Cracking the SAT Chemistry Subject Test
Part I
Orientation
Chapter 2
Test Strategies
The SAT Chemistry Subject Test always examines the same areas of chemistry using three different question types. Your job is to know what to expect from the test, not only in terms of specific content, but also in terms of how the test is written and how you can use that information to your advantage. In this chapter we will discuss the breakdown of the test and general strategies you can use.
CRACKING THE SAT CHEMISTRY TEST
The SAT Chemistry Subject Test is made up of 85 multiple-choice questions, and you have one hour to answer them. You're not allowed to use a calculator on this test, but you won't need one. The test is divided into three sections: Parts A, B, and C, and each section is made up of a different type of question. Let's take a closer look at these parts.
Part A: Classification Questions
The first 20 to 25 questions you'll see on the exam fall under the category of what ETS calls classification questions. In this type of question, you'll see a list of five words or phrases lettered A through E, followed by three to five questions. But sometimes the questions aren't really questions; they're phrases. Your job is to match the phrase in the “question” with a word or phrase that appears in the list A through E. Forget about chemistry for a minute, and see how it works.
Directions: Each set of lettered choices below refers to the numbered statements or questions immediately following it. Select the one lettered choice that best fits each statement or answers each question and then fill in the corresponding oval on the answer sheet. A choice may be used once, more than once, or not at all in each set.
Questions 1–4 refer to the following.
(A) Red light
(B) Swimming pool
(C) Piano
(D) Fire engine
(E) Ocean liner
1. Musical instrument that involves keyboard outside and strings inside
2. Motor vehicle designed to assist in effort to extinguish flames
3. Sea vessel that carries passengers across large bodies of water
4. Water-filled pit designed for recreational or athletic activities
The answers, of course, are C, D, E, and B. Now let's move on to see what the questions in Part B look like.
Part B: Relationship Analysis Questions
The questions that make up Part B of the exam won't ask you to decide among choices A, B, C, D, or E. Relationship analysis questions consist of two statements with the word BECAUSE in between them. You're supposed to figure out if the statements are true or false. If both are true, you're also expected to figure out whether the word BECAUSE belongs there. Once again, forget about chemistry for a second so we can show you how the questions work. The questions in part B of the exam are numbered in a peculiar manner. This section begins with the number 101, though there are only 85 questions in the whole test!
CE?
On your answer sheet
for Part B, you'll see
ovals marked T and F for
true and false, but the
“because” circle is marked
CE, which stands for
Cause / Effect. You're
looking to see if I and II
have a cause-and-effect
relationship.
Directions: Each question below consists of two statements, I in the left-hand column and II in the right-hand column. For each question, determine whether statement I is true or false and whether statement II is true or false and fill in the corresponding T or F ovals on your answer sheet. Fill in oval CE only if statement II is a correct explanation of statement I.
![]()
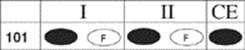
101.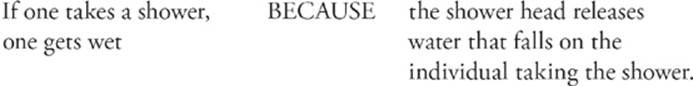
102.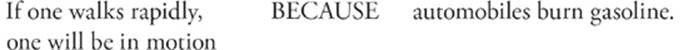
103.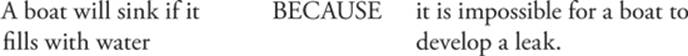
104.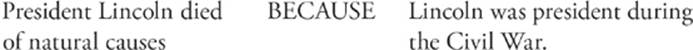
105.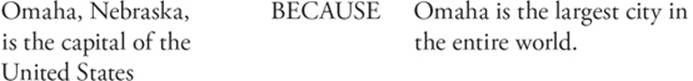
Question 101 Both statements are true, and the “because” belongs there. You get wet in a shower because the shower pours water on you.
Question 102 Both statements are true. If you walk, you move, and automobiles do burn gasoline. But the “because” doesn't belong there. A walker doesn't move because automobiles burn gasoline. The statements have nothing to do with one another.

Question 103 A boat will sink if it fills with water. That's true. But the second statement is false. Boats can develop leaks.

Question 104 The first statement is false. Lincoln was murdered. The second statement is true. He was president during the Civil War.

Question 105 Both statements are false. Omaha is not the capittal of the United States, and it isn't the largest city in the world.

Now let's talk about the third and final section of the exam, Part C.
Part C: Five-Choice Completion Questions
The question type that makes up the majority of the test (40–50 questions) is ordinary looking multiple-choice questions such as the one below.
28. Which is the formula of a compound?
(A) HCl
(B) He
(C) Cu
(D) O2
(E) Br2
Here, the answer is A. A compound is a chemical combination of two or more elements. (We'll talk more about that later.) This type of question is straightforward: You read the question and choose the answer choice that best answers the question.
Now that you know what kinds of questions you'll see on the SAT Chemistry Subject Test, let's talk about the strategies you can use to tackle these questions.
STRATEGY #1: STUDY THE RIGHT STUFF IN THE RIGHT WAY
One important strategy for preparing to take this exam is to study only the concepts that will be tested. In Chapters 3 through 14, we will take a look at all the subjects that are certain to appear on the test and explain them in a way that's specifically designed to help you answer the test questions.
What topics do we cover? Well, the same topics that the College Board lists on their website as being covered.
I. Structure of Matter (25% of the questions will be on this topic): atomic theory and structure; periodic relationships; chemical bonding and molecular structure; nuclear reactions
II. States of Matter (15%): kinetic molecular theory of gases; gas laws; liquids, solids, and phase changes; solutions, concentration units, solubility, conductivity, and colligative properties
III. Reaction Types (14%): acids and bases; oxidation-reduction; precipitation
IV. Stoichiometry (12%): mole concept and Avogadro's number; empirical formula and molecular formulas; percent composition, stoichiometric formulas; limiting reagents
V. Equilibrium and Reaction Rates (7%): gas equilibria and ionic equilibria; Le Chatelier's principle; equilibrium expressions; rate of reaction
VI. Thermodynamics (6%): energy changes in chemical reactions and physical properties; Hess's law; randomness
VII. Descriptive Chemistry (13%): physical and chemical properties of elements and their familiar compounds; chemical reactivity and products of chemical reactions; simple examples from organic chemistry and environmental chemistry
VIII. Laboratory (8%): equipment; measurements; procedures; observations; safety; calculations; interpretations of results
As you can see, we do not include everything there is to know about chemistry; your chemistry textbook does that. We just hope to strengthen and refresh your knowledge in the specific areas that will be important on the test.
STRATEGY #2: DO THE EASY ONES FIRST
In each of the three sections of the SAT Chemistry Subject Test, the easier questions tend to come first and the harder ones come later. When you begin each section, answer as many of the “easy” questions as you can, but when they start to become more difficult, go on to the next section and do the same. Once you've answered all of the relatively easy questions in all the sections, go back to each section and start answering the more difficult ones.
This strategy makes sense because all questions are worth the same amount; answering a hard question correctly won't get you more points than answering an easy one correctly. If there's a chance that you might not be able to get to every question on the exam in the 60 minutes you're given, make sure you at least answer the ones you're sure to get right, first!
You don't need to answer every question to get a good score on the SAT Chemistry. It's possible to leave 30 questions blank and still score near 600 if you do well on the questions you do answer.
STRATEGY #3: TAKE A GUESS!
As we told you in the last chapter, in calculating your “raw score” (from which it then calculates your scaled score), ETS does the following:
1. Gives you one point for each question you answer correctly
2. Deducts ![]() of a point for each question you answer incorrectly
of a point for each question you answer incorrectly
3. Doesn't count questions you didn't answer
Because ETS deducts one-quarter of a point for any question you answer incorrectly, you should definitely guess the answer to any question for which you can eliminate at least one of the five answer choices. If you can eliminate one answer choice and then take a guess, then you will have a one-in-four chance of choosing the correct answer. If you can eliminate two answer choices, your odds of choosing correctly go to one-in-three.
As you read Chapters 3 through 14, you'll see that all of our techniques and strategies teach you to eliminate wrong choices. After you've done that, use guessing to your advantage.
STRATEGY #4: MAKING ASSOCIATIONS (TYPE A, B, AND C QUESTIONS)
One helpful strategy for learning the key chemistry concepts that will show up on the test is to make associations between terms and concepts. What are we talking about? Well, let's forget chemistry, just to make the point. You may have learned in school that Teddy Roosevelt was a “trustbuster.” You might not know what trusts are, how he busted them, why he wanted to bust them, or why anyone cares if trusts get busted. But if you learned to associate the name Teddy Roosevelt with the phrase “trustbuster,” you would be able to answer a test question that looks like this.
3. Theodore Roosevelt believed in
(A) creating trusts
(B) destroying trusts
(C) making trusts larger
(D) communism
(E) socialistic economics
The association you learned to make—;“Teddy Roosevelt” with “trustbuster”—;allowed you to choose the correct answer: B.
This strategy will also be useful on the SAT Chemistry Test; many questions will test your ability to associate one word or phrase with another. For example, suppose you had no idea what was meant by “pH,” “acid,” or “base,” and you had just learned to associate
pH less than 7 with: acid
pH greater than 7 with: base
You'd be able to answer a test question that looked like this:
28. Which of the following solutions is most acidic?
(A) A solution of phosphoric acid at pH 4
(B) A solution of sodium hydroxide at pH 11
(C) A solution of hydrochloric acid at pH 5
(D) A solution of acetic acid at pH 6
(E) A solution of aqueous ammonia at pH 9
Whatever else you know about acids and bases, you know that choice A is right because, among the listed solutions, it has the lowest pH.
Throughout our teaching of SAT Chemistry, we will show you what associations to make, how to make them, and how they will point you to the right answers on test day.
STRATEGY #5: REPHRASE THE QUESTION (TYPE A AND C QUESTIONS)
In the questions seen in Parts A and C on the exam, the test writers will obscure the information in the question to make it harder to read. They are trying to mess you up! Don't fall for it.
To understand this type of trap, read the two sentences below.
1. In any dynamic chemical equilibrium, the removal of product will drive the equilibrium to the right and thus increase the concentration of product, while the concentration of reactants will decrease.
2. If a dynamic chemical equilibrium is subjected to withdrawal of product, the concentration of reactants will diminish, and the concentration of product will become greater.
These two statements mean exactly the same thing, but their wording is very different. Many of the words and phrases in statement 2 have the same meaning as those in statement 1, but they're disguised—;they're camouflaged.
|
subjected to withdrawal of product |
means |
the removal of product |
|
concentration of product will become greater |
means |
increase the concentration of product |
|
concentration of reactants will diminish |
means |
the concentration of reactants will decrease |
So, What About It?
When you learn something, whether it's chemistry or anything else, you tend to learn it in certain particular phrases. For instance, maybe you think of an element as “a substance that cannot be broken into any simpler substance.” Fine. But if you're too attached to that particular way of stating it, look what happens when you try to answer the following question.
25. Which of the following best describes the characteristics of an element?
(A) It is capable of existing in relatively simple molecular forms.
(B) It exists only in molar quantities.
(C) It will always react with any other element.
(D) It is a fundamental form of matter.
(E) It is more reactive if the surrounding entropy is high.
If you're too attached to the way that you usually describe elements, you might not see the right answer although (a) you do know it, and (b) it's staring you in the face. The right answer to this question is D. To say that an element is a fundamental form of matter is, more or less, to say it can't be broken down into simpler substances. The words aren't the same, but the meaning is.
Many students who know what an element is might still not answer this question correctly. This is because they'll look quickly through the choices and not see anything they recognize—;this throws them into “answer-choice panic,” and they'll pick something that “sounds right”—;something that has the word “simple” in it, such as choice A.
That's too bad. Students who do know the content sometimes choose the wrong answer simply because they fall for the camouflage trap.
Here's another example from a type C question. Suppose you know that if you add heat to a sample of gas molecules, each molecule, on average, starts bouncing around faster than it did before. But you're accustomed to stating it this way:
The average kinetic energy of gas molecules is directly proportional to the absolute temperature of the gas.
