Chemistry: A Self-Teaching Guide - Post R., Snyder C., Houk C.C. 2020
Atomic Weights
Now that you have some idea of what is in an atom, let's look at the weight of atoms. Each atom has a definite and characteristic weight. This weight provides a very convenient way to state the amount of substance required for a chemical reaction.
In this chapter we discuss how the weights of atoms were determined experimentally. You will encounter for the first time a very formidable number — 602,200,000,000,000,000,000,000 (6.022 × 1023) — called Avogadro's number. The number becomes very important in later chapters, so be sure you understand its significance!
OBJECTIVES
After completing this chapter, you will be able to
· recognize and apply or illustrate: isotope, atomic weight, atomic mass unit (amu), mass spectrograph, gram atomic weight, and Avogadro's number;
· explain the fractional atomic weights listed in the periodic table;
· calculate the number of atoms in a given weight of an element and vice versa;
· calculate the approximate atomic weight of an element when given the abundance and approximate mass of its isotopes;
· distinguish between atomic weight expressed in amu and gram atomic weight.
![]() Let's review a bit. The notation
Let's review a bit. The notation ![]() indicates a neutral atom of chlorine.
indicates a neutral atom of chlorine.
1. What is its atomic number? _________
2. What is its mass number? _________
3. How many protons does it have? _________
4. How many electrons? _________
5. How many neutrons? _________
Answer: (a) 17; (b) 35; (c) 17; (d) 17; (e) 18
![]() Different atoms of the same element can have different numbers of neutrons and, therefore, different mass numbers. Here is another neutral chlorine atom:
Different atoms of the same element can have different numbers of neutrons and, therefore, different mass numbers. Here is another neutral chlorine atom: ![]() .
.
1. What is its mass number? _________
2. How many protons does it have? _________
3. How many neutrons? _________
Answer: (a) 37; (b) 17; (c) 20
![]() Since neutrons and protons combine to make up the mass number, two atoms of the same element can have different mass numbers. Chlorine can exist as
Since neutrons and protons combine to make up the mass number, two atoms of the same element can have different mass numbers. Chlorine can exist as ![]() and as
and as ![]() . The only difference between these atoms of chlorine is that
. The only difference between these atoms of chlorine is that ![]() contains two more neutrons than
contains two more neutrons than ![]() .
.
Antimony can exist as ![]() and
and ![]() . The only difference between these atoms of antimony is that
. The only difference between these atoms of antimony is that ![]() contