temperature is
Q = mCpΔT
where Q is the energy, m is the mass, Cp is the specific heat, and ΔT is the change in temperature. You want to find the heat (energy), so simply enter the numbers and solve:
Q = (0.326 g)(0.986 J/g°C)(–1.37°C) = –0.400 J
The sample lost 0.400 J of energy.
Note: Watch the value of ΔT. It’s negative because the temperature decreased, which results in a negative heat change.
760. 1.60 g
The basic equation relating energy to mass, specific heat, and temperature is
![]()
where m is the mass, Cp is the specific heat, and ΔT is the change in temperature for Samples 1 and 2. The negative sign appears on the right side of the equation because Sample 2 lost heat to Sample 1.
You want to find the mass of Sample 2, so solve the equation for m2:
![]()
Now enter the appropriate values and do the math:
![]()
Sample 2 has a mass of 1.60 g.
761. 2.60 J/g°C
The basic equation relating energy to mass, specific heat, and temperature is
![]()
where m is the mass, Cp is the specific heat, and ΔT is the change in temperature for Samples 1 and 2. The negative sign appears on the right side of the equation because Sample 2 lost heat to Sample 1.
Rearrange the equation to solve for Cp1, the specific heat of Sample 1:
![]()
Entering the appropriate values and doing the math gives you
![]()
The specific heat of Sample 1 is 2.60 J/g°C.
762. 53.1°C
The basic equation relating energy to mass, specific heat, and temperature is
![]()
where m is the mass, Cp is the specific heat, and ΔT is the change in temperature for Samples 1 and 2. The negative sign on the left side of the equation indicates that Sample 1 lost heat to Sample 2.
You want to find the final temperature of Sample 2, so rewrite ΔT2 as Tf – Ti, the final temperature minus the initial temperature:
![]()
Solve the equation for Tf2. This equation rearranges to
![]()
Entering the appropriate values and doing the calculations gives you
![]()
The final temperature of Sample 2 is 53.1°C.
763. 2 kJ
This reaction deals with the conversion of graphite (pencil lead) to diamonds. The basic equation for the heat of reaction is
![]()
Use the table to look up the standard heat of formation (ΔH°f) for diamonds and graphite and then enter them in the equation:
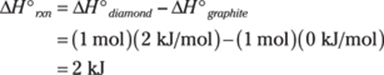
This reaction requires 2 kJ of heat.
764. –396 kJ
This reaction shows the burning of diamonds. The basic equation for the heat of reaction is
![]()
Use the table to look up the standard heats of formation (ΔH°f) for carbon dioxide, diamonds, and oxygen. Enter the numbers in the equation and do the math:
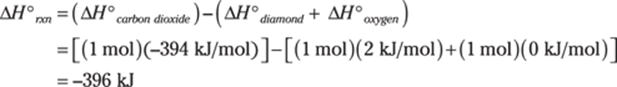
The answer is negative, so this reaction releases 396 kJ of heat.
765. –566 kJ
This reaction shows the combustion of toxic carbon monoxide to form carbon dioxide. The basic equation for the heat of reaction is
![]()
Use the table to look up the standard heat of formation (ΔH°f) for each substance involved — carbon dioxide, carbon monoxide, and oxygen — and enter the numbers in the equation:
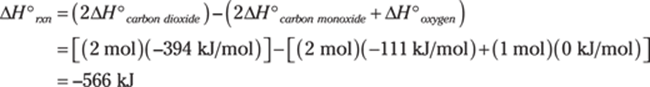
The answer is negative, so this reaction releases 566 kJ of heat.
766. –904 kJ
This reaction shows the combustion of ammonia, an important step in the synthesis of nitric acid and many fertilizers. The basic equation for the heat of reaction is
![]()
Use the table to determine the standard heats of formation (ΔH°f) for each of the substances involved — nitrogen oxide, water vapor, ammonia, and oxygen — and enter them in the equation:
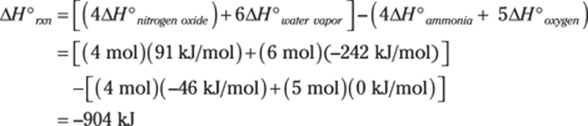
The answer is negative, so this reaction releases 904 kJ of heat.
767. –1,369 kJ
The basic equation for the heat of reaction is
![]()
Use the table to determine the standard heats of formation (ΔH°f) for each substance involved — carbon dioxide, water, ethyl alcohol, and oxygen — and enter them in the equation:
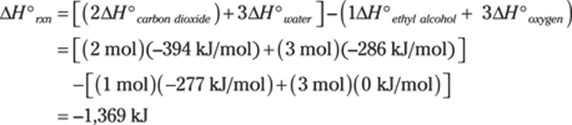
The answer is negative, so this reaction releases 1,369 kJ of heat.
768. –2,807 kJ
This reaction is the basic reaction known as respiration. The basic equation for the heat of reaction is
![]()
Use the table to determine the standard heat of formation (ΔH°f) for each substance involved — carbon dioxide, water, glucose, and oxygen — and enter them in the equation:
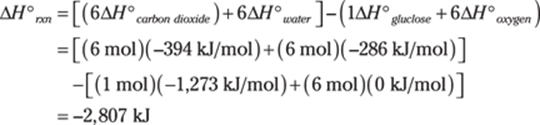
The answer is negative, so this reaction releases 2,807 kJ of heat.
769. 53 kJ/mol
The basic equation for the heat of reaction is
![]()
Use the table to determine the standard heat of formation (ΔH°f) for each substance where the value is known — nitrogen oxide and chlorine — and enter these values and the given heat of reaction (ΔH°rxn) in the equation. This gives you the following:
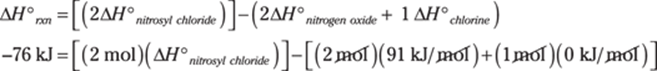
Now solve for ΔH°nitrosyl chloride:
![]()
The standard heat of formation for nitrosyl chloride is 53 kJ/mol.
770. –105 kJ/mol
The basic equation for the heat of reaction is
![]()
Use the table to determine the standard heat of formation (ΔH°f) for each substance where the value is known — carbon dioxide, water vapor, and oxygen — and enter these values and the given heat of reaction (ΔH°rxn) in the equation:
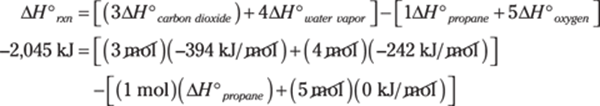
Then solve for ΔH°propane:
![]()
The standard heat of formation for propane is –105 kJ/mol.
771. 73 kJ/mol
The basic equation for the heat of reaction is
![]()
Now use the table to determine the standard heat of formation (ΔH°f) for each substance where the value is known — boron oxide, water, and oxygen — and enter these values and the given heat of reaction (ΔHrxn) in the equation:
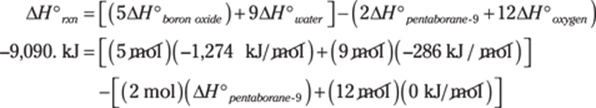
Solve for ΔHpentaborane-9:
![]()
The standard heat of formation for pentaborane-9 is 73 kJ/mol.
772. 170 kJ
Simply add the two thermochemical equations and cancel substances appearing on both sides to get the desired equation. Adding the individual heats of reaction gives you the overall heat of reaction:
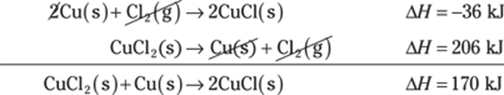
The heat of reaction is 170 kJ.
773. –512 kJ
Adding the individual heats of reaction gives you the overall heat of reaction.
Double the first thermochemical equation, including its energy, before adding the two equations together, because the fluorine needs to have a coefficient of 2 to match the coefficient in the desired reaction. Cancel any substance that appears in equal amounts on both sides of the reaction arrows.
Here are the calculations, beginning with the doubled first equation:
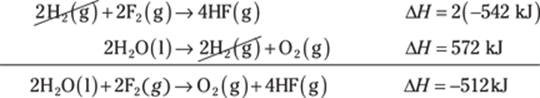
The heat of reaction is –512 kJ.
774. –566 kJ
Adding the individual heats of reaction gives you the overall heat of reaction.
Double the first thermochemical equation, including its energy, before adding the two equations together, because the CO2 needs to have a coefficient of 2 to match the coefficient in the desired reaction. Cancel any substance that appears in equal amounts on both sides of the reaction arrows.
Here are the calculations, beginning with the doubled first equation:
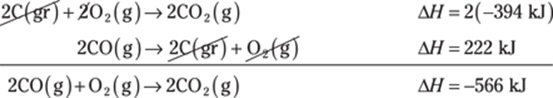
The heat of reaction is –566 kJ.
775. –1,561 kJ
To get the desired equation, simply add the three thermochemical equations and cancel substances that appear in equal amounts on both sides of the reaction arrow. Adding the individual heats of reaction gives you the overall heat of reaction:
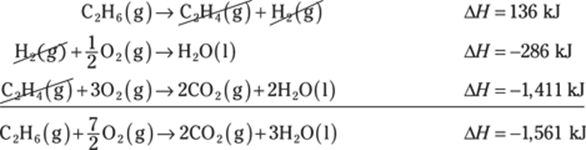
The heat of reaction is –1,561 kJ.
776. –925 kJ
Adding the individual heats of reaction gives you the overall heat of reaction.
The desired equation uses hydrogen as a reactant, so reverse the last thermochemical equation to get the hydrogen on the left side, which means reversing the sign on the energy. You also need to multiply the last two equations and their energies by 1/2 to reduce their coefficients to agree with the ones in the final equation.
Here’s what you get when you apply these changes and add the equations, canceling any substances that appear in equal amounts on both sides of the reaction arrows:
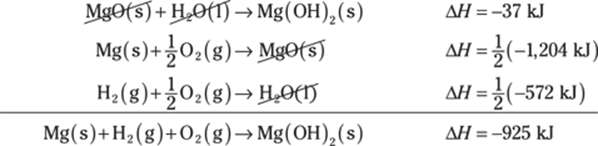
The heat of reaction is –925 kJ.
777. –906 kJ
Adding the individual heats of reaction gives you the overall heat of reaction.
The desired reaction uses ammonia (NH3) as a reactant, so reverse the first equation to get the ammonia on the left side, which means reversing the sign on the energy; you also need to double the first equation along with its energy, because the desired equation has 4NH3, not 2NH3. Triple the second equation (to convert 2H2O to 6H2O) and double the third equation (to convert N2 to 2N2), along with doubling and tripling their energies.
Here’s what you get when you apply these changes and add the equations, canceling any substances that appear in equal amounts on both sides of the reaction arrows:
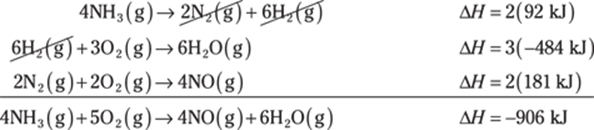
The heat of reaction is –906 kJ.
778. 256 kJ
Adding the individual heats of reaction gives you the overall heat of reaction.
To get the sum of the three thermochemical equations to match the desired equation, you need to reverse both the second and third equations (to get the reactants and products on the same side as they’re on in the desired equation); this means reversing the signs on their energies. You also need to multiply the first and third equations (including their associated energies) by 1/2 to get the coefficients on HCN and NH3 to match those in the desired equation.
Here’s what you get when you apply these changes and add the equations, canceling any substances that appear in equal amounts on both sides of the reaction arrows:
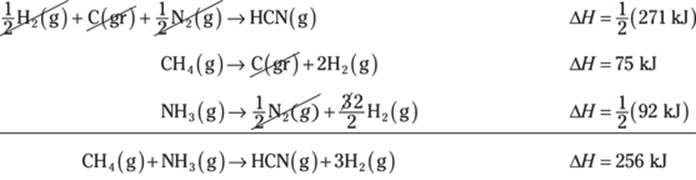
The heat of reaction is 256 kJ.
779. –205 kJ
Adding the individual heats of reaction gives you the overall heat of reaction.
To get the sum of the three thermochemical equations to match the desired equation, you need to double the first equation and its energy. You also need to reverse the third equation, which means reversing the sign on the energy. No changes are needed for the second equation.
Here’s what you get when you apply these changes and add the equations, canceling any substances that appear in equal amounts on both sides of the reaction arrows:
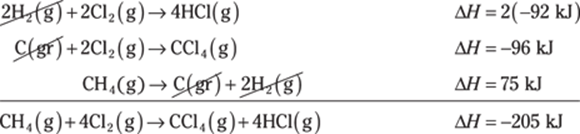
The heat of reaction is –205 kJ.
780. –128 kJ
Adding the individual heats of reaction gives you the overall heat of reaction.
To get the sum of the four thermochemical equations to match the desired equation, you need to reverse the second and fourth equations (to get the CO and CH3OH on the same side as in the final equation), which also means reversing the sign on their energies. You also need to double the first equation, including its associated energy, because the desired equation has a coefficient of 2 on the H2.
Here’s what you get when you apply these changes and add the equations, canceling any substances that appear in equal amounts on both sides of the reaction arrows:
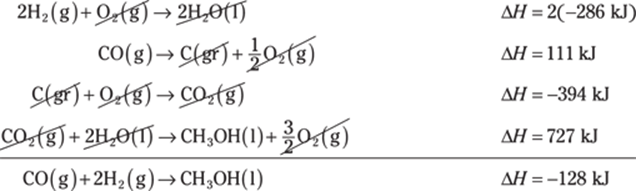
The heat of reaction is –128 kJ.
781. –49 kJ
Adding the individual heats of reaction gives you the overall heat of reaction.
To get the sum of the three thermochemical equations to match the desired equation, you need to reverse the first and third equations, which also means reversing the signs on the energy. You also need to multiply the second equation and its associated energy by 1/2 and to multiply the first equation and its associated energy by 3/2.
Here’s what you get when you apply these changes and add the equations, canceling any substances that appear in equal amounts on both sides of the reaction arrows:
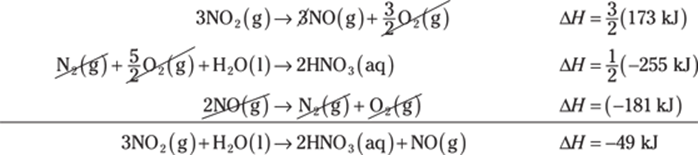
The heat of the reaction is –49 kJ.
782. 1,900 torr
To convert from atmospheres to torr, multiply the given pressure by 760 torr/1 atm:
![]()
783. 76 kPa
To convert from atmospheres to kilopascals, multiply the given pressure by 101 kPa/1 atm:
![]()
784. 0.695 atm
To find how many atmospheres are in a certain number of millimeters of mercury, multiply the given pressure by 1 atm/760 mm Hg:
![]()
785. 2.63 atm
To find how many atmospheres are in a certain number of pounds per square inch, multiply the given pressure by 1 atm/14.7 psi:
![]()
786. 3,800 mm Hg
To convert from atmospheres to millimeters of mercury, multiply the given pressure by 760 mm Hg/1 atm:
![]()
787. 1,050 mm Hg
To convert torr to millimeters of mercury, you can convert torr to atmospheres (760 torr = 1 atm) and then convert atmospheres to millimeters of mercury (1 atm = 760 mm Hg):
![]()
Or you can remember that 1 torr = 1 mm Hg:
![]()
788. 23.4 torr
To find how many torr are in a certain number of kilopascals, convert kilopascals to atmospheres (by multiplying by 1 atm/101 kPa) and then convert atmospheres to torr (by multiplying by 760 torr/1 atm):
![]()
789. 344 kPa
To find how many kilopascals are in a certain number of pounds per square inch, convert pounds per square inch to atmospheres (by multiplying by 1 atm/14.7 psi) and then convert atmospheres to kilopascals (by multiplying by 101 kPa/1 atm):
![]()
790. 1.5 L
When you’re given a combination of initial and final pressures and volumes at a constant temperature, the calculations will involve Boyle’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 3.0 atm |
P2 = 10.0 atm |
|
V1 = 5.01 L |
V2 = ? L |
Make sure the units match, substitute the values into the equation from Boyle’s law, and solve for V2, giving your answer two significant figures:
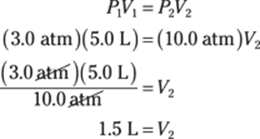
Now make sure your answer is reasonable. The pressure is increasing, so the volume should decrease. The new volume is less than the original volume, so this answer makes sense.
791. 1,070 torr
When you’re given a combination of initial and final pressures and volumes at a constant temperature, the calculations will involve Boyle’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 765 torr |
P2 = ? torr |
|
V1 = 17.5 L |
V2 = 12.5 L |
Make sure the units match, substitute the values into the equation from Boyle’s law, and solve for P2, giving your answer three significant figures:
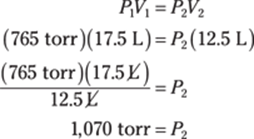
Now make sure your answer is reasonable. The volume is decreasing, so the pressure should increase. The new pressure is greater than the original pressure, so this answer makes sense.
792. 130 L
When you’re given a combination of initial and final pressures and volumes at a constant temperature, the calculations will involve Boyle’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 0.75 atm |
P2 = 0.25 atm |
|
V1 = 44.8 L |
V2 = ? L |
Make sure the units match, substitute the values into the equation from Boyle’s law, and solve for V2, giving your answer two significant figures:
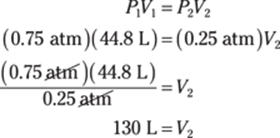
Last, make sure your answer is reasonable. The pressure is decreasing, so the volume should increase. The new volume is larger than the original volume, so this answer makes sense.
793. 1.11 atm
When you’re given a combination of initial and final pressures and volumes at a constant temperature, the calculations will involve Boyle’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 1.75 atm |
P2 = ? atm |
|
V1 = 547 mL |
V2 = 861 mL |
Make sure the units match, substitute the values into the equation from Boyle’s law, and solve for P2, giving your answer three significant figures:
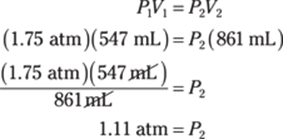
Now make sure your answer is reasonable. The volume is increasing, so the pressure should decrease. The new pressure is lower than the original pressure, so this answer makes sense.
794. 99.9 mL
When you’re given a combination of initial and final pressures and volumes at a constant temperature, the calculations will involve Boyle’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 95.0 kPa |
P2 = 211 kPa |
|
V1 = ? mL |
V2 = 45.0 mL |
Make sure the units match, substitute the values into the equation from Boyle’s law, and solve for V1, giving your answer three significant figures:
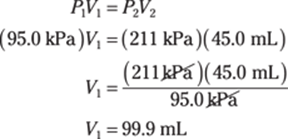
Now make sure your answer is reasonable. The pressure increased, so the volume should decrease. The original volume is larger than the final volume, so this answer makes sense.
795. 555 torr
When you’re given a combination of initial and final pressures and volumes at a constant temperature, the calculations will involve Boyle’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = ? torr |
P2 = 5.10 atm |
|
V1 = 2,645 mL |
V2 = 379 mL |
Next, make sure the units match. The answer needs to be in torr, so you can change the atmospheres to torr now:
![]()
Then substitute the values into the equation from Boyle’s law and solve for P1, giving your answer three significant figures:
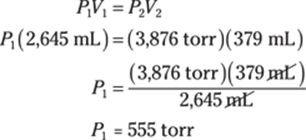
Your other option is to use this equation to find P1 in atmospheres and convert to torr at the end.
Last, make sure your answer is reasonable. The volume decreased, so the pressure should increase. The original pressure, 555 torr, is less than the final pressure, 3,876 torr, so this answer makes sense.
796. 1,580 mL
When you’re given a combination of initial and final pressures and volumes at a constant temperature, the calculations will involve Boyle’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 1,020 torr |
P2 = 7,660 torr |
|
V1 = ? mL |
V2 = 0.210 L |
Next, make sure the units match. The answer needs to be in milliliters, so you can change the liters to milliliters now:
![]()
Then substitute the values into the equation from Boyle’s law and solve for V1, giving your answer three significant figures:
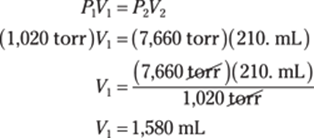
Your other option is to use this equation to find V1 in liters and convert to milliliters at the end.
Last, make sure your answer makes sense. The pressure is increasing, so the volume should decrease. The original volume is 1,580 mL, or 1.58 L, which is larger than the final volume, 0.210 L, so this answer makes sense.
797. 0.568 atm
When you’re given a combination of initial and final pressures and volumes at a constant temperature, the calculations will involve Boyle’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = ? atm |
P2 = 720. torr |
|
V1 = 1.00 L |
V2 = 600. mL |
Next, make sure the units match. In this problem, the volumes are in milliliters and liters, and the pressures are in torr and atmospheres. The volume conversion is easy when you remember that 1.00 L = 1,000 mL; the other conversion requires changing torr to atmospheres using 1 atm = 760 torr:
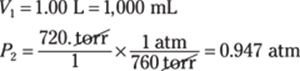
Now substitute the values into the equation from Boyle’s law and solve for P1, giving your answer three significant figures:
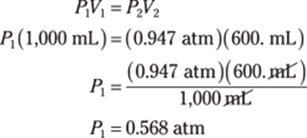
Now make sure your answer is reasonable. The volume decreased, so the pressure should increase. The original pressure is smaller than the final pressure, so this answer makes sense.
798. 0.35 L
When you’re given a combination of initial and final pressures and volumes at a constant temperature, the calculations will involve Boyle’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 1.50 atm |
P2 = 540 torr |
|
V1 = ? L |
V2 = 730 mL |
Next, make sure the units match. In this problem, the pressures are in atmospheres and torr, and the volumes are in milliliters and liters. Completing the pressure conversion first will allow the units to cancel when you place the pressures into the equation from Boyle’s law:
![]()
Substitute the values into the equation and solve for V1:
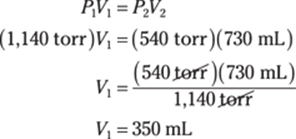
Give the final answer in liters, with two significant figures:
![]()
Make sure your answer is reasonable. The pressure decreased, so the volume should increase. The original volume is smaller than the final volume, so this answer makes sense.
799. 7.0 × 102 mL
When you’re given a combination of initial and final pressures and volumes at a constant temperature, the calculations will involve Boyle’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 920 mm Hg |
P2 = 2.5 atm |
|
V1 = ? mL |
V2 = 0.34 L |
Next, make sure the units match. In this problem, the pressures are in millimeters of mercury and atmospheres, and the volumes are in liters and milliliters. Do the pressure conversion first so the units will cancel when you enter them in the equation from Boyle’s law:
![]()
Then substitute the values into the equation and solve for V1:
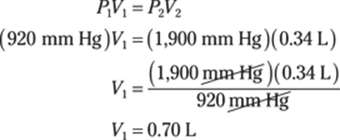
Change the answer from liters to milliliters, giving the answer two significant figures:
![]()
Last, make sure your answer is reasonable. The pressure increased, so the volume should decrease. The original volume is larger than the final volume, so this answer makes sense.
800. 8.4 L
When you’re given a combination of initial and final volumes and temperatures at a constant pressure, the calculations will involve Charles’s law. Draw a little chart to keep track of the quantities and the units:
|
V1 = 4.2 L |
V2 = ? L |
|
T1 = 200. K |
T2 = 400. K |
Next, make sure the temperatures are in kelvins. They are, so substitute the values into the equation from Charles’s law and solve for V2, giving your answer two significant figures:
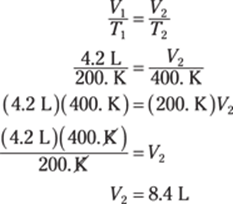
This answer makes sense. Volume and temperature are directly proportional, so if the temperature increases, then the volume must increase, too.
801. 240 mL
When you’re given a combination of initial and final volumes and temperatures at a constant pressure, the calculations will involve Charles’s law. Draw a little chart to keep track of the quantities and the units:
|
V1 = ? mL |
V2 = 473 mL |
|
T1 = 20. K |
T2 = 40. K |
Next, make sure the temperatures are in kelvins. They are, so substitute the values into the equation from Charles’s law and solve for V1, giving your answer two significant figures:
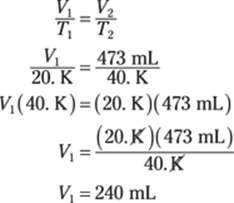
This answer makes sense. Volume and temperature are directly proportional, so if the original temperature was lower than the final temperature, then the original volume was smaller than the final volume, too.
802. 3.62 L
When you’re given a combination of initial and final volumes and temperatures at a constant pressure, the calculations will involve Charles’s law. Draw a little chart to keep track of the quantities and the units:
|
V1 = ? L |
V2 = 2.50 L |
|
T1 = 323 K |
T2 = 223 K |
Next, make sure the temperatures are in kelvins. They are, so substitute the values into the equation from Charles’s law and solve for the V1, giving your answer three significant figures:
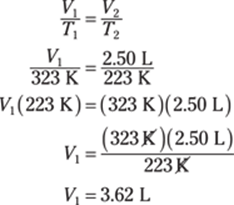
This answer makes sense. Because volume and temperature are directly proportional, an initial temperature that’s higher than the final temperature indicates that the initial volume was larger than the final volume.
803. 5.00 L
When you’re given a combination of initial and final volumes and temperatures at a constant pressure, the calculations will involve Charles’s law. Draw a little chart to keep track of the quantities and the units:
|
V1 = 20.0 L |
V2 = ? L |
|
T1 = 300. K |
T2 = 75.0 K |
Next, make sure the temperatures are in kelvins. They are, so substitute the values into the equation from Charles’s law and solve for V2, giving your answer three significant figures:
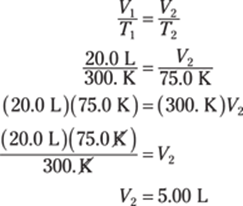
This answer makes sense. The temperature is decreasing, so the volume should be decreasing, because at a constant pressure, volume and temperature are directly proportional.
804. 43.3 mL
When you’re given a combination of initial and final volumes and temperatures at a constant pressure, the calculations will involve Charles’s law. Draw a little chart to keep track of the quantities and the units:
|
V1 = 50.0 mL |
V2 = ? mL |
|
T1 = 100.0°C |
T2 = 50.0°C |
Next, make sure the temperatures are in kelvins. To convert to kelvins, add 273.2 to the Celsius temperatures:
T1 = 100.0°C + 273.2 = 373.2 K
T2 = 50.0°C + 273.2 = 323.2 K
Substitute the values into the equation from Charles’s law and solve for V2, giving your answer three significant figures:
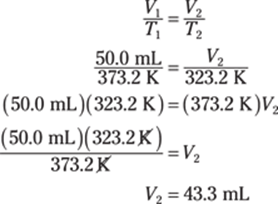
Volume and pressure are directly proportional, so when the temperature decreases, the volume also decreases. The final volume of 43.3 mL is less than 50.0 mL, so the answer is reasonable.
805. 114 K
When you’re given a combination of initial and final volumes and temperatures at a constant pressure, the calculations will involve Charles’s law. Draw a little chart to keep track of the quantities and the units:
|
V1 = 350. mL |
V2 = 0.100 L |
|
T1 = 127°C |
T2 = ? K |
Next, make sure the temperature is in kelvins. To convert the Celsius temperature into kelvins, add 273:
T1 = 127°C + 273 = 400. K
The volumes also need to be in the same unit, so convert the liters into milliliters (or alternatively, the milliliters into liters):
![]()
Next, substitute the values into the equation from Charles’s law and solve for T2:
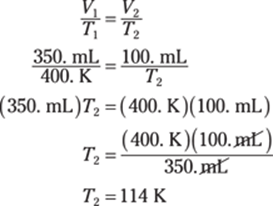
In this problem, the volume is decreasing, so the temperature must also decrease. The final temperature of 114 K is definitely lower than 400. K, so this answer makes sense.
806. 519 K
When you’re given a combination of initial and final volumes and temperatures at a constant pressure, the calculations will involve Charles’s law. Draw a little chart to keep track of the quantities and the units:
|
V1 = 981 mL |
V2 = 1,520 mL |
|
T1 = 335 K |
T2 = ? K |
Next, make sure the temperature is in kelvins. It is, so substitute the values into the equation from Charles’s law and solve for T2, giving your answer three significant figures:
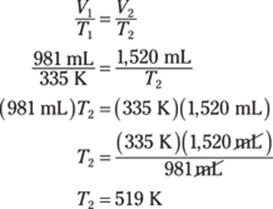
The volume increased in this problem, so the temperature should also increase. The final temperature of 519 K is greater than 335 K, so the answer is reasonable.
807. 1,150 K
When you’re given a combination of initial and final volumes and temperatures at a constant pressure, the calculations will involve Charles’s law. Draw a little chart to keep track of the quantities and the units:
|
V1 = 2.63 L |
V2 = 627 mL |
|
T1 = ? K |
T2 = 275 K |
Next, make sure the temperature is in kelvins, as it is here. Also, both volumes need to have the same units so they’ll cancel during the calculations. Converting liters to milliliters requires multiplying by 1,000 mL/1 L:
![]()
Substitute the values into the equation from Charles’s law and solve for T1, giving your answer three significant figures:
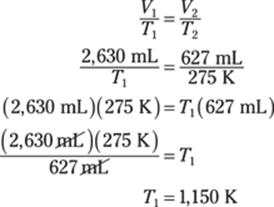
In this problem, the volume is decreasing, so the temperature must also decrease. The final temperature of 275 K is definitely smaller than 1,150 K, so this answer makes sense.
808. 2,140 K
When you’re given a combination of initial and final volumes and temperatures at a constant pressure, the calculations will involve Charles’s law. Draw a little chart to keep track of the quantities and the units:
|
V1 = 90.0 mL |
V2 = 10.0 mL |
|
T1 = ? K |
T2 = –35°C |
Next, make sure the temperature is in kelvins. It isn’t, so add 273 to the Celsius temperature:
T2 = –35°C + 273 = 238 K
Substitute the values into the equation from Charles’s law and solve for T1, giving the answer three significant figures:
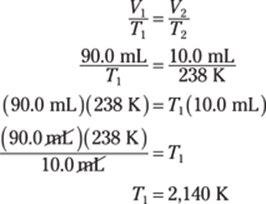
In this problem, the volume is decreasing, so the temperature must also decrease. The final temperature of 238 K is definitely smaller than 2,140 K, so this answer makes sense.
809. 1,500 K
When you’re given a combination of initial and final volumes and temperatures at a constant pressure, the calculations will involve Charles’s law. Draw a little chart to keep track of the quantities and the units:
|
V1 = 750 mL |
V2 = 3.0 L |
|
T1 = 95°C |
T2 = ? K |
Next, make sure the temperature is in kelvins. It isn’t, so add 273 to the Celsius temperature:
T1 = 95°C + 273 = 368 K
You also need to make sure that the volume units are the same, so change the liters to milliliters by multiplying by 1,000 mL/1 L (or alternatively, change the milliliters to liters):
![]()
Substitute the values into the equation from Charles’s law and solve for T2, giving your answer two significant figures:
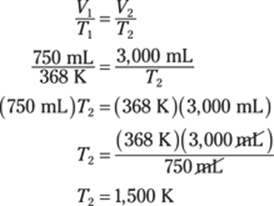
In this problem, the volume is increasing, so the temperature should also increase. The final temperature of 1,500 K is definitely larger than 368 K, so this answer makes sense.
810. 1,350 torr
When you’re given a combination of initial and final pressures and temperatures at a constant volume, the calculations will involve Gay-Lussac’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 900. torr |
P2 = ? torr |
|
T1 = 300. K |
T2 = 450. K |
Next, make sure the temperatures are in kelvins. They are, so substitute the values into the equation from Gay-Lussac’s law and solve for P2, giving your answer three significant figures:
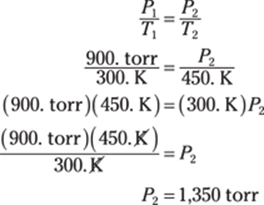
In this problem, the temperature is increasing, so the pressure should increase. The final pressure of 1,350 torr is greater than 900. torr, so this answer makes sense.
811. 1.5 atm
When you’re given a combination of initial and final pressures and temperatures at a constant volume, the calculations will involve Gay-Lussac’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 2.5 atm |
P2 = ? atm |
|
T1 = 500. K |
T2 = 300. K |
Next, make sure the temperatures are in kelvins. They are, so substitute the values into the equation from Gay-Lussac’s law and solve for P2, giving your answer two significant figures:
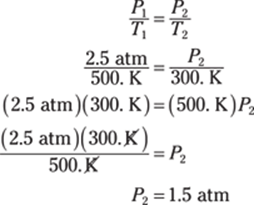
In this problem, the temperature is decreasing, so the pressure should decrease. The final pressure of 1.5 atm is less than 2.5 atm, so this answer makes sense.
812. 1,280 torr
When you’re given a combination of initial and final pressures and temperatures at a constant volume, the calculations will involve Gay-Lussac’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = ? torr |
P2 = 675 torr |
|
T1 = 425 K |
T2 = 225 K |
Next, make sure the temperatures are in kelvins. They are, so substitute the values into the equation from Gay-Lussac’s law and solve for P1, giving your answer three significant figures:
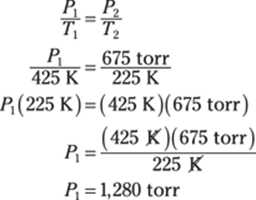
In this question, the temperature is decreasing, so the pressure should decrease. The initial pressure of 1,280 torr is greater than 675 torr, so the answer makes sense.
813. 5.05 atm
When you’re given a combination of initial and final pressures and temperatures at a constant volume, the calculations will involve Gay-Lussac’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = ? atm |
P2 = 8.10 atm |
|
T1 = 315 K |
T2 = 505 K |
Next, make sure the temperatures are in kelvins. They are, so substitute the values into the equation from Gay-Lussac’s law and solve for P1, giving your answer three significant figures:
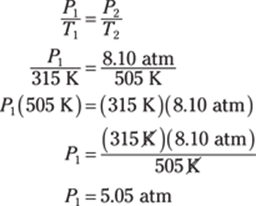
The temperature in this question is increasing, so the pressure should increase. The answer checks, because the initial pressure of 5.05 atm is less than the final pressure of 8.10 atm.
814. 420 kPa
When you’re given a combination of initial and final pressures and temperatures at a constant volume, the calculations will involve Gay-Lussac’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 470 kPa |
P2 = ? kPa |
|
T1 = 310 K |
T2 = 280 K |
Next, make sure the temperatures are in kelvins. They are, so substitute the values into the equation from Gay-Lussac’s law and solve for P2, giving your answer two significant figures:
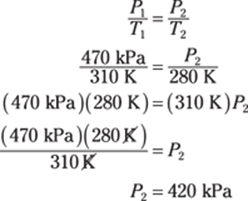
To check this answer, compare the initial temperatures. The temperature is decreasing, so the pressure should decrease. The final pressure of 420 kPa is less than 470 kPa, so this answer makes sense.
815. 1,760 K
When you’re given a combination of initial and final pressures and temperatures at a constant volume, the calculations will involve Gay-Lussac’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 780. mm Hg |
P2 = 1,280 mm Hg |
|
T1 = 801°C |
T2 = ? K |
Next, make sure the temperature is in kelvins. It isn’t, so add 273 to the Celsius temperature:
T1 = 801°C + 273 = 1,074 K
Substitute the values into the equation from Gay-Lussac’s law and solve for T2, giving your answer three significant figures:
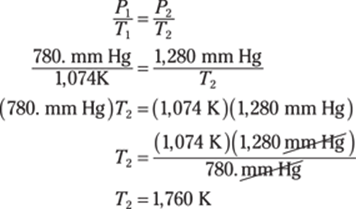
The pressure is increasing, so the temperature should increase. The final temperature of 1,760 K is approximately 1,490°C, which is greater than 801°C, so this answer makes sense.
816. 660.°C
When you’re given a combination of initial and final pressures and temperatures at a constant volume, the calculations will involve Gay-Lussac’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 1.00 atm |
P2 = 2.50 atm |
|
T1 = 100.°C |
T2 = ? °C |
Next, make sure the temperature is in kelvins. It isn’t, so add 273 to the Celsius temperature:
T1 = 100.°C + 273 = 373 K
Substitute the values into the equation from Gay-Lussac’s law and solve for T2, giving your answer three significant figures:
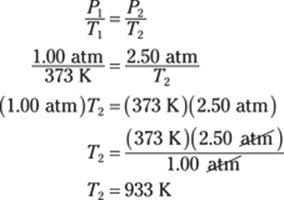
The final temperature needs to be in degrees Celsius, so subtract 273 from the Kelvin temperature:
T2 = 933 K – 273 = 660.°C
To check the answer, note that the pressure increased, so the temperature should increase as well. The final temperature of 660.°C is greater than 100.°C, so this answer makes sense.
817. 235 K
When you’re given a combination of initial and final pressures and temperatures at a constant volume, the calculations will involve Gay-Lussac’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 428 torr |
P2 = 1.25 atm |
|
T1 = ? K |
T2 = 521 K |
The pressures aren’t in the same unit, so convert one unit to the other. Here’s the conversion from torr to atmospheres:
![]()
The temperature is already in kelvins, so substitute the values into the equation from Gay-Lussac’s law and solve for ![]() , giving your answer three significant figures:
, giving your answer three significant figures:
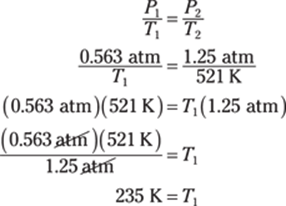
The pressure is increasing (going from 0.563 atm to 1.25 atm), so the temperature should increase as well. The temperature goes from 235 K to 521 K, so this answer is reasonable.
818. –76°C
When you’re given a combination of initial and final pressures and temperatures at a constant volume, the calculations will involve Gay-Lussac’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 44.1 psi |
P2 = 2,028 torr |
|
T1 = ? °C |
T2 = 175 K |
The pressures aren’t in the same unit, so convert one unit to the other. Here’s the conversion from pounds per square inch to torr:
![]()
The given temperature is already in kelvins, so substitute the values into the equation from Gay-Lussac’s law and solve for T1, giving your answer three significant figures:
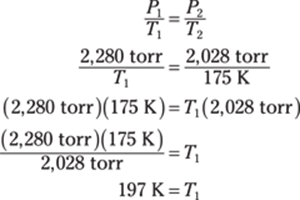
The answer needs to be in degrees Celsius, so take the temperature in kelvins and subtract 273:
T1 = 197 K – 273 = –76°C
In this problem, the pressure decreases from 2,280 torr to 2,028 torr, so the temperature should also decrease. You see the temperature decreasing from 197 K to 175 K, so this answer is reasonable.
819. 391°C
When you’re given a combination of initial and final pressures and temperatures at a constant volume, the calculations will involve Gay-Lussac’s law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 0.612 atm |
P2 = 34.9 in. Hg |
|
T1 = 75°C |
T2 = ? °C |
First, convert the initial temperature from degrees Celsius to kelvins by adding 273:
T1 = 75°C + 273 = 348 K
The pressures aren’t in the same unit, so convert one unit to the other. Here’s the conversion from inches of mercury to atmospheres (1 atm = 29.9 in. Hg):
![]()
Substitute the values into the equation from Gay-Lussac’s law and solve for T2, giving your answer three significant figures:
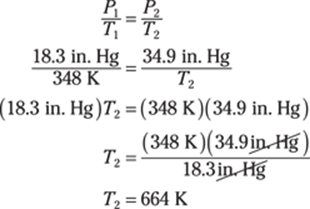
The final temperature needs to be in degrees Celsius, so subtract 273 from the Kelvin temperature:
T2 = 664 K – 273 = 391°C
To check the answer, note that the pressure increases, so the temperature should increase as well. The final temperature of 391°C is greater than the initial temperature of 75°C, so the final answer makes sense.
820. 1.47 atm
When you’re given a combination of initial and final pressures, volumes, and temperatures for a gas sample, the calculations will involve the combined gas law. The units match and the temperatures are in kelvins, so substitute the given values into the equation and solve for P2, giving your answer three significant figures:
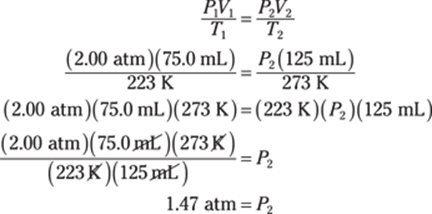
The final pressure is 1.47 atm.
821. 7.7 L
When you’re given a combination of initial and final pressures, volumes, and temperatures for a gas sample, the calculations will involve the combined gas law. The units match and the temperatures are in kelvins, so substitute the given values into the equation and solve for V2, giving your answer two significant figures:
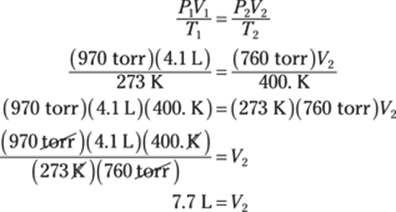
The final volume is 7.7 L.
822. 1.7 atm
When you’re given a combination of initial and final pressures, volumes, and temperatures for a gas sample, the calculations will involve the combined gas law. The units match and the temperatures are in kelvins, so substitute the given values into the equation and solve for P1, giving your answer two significant figures:
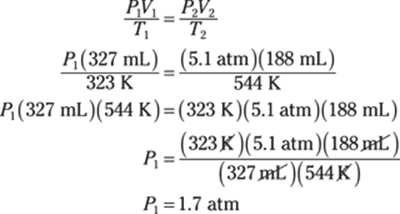
The initial pressure is 1.7 atm.
823. 320 mL
When you’re given a combination of initial and final pressures, volumes, and temperatures for a gas sample, the calculations will involve the combined gas law. The units match and the temperatures are in kelvins, so substitute the given values into the equation and solve for V1, giving your answer two significant figures:
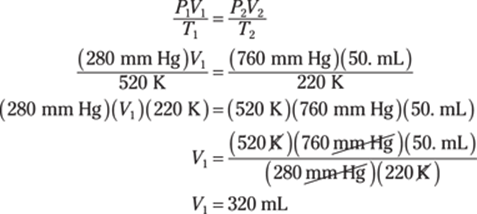
The initial volume is 320 mL.
824. 1.68 atm
When you’re given a combination of initial and final pressures, volumes, and temperatures for a gas sample, the calculations will involve the combined gas law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 4.23 atm |
P2 = ? atm |
|
V1 = 1,870 mL |
V2 = 6.01 L |
|
T1 = 293 K |
T2 = 373 K |
The volumes aren’t in the same units, so convert the milliliters to liters or the liters to milliliters. Here’s the conversion for milliliters to liters:
![]()
The temperatures are already in kelvins, so substitute the given values into the equation for the combined gas law and solve for P2, giving your answer three significant figures:
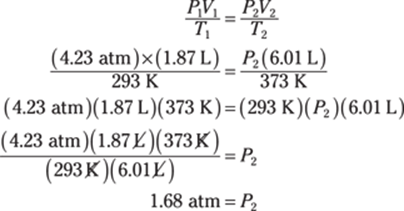
The final pressure is 1.68 atm.
825. 2.1 L
Remember that standard temperature is 273 K and standard pressure is 1 atm (Note: The pressure is an exact number, so it doesn’t affect the number of significant figures in the answer). With this info, you have a combination of initial and final pressures, volumes, and temperatures for a gas sample, so the calculations will involve the combined gas law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 450 torr |
P2 = 1 atm |
|
V1 = 10.0 L |
V2 = ? L |
|
T1 = 773 K |
T2 = 273 K |
The pressures aren’t in the same units, so convert atmospheres to torr: P2 = 1 atm = 760 torr. The temperatures are already in kelvins, so substitute the values into the equation for the combined gas law and solve for V2, giving your answer two significant figures:
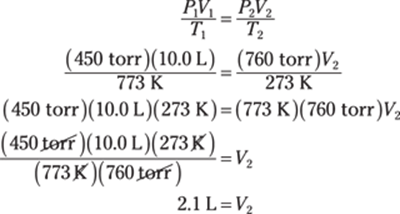
The final volume will be 2.1 L.
826. 16 atm
When you’re given a combination of initial and final pressures, volumes, and temperatures for a gas sample, the calculations will involve the combined gas law. Draw a little chart to keep track of the quantities and the units:
|
P1 = ? atm |
P2 = 5.5 atm |
|
V1 = 0.75 L |
V2 = 2,647 mL |
|
T1 = 25°C |
T2 = 100.°C |
Next, make sure the temperatures are in kelvins. They aren’t, so add 273 to the Celsius temperatures:
T1 = 25°C + 273 = 298 K
T2 = 100.°C + 273 = 373 K
Also, you need to convert one of the volume units so that the units match. Here’s the conversion from liters to milliliters:
![]()
Substitute the values into the equation for the combined gas law and solve for P1, giving your answer two significant figures:
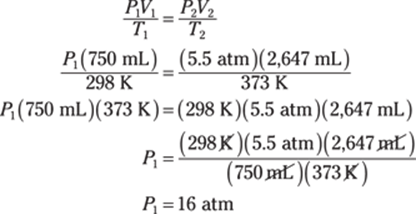
The original pressure was 16 atm.
827. 1,220 mL
When you’re given a combination of initial and final pressures, volumes, and temperatures for a gas sample, the calculations will involve the combined gas law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 3.00 atm |
P2 = 3,862 mm Hg |
|
V1 = ? mL |
V2 = 594 mL |
|
T1 = 300.°C |
T2 = 200.°C |
Next, make sure the temperatures are in kelvins. They aren’t, so add 273 to the Celsius temperatures given:
T1 = 300.°C + 273 = 573 K
T2 = 200.°C + 273 = 473 K
Also, the pressures need to be in the same unit. Here’s the conversion from atmospheres to millimeters of mercury:
![]()
Substitute the values into the equation for the combined gas law and solve for V1, giving your answer three significant figures:
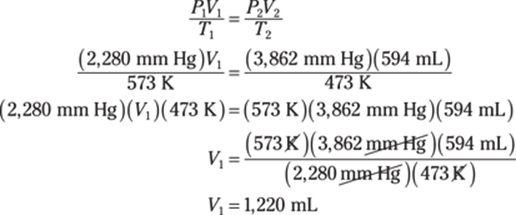
The original volume was 1,220 mL.
828. 330 K
When you’re given a combination of initial and final pressures, volumes, and temperatures for a gas sample, the calculations will involve the combined gas law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 970 torr |
P2 = 0.78 atm |
|
V1 = 220. mL |
V2 = 2.4 L |
|
T1 = 50. K |
T2 = ? K |
You need to convert the pressure and volume units so they’ll cancel later. Here are the conversions from atmospheres to torr and from liters to milliliters:
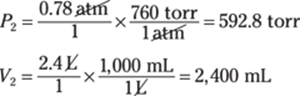
Next, substitute the given and converted values into the equation for the combined gas law and solve for T2, giving your answer two significant figures:
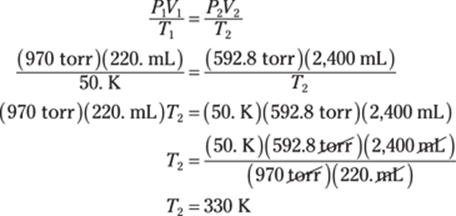
The gas’s final temperature is 330 K.
829. 518 K
When you’re given a combination of initial and final pressures, volumes, and temperatures for a gas sample, the calculations will involve the combined gas law. Draw a little chart to keep track of the quantities and the units:
|
P1 = 35.3 psi |
P2 = 6.18 atm |
|
V1 = 10.0 L |
V2 = 4,290 mL |
|
T1 = ? K |
T2 = 572 K |
You need to convert the pressure and the volume units so they’ll cancel later. Here are the conversions from pounds per square inch to atmospheres and from milliliters to liters:
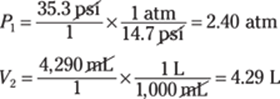
The temperature is already in kelvins, so substitute the values into the equation for the combined gas law and solve for T1, giving your answer three significant figures:
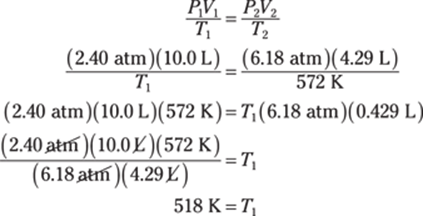
The initial temperature was 518 K.
830. 560. L
To use Avogadro’s law, you need to know the number of moles of the gas, so use the molar mass of helium to convert from grams to moles. Then multiply by 22.4 L/1 mol to find the volume of the gas:
![]()
831. 12.7 L
To use Avogadro’s law, you need to know the number of moles of the gas, so use the molar mass of carbon dioxide to convert from grams to moles. Then multiply by 22.4 L/1 mol to find the volume of the gas:
![]()
832. 2.46 × 1023 molecules
Convert the volume of the nitrogen gas to moles using 22.4 L = 1 mol (at standard temperature and pressure). Then multiply by Avogadro’s number to find the number of molecules:
![]()
833. 27.14 g
First, determine the number of moles of methane gas using 22.4 L = 1 mol (at standard temperature and pressure). The multiply by the molar mass of CH4 to find the mass of the sample:
![]()
834. 110. L
Avogadro’s law states that the volume of a gas is proportional to the number of moles present (assuming the pressure and temperature are constant). One way of using Avogadro’s law is through the following relationship:
![]()
Here are the given values from the question:
|
V1 = 55.0 L |
V2 = ? L |
|
n1 = 2.10 mol |
n2 = ? mol |
Calculate the number of moles of NO2 produced using the mole ratio from the balanced equation. You have 2 mol of NO2 for every mole of N2O4:
![]()
Enter your numbers in Avogadro’s relationship and solve for V2, the volume of NO2 gas produced. Give your answer three significant figures.
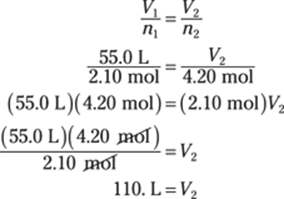
The reaction produces 110. L of NO2.
835. 18.6 L
Avogadro’s law states that the volume of a gas is proportional to the number of moles present (assuming the pressure and temperature are constant). One way of using Avogadro’s law is through the following relationship:
![]()
Here are the given values from the question:
|
V1 = 12.4 L |
V2 = ? L |
|
n1 = 0.296 mol |
n2 = ? mol |
Calculate the number of moles produced using the mole ratio from the balanced equation. You have 3 mol of O2 for every 2 mol of O3:
![]()
Next, substitute the values into Avogadro’s relationship and solve for V2, the volume of O2 gas produced. Give your answer three significant figures.
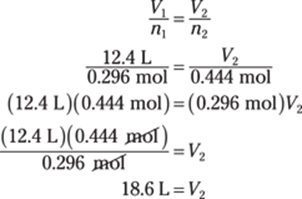
The reaction produces 18.6 L of O2.
836. 13.1 atm
After reading the question, list all the given information. You know the volume, number of moles of the gas, and temperature, and you want to find the pressure, so using the ideal gas law makes sense.
|
P = ? atm |
|
V = 12.5 L |
|
n = 5.00 mol |
|
T = 400. K |
Rearrange the ideal gas law to solve for P, and then enter the given values and R = 0.0821 L·atm/K·mol in the equation. Give your answer three significant figures:
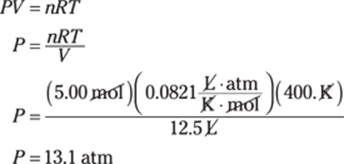
The pressure of the gas sample is 13.1 atm.
837. 196 L
After reading the question, list all the given information. You know the pressure, number of moles of the gas, and temperature, and you want to find the volume, so using the ideal gas law makes sense.
|
P = 0.747 atm |
|
V = ? L |
|
n = 8.91 mol |
|
T = 200. K |
Rearrange the ideal gas law to solve for V, and then enter the given values and R = 0.0821 L·atm/K·mol in the equation. Give your answer three significant figures:
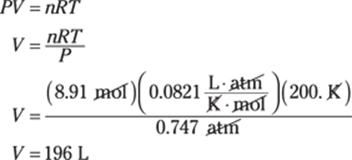
The volume of the gas is 196 L.
838. 0.0613 mol
After reading the question, list all the given information. You know the pressure, volume, and temperature, and you want to find the number of moles, so using the ideal gas law makes sense.
|
P = 3.00 atm |
|
V = 500.0 mL |
|
n = ? mol |
|
T = 298 K |
Rearrange the ideal gas law to solve for n, the number of moles, and then enter the given values (converting milliliters to liters) and R = 0.0821 L·atm/K·mol in the equation. Give your answer three significant figures:
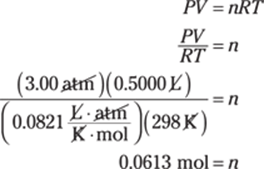
The gas sample contains 0.0613 mol of helium.
839. 230 K
After reading the question, list all the given information. You know the pressure, volume, and number of moles of the gas, and you want to find the temperature, so using the ideal gas law makes sense.
|
P = 4.1 atm |
|
V = 2.8 L |
|
n = 0.60 mol |
|
T = ? K |
Rearrange the ideal gas law to solve for the temperature T, and then enter the given values and R = 0.0821 L·atm/K·mol in the equation. Give your answer two significant figures:
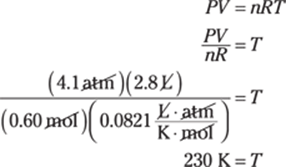
The gas’s temperature is 230 K.
840. 1,060 L
After reading the question, list all the given information. You know the pressure, number of moles of the gas, and temperature, and you want to find the volume, so using the ideal gas law makes sense.
|
P = 1.50 atm |
|
V = ? L |
|
n = 40.6 mol |
|
T = 205°C |
Convert the temperature from degrees Celsius to kelvins:
T = 205°C + 273 = 478 K
Rearrange the ideal gas law to solve for V, and then enter the given and calculated values and R = 0.0821 L·atm/K·mol in the equation. Give your answer three significant figures:
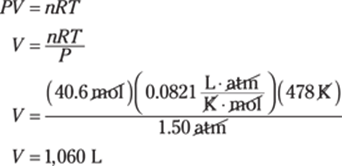
The volume of the carbon dioxide gas is 1,060 L.
841. 195 K
After reading the question, list all the given information. You know the pressure, volume, and number of moles of the gas, and you want to find the temperature, so using the ideal gas law makes sense.
|
P = 6.98 psi |
|
V = 11.2 L |
|
n = 0.333 mol |
|
T = ? K |
Convert the pressure to atmospheres by multiplying the pounds per square inch by 1 atm/14.7 psi:
![]()
Rearrange the ideal gas law to solve for T, and then enter the given and calculated values and R = 0.0821 L·atm/K·mol in the equation. Give your answer three significant figures:
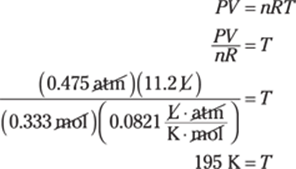
The temperature of the gas sample is 195 K.
842. 0.0944 mol
After reading the question, list all the given information. You know the pressure, volume, and temperature, and you want to find the number of moles of the gas, so using the ideal gas law makes sense.
|
P = 672 torr |
|
V = 3,050 mL |
|
n = ? mol |
|
T = 75°C |
Convert the pressure, volume, and temperature into units that will cancel with the units in the gas constant R: atmospheres, liters, and kelvins:
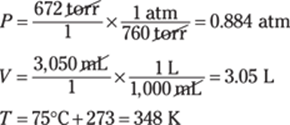
Rearrange the ideal gas law to solve for n, the number of moles, and then enter the given and calculated values and R = 0.0821 L·atm/K·mol in the equation. Give your answer three significant figures:
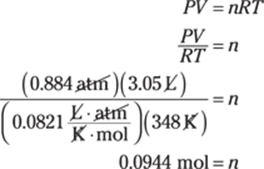
The sample contains 0.0944 mol of oxygen gas.
843. 2.43 atm
After reading the question, list all the given information. You know the volume, number of grams of the gas (which you can convert to moles), and temperature, and you want to find the pressure, so using the ideal gas law makes sense.
|
P = ? atm |
|
V = 75.0 mL |
|
n = ? mol |
|
m = 0.618 g |
|
T = 255 K |
Convert the volume from milliliters to liters (so that the unit will cancel with the volume unit in the gas constant R). Also convert the grams to moles, using twice the molar mass of chlorine because there are two chlorine atoms per molecule of chlorine gas:
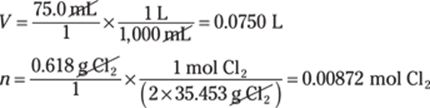
After all that, rearrange the equation for the ideal gas law to solve for P, and then enter the given and calculated values and R = 0.0821 L·atm/K·mol in the equation. Give your answer three significant figures:
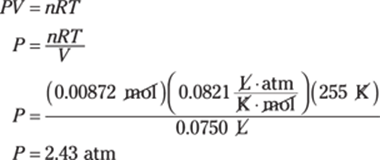
The pressure of the chlorine gas is 2.43 atm.
844. 16 g/mol
To find the molar mass given the number of grams, the pressure, the volume, and the temperature, you need to find the number of moles of the gas. Identify the given information first:
|
P = 0.90 atm |
|
V = 555 L |
|
n = ? mol |
|
m = 261 g |
|
T = 373 K |
Solve the ideal gas law for n, and then enter the given values and R = 0.0821 L·atm/K·mol in the equation:
To find the gas’s molar mass, divide the number of grams by the number of moles. Give your answer two significant figures (because the pressure has only two significant figures):
![]()
The molar mass is 16 g/mol.
845. 44.0 g/mol
To find the molar mass given the number of grams, the pressure, the volume, and the temperature, you need to find the number of moles of the gas. Identify the given information first:
|
P = 1.05 atm |
|
V = 11.3 L |
|
n = ? mol |
|
m = 12.15 g |
|
T = 523 K |
Solve the ideal gas law for n, the number of moles, and then enter the given values and R = 0.0821 L·atm/K·mol in the equation:
To find the gas’s molar mass, divide the number of grams by the number of moles. Give your answer three significant figures:
![]()
The molar mass of the gas is 44.0 g/mol.
846. 847 torr
Given the individual partial pressures, using Dalton’s law is the best way to solve this problem. Enter the partial pressures in the equation and do the math:
847. 3.38 atm
Given the individual partial pressures, using Dalton’s law is the best way to solve this problem. First, convert the torr to atmospheres:
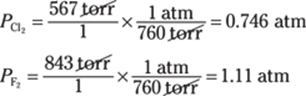
Then enter your numbers into the formula for Dalton’s law, giving your answer two decimal places:
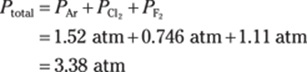
848. 2.99 atm
Given the ratio of the partial pressures (7:3:2) and the total pressure, set up an algebraic equation to determine the partial pressure of any of the gases:
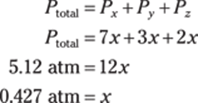
Now substitute in the value of x and calculate the partial pressure of Px, giving your answer three significant figures:
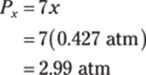
849. 1.63 atm
Given the total pressure and some partial pressures, using Dalton’s law is the best way to solve this problem. Substitute in the given values and solve for the partial pressure of O2, giving your answer two decimal places:
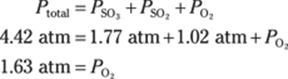
850. 493 torr
The number of moles is proportional to the pressure of the gas (from the ideal gas law), so you can use the mole fraction (χn) to solve for the partial pressure of the propane. First, find the total number of moles in the sample by adding the given values:
ntotal = 0.822 mol + 0.282 mol + 0.550 mol = 1.654 mol
Next, multiply the mole fraction of propane by the total pressure to get the partial pressure due to propane. Give your answer three significant figures:
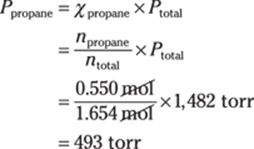
851. 1.31 atm
Each gas exerts a partial pressure proportional to the amount of gas present, so first determine the number of moles of N2:
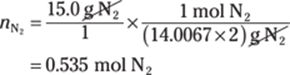
Then you can use the ideal gas law to determine the partial pressure due to the nitrogen gas. Don’t forget to convert the temperature to kelvins by adding 273 to the Celsius temperature:
T = 25°C + 273 = 298 K
Then enter the numbers in the ideal gas law and solve for the partial pressure of N2. Give your answer three significant figures:
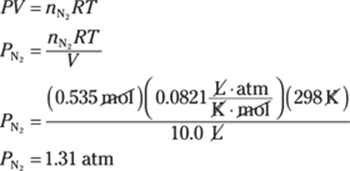
852. 2.84 atm
Each gas exerts a partial pressure proportional to the amount of gas present, so first determine the number of moles of each gas. Divide the given masses by the molar masses of N2 and O2:
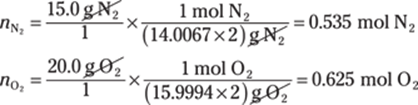
Then use the ideal gas law, PV = nRT, to determine the partial pressure of each gas. Don’t forget to convert the temperature to kelvins by adding 273 to the Celsius temperature:
T = 25°C + 273 = 298 K
Now substitute the values for N2 into the ideal gas law and solve for the pressure (remember that the gas constant R = 0.0821 L·atm/K·mol):
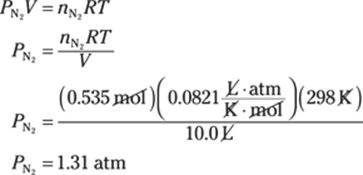
Use the ideal gas law again to find the partial pressure of O2:
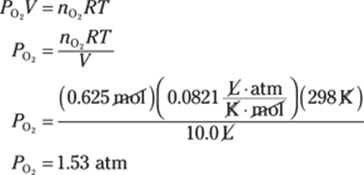
Then use Dalton’s law. Determine the total pressure by adding together the two partial pressures, giving your final answer two decimal places:
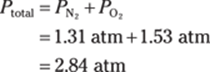
853. 731.8 torr and 10.7 g NH4NO2
Find the partial pressure due to the nitrogen gas using Dalton’s law:
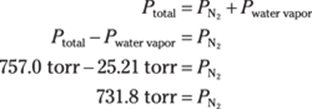
Next, calculate the number of moles of ammonium nitrate using the ideal gas law, PV = nRT (remember that the gas constant R = 0.0821 L·atm/K·mol):
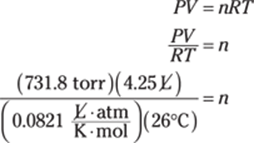
The units need to cancel, so convert the torr to atmospheres by multiplying by 1 atm/760 torr and convert the 26°C to kelvins by adding 273:
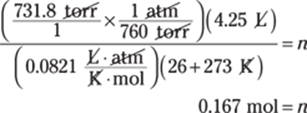
Last, use stoichiometry to find the mass of ammonium nitrite using the mole ratio and the molar mass of ammonium nitrite. Give your answer three significant figures:
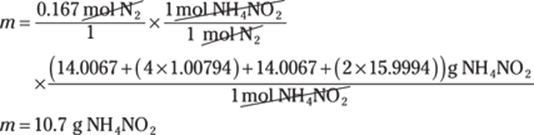
854. 742.6 torr and 0.485 g KCl
First, write and balance the chemical equation that represents the reaction. Here’s the unbalanced equation:
![]()
Balance the oxygen by multiplying the KClO3 by 2 and the O2 by 3:
![]()
And then balance the potassium and the chlorine by multiplying the KCl by 2:
![]()
Now find the partial pressure due to the oxygen gas using Dalton’s law:
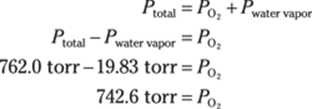
Next, calculate the number of moles of oxygen gas using the ideal gas law, PV = nRT, where R is a constant equal to 0.0821 L·atm/K·mol:
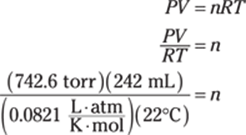
The units need to cancel, so convert the torr to atmospheres by multiplying by 1 atm/760 torr, convert the milliliters to liters by multiplying by 1 L/1,000 mL, and convert the 22°C to kelvins by adding 273:
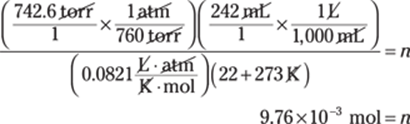
Last, use stoichiometry to find the mass of potassium chloride using the mole ratio and the molar mass of potassium chloride. Give your answer three significant figures:
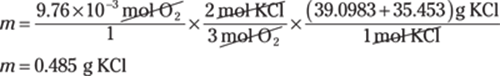
855. O2, because it has the smaller molar mass
According to Graham’s law, the gas with the smaller molar mass effuses/diffuses faster, so the gas with the larger molar mass must diffuse at a slower rate. O2 has a molar mass of about 2(16 g/mol) = 32 g/mol, and argon has a molar mass of about 39 g/mol, so the oxygen gas must effuse faster.
856. CO2, because it has the larger molar mass
According to Graham’s law, the gas with the smaller molar mass effuses/diffuses faster, so the gas with the larger molar mass must effuse at a slower rate. The molar mass of CO2 is about 12 g/mol + 2(16 g/mol) = 44 g/mol, and the molar mass of NH3 is about 14 g/mol + 3(1 g/mol) = 17 g/mol, so the carbon dioxide must effuse more slowly.
857. SO2 effuses 1.0261 times as fast as ClO2.
According to Graham’s law, the gas with the smaller molar mass, SO2, will effuse faster. To find the ratio of the rates of the two gases, substitute one molar mass (MM) of each gas into the formula:
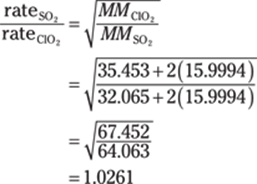
The answer must be greater than 1, because you’re comparing the rate of the faster gas to the rate of the slower one. If your answer is less than 1, you likely placed the wrong gas in the denominator, and the answer is the reciprocal to the one you obtained.
858. 122 L HCl
At standard temperature and pressure, you can use the molar volume of a gas for stoichiometry, because Avogadro’s law proportionally relates the volume to the number of moles of a gas. Using 22.4 L = 1 mol of a gas and the mole ratio from the balanced equation, you can convert from liters H2 to liters HCl using the following map:
liters H2 → moles H2 → moles HCl → liters HCl
Here are the calculations:
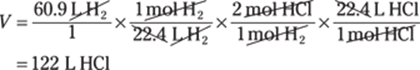
Or you can use the relationship that for every liter of H2 consumed, 2 L of HCl is formed:
![]()
859. 1,790 g Hg
First, write the reaction:
![]()
Now balance it. To balance the oxygen atoms, multiply the HgO by 2:
![]()
Next, balance the mercury atoms by multiplying the Hg on the products side by 2:
![]()
Verify that the atoms on each side of the arrow are equal to each other.
Next, calculate the mass of mercury using molar volume (22.4 L/mol), the mole ratio (from the balanced chemical equation), and molar mass (from the atomic masses on the periodic table):
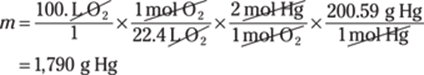
860. 284 L O2
First, write the reaction:
![]()
Then balance it. To balance the oxygen atoms, multiply the water by 2; this gives you an even number of oxygen atoms on the products side:
![]()
Next, balance the hydrogen atoms by multiplying the hydrogen peroxide on the reactant side by 2:
![]()
Verify that the atoms on each side of the arrow are equal to each other.
Next, calculate the volume of oxygen gas produced using molar volume (22.4 L/mol), the mole ratio (from the balanced chemical equation), and molar mass (from the atomic masses on the periodic table):
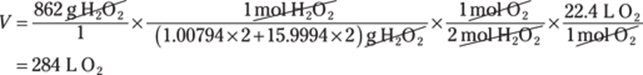
861. 33.3 L NH3
First, write the reaction:
![]()
The balance it. Multiply the ammonia by 2 to balance the two atoms of nitrogen on the reactants side:
![]()
Multiply the hydrogen on the reactants side by 3 to balance the six atoms of hydrogen on the product side:
![]()
Verify that the atoms on each side of the arrow are equal to each other.
Next, find the limiting reactant by comparing the number of moles of each reactant:
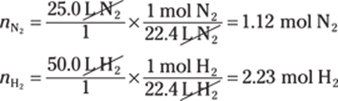
You need three times as much H2 as N2 according to the mole ratio from the balanced reaction, so the H2 is the limiting reactant. Next, solve for the volume of NH3:
liters H2 → moles H2 → moles NH3 → liters NH3
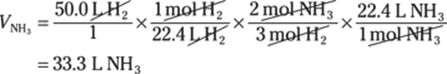
862. the substance dissolved
In a solution, the solute is the substance dissolved. It’s normally the component of a solution present in a lesser quantity.
863. the substance doing the dissolving
In a solution, the solvent is the substance doing the dissolving. It’s normally the component of a solution present in a greater quantity.
864. nitrogen
Air is a solution of gases. The major component (solvent) is nitrogen, which comprises about 80% of air.
865. iron
High-carbon steel — actually, all steel — is a solution containing carbon and other elements dissolved in iron. You can consider all alloys to be solutions. For example, sterling silver is copper dissolved in silver.
866. water
Regular, unflavored vodka is mostly alcohol and water. Because less than half of the vodka (40%) is alcohol, water (the major component) must be the solvent.
867. a saturated solution
The solution above the undissolved solid is a saturated solution. No more solid can dissolve in it.
868. an unsaturated solution
The solution above the undissolved solid was a saturated solution. Separating this solution and then adding more solvent lowers the concentration to something less than saturated: unsaturated.
869. a supersaturated solution
The solution must have been supersaturated. A supersaturated solution is only partially stable; such solutions spontaneously break down, rejecting any solute in excess of what’s present in the saturated solution. If the excess solute is a solid, it appears as a precipitate, a solid separating from the solution.
870. volume of the solution
Molarity (M) is the moles of solute per liter (volume) of solution. Multiplying the molarity of the solution by its volume in liters yields the moles of solute.
871. mass of the solvent
Molality (m) is the moles of solute per kilogram (mass) of solvent. Multiplying the molality of the solution by the solvent’s mass in kilograms yields the moles of solute.
872. 1.2 × 10–2 ppt Pb2+
To give the concentration of a solution in terms of parts per thousand, express the quantity of solute over the quantity of solvent in the same units and multiply the result by 1,000. The amount of lead is in grams, so you need to determine the grams of solution; the grams of solution is the product of the volume of the solution (mL) and its density (g/mL).
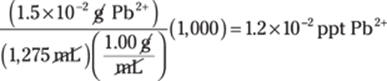
The given concentration of the lead is expressed to two significant figures, which limits the final answer to two significant figures.
873. 2.6 ppm DDT
To give the concentration of a solution in terms of parts per million, express the quantity of solute over the quantity of solvent in the same units and multiply the result by 1,000,000, or 106. The amount of DDT is in grams, so you need to determine the grams of solution; the grams of solution is the product of the volume of the solution (mL) and its density (g/mL).
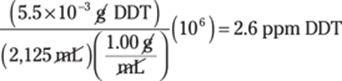
The given mass of the DDT has two significant figures, which limits the final answer to two significant figures.
874. 11 ppb Hg
To express the concentration of a solution in terms of parts per billion, express the quantity of solute over the quantity of solvent in the same units and multiply the result by 1,000,000,000, or 109.
In this case, a convenient unit for the quantity is grams. To convert the moles of mercury to grams, multiply by the molar mass of mercury from the periodic table (200.6 g/mol). To find the quantity of solvent, multiply the volume of the solution by its density. Note that to use the density, you first need to convert the volume (liters) to milliliters. (Another option is to convert the density to g/L.)
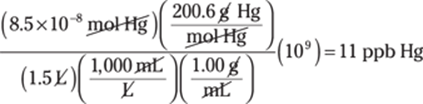
The given concentration of the mercury has two significant figures, which limits the final answer to two significant figures.
875. 0.080 M NaNO3
To determine the molarity of a solution, divide the moles of the solute by the liters of solution:
![]()
The given volume (2.5 L) has two significant figures, which limits the answer to two significant figures.
876. 0.07246 M NaOH
To determine the molarity of a solution, divide the moles of the solute by the liters of solution. For this problem, you need to convert the milliliters to liters.

The given quantities have four significant figures, which limits the answer to four significant figures.
877. 0.30 M NaCl
The molarity is the moles of solute divided by the liters of solution. In this case, you need to use the molar mass (58 g/mol) to convert the grams of NaCl to moles. Begin the problem by expressing the given information as the grams of NaCl over the volume of the solution (L). From this setup, you can see that only the molar mass is needed to convert to the appropriate units, because mol/L = M.
![]()
The given values all have two significant figures, so the answer has two significant figures.
878. 0.58 mol LiCl
Multiplying the molarity of a solution by its volume (in liters) gives you the moles of solute. In this type of problem, expressing the molarity of the solution in terms of its definition (mol/L) is useful:
![]()
The given values all have two significant figures, so the answer has two significant figures.
879. 0.61 L
Convert the mass (15 g) to moles using the molar mass of sulfuric acid (98 g/mol) and then multiply the result by the reciprocal of the molarity (1/M = L/mol):
![]()
The given values all have two significant figures, so the answer has two significant figures.
880. 94.0 g MgCl2
First, determine the moles of MgCl2 by multiplying the molarity of the solution by its volume (in liters). In this type of problem, expressing the molarity of the solution in terms of its definition (mol/L) is useful. Multiplying the moles of MgCl2 by its molar mass yields the grams of solute.
![]()
The given values all have three significant figures, so the answer has three significant figures.
881. 8.0 L
You want the volume of the diluted solution, so rearrange the equation M1V1 = M2V2 to solve for V2. Then enter the numbers and do the math:
![]()
The given values all have two significant figures, so the answer has two significant figures.
882. 3.8 L
The question asks for the volume of the diluted solution, so rearrange the equation M1V1 = M2V2 to solve for V2. Then enter the numbers:
![]()
The given values all have two significant figures, so the answer has two significant figures.
883. 1.4 M HNO3
You want the molarity of the diluted solution, so rearrange the equation M1V1 = M2V2 to solve for M2. Then enter the numbers and do the math:
![]()
The given values all have two significant figures, so the answer has two significant figures.
884. 0.60 M H3PO4
The question asks for the molarity of the diluted solution, so rearrange the equation M1V1 = M2V2 to solve for M2. Both volumes are in milliliters, so you don’t need to convert the volume units. However, you do need to remember that the final volume, V2, is the sum of the two volumes given in the problem.
![]()
The given values have a minimum of two significant figures, so the answer has two significant figures.
885. 0.42 L HCl
You want the volume of the starting solution, so rearrange the equation M1V1 = M2V2 to solve for V1. Then enter the numbers and do the math:
![]()
The given values all have two significant figures, so the answer has two significant figures.
886. 1.9 × 102 mL
The question asks for the volume of the starting solution, so rearrange the equation M1V1 = M2V2 to solve for V1. Both the given volume and the volume you want to find are in milliliters, so you don’t need to convert the volume units.
![]()
The given values have a minimum of two significant figures, so the answer has two significant figures.
887. 6.49 L
You want the volume of the diluted solution, so rearrange the equation M1V1 = M2V2 to find V2:
![]() = 6.49 L
= 6.49 L
The given values all have three significant figures, so the answer has three significant figures.
888. 20.0 L
The question asks for the volume of the diluted solution, so rearrange the equation M1V1 = M2V2 to find V2:
![]()
The given values all have three significant figures, so the answer has three significant figures.
889. 0.20 m KBr
Molality (m) is the moles of solute divided by the kilograms of solvent:
![]()
The given values have a minimum of two significant figures, so the answer has two significant figures.
890. 7.9 × 10–3 m Ca(NO3)2
Molality (m) is the moles of solute divided by the kilograms of solvent. The mass of the solvent is in grams, so convert the grams of water to kilograms.
![]()
The given values have a minimum of two significant figures, so the answer has two significant figures.
891. 0.384 m HCl
Molality (m) is the moles of solute divided by the kilograms of solvent. Use the molar mass of HCl (36.5 g/mol) to convert the grams of the solute, HCl, to moles. The grams cancel out, so you don’t have to convert grams to kilograms.
![]()
The given values all have three significant figures, so the answer has three significant figures.
892. 0.24 mol ZnCl2
The question asks for the moles of the solute. Molality (m) is the moles of solute divided by the kilograms of solvent; therefore, the number of moles present is the molality of the solution (0.16 m) multiplied by the kilograms of solvent (1.5 kg). Enter the numbers and do the math:
![]()
The given values have two significant figures, so the answer has two significant figures.
893. 6.9 g CdCl2
You want to find the mass of the solute. Molality is the moles of solute divided by the kilograms of solvent; therefore, the number of moles present is the molality of the solution (0.015 m) multiplied by the kilograms of the solvent (2.5 kg). Multiplying the number of moles by the molar mass gives you the mass of the solute in grams.
![]()
The given values have a minimum of two significant figures; therefore, the answer has two significant figures.
894. 3.94 g C6H12O6
The question asks for the mass of the solute, glucose, in grams. Molality is the moles of solute divided by the kilograms of solvent; therefore, the number of moles present is the molality of the solution (0.125 m) multiplied by the kilograms of the solvent, water. Multiplying the number of moles by the molar mass gives you the amount of the solute in grams. Note that for the units to cancel correctly, you need to convert the grams of solvent (175 g) to kilograms; this conversion appears in the second and third terms in the following equation:
![]()
The given values have a minimum of three significant figures, so the answer has three significant figures.
895. 3
Calcium nitrate is a strong electrolyte, so in solution, it separates into calcium ions and nitrate ions. Based on the formula Ca(NO3)2, calcium nitrate gives you one calcium ion and two nitrate ions, for a total of three ions. The formation of three ions means that the van’t Hoff factor, i, is 3.
896. It’s lower.
Weak electrolytes produce fewer ions than similar strong electrolytes; therefore, the van’t Hoff factor for weak electrolytes is always less than the van’t Hoff factor for a similar strong electrolyte. Nonelectrolytes can’t produce any ions in solution, so they always have a van’t Hoff factor of 1.
897. 2.79°C
The general equation for freezing point depression is ΔT = iKfm. For nonelectrolytes, the van’t Hoff factor, i, is 1 because a nonelectrolyte can produce only one molecule in solution. Enter the numbers and do the math:
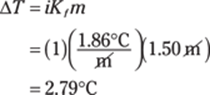
The given values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
A freezing point depression of 2.79°C means that the solution freezes 2.79°C below the normal freezing point of the solvent.
898. 23.4°C
The general equation for freezing point depression is ΔT = iKfm. For nonelectrolytes, the van’t Hoff factor, i, is 1.
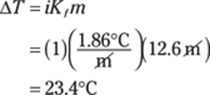
The given values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
A freezing point depression of 23.4°C means that the solution freezes 23.4°C below the normal freezing point of the solvent.
899. 0.102°C
The general equation for boiling point elevation is ΔT = iKbm. For nonelectrolytes, the van’t Hoff factor, i, is 1. To determine the molality of the solution, m, divide the moles of solute (isopropyl alcohol) by the kilograms of solvent (water). Here are the calculations:
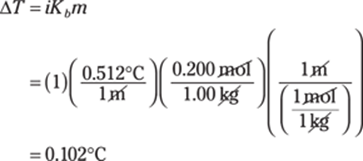
The given values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
A boiling point elevation of 0.102°C means that the solution boils 0.102°C above the normal boiling point of the solvent.
900. 0.108°C
The general equation for boiling point elevation is ΔT = iKbm. For nonelectrolytes, the van’t Hoff factor, i, is 1. To determine the molality of the solution, m, divide the moles of solute (propyl alcohol) by the kilograms of solvent (water). Here are the calculations:
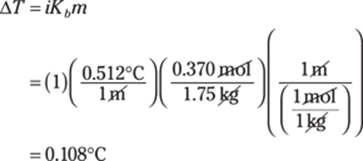
The given values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
A boiling point elevation of 0.108°C means that the solution boils 0.108°C above the normal boiling point of the solvent.
901. 0.915°C
The general equation for freezing point depression is ΔT = iKfm. For nonelectrolytes, the van’t Hoff factor, i, is 1. To determine the molality of the solution, m, convert the grams of ethyl alcohol to moles and divide the result by the kilograms of the solvent. Here are the calculations:
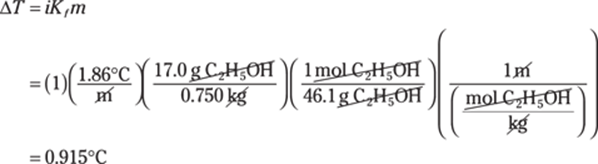
The given values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
A freezing point depression of 0.915°C means that the solution freezes 0.915°C below the normal freezing point of the solvent.
902. 100.673°C
The general equation for boiling point elevation is ΔT = iKbm. For nonelectrolytes, the van’t Hoff factor, i, is 1. To determine the molality of the solution, m, convert the grams of propylene glycol to moles and divide the result by the kilograms of the solvent. Here are the calculations:
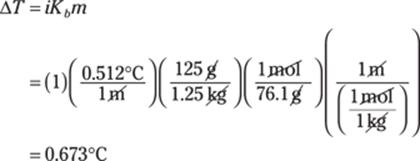
The given values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
A boiling point elevation of 0.673°C means that the solution boils 0.673°C above the normal boiling point of the solvent, which is water. This solution will boil at 100.000°C + 0.673°C = 100.673°C.
903. 0.904°C
The general equation for boiling point elevation is ΔT = iKbm. Hydrogen bromide is a strong electrolyte producing two ions when put into solution; therefore, the van’t Hoff factor, i, is 2. To determine the molality of the solution, m, convert the grams of hydrogen bromide to moles and divide the result by the kilograms of the solvent.
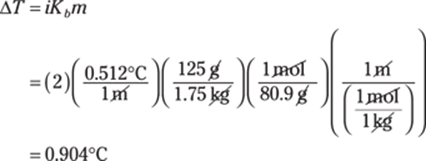
The given values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
A boiling point elevation of 0.904°C means that the solution boils 904°C above the normal boiling point of the solvent.
904. –0.969°C
The general equation is for freezing point depression is ΔT = iKfm. For nonelectrolytes, the van’t Hoff factor, i, is 1.To determine the molality of the solution, m, convert the grams of methyl alcohol to moles and divide the result by the kilograms of the solvent. Here are the calculations:
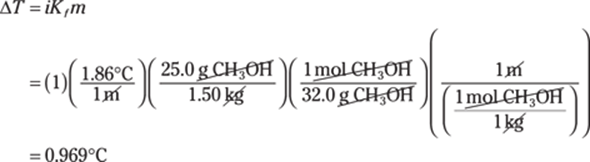
The given values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
A freezing point depression of 0.969°C means that the solution freezes 0.969°C below the normal freezing point of the solvent, which is water. Therefore, the freezing point is
0°C – 0.969°C = –0.969°C
905. –3.69°C
The general equation for freezing point depression is ΔT = iKfm. Rubidium chloride is a strong electrolyte producing two ions when put into solution, so the van’t Hoff factor is 2. To determine the molality of the solution, m, convert the grams of rubidium chloride to moles and divide the result by the kilograms of the solvent. Here are the calculations:
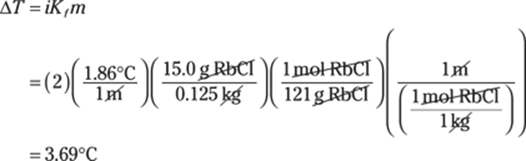
The given values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
A freezing point depression of 3.69°C means that the solution freezes 3.69°C below the normal freezing point of the solvent, which is water. This solution will freeze at
0°C – 3.69°C = –3.69°C
906. 83.40°C
The general equation for boiling point elevation is ΔT = iKbm. For nonelectrolytes, the van’t Hoff factor, i, is 1. To determine the molality of the solution, m, convert the grams of hexane to moles and divide the result by the kilograms of the solvent. Here are the calculations:

The given values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
A boiling point elevation of 2.70°C means that the solution boils 2.70°C above the normal boiling point of the solvent, which is cyclohexane. Thus, the boiling point is
80.70°C + 2.70°C = 83.40°C
907. 6.12 atm
The general equation for osmotic pressure is ΠV = inRT or Π = iMRT, where Π is the osmotic pressure, V is the volume, i is the van’t Hoff factor, n is the number of moles, R is the gas constant, T is the temperature in kelvins, and M is the molarity.
The problem gives you the number of moles, so use the ΠV = inRT form. For nonelectrolytes, the van’t Hoff factor is 1. The temperature must be in kelvins, so convert from degrees Celsius: T = °C + 273 = 25 + 273 = 298 K.
Solve the equation ΠV = inRT for Π, enter the appropriate values, and do the math:
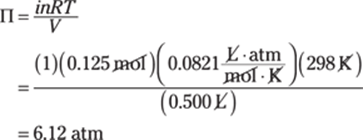
The values in the calculation all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
908. 30.6 atm
The general equation for osmotic pressure is ΠV = inRT or Π = iMRT, where Π is the osmotic pressure, V is the volume, i is the van’t Hoff factor, n is the number of moles, R is the gas constant, T is the temperature, and Mis the molarity.
You’re given the molarity, so use Π = iMRT. For nonelectrolytes, the van’t Hoff factor is 1. The temperature must be in kelvins, so convert from degrees Celsius: T = °C + 273 = 25 + 273 = 298 K.
Enter the appropriate values in the equation and do the math. Expressing the molarity in terms of its definition (mol/L) can help you set up the problem correctly.

The values in the calculation all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
909. 1.75 atm
The general equation for osmotic pressure is ΠV = inRT or Π = iMRT, where Π is the osmotic pressure, V is the volume, i is the van’t Hoff factor, n is the number of moles, R is the gas constant, T is the temperature, and Mis the molarity.
You can find the number of moles from the mass and molar mass of the solute, so use the ΠV = inRT form. For nonelectrolytes, the van’t Hoff factor is 1. The temperature must be in kelvins, so convert from degrees Celsius: T = °C + 273 = 45 + 273 = 318 K. To convert the mass of the solute to moles, multiply by the molar mass.
Solve the equation ΠV = inRT for Π, enter the appropriate values, and do the math:
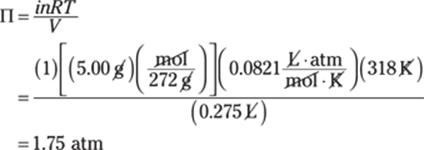
The values all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
910. 3.48 × 103 g/mol
The general equation for osmotic pressure is ΠV = inRT or Π = iMRT, where Π is the osmotic pressure, V is the volume, i is the van’t Hoff factor, n is the number of moles, R is the gas constant, T is the temperature, and Mis the molarity.
To find the molar mass, you first need to calculate the number of moles, n, so use the ΠV = inRT form of the equation. For nonelectrolytes, the van’t Hoff factor is 1. The temperature must be in kelvins, so convert from degrees Celsius: T = °C + 273 = 37 + 273 = 310. K.
Rearrange ΠV = inRT and solve for n to find the number of moles:
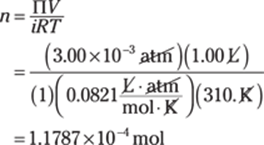
To get the molar mass of the polymer, divide the mass of the sample by the number of moles:
![]()
The values in the calculation all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.) Extra numbers appear in the calculations for the intermediate value (moles) to minimize errors due to intermediate rounding.
911. 17.7 atm
The general equation for osmotic pressure is ΠV = inRT or Π = iMRT, where Π is the osmotic pressure, V is the volume, i is the van’t Hoff factor, n is the number of moles, R is the gas constant, T is the temperature, and Mis the molarity.
You can find the number of moles, n, by multiplying the grams of calcium nitrate by its molar mass, so use the ΠV = inRT form. This is a strong electrolyte that produces three ions per molecule in solution (one calcium ion and two nitrate ions), so the van’t Hoff factor is 3. The temperature, 37°C, must be in kelvins, so do the temperature conversion: T = °C + 273 = 37 + 273 = 310. K.
To finish the problem, rearrange the equation and enter the appropriate values:
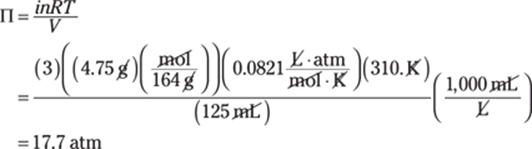
The values in the calculation all have three significant figures, so the answer has three significant figures. (The van’t Hoff number has no effect on significant figures because it’s an exact number in this case.)
912. HNO3
Nitric acid (HNO3) is a strong acid. It’s one of the most commonly encountered strong acids, along with hydrochloric acid (HCl), hydrobromic acid (HBr), hydroiodic acid (HI), chloric acid (HClO3), perchloric acid (HClO4), and sulfuric acid (H2SO4).
913. KOH
Potassium hydroxide (KOH) is a commonly encountered strong base, as are sodium hydroxide (NaOH) and calcium hydroxide (Ca(OH)2). Other strong bases are lithium hydroxide (LiOH), rubidium hydroxide (RbOH), cesium hydroxide (CsOH), strontium hydroxide (Sr(OH)2), and barium hydroxide, (Ba(OH)2).
914. HNO2
HNO2 is nitrous acid. In most cases, if the formula begins with H, the compound is an acid. Water (H2O) is a common exception. Seven acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4) are strong acids — assume all other acids are weak unless told otherwise.
915. NH3
Ammonia (NH3) and similar compounds are common weak bases. Similar substances, including amines such as methylamine (CH3NH2), are organic bases that are also weak.
916. CH3OH
Methanol, CH3OH, is not an acid or a base. Metal hydroxides, such as NaOH, are well-known bases. However, when the –OH group is attached to a nonmetal, the hydroxides are often neutral compounds. The presence of very electronegative elements can cause these otherwise neutral compounds to be acidic.
917. CH3NH2
The compound methylamine (CH3NH2) is similar to ammonia (NH3), and like ammonia, it’s a weak base. Many compounds containing nitrogen and carbon are weak bases.
918. Cl–
To determine the conjugate base of any acid, simply remove one H+. In this case, HCl – H+ leaves Cl–. Never remove more than one H+ to determine the conjugate base.
919. HC2H3O2
To determine the conjugate acid of any base, simply add one H+. In this case, ![]() forms HC2H3O2. Never add more than one H+ to determine the conjugate acid.
forms HC2H3O2. Never add more than one H+ to determine the conjugate acid.
920. ![]()
To determine the conjugate acid of any base, simply add one H+. In this case, NH3 + H+ forms ![]() . Never add more than one H+ to determine the conjugate acid.
. Never add more than one H+ to determine the conjugate acid.
921. ![]()
To determine the conjugate base of any acid, simply remove one H+. In this case, NH3 – H+ leaves ![]() . Never remove more than one H+ to determine the conjugate base.
. Never remove more than one H+ to determine the conjugate base.
922. ![]()
To determine the conjugate base of any acid, simply remove one H+. In this case, H3PO4 – H+ leaves ![]() . Never remove more than one H+ to determine the conjugate base.
. Never remove more than one H+ to determine the conjugate base.
923. ![]()
To determine the conjugate acid of any base, simply add one H+. In this case, H2SO4 + H+ forms ![]() .
.
Note: In aqueous solutions, H2SO4 is a strong acid; however, stronger acids, such as HClO4, exist. The stronger acid is capable of transferring an H+ not only to a base but also to a weaker acid. This occurs in many aqueous solutions where an acid transfers an H+ to the base H2O to produce the conjugate acid H3O+.
924. HPO42–
The ion HPO42– may either lose a hydrogen ion to form PO43– or gain a hydrogen ion to form ![]() . The ability to lose a hydrogen ion makes it a conjugate acid, and the ability to gain a hydrogen ion makes it a conjugate base.
. The ability to lose a hydrogen ion makes it a conjugate acid, and the ability to gain a hydrogen ion makes it a conjugate base.
925. 2.0
To determine the pH of a solution, you need to know the hydrogen ion concentration. For a strong acid such as HNO3 (nitric acid), the hydrogen ion concentration is equal to the concentration of the acid. Here, [H+] = 0.01 M. Inserting this concentration in the pH relationship gives you the following:
![]()
In this case, converting the concentration to scientific notation, 10–2, and taking the negative of the exponent also gives the pH. Because this is an acid, the pH should be below 7, which it is.
If this compound were a weak acid, you’d have to use the Ka of the acid to determine the hydrogen ion concentration. Ka is the equilibrium constant for the ionization of a weak acid.
926. 3.0
To determine the pOH of a solution, you need to know the hydroxide ion concentration. For a strong base such as KOH (potassium hydroxide), the hydroxide ion concentration is equal to the concentration of the base. In this case, [OH–] = 0.001 M. Inserting this concentration in the pOH relationship gives you the following:
![]()
In this case, converting the concentration to scientific notation, 10–3, and taking the negative of the exponent also gives you the pOH. Because this is an acid, the pOH should be below 7, which it is.
If this compound were a weak base, you’d have to use the Kb of the base to determine the hydroxide ion concentration. Kb is the equilibrium constant for the ionization of a weak base.
927. 12.18
To determine the pH of a base solution, it’s helpful to find the pOH of the solution first. To determine the pOH, find the hydroxide ion concentration. For a strong base such as NaOH (sodium hydroxide), the hydroxide ion concentration is equal to the concentration of the base. Here, [OH–] = 0.015 M. Inserting this concentration in the pOH relationship gives you the following:
![]()
The last step is to find the pH using the relationship pKw = pH + pOH, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 1.82 = 12.18
Another way to solve the problem is to use the relationship Kw = [H+][OH–] = 1.00 × 10–14:
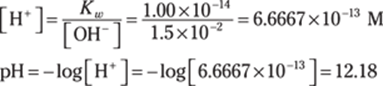
Because this solution is a base, the pH should be above 7, which it is.
If this compound were a weak base, you’d have to use the Kb of the base to determine the hydroxide ion concentration. Kb is the equilibrium constant for the ionization of a weak base.
928. 11.40
To determine the pOH of an acid solution, it’s helpful to find the pH of the solution first. To determine the pH, find the hydrogen ion concentration. For a strong acid such as HCl (hydrochloric acid), the hydrogen ion concentration is equal to the concentration of the acid. In this case, [H+] = 0.0025 M. Inserting this concentration in the pH relationship gives you the following:
![]()
The last step is to find the pOH using the relationship pKw = pH + pOH, where pKw = 14.000:
pOH = 14.000 – pH = 14.000 – 2.60 = 11.40
Another way to solve the problem is to use the relationship Kw = [H+][OH–] = 1.00 × 10–14, where Kw is the autoionization equilibrium constant for water:
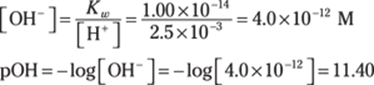
Because this is an acid, the pH should be below 7 and the pOH should be above 7, which they are.
If this compound were a weak acid, you’d have to use the acid’s ionization constant, Ka, to determine the hydrogen ion concentration.
929. 11.8
To determine the pH of a base solution, it’s helpful to find the pOH of the solution first. To determine the pOH, find the hydroxide ion concentration. For a strong base such as Ba(OH)2 (barium hydroxide), the hydroxide ion concentration is double the concentration of the base (double because of the subscript 2). In this case, [OH–] = 2(0.003) = 0.006 M. Inserting this concentration in the pOH relationship gives you the following:
![]()
The last step is to find the pH using the relationship pKw = pH + pOH, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 2.2 = 11.8
Because this compound is a base, the pH should be above 7, which it is.
You can also solve the problem by using the relationship Kw = [H+][OH–] = 1.00 × 10–14:
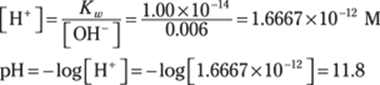
If this compound were a weak base, you’d have to use the base’s ionization constant, Kb, to determine the hydroxide ion concentration.
930. 11.30
To determine the pH of a base solution, it’s helpful to find the pOH of the solution first. To determine the pOH, find the hydroxide ion concentration. For a strong base such as Sr(OH)2 (strontium hydroxide), the hydroxide ion concentration is double the concentration of the base (double because of the subscript 2). In this case, [OH–] = 2(1.0 × 10–3) = 2.0 × 10–3 M. Inserting this concentration in the pOH relationship gives you the following:
![]()
The last step is to find the pH using the relationship pKw = pH + pOH, where pKw = 14.000:
pH = 14.000 – pOH = 14.000 – 2.70 = 11.30
Because this is a base, the pH should be above 7 and the pOH should be below 7, which they are.
You can also solve the problem by using the relationship Kw = [H+][OH–] = 1.00 × 10–14. In this case,
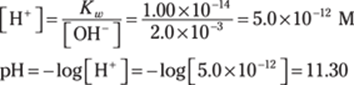
If this compound were a weak base, you’d have to use the base’s ionization constant, Kb, to determine the hydroxide ion concentration.
931. 0
To determine the pH of a solution, you need to know the hydrogen ion concentration. For a strong acid such as HBr (hydrobromic acid), the hydrogen ion concentration is equal to the concentration of the acid. Here, [H+] = 1.0 M. Inserting this concentration in the pH relationship gives you the following:
![]()
In this case, converting the concentration to scientific notation, 100, and taking the negative of the exponent also gives you the pH.
Because the compound is an acid, the pH should be below 7, which it is.
If this compound were a weak acid, you’d need to use the acid’s ionization constant, Ka, to determine the hydrogen ion concentration.
932. 15.18
To determine the pH of a base solution, it’s helpful to find the pOH of the solution first. To determine the pOH, you need the hydroxide ion concentration. For a strong base such as NaOH (sodium hydroxide), the hydroxide ion concentration is equal to the concentration of the base. In this case, [OH–] = 15 M. Inserting this concentration in the pOH relationship gives you the following:
![]()
The last step is to find the pH using the relationship pKw = pH + pOH, where pKw = 14.000:
pH = 14.000 – pOH = 14.000 – (–1.18) = 15.18
You can also solve the problem by using the relationship Kw = [H+][OH–] = 1.00 × 10–14:
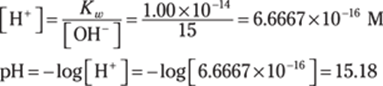
Because this compound is a base, the pH should be above 7, which it is. You have a very high concentration of a strong base, so it’s possible for the pH to be above 14.
If this compound were a weak base, you’d have to use the base’s ionization constant, Kb, to determine the hydroxide ion concentration.
933. 2.39
Acetic acid isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
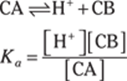
where CA refers to the conjugate acid and CB refers to the conjugate base. In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
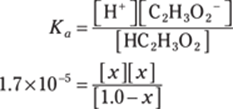
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –5, and the exponent on the concentration is 0. If the exponent on theK is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
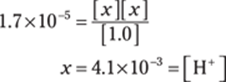
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, use the pH definition:
![]()
934. 1.66
Nitrous acid isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
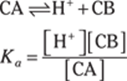
where CA refers to the conjugate acid and CB refers to the conjugate base. In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
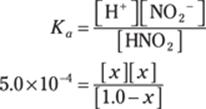
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –4, and the exponent on the concentration is 0. If the exponent on theK is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
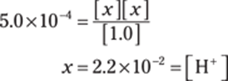
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, use the pH definition:
![]()
935. 2.38
Ammonia isn’t one of the strong bases (NaOH, KOH, LiOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, and Ba(OH)2), so it must be a weak base. Weak base calculations need a base equilibrium constant, Kb. The generic form of every Kb problem is
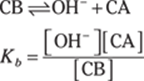
where CA refers to the conjugate acid and CB refers to the conjugate base. For balancing purposes, water may be present in the equilibrium chemical equation; however, water is also the solvent, so it shouldn’t be in the equilibrium expression.
In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Kb expression gives you the following:
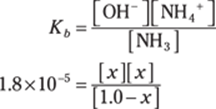
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –5, and the exponent on the concentration is 0. If the exponent on theK is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
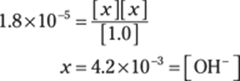
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, use the pOH definition:
![]()
936. 5.10
Hydrocyanic acid isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Kaproblem is
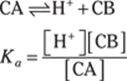
where CA refers to the conjugate acid and CB refers to the conjugate base. In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
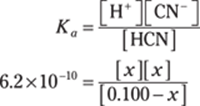
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –10, and the exponent on the concentration is –1. If the exponent on the K is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
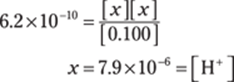
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, use the pH definition:
![]()
937. 2.74
Acetic acid isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
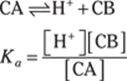
where CA refers to the conjugate acid and CB refers to the conjugate base. In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
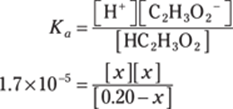
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –5, and the exponent on the concentration is –1. If the exponent on theK is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
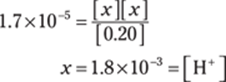
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, use the pH definition:
![]()
938. 2.85
Periodic acid isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
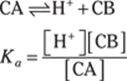
where CA refers to the conjugate acid and CB refers to the conjugate base. In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
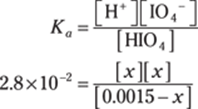
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –2, and the exponent on the concentration is –3. If the exponent on theK is at least 3 less than the exponent on the concentration, you can assume that –x in the denominator is insignificant. That doesn’t occur in this case, so you have to solve this equation as a quadratic.
Write the quadratic equation in standard form:
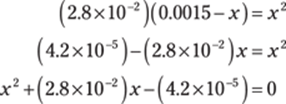
Then solve for x using the quadratic formula. Here, a = 1, b = 2.8 × 10–2, and c = –4.2 × 10–5:
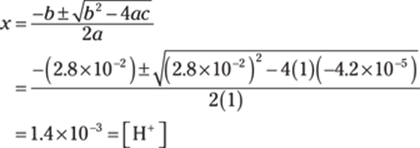
You can neglect the negative root because negative concentrations aren’t possible.
Finally, use the pH definition:
![]()
939. 2.68
Chlorous acid isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
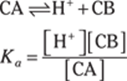
where CA refers to the conjugate acid and CB refers to the conjugate base. In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
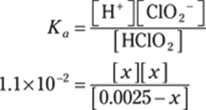
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –2, and the exponent on the concentration is –3. If the exponent on theK is at least 3 less than the exponent on the concentration, you can assume that –x in the denominator is insignificant. That doesn’t occur in this case, so you have to solve this equation as a quadratic.
Write the quadratic equation in standard form:
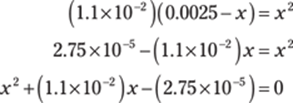
Then solve for x using the quadratic formula. Here, a = 1, b = 1.1 × 10–2, and c = –2.75 × 10–5:
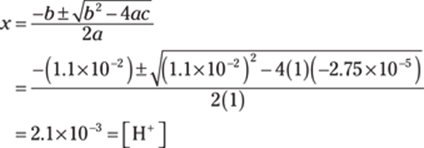
You can neglect the negative root because negative concentrations aren’t possible.
Finally, use the pH definition:
![]()
940. 4.39
Pyridine isn’t one of the strong bases (NaOH, KOH, LiOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, and Ba(OH)2), so it must be a weak base. Weak base calculations need a base equilibrium constant, Kb. The generic form of every Kb problem is
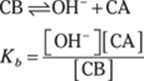
where CA refers to the conjugate acid and CB refers to the conjugate base. For balancing purposes, water may be present in the equilibrium chemical equation; however, water is also the solvent, so it shouldn’t be in the equilibrium expression.
In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Kb expression gives you the following:
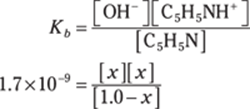
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –9, and the exponent on the concentration is 0. If the exponent on theK is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
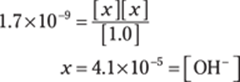
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, use the pOH definition:
![]()
941. 11.20
Ammonia isn’t one of the strong bases (NaOH, KOH, LiOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, and Ba(OH)2), so it must be a weak base. Weak base calculations need a base equilibrium constant, Kb. The generic form of every Kb problem is
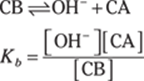
where CA refers to the conjugate acid and CB refers to the conjugate base. For balancing purposes, water may be present in the equilibrium chemical equation; however, water is also the solvent, so it shouldn’t be in the equilibrium expression.
In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Kb expression gives you the following:
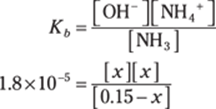
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to that of the concentration (in scientific notation). In this case, the exponent on the K is –5, and the exponent on the concentration is –1. If the exponent on the K is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
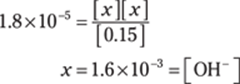
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Find the pOH using the pOH definition:
![]()
Finally, find the pH using the relationship pKw = pH + pOH, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 2.80 = 11.20
942. 12.56
Methylamine isn’t one of the strong bases (NaOH, KOH, LiOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, and Ba(OH)2), so it must be a weak base. Weak base calculations need a base equilibrium constant, Kb. The generic form of every Kb problem is
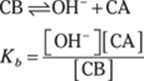
where CA refers to the conjugate acid and CB refers to the conjugate base. For balancing purposes, water may be present in the equilibrium chemical equation; however, water is also the solvent, so it shouldn’t be in the equilibrium expression.
In this problem, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Kb expression gives you the following:
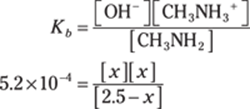
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –4, and the exponent on the concentration is 0. If the exponent on theK is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
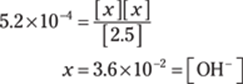
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Use the pOH definition to find the pOH:
![]()
Finally, find the pH using the relationship pKw = pH + pOH, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 1.44 = 12.56
943. 1.38
Chlorous acid isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
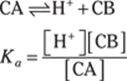
where CA refers to the conjugate acid and CB refers to the conjugate base. In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
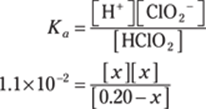
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –2, and the exponent on the concentration is –1. If the exponent on theK is at least 3 less than the exponent on the concentration, you can assume that –x in the denominator is insignificant. That doesn’t occur in this case, so you have to solve this equation as a quadratic.
Write the quadratic equation in standard form:
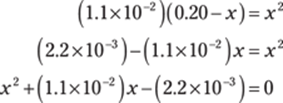
Then solve for x using the quadratic formula. Here, a = 1, b = 1.1 × 10–2, and c = –2.2 × 10–3:
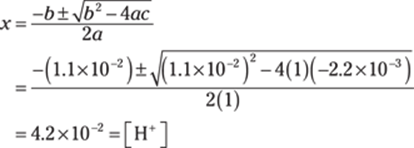
You can neglect the negative root because negative concentrations aren’t possible.
Finally, find the pH using the pH definition:
![]()
944. 2.68
Cyanic acid isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
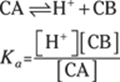
where CA refers to the conjugate acid and CB refers to the conjugate base.
The problem gives you the pKa, so convert that to a Ka:
![]()
In this problem, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
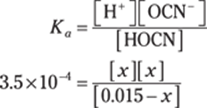
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –4, and the exponent on the concentration is –2. If the exponent on theK is at least 3 less than the exponent on the concentration, you can assume that –x in the denominator is insignificant. That doesn’t occur in this case, so you have to solve this equation as a quadratic.
Write the quadratic equation in standard form:
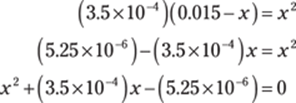
Then solve for x using the quadratic formula. Here, a = 1, b = 3.5 × 10–4, and c = –5.25 × 10–6:
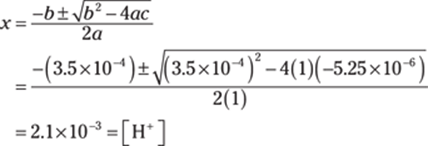
You can neglect the negative root because negative concentrations aren’t possible.
Finally, find the pH using the pH definition:
![]()
945. 1.64
Methylamine isn’t one of the strong bases (NaOH, KOH, LiOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, and Ba(OH)2), so it must be a weak base. Weak base calculations need a base equilibrium constant, Kb. The generic form of every Kb problem is
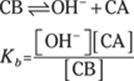
where CA refers to the conjugate acid and CB refers to the conjugate base. For balancing purposes, water may be present in the equilibrium chemical equation; however, water is also the solvent, so it shouldn’t be in the equilibrium expression.
The problem gives you the pKb, so convert that to a Kb:
![]()
In this problem, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Kb expression gives you the following:
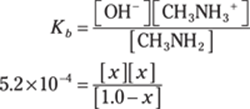
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –4, and the exponent on the concentration is 0. If the exponent on theK is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
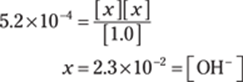
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, find the pOH using the pOH definition:
![]()
946. 1.33
NaHSO4 is a strong electrolyte that completely separates into Na+ and ![]() . The hydrogen sulfate ion, due to the presence of an acidic hydrogen, isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
. The hydrogen sulfate ion, due to the presence of an acidic hydrogen, isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
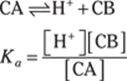
where CA refers to the conjugate acid and CB refers to the conjugate base. In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
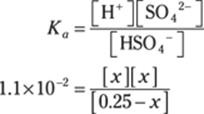
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –2, and the exponent on the concentration is –1. If the exponent on theK is at least 3 less than the exponent on the concentration, you can assume that –x in the denominator is insignificant. That doesn’t occur in this case, so you have to solve this equation as a quadratic.
Write the quadratic equation in standard form:
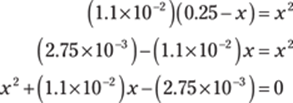
Then solve for x using the quadratic formula. Here, a = 1, b = 1.1 × 10–2, and c = –2.75 × 10–3:
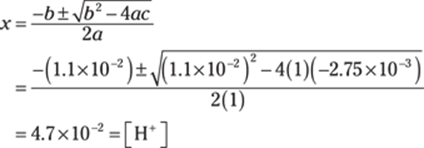
You can neglect the negative root because negative concentrations aren’t possible.
Finally, use the pH definition to find the pH:
![]()
947. 9.38
Calcium acetate is a strong electrolyte, which completely separates into Ca2+ and ![]() . The acetate ion is a conjugate base of a weak acid. The acetate ion isn’t one of the strong bases (NaOH, KOH, LiOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, and Ba(OH)2), so it must be a weak base. Weak base calculations need a base equilibrium constant, Kb. The generic form of every Kb problem is
. The acetate ion is a conjugate base of a weak acid. The acetate ion isn’t one of the strong bases (NaOH, KOH, LiOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, and Ba(OH)2), so it must be a weak base. Weak base calculations need a base equilibrium constant, Kb. The generic form of every Kb problem is
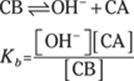
where CA refers to the conjugate acid and CB refers to the conjugate base. For balancing purposes, water may be present in the equilibrium chemical equation; however, water is also the solvent, so it shouldn’t be in the equilibrium expression.
The problem gives you the Ka, so convert the Ka to Kb using the following relationship, where Kw = 1.00 × 10–14:
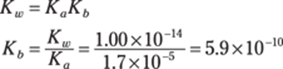
The formula of calcium acetate, Ca(C2H3O2)2, contains two acetate ions; therefore, the concentration of the acetate ion is twice that of the calcium acetate, or 1.0 M. In this problem, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Kb expression gives you the following:
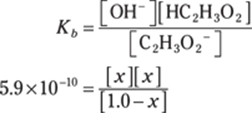
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –10, and the exponent on the concentration is 0. If the exponent on theK is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
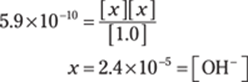
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Next, find the pOH using the pOH definition:
![]()
Finally, find the pH using the relationship pKw = pH + pOH, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 4.62 = 9.38
948. 2.500 × 10–3 mol HCl
First, add the information from the problem to the balanced chemical equation:
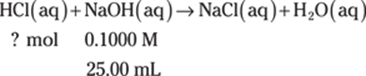
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
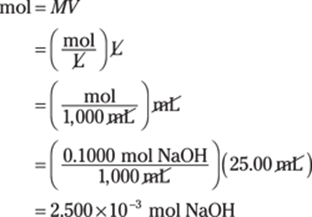
Now convert from moles of NaOH to moles of HCl using the mole ratio, which is based on the coefficients from the balanced chemical equation. Here are all the calculations for this problem:
mol NaOH/mL → mol NaOH → mol HCl
![]()
949. 0.1771 M HClO
First, add the information from the question to the balanced chemical equation:
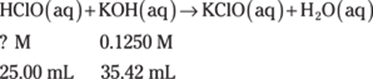
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
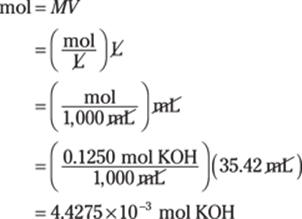
Now convert from moles of KOH to moles of HClO using the mole ratio, which is based on the coefficients from the balanced chemical equation:
![]()
Dividing by the volume of the HClO solution (in liters) yields the molarity. Here are all the calculations for this problem:
mol KOH/mL → mol KOH → mol HClO → mol HClO/L
![]()
950. 0.2123 M HClO2
First, add the information from the problem to the balanced chemical equation:
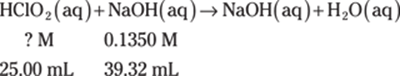
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
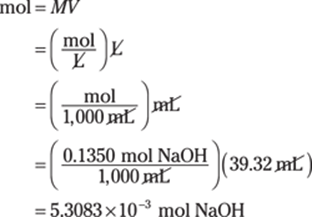
Now convert from moles of NaOH to moles of HClO2 using the mole ratio, which is based on the coefficients from the balanced chemical equation:
![]()
Dividing by the volume of the HClO2 solution (in liters) yields the molarity. Here are all the calculations for this problem:
mol NaOH/mL → mol NaOH → mol HClO2 → mol HClO2/L
![]()
951. 1.250 × 10–2 mol LiOH
First, add the information from the problem to the balanced chemical equation:
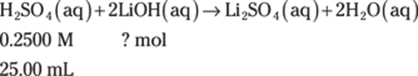
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
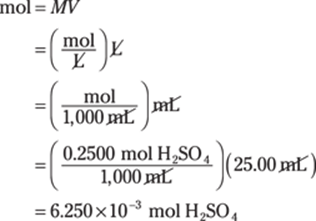
Now convert from moles of H2SO4 to moles of LiOH using the mole ratio, which is based on the coefficients from the balanced chemical equation. Here are all the calculations for this problem:
mol H2SO4/mL → mol H2SO4 → mol LiOH
![]()
952. 0.09496 M Ca(OH)2
First, add the information from the problem to the balanced chemical equation:
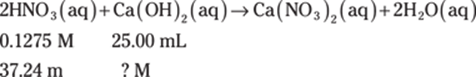
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
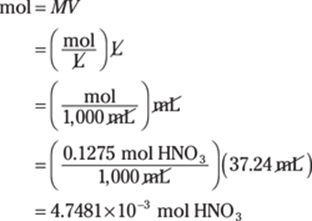
Now convert from moles of HNO3 to moles of Ca(OH)2 using the mole ratio, which is based on the coefficients from the balanced chemical equation:
![]()
Dividing by the volume of the Ca(OH)2 solution (in liters) yields the molarity. Here are all the calculations for this problem:
mol HNO3/mL → mol HNO3 → mol Ca(OH)2 → mol Ca(OH)2/L
![]()
953. 0.5889 M Sr(OH)2
First, add the information from the problem to the balanced chemical equation:
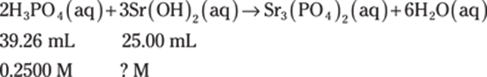
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
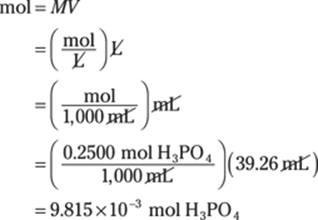
Now convert from moles of H3PO4 to moles of Sr(OH)2 using the mole ratio, which is based on the coefficients from the balanced chemical equation:
![]()
Dividing by the volume of the Sr(OH)2 solution (in liters) yields the molarity. Here are all the calculations for this problem:
mol H3PO4/mL → mol H3PO4 → mol Sr(OH)2 → mol Sr(OH)2/L
![]()
954. 3.500 × 10–3 mol HNO2
The problem doesn’t give you an equation, so the first step is to write a balanced chemical equation:
![]()
Add the information from the problem to the balanced chemical equation:
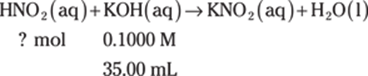
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
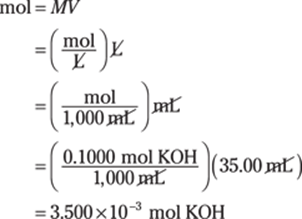
Now convert from moles of KOH to moles of HNO2 using the mole ratio, which is based on the coefficients from the balanced chemical equation. Here are all the calculations for this problem:
mol KOH/mL → mol KOH → mol HNO2
![]()
955. 0.4277 M RbOH
The problem doesn’t give you an equation, so the first step is to write a balanced chemical equation:
![]()
Add the information from the problem to the balanced chemical equation:
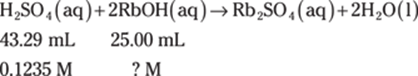
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
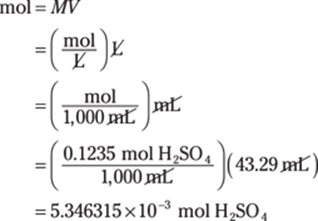
Now convert from moles of H2SO4 to moles of RbOH using the mole ratio, which is based on the coefficients from the balanced chemical equation:
![]()
Dividing by the volume of the RbOH solution (in liters) yields the molarity. Here are all the calculations for this problem:
mol H2SO4/mL → mol H2SO4 → mol RbOH → mol RbOH/L
![]()
956. 1.865 × 10–2 M H3PO4
The problem doesn’t give you an equation, so the first step is to write a balanced chemical equation:
![]()
Add the information from the problem to the balanced chemical equation:
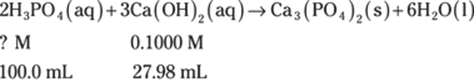
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
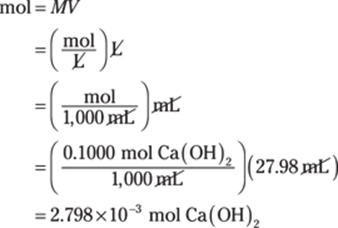
Now convert from moles of Ca(OH)2 to moles of H3PO4 using the mole ratio, which is based on the coefficients from the balanced chemical equation:
![]()
Dividing by the volume of the H3PO4 solution (in liters) yields the molarity. Here are all the calculations for this problem:
mol Ca(OH)2/mL → mol Ca(OH)2 → mol H3PO4 → mol H3PO4/L
![]()
957. 0.6170 M NH3
The problem doesn’t give you an equation, so the first step is to write a balanced chemical equation:
![]()
Add the information from the problem to the balanced chemical equation:
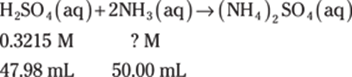
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
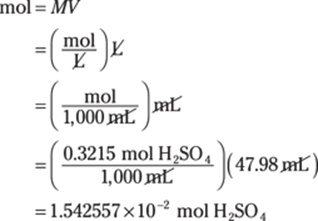
Now convert from moles of H2SO4 to moles of NH3 using the mole ratio, which is based on the coefficients from the balanced chemical equation:
![]()
Dividing by the volume of the NH3 solution (in liters) yields the molarity. Here are all the calculations for this problem:
mol H2SO4/mL → mol H2SO4 → mol NH3 → mol NH3/L
![]()
958. 0.2303 g HC2H3O2
The problem doesn’t give you an equation, so the first step is to write a balanced chemical equation:
![]()
Add the information from the problem to the balanced chemical equation:
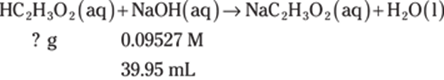
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
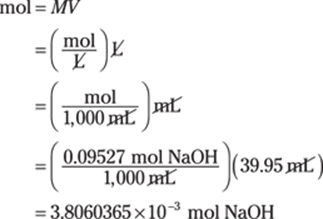
Now convert from moles of NaOH to moles of HC2H3O2 using the mole ratio, which is based on the coefficients from the balanced chemical equation:
![]()
To finish the problem, multiply the moles by the molar mass. Here are all the calculations for this problem:
mol NaOH/mL → mol NaOH → mol HC2H3O2 → mol HC2H3O2/L
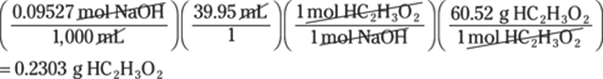
959. 0.05333 g NH3
The problem doesn’t give you an equation, so the first step is to write a balanced chemical equation:
![]()
Add the information from the problem to the balanced chemical equation:
Next, change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
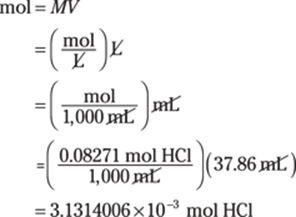
Now convert from moles of HCl to moles of NH3 using the mole ratio, which is based on the coefficients from the balanced chemical equation:
![]()
To finish the problem, multiply the moles by the molar mass. Here are all the calculations for this problem:
mol HCl/mL → mol HCl → mol NH3 → g NH3
![]()
960. HNO2
A buffer solution must contain a conjugate acid-base pair of a weak acid or base. Sodium nitrite (NaNO2) contains the nitrite ion, which is the conjugate base of nitrous acid (HNO2), a weak acid.