If you're married to that statement, what's going to happen when you see the question below?
26. Which of the following is always increased by the addition of thermal energy to a sample of gas in a closed container?
(A) Ideal gas constant
(B) Average speed of gas molecules
(C) Molecular weight of gas molecules
(D) Volume of gas sample
(E) Volatility
You know that increased heat increases average kinetic energy, but the answer has been camouflaged.
|
average speed of gas |
means |
average kinetic energy of molecules |
|
addition of thermal energy |
means |
increasing the temperature |
Maybe you're not used to thinking of the “addition of thermal energy” resulting in an increase in temperature. When it comes to gases, you're also not accustomed to thinking of “average speed of gas molecules” as reflecting the average kinetic energy. Although you know your chemistry, you might not realize that the right answer to this question is B. You might decide to pick some crazy answer such as A or D. Why? Because you fell into the trap.
Here's the Good News: You Can Rephrase It Yourself
To avoid the trap, keep some simple rules in mind.
• Remember that there's usually more than one way to say something.
• When you see a question that asks about a topic you've studied, don't fall apart just because the answer doesn't leap out at you right away.
• Relax. Realize that the right answer is probably camouflaged by words that are different from the ones you have in mind. Calmly search for them, and chances are, they will leap out at you.
In other words, keep an open mind. Don't expect test makers to use your words. Remember: The same concept or idea can be expressed in many different ways. Keep the concepts you know in mind, and don't get too attached to the words you use to express them.
Another Way out of the Trap: Translate and Work Backward
Suppose you do try to keep an open mind on a particular question, and it just doesn't seem to work; the right answer isn't coming to you, although you know your chemistry. For questions in Part A, here's what you can do: Look through all of the answer choices, and restate them in your own words. Below, we've listed five possible answer choices you might see in Part A of the exam. Below each answer choice, we've included one way of restating the answer.
(A) ideal gas constant
It's the letter R in the equation PV = nRT. It equals about 0.082 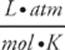 .
.
(B) average speed of gas molecules
It's the speed at which gas molecules are moving around in a tank or container—;it has to do with how much energy they have. It goes up with higher temperature and down with lower temperature.
(C) molecular weight of gas molecules
It's the weight (expressed in amu) of a gas molecule.
(D) volume of gas sample
It's the space the gas sample takes up—;equal to the size of the container.
(E) volatility
It has to do with how easily a liquid below its boiling point evaporates when it's sitting around.
Now look at each of the following questions:
Questions 1–4 refer to the following.
1. Is always increased by the addition of thermal energy to a sample of gas in a closed container
2. Can be related to the pressure of a gas sample by the ideal gas law
3. Property associated with vapor pressure
4. Depends on the formula of a gas but not its temperature
Read the questions carefully, one by one, and compare them to the answer choices that you've put into your own words.
1. Hopefully you realize that “thermal energy” means heat. Now, which of the answer choices (stated in your own words) has to do with a factor that's affected by the addition of heat? (B) does—;it says that the average speed of gas molecules increases as heat (thermal energy) is added to the system and decreases as heat is taken away from a system. The answer to question 1 is (B).
2. If you know the ideal gas law (covered in Chapter 8), you would simply look among the answer choices for one of the variables from the equation, PV = nRT. In this equation, P = pressure, V = volume, n = number of moles of gas, R = the ideal gas constant, and T = temperature. Volume is the variable listed among the answer choices, so the correct answer is (D).
3. You should associate vapor pressure with the degree to which a liquid will evaporate at a temperature that's below its normal boiling point—;and lo and behold, this is similar to how you've paraphrased choice (E): volatility. The answer is (E).
4. Read the question, and then look at the answer choices. Which of the choices, when stated in your words, mentions a characteristic of a gas that depends on its formula but not its temperature? Well, choice (C) looks like the most likely—;the weight of a gas molecule does depend on its identity (formula) but not on its temperature. None of the other answer choices make any sense, so choose (C).
STRATEGY #6: AVOIDING THE TEMPTATION TRAP
Suppose we gave this question to a seven-year-old:
27. Which of the following best expresses the effect of Gibbs free energy and the spontaneity of a chemical reaction?
(A) When Gibbs free energy is negative, the reaction proceeds spontaneously in the forward direction.
(B) When Gibbs free energy is negative, the reaction proceeds spontaneously in the reverse direction.
(C) George Washington was the first president of the United States.
(D) Gibbs free energy affects the spontaneity only of exothermic reactions.
(E) Gibbs free energy affects the spontaneity only of endothermic reactions.
The child won't know what any of this means, but she will probably know that George Washington was the first president. So she'll choose C; it's something she knows. She fell into the “temptation trap.” The test writer stuck something into the answer choices that was familiar to the student; it was so familiar that the student chose it although it has nothing to do with the question.
Truth Isn't Always Right
Although answer choice
(C) is a true statement, it
doesn't answer the question
that's being asked.
That's how ETS traps you.
Make sure you're always
answering the question
posed on the test.
What's That Got to Do with Me and the SAT Chemistry Test?
A lot. The temptation trap usually rears its head on questions from Parts A and C of the exam. On the day you take the test, there will be many things you know and some that you don't know. When you meet up with a type C question that's stumping you, you might reach out and grab an answer choice that “sounds familiar” although it has nothing to do with the question.
Suppose you know that adding an acid to a base increases the hydrogen ion concentration of the solution. Now, look at this question.
26. Which of the following will definitely occur if a quantity of acetic acid is added to a solution of potassium hydroxide at pH 11?
(A) The number of free protons per liter of solution will increase.
(B) Titration will tend to neutralize the solution.
(C) The acetic acid will act as a weak base.
(D) Acetate ion will precipitate out of solution.
(E) The pH will remain constant.
The correct answer is A, but if the answer to this question doesn't leap right out at you, you might decide to make a dash for something you know. Choice B, by itself, is a true statement with which you might be familiar; titration between an acid and a base does tend to neutralize a solution. You might say to yourself, quickly, quietly, and almost unconsciously: “I've heard that statement. It sounds right.” But B is wrong because it doesn't answer the question.
You Can Avoid the Temptation Trap
When you find yourself ready to choose an answer because it sounds right, stop to look at the question again carefully. Then take another look at the answer choices to see if another of them, although in camouflage, is really a better answer to the question.
Let's think about the question we just looked at. We're adding an acid to a base. We know that we'll be lowering the pH of the solution—;increasing the hydrogen ion concentration. Choice A says exactly that—;in camouflage. Instead of referring to hydrogen ions, it refers to free protons. Instead of referring directly to concentration, or pH, it talks about increasing the number of protons per liter of solution. The right answer is A, and you knew it, but you might not have chosen it. Why? Because panic led you straight into the temptation trap. Don't let that happen!
How Would You Say It?
Put the answers in your
own words to avoid the
camouflage trap.
STRATEGY #7: DIVIDE AND CONQUER
Let's take another look at the instructions to Part B.
Directions: Each question below consists of two statements, I in the left-hand column and II in the right-hand column. For each question, determine whether statement I is true or false and whether statement II is true or false and fill in the corresponding T or F ovals on your answer sheet. Fill in oval CE only if statement II is a correct explanation of statement I.
Now let's look at a question that has nothing to do with chemistry, to show how the divide and conquer strategy works.
![]()
101.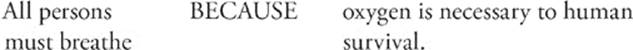
Here's what to do.
Step 1: Look at the first statement by itself and decide whether it's true or false. It's true. That means we fill in the oval marked T.
Step 2: Look at the second statement by itself. Is it true or false? It's true. That means we fill in the second oval marked T.
Step 3: Put the statements together, join them with the “because,” and then decide if the sentence makes sense. “All persons must breathe because oxygen is necessary to human survival.”
Cover Up!
When dealing with Part B
questions, it often helps
to cover up the statement
you're not looking at so
that it doesn't influence
your thinking.
Does it make sense? Yes. So you would fill in the oval marked CE.

Try this question.