961. (NH4)2SO4
A buffer solution must contain a conjugate acid-base pair of a weak acid or base. Ammonia (NH3) is a weak base, so to create a buffer, you need to add the conjugate acid. The conjugate acid of ammonia acid is the ammonium ion ![]() .
.
962. KHCO3
A buffer solution must contain a conjugate acid-base pair of a weak acid or base. Sodium carbonate (Na2CO3) contains the carbonate ion, which is the conjugate base of the hydrogen carbonate ion ![]() , a weak acid.
, a weak acid.
963. 4.58
This is a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is acetic acid (0.75 M), and the conjugate base, CB, is the acetate ion (0.50 M). The acetate ion comes from sodium acetate, which is a strong electrolyte.
Entering the given information in the Henderson-Hasselbalch equation gives you the answer:
![]()
You can also solve any buffer problem as a Ka or Kb problem. This normally takes more time than using the Henderson-Hasselbalch equation and results in the same answer.
Acetic acid isn’t of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
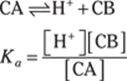
The problem gives you the pKa, so convert the pKa to a Ka:
![]()
In this case, the equilibrium equation and the change in the concentrations (calculated previously) are
![]()
Entering this information in the Ka expression gives you the following:
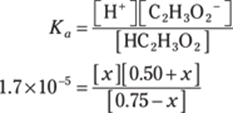
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the changes in the numerator and denominator are insignificant, as they are here, you can drop the +x and –x, simplifying the problem:
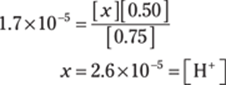
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, find the pH using the pH definition:
![]()
964. 4.90
This is a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is the ammonium ion (0.35 M), and the conjugate base, CB, is ammonia (0.25 M). The ammonium ion comes from ammonium chloride, which is a strong electrolyte.
Entering the given information in the Henderson-Hasselbalch equation gives you the answer:
![]()
You can also solve any buffer problem as a Ka or Kb problem (see Question 963 for an example of this approach). This normally takes more time than using the Henderson-Hasselbalch equation and results in the same answer.
965. 3.32
This is a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is hydrofluoric acid (0.25 M), and the conjugate base, CB, is the fluoride ion (0.35 M). The fluoride ion comes from sodium fluoride, which is a strong electrolyte.
Entering the given information in the Henderson-Hasselbalch equation gives you the answer:
![]()
You can also solve any buffer problem as a Ka or Kb problem (see Question 963 for an example of this approach).
966. 3.10
This is a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is the methylammonium ion (0.50 M), and the conjugate base, CB, is methylamine (0.75 M). The methylammonium ion comes from methylammonium chloride, which is a strong electrolyte.
Entering the given information in the Henderson-Hasselbalch equation gives you the following:
![]()
You can also solve any buffer problem as a Ka or Kb problem (see Question 963 for an example of this approach).
967. 9.40
This is a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is the ammonium ion (0.25 M), and the conjugate base, CB, is ammonia (0.35 M). The ammonium ion comes from ammonium chloride, which is a strong electrolyte.
Entering the given information in the Henderson-Hasselbalch equation gives you the following:
![]()
To convert the pOH to pH, use the relationship pH + pOH = pKw, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 4.60 = 9.40
You can also solve any buffer problem as a Ka or Kb problem (see Question 963 for an example of this approach).
968. 11.02
This is a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is the methylammonium ion (0.25 M), and the conjugate base, CB, is methylamine (0.50 M). The methylammonium ion comes from methylammonium chloride, which is a strong electrolyte.
![]()
To finish the problem, convert the pOH to pH using the relationship pH + pOH = pKw, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 2.98 = 11.02
You can also solve any buffer problem as a Ka or Kb problem (see Question 963 for an example of this approach).
969. 2.69
The problem doesn’t give you an equation, so the first step to write a balanced chemical equation:
![]()
Write the sodium fluoride as the separated ions, because the fluoride ion will be necessary for later calculations.
Adding the given amounts of material to the balanced chemical equation gives you the following:
![]()
Quantities of two reactants are present, so this is a limiting reactant problem. Because the stoichiometry is 1:1, the substance in the lesser amount is the limiting reactant.
The limiting reactant will go to zero and take an equal amount of the other reactant with it. This means 0.75 mol of HF will remain. The reactant will produce an equal number of moles of product. The product of interest is the fluoride ion, and 0.25 mol of this ion will form. Dividing each of the mole values by the volume of solution gives the molarities: The solution is 0.75 M HF and 0.25 M F–.
You now have a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is hydrofluoric acid (0.75 M), and the conjugate base, CB, is the fluoride ion (0.25 M). The fluoride ion comes from sodium fluoride, which is a strong electrolyte.
Entering the given information in the Henderson-Hasselbalch equation gives you the answer:
![]()
You can also solve any buffer problem as a Ka or Kb problem (see Question 963 for an example of this approach). This normally takes more time than using the Henderson-Hasselbalch equation and results in the same answer.
970. 9.55
The problem doesn’t give you an equation, so the first step in this problem is to write a balanced chemical equation:
![]()
Write the ammonium as separated ions, because the fluoride ion will be necessary for later calculations.
Adding the given amounts of material to the balanced chemical equation gives you the following:
![]()
Quantities of two reactants are present, so this is a limiting reactant problem. Because the stoichiometry is 1:1, the substance in the lesser amount is the limiting reactant.
The limiting reactant will go to zero and take an equal amount of the other reactant with it. This means 0.50 mol of NH3 will remain. The reactant will produce an equal number of moles of product. The product of interest is the ammonium ion, and 0.25 mol of this ion will form. Dividing each of the mole values by the volume of solution gives the molarities: The solution is 0.50 M NH3 and 0.25 M NH4+.
You now have a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is the ammonium ion (0.25 M), and the conjugate base, CB, is ammonia (0.50 M). The ammonium ion comes from ammonium chloride, which is a strong electrolyte.
Entering the given information in the Henderson-Hasselbalch equation gives you the following:
![]()
To convert the pOH to pH and finish the problem, use the relationship pH + pOH = pKw, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 4.45 = 9.55
You can also solve any buffer problem as a Ka or Kb problem (see Question 963 for an example of this approach).
971. 2.91
The initial pH is the pH before the addition of any base; therefore, only the acetic acid will affect the pH of the solution.
Acetic acid isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
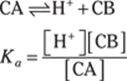
where CA refers to the conjugate acid and CB refers to the conjugate base. In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
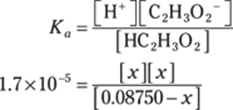
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to that of the concentration (in scientific notation). In this case, the exponent on the K is –5, and the exponent on the concentration is –2. If the exponent on the K is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. Therefore, you can rewrite the K equation as
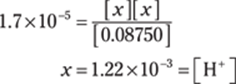
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, find the pH using the pH definition:
![]()
972. 11.11
The initial pH is the pH before the addition of any HCl; therefore, the only substance present that will affect the pH is the ammonia.
Ammonia isn’t one of the strong bases (NaOH, KOH, LiOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, and Ba(OH)2), so it must be a weak base. Weak base calculations need a base equilibrium constant, Kb. The generic form of every Kb problem is
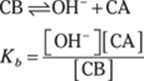
where CA refers to the conjugate acid and CB refers to the conjugate base. For balancing purposes, water may be present in the equilibrium chemical equation; however, water is also the solvent, so it shouldn’t be in the equilibrium expression.
The problem gives you the pKb, so convert the pKa to the Kb:
Kb = 10–4.75 = 1.8 × 10–5
In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Kb expression gives you the following:
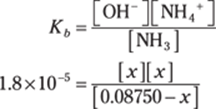
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –5, and the exponent on the concentration is –2. If the exponent on theK is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
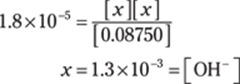
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Find the pOH using the pOH definition:
![]()
Finally, find the pH using the relationship pKw = pH + pOH, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 2.89 = 11.11
973. 7.00
Sodium hydroxide is one of the strong bases (NaOH, KOH, LiOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, and Ba(OH)2), and hydrochloric acid is one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4). No calculations are necessary for strong acid-strong base titrations, because the equivalence point is always at pH = 7.00.
974. 4.76
First, add the information from the problem to the balanced chemical equation:
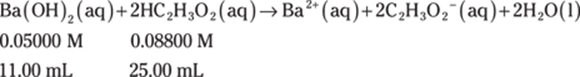
The problem gives you the quantities of two reactants, so you need to determine which reactant is the limiting reactant.
First, find the number of moles of each reactant. Change the molarity to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
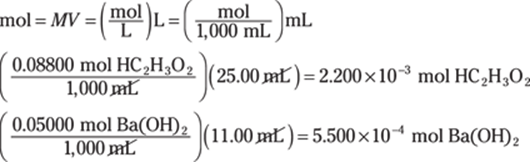
Due to the stoichiometry of the reaction, 5.500 × 10–4 mol of Ba(OH)2 will react completely with twice as many moles (1.100 × 10–3 mol) of HC2H3O2; because more HC2H3O2 is present, the Ba(OH)2 is the limiting reactant.
The amount of limiting reactant will decrease to zero, taking some of the excess reactant away and producing some of the product. The only product of importance is the conjugate base of the weak acid (HC2H3O2).
The moles of the nonlimiting reactant (conjugate acid) remaining after the reaction is the original number of moles minus the moles reacted:
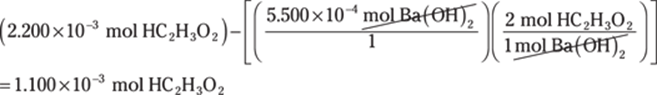
Next, use the moles of the limiting reactant to determine the moles of acetate ion (conjugate base) formed:
![]()
At this point, the volume of the solution is 25.00 mL + 11.00 mL = 36.00 mL = 0.03600 L.
Next, determine the concentrations of the conjugate acid and the conjugate base by dividing the moles of each substance by the volume of the solution in liters:
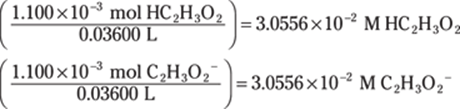
This is now a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is acetic acid (3.0556 × 10–2 M), and the conjugate base, CB, is the acetate ion (3.0556 × 10–2 M). The acetate ion comes from sodium acetate, which is a strong electrolyte.
Entering the given information in the Henderson-Hasselbalch equation gives you the final answer:
![]()
Note this is a special situation. Whenever the concentrations of the conjugate base and conjugate acid are the same, pH = pKa
You can also solve any buffer problem as a Ka or Kb problem. This normally takes more time than using the Henderson-Hasselbalch equation and results in the same answer.
Acetic acid isn’t one of the strong acids (HNO3, HCl, HBr, HI, HClO3, HClO4, and H2SO4), so it must be a weak acid. Weak acid calculations need an acid equilibrium constant, Ka. The generic form of every Ka problem is
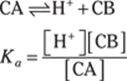
The problem gives you the pKa, so convert the pKa to a Ka:
![]()
In this case, the equilibrium equation and the change in the concentrations (calculated previously) are
![]()
Entering this information in the Ka expression gives you the following:
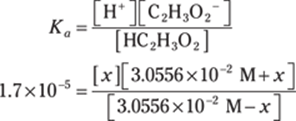
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the changes in the numerator and denominator are insignificant, as they are here, you can drop the +x and –x, simplifying the problem:
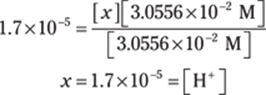
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, find the pH using the pH definition:
![]()
975. 9.24
First, add the information from the problem to the balanced chemical equation:
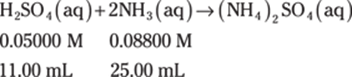
The problem gives you the quantities of two reactants, so you need to determine which reactant is the limiting reactant.
First, find the number of moles of each reactant. Change the molarities to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
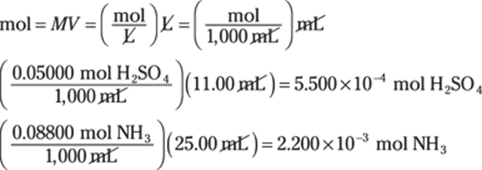
Due to the stoichiometry of the reaction, 5.500 × 10–4 mol of H2SO4 will react completely with twice as many moles (1.100 × 10–3 mol) of NH3; because more NH3 is present, H2SO4 is the limiting reactant.
The amount of limiting reactant will decrease to zero, taking some of the excess reactant away and producing some of the product. The only product of importance is the conjugate acid of the weak base (NH3).
The moles of nonlimiting reactant remaining (conjugate acid) after the reaction is the original number of moles minus the moles reacted:
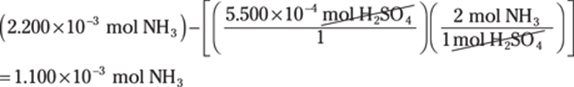
Next, use the moles of the limiting reactant to determine the moles of ammonium ion (conjugate acid) formed:
![]()
At this point, the volume of the solution is 25.00 mL + 11.00 mL = 36.00 mL = 0.03600 L.
Next, determine the concentrations of the conjugate acid and the conjugate base by dividing the moles of the substance by the volume of the solution in liters:
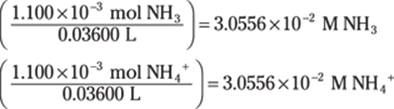
This is now a buffer solution problem, which is easiest to solve by using the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is the ammonium ion (3.0556 × 10–2 M), and the conjugate base, CB, is ammonia (3.0556 × 10–2 M).
Entering the given information in the Henderson-Hasselbalch equation gives you the pOH:
![]()
Finally, find the pH using the relationship, pKw = pH + pOH, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 4.76 = 9.24
You can also solve any buffer problem as a Ka or Kb problem (see Question 974 for an example of this approach). This normally takes more time than using the Henderson-Hasselbalch equation and results in the same answer.
976. 5.76
First, add the information from the problem to the balanced chemical equation:
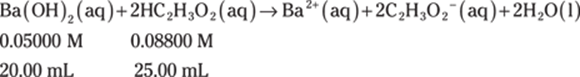
The problem gives you the quantities of two reactants, so you need to determine which one is the limiting reactant.
First, find the number of moles of each reactant. Change the molarities to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
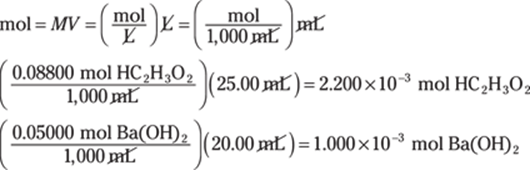
Due to the stoichiometry of the reaction, 1.000 × 10–3 mol Ba(OH)2 will react completely with twice as many moles (2.000 × 10–3 mol) of HC2H3O2; because more HC2H3O2 is present, the Ba(OH)2 is the limiting reactant.
The amount of limiting reactant will decrease to zero, taking some of the excess reactant away and producing some of the product. The only product of importance is the conjugate base of the weak acid (HC2H3O2).
The moles of nonlimiting reactant remaining (conjugate acid) after the reaction is the original number of moles minus the moles reacted:
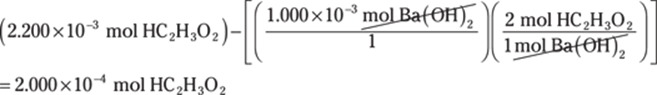
Next, use the moles of the limiting reactant to determine the moles of acetate ion (conjugate base) formed:
![]()
At this point, the volume of the solution is 25.00 mL + 20.00 mL = 45.00 mL = 0.04500 L.
Next, determine the concentrations of the conjugate acid and the conjugate base by dividing the moles of the substance by the volume of the solution in liters:
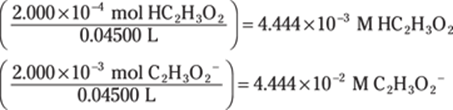
This is now a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is acetic acid (4.444 × 10–3 M), and the conjugate base, CB, is the acetate ion (4.444 × 10–3 M). The acetate ion comes from sodium acetate, which is a strong electrolyte.
Entering the given information in the Henderson-Hasselbalch equation gives you the final answer:
![]()
You can also solve any buffer problem as a Ka or Kb problem (see Question 974 for an example of this approach). This normally takes more time than using the Henderson-Hasselbalch equation and results in the same answer.
977. 8.72
First, add the information from the problem to the balanced chemical equation:
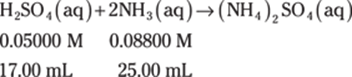
The problem gives you the quantities of two reactants, so you need to determine which one is the limiting reactant.
First, find the number of moles of each reactant. Change the molarities to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL:
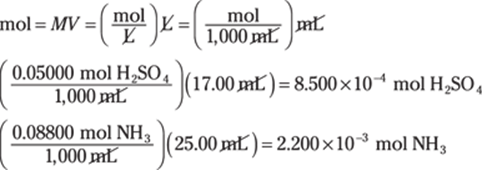
Due to the stoichiometry of the reaction, 8.500 × 10–4 mol of H2SO4 will react completely with twice as many moles (1.700 × 10–3 mol) of NH3; because more NH3 is present, the H2SO4 is the limiting reactant.