![]()
102.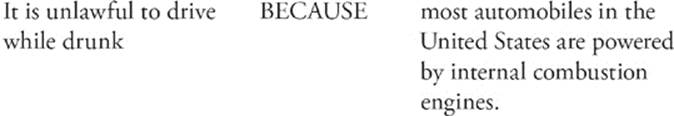
Step 1: Look at statement I by itself. Is it true or false? It's true. We fill in the first oval marked T.
Step 2: Look at statement II by itself. Is it true or false? It's true. We fill in the second oval marked T.
Step 3: Now put them together. “It is unlawful to drive while drunk because most automobiles in the United States are powered by internal combustion engines.”
Does it make sense? No. So we do not fill in the oval marked CE.

Let's do another one.
![]()
103.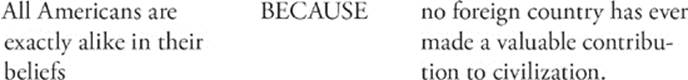
Step 1: Look at statement I by itself. Is it true or false? It's false.
Step 2: Look at statement II by itself. Is it true or false? It's false.

Notice that if either statement I or statement II is false, then there can be no cause-and-effect relationship, and you don't have to worry about filling in the CE oval.
When it comes to the divide and conquer strategy, use step 3 only if you determine that both statements are true.
STRATEGY #8: PROCESS OF ELIMINATION
We saved the best for last! Process of Elimination (or POE) is the most important strategy you have for the SAT Chemistry test. It involves deciding what the bad answers are and crossing them off, instead of just looking for the right answer. Here's how it works.
Try the following question:
1. What is the French word for “eggplant”?
What? You don't know? Well then, you'd better guess at random. (By the way, there are no questions about vegetables, French or otherwise, on the SAT Chemistry Exam. We're just using this question to make a point.)
If you really don't know the answer to a question, of course, you should always guess. But before you choose an answer at random, take a look at the problem the way you would see it on the test.
1. What is the French word for “eggplant?”
A. ![]()
B. ![]()
C. Aubergine
D. ![]()
E. $&%()@@
Suddenly the question looks a lot easier, doesn't it? You may not have known the correct answer to this question, but you certainly knew four answers that were incorrect.
POE in Practice
The Process of Elimination (POE for short) enables you to make your guesses really count. Incorrect answer choices are often easier to spot than correct ones. Sometimes they are logically absurd; sometimes they are the opposite of the correct answer. If you find a wrong answer, eliminate it. While you will rarely be able to eliminate all of the incorrect answer choices, it is often possible to eliminate one or two, and each time you can eliminate an answer choice, your odds of guessing correctly get better.
A Moral Dilemma
What if someone approached
you moments
before the test began
and offered to give you
the answers to the test?
You'd be SHOCKED, right?
But what if we told you
that the person making
the offer was the proctor
running the test? The fact
is that every student who
takes the test gets to see
all the answers ahead of
time; they're printed in the
test booklet, right underneath
each question.
Try another question.
1. What is the capital of Malawi?
A. New York
B. Lilongwe
C. Paris
D. Kinshasa
E. Chicago
This time you could probably eliminate only three of the answer choices. However, this means that you are down to a fifty-fifty guess—;much better than random guessing.
The Process of Elimination is a tremendously powerful tool. We refer to it in every single chapter of this book, and explain how to use it on a variety of specific question types.
Letter of the Day
Which makes more sense—;guessing the same letter every time or switching around? If you think you're better off switching around, think again. As counterintuitive as it may seem, you will pick up more points consistently if you always guess the same letter. Sure, you won't get all of your random guesses correct, but you'll get some points. On the contrary, if you vary your guess answer, you might get some correct, but you might miss all of them just as easily.
It doesn't matter what letter you pick as your Letter of the Day. Contrary to popular opinion, you won't get more questions right if you guess (C) rather than any other choice. Go crazy, guess (A) or (F) on the next test you take. Just be consistent.
LET'S GET GOING
In Chapters 3 through 14, we'll teach you chemistry with our own special tailored-to-the-subject-test method. All along the way we'll ask you subject test–type questions. Then, in Chapter 15, we'll explain the answers, showing you how to use knowledge and strategy to earn a high score.