The amount of limiting reactant will decrease to zero, taking some of the excess reactant away and producing some of the product. The only product of importance is the conjugate acid of the weak base (NH3).
The moles of nonlimiting reactant remaining (conjugate acid) after the reaction is the original number of moles minus the moles reacted:
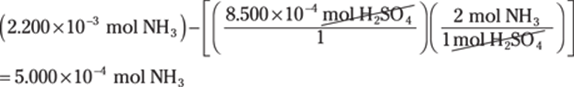
Next, use the moles of the limiting reactant to determine the moles of ammonium ion (conjugate acid) formed:
![]()
At this point, the volume of the solution is 25.00 mL + 17.00 mL = 42.00 mL = 0.04200 L.
Now determine the concentrations of the conjugate acid and the conjugate base by dividing the moles of the substance by the volume of the solution in liters:
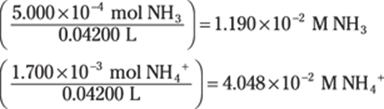
This is now a buffer solution problem, which is easiest to solve with the Henderson-Hasselbalch equation:
![]()
The conjugate acid, CA, is the ammonium ion (4.048 × 10–2 M), and the conjugate base, CB, is ammonia (1.190 × 10–2 M).
Entering the given information in the Henderson-Hasselbalch equation gives you the following:
![]()
Finally, get the pH by using the relationship pKw = pH + pOH, where pKw = 14.000:
pH = pKw – pOH = 14.000 – 5.28 = 8.72
You can also solve any buffer problem as a Ka or Kb problem (see Question 974 for an example of this approach). This normally takes more time than using the Henderson-Hasselbalch equation and results in the same answer.
978. 8.72
The problem doesn’t give you an equation, so the first step is to write a balanced chemical equation:
![]()
Then add the information from the problem to the balanced chemical equation:
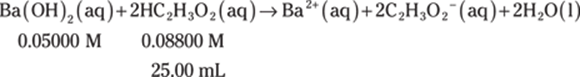
This reaction is at the equivalence point, so none of the reactants will remain. Therefore, the only thing present that might affect the pH is the conjugate base of the weak acid, which is the acetate ion.
You need to calculate the molarity of the acetate ion in the solution at the equivalence point, and to do this, you need to know the moles present and the total volume of the solution. But you have more information about the acetic acid, so begin calculations with the acetic acid.
First, change the molarity of the acetic acid to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL.
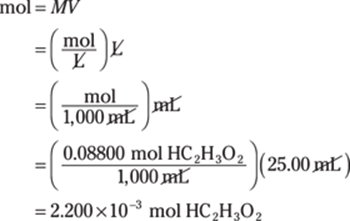
From the moles of acetic acid, you can determine both the number of moles of acetate ion formed and the volume of barium hydroxide solution necessary to do this.
The moles of acetate ion (the conjugate base) formed comes from multiplying the moles of acetic acid by the mole ratio from the balanced chemical equation:
![]()
Find the volume of barium hydroxide solution added using the moles of acetic acid, the mole ratio from the balanced chemical equation, and the molarity of the barium hydroxide solution:
![]()
At this point, the volume of the solution is 25.00 mL + 22.00 mL = 47.00 mL = 0.04700 L.
Next, determine the concentration of the conjugate base by dividing the moles of the substance by the volume of the solution in liters:
![]()
Next is the equilibrium part of the problem. The acetate ion is the conjugate base of a weak acid, so it will be part of a Kb equilibrium. The generic form of every Kb problem is
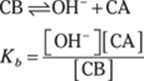
where CA refers to the conjugate acid and CB refers to the conjugate base. For balancing purposes, water may be present in the equilibrium chemical equation; however, water is also the solvent, so it shouldn’t be in the equilibrium expression.
You need to convert the pKa given in the problem to a Kb. This is a two-part conversion. First find pKb using the relationship pKw = pKa + pKb, where pKw = 14.000:
pKb = pKw – pKa = 14.000 – 4.76 = 9.24
Then change pKb to Kb:
![]()
In this problem, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Kb expression gives you the following:
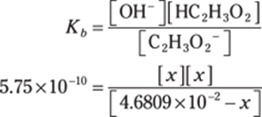
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to the exponent on the concentration (in scientific notation). In this case, the exponent on the K is –10, and the exponent on the concentration is –2. If the exponent on the K is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
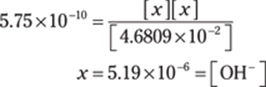
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Find the pOH using the pOH definition:
![]()
Finally, find the pH using the relationship pKw = pH + pOH:
pH = pKw – pOH = 14.000 – 5.28 = 8.72
979. 9.19
The problem doesn’t give you an equation, so the first step is to write a balanced chemical equation:
![]()
Then add the information from the problem to the balanced chemical equation:
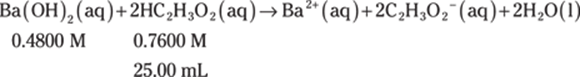
This reaction is at the equivalence point, so none of the reactants will remain. Therefore, the only thing present that might affect the pH is the acetate ion, the conjugate base of the weak acid. You need to calculate the molarity of the acetate ion in the solution at the equivalence point, which requires knowing the moles present and the total volume of the solution. The problem gives you more information about the acetic acid, so begin your calculations with acetic acid.
Change the molarity of the acetic acid to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit M in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL.
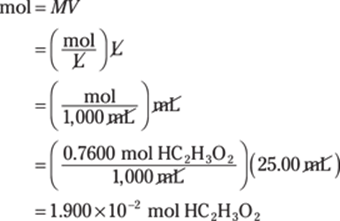
From the moles of acetic acid, you can determine both the number of moles of acetate ions formed and the volume of barium hydroxide solution.
The moles of acetate ion (conjugate base) formed comes from multiplying the moles of acetic acid by the mole ratio from the balanced chemical equation:
![]()
Find the volume of barium hydroxide solution added using the moles of acetic acid, the mole ratio from the balanced chemical equation, and the molarity of the barium hydroxide solution:
![]()
At this point, the volume of the solution is 25.00 mL + 19.79 mL = 44.79 mL = 0.04479 L.
Now determine the concentration of the conjugate base by dividing the moles of the substance by the volume of the solution in liters:
![]()
Next is the equilibrium part of the problem. The acetate ion is the conjugate base of a weak acid, so it will be part of a Kb equilibrium. The generic form of every Kb problem is
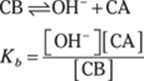
where CA refers to the conjugate acid and CB refers to the conjugate base. For balancing purposes, water may be present in the equilibrium chemical equation; however, water is also the solvent, so it shouldn’t be in the equilibrium expression.
You need to convert the pKa given in the problem to a Kb. This is a two-part conversion. First find pKb using the relationship pKw = pKa + pKb, where pKw = 14.000:
pKb = pKw – pKa = 14.000 – 4.76 = 9.24
Then change pKb to Kb:
![]()
In this problem, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
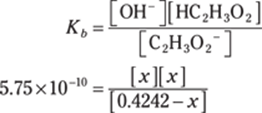
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to that of the concentration (in scientific notation). In this case, the exponent on the K is –10, and the exponent on the concentration is –1. If the exponent on the K is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant. That means you can rewrite the K equation as
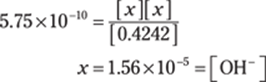
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Find the pOH using the pOH definition:
![]()
Finally, find the pH using the relationship pKw = pH + pOH:
pH = pKw – pOH = 14.000 – 4.81 = 9.19
980. 5.26
The problem doesn’t give you an equation, so the first step is to write a balanced chemical equation:
![]()
Then add the information from the problem to the balanced chemical equation:
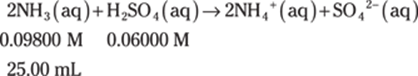
This reaction is at the equivalence point, so none of the reactants will remain. Therefore, the only thing present that might affect the pH is the ammonium ion, the conjugate acid of the weak base.
You need to calculate the molarity of the ammonium ion in the solution at the equivalence point, which requires knowing the moles present and the total volume of the solution. The problem gives you more information about the ammonia, so begin calculations with ammonia.
Change the molarity of the ammonia to moles by multiplying the molarity by the volume. The conversion is easier to see if you write the molarity unit in terms of its definition (mol/L). The given volume is in milliliters, so remember that a liter is 1,000 mL.
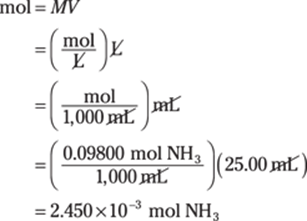
From the moles of ammonia, you can determine both the number of moles of ammonium ion formed and the volume of the sulfuric acid solution.
The moles of ammonium ion (conjugate acid) formed comes from multiplying the moles of ammonia by the mole ratio from the balanced chemical equation:
![]()
Find the volume of barium hydroxide solution added using the moles of ammonia, the mole ratio from the balanced chemical equation, and the molarity of the sulfuric acid solution:
![]()
At this point, the volume of the solution is 25.00 mL + 20.42 mL = 45.42 mL = 0.04542 L.
Now determine the concentration of the conjugate base by dividing the moles of the substance by the volume of the solution in liters:
![]()
Next is the equilibrium part of the problem. The ammonium ion is the conjugate acid of a weak base, so it will be part of a Ka equilibrium. The generic form of every Ka problem is
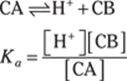
where CA refers to the conjugate acid and CB refers to the conjugate base.
You need to convert the pKa given in the problem to a Kb. This is a two-part conversion. First find pKb using the relationship pKw = pKa + pKb, where pKw = 14.000:
pKb = pKw – pKa = 14.000 – 4.75 = 9.25
Then change pKb to Kb:
![]()
In this case, the equilibrium equation and the change in the concentrations are
![]()
Entering this information in the Ka expression gives you the following:
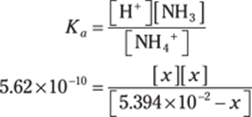
This is a quadratic equation, and you can solve it as such. But before doing the math, think about the problem logically. If the change in the denominator is insignificant, you can drop the –x and make the problem easier to solve.
A simple check is to compare the exponent on the K to that of the concentration (in scientific notation). In this case, the exponent on the K is –10, and the exponent on the concentration is –2. If the exponent on the K is at least 3 less than the exponent on the concentration (as it is in this case), you can assume that –x in the denominator is insignificant, simplifying the K equation to
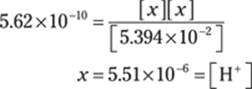
The x value is sufficiently small that 1.0 – x ≈ 1.0, which validates the assumption.
Finally, find the pH using the pH definition:
![]()
981. origin
The coordinates of the origin are (0, 0).
982. interpolation
Finding a point that falls between the minimum and maximum values plotted on a line or curve is interpolation; you’re finding a value inside your data.
Interpolation is an important method for estimating values based on data trends from the experiment. You can find an approximate value for a point that falls between the tested data points without doing an additional experiment, which can save a significant amount of time.
983. extrapolation
Estimating a value that falls below the minimum values or above the maximum values but follows the trend shown in the graph is called extrapolation.
Extrapolation allows you to make an educated guess about a result of a future experiment. The method assumes that the trend observed in the given values will continue beyond this range, which isn’t always a valid assumption. One important aspect of science is that a theory should be able to predict future events (extrapolation).
984. the origin
The line crosses the x-axis when the volume equals zero. This graph should pass through the origin because zero volume corresponds to zero mass.
985. absolute zero
This graph is a plot of Charles’s gas law. The x-intercept represents the point where the volume of an ideal gas would be zero, which occurs when the temperature is absolute zero.
986. speed
The slope is rise over run: the change in y-values (distance) divided by the change in x-values (time). Therefore, the line has units of distance/time, such as miles per hour, which correspond to units of speed.
987. density
The slope is rise over run: the change in y-values (mass) divided by the change in x-values (volume). Therefore, the line has units of mass/volume, such as grams per cubic centimeter, which correspond to units of density.Density is the mass of something divided by its volume.
988. heat capacity
The slope is rise over run: the change in y-values (heat energy) divided by the change in x-values (temperature). Therefore, the line has units of energy/temperature, such as joules per degree Celsius. This unit corresponds to heat capacity, which is the amount of energy necessary to increase the temperature of a substance by 1°C.
989. straight line with a positive slope
A directly proportional relationship produces a straight line with a positive slope. You can write the relationship from Charles’s law as V ∝ T, where ∝ means proportional. With this relationship, as the gas’s temperature increases, the volume increases by the same factor. For example, if you double the temperature, the volume doubles as well, and the volume triples if you triple the temperature.
This law assumes that the pressure and the amount of gas are constant.
990. a line curving downward
A graph showing an inversely proportional relationship is a smooth curve that slopes downward. You can write the relationship from Boyle’s law as ![]() , or PV = constant. With this relationship, if the pressure doubles, the volume is halved, and if you triple the pressure, you get a third of the volume.
, or PV = constant. With this relationship, if the pressure doubles, the volume is halved, and if you triple the pressure, you get a third of the volume.
This law assumes that the temperature and the amount of gas are constant.
991. a line curving upward
The volume is related to the edge of a cube (s). The relationship is V = s3. Cubic relationships give curved lines. The graph curves upward because a larger cube has more volume.
992. 27 pennies
After plotting the data, draw the line of best fit. For a linear set of data, the line of best fit is a straight line that splits the difference between the data points. Points will be equally distributed on each side of this line. In science, graphs very rarely connect the dots.
The following graph shows the plotted data points and the best-fit line. Your graph should have individual points above and below this line.
To answer this question, go up to about 84.1 on the y-axis and then go right until you hit the line of best fit. Next, go down to the x-axis and read the value.
Note: The smallest division on your x-axis could be as small as 0.1 penny, but for practicality, it will probably be 1 penny or larger to avoid cutting a penny into pieces.
993. the average mass of one penny
To find the slope of a line, you choose two points on the line and divide the difference in the y-coordinates by the difference in the x-coordinates. The y-values represent mass, and the x-values represent the number of pennies:
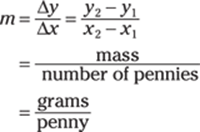
The units (grams/penny) indicate that the slope represents the average mass of one penny.
994. 4.6 mL
After plotting the data, draw the line of best fit. For a linear set of data, the line of best fit is a straight line that splits the difference between the data points. Points will be equally distributed on each side of this line. In science, graphs very rarely connect the dots.
The following graph shows the plotted data points and the best-fit line. Your graph should have individual points above and below this line. The smallest division on your x-axis could be anything as small as 0.1 mL, but for practicality, it’s more likely to be 0.2 mL or 0.5 mL.
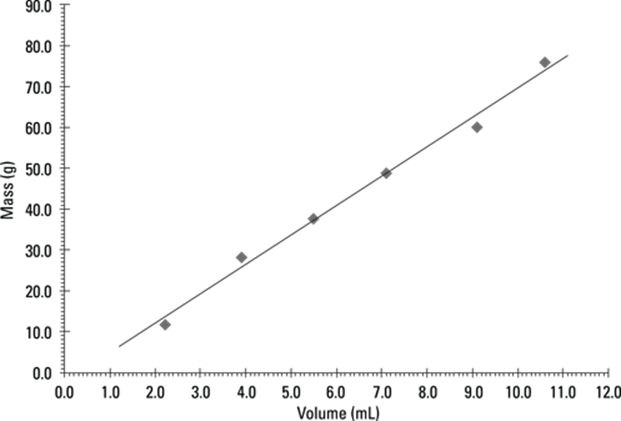
To answer this question, go up to about 31.0 g on the y-axis and then go right until you hit the line of best fit. Next, go down to the x-axis and read the value.
995. 70.1 g
After plotting the data, draw the line of best fit. For a linear set of data, the line of best fit is a straight line that splits the difference between the data points. Points will be equally distributed on each side of this line. In science, graphs very rarely connect the dots.
The following graph shows the plotted data points and the best-fit line. Your graph should have individual points above and below this line. The smallest division on your y-axis could be anything as small as 0.01 g, but for practicality, it’s more likely to be 0.1 g, 0.5 g, or 1 g.

To answer this question, go over to about 10.0 mL on the x-axis and then go up until you hit the line of best fit. Next, go over to the y-axis and read the value.
996. 0.083 M
After plotting the data, draw the line of best fit. For a linear set of data, the line of best fit is a straight line that splits the difference between the data points. Points will be equally distributed on each side of this line. In science, graphs very rarely connect the dots.
The following graph shows the plotted data points and the best-fit line. Your graph should have individual points above and below this line. The smallest division on your x-axis could be anything as small as 0.001 M, but for practicality, it’s more likely to be 0.0025 M or 0.0050 M.
To answer this question, go up to about 0.318 on the y-axis and then go right until you hit the line of best fit. Next, go down to the x-axis and read the value.
This graph illustrates Beer’s law. The absorbance is directly proportional to the concentration.
997. 0.479
After plotting the data, draw the line of best fit. For a linear set of data, the line of best fit is a straight line that splits the difference between the data points. Points will be equally distributed on each side of this line. In science, graphs very rarely connect the dots.
The following graph shows the plotted data points and the best-fit line. Your graph should have individual points above and below this line. The smallest division on your y-axis could be anything as small as 0.001, but for practicality, it’s more likely to be 0.025, 0.05, or 0.1. Absorbance doesn’t have a unit.

To answer this question, go over to 0.125 M on the x-axis (this is an extrapolated value, beyond the data points from the experiment); then go up until you hit the line of best fit. Next, go over to the y-axis and read the value.
This graph illustrates Beer’s law. The absorbance is directly proportional to the concentration.
998. –271.5°C
After plotting the data, draw the line of best fit. For a linear set of data, the line of best fit is a straight line that splits the difference between the data points. Points will be equally distributed on each side of this line. In science, graphs are very rarely connect the dots.
The following graph shows the best-fit line. Your graph should have individual points above and below this line. The smallest division on your x-axis could be anything as small as 0.1°C, but for practicality, it’s more likely to be 10°C or 25°C (although numbers this big will make it difficult to estimate the value).

To answer this question, go over on the line of best fit until it intersects with the x-axis and read the value.
This graph illustrates Charles’s law and the method for determining absolute zero, the lowest possible temperature. Data from other experiments have shown that the actual value of absolute zero, which is 0 kelvins on the Kelvin scale, is –273.15°C.
999. the line has a gradual positive slope, shoots up, and then levels out
This graph is shaped like an s-curve that has had the top and bottom pulled so that the middle of the s is almost a vertical line:
The graph shows data from an acid-base titration in which the base is the titrant. In an acid-base titration, the pH of the solution is plotted versus the volume of a substance added. The center of the sharp rise is the equivalence point of the titration.
1,000. 4.64
Plot the data and draw the line of best fit, which is a smooth curve. The smallest division on your y-axis could be anything as small as 0.01, but for practicality, it’s more likely to be 1.00. Note that pH has no unit.

To answer this question, go over to 12.25 mL on the x-axis and then go up until you hit the line of best fit. Next, go over to the y-axis and read the value.
The pH at 12.25 mL is halfway to the equivalence point. At the equivalence point, the pH is equal to the pKa of the acid. The pKa is equal to –log Ka, where Ka is the equilibrium constant for the ionization of a weak acid. If you have a list of possible acids, the Ka value can help you identify an unknown acid used in the titration.
1,001. ![]() versus time
versus time
Graphing 1/[A] versus time should give you a line that looks like the one in the following graph. [A] stands for the molar concentration of one of the reactants, so the units for 1/[A] are M–1, or 1/M. Sketching graphs (or graphing the data with a program such as Excel) quickly shows that the other combinations aren’t linear.
A second-order kinetics reaction always gives you a positive linear plot of 1/[A] versus time. Identifying this relationship is a useful way to show that a reaction follows second-order kinetics. The slope of the line is related to the rate constant for the reaction. A first-order reaction would give you a negative linear plot for ln[A] versus time, and a zero-order reaction would give you a negative linear plot for [A] versus time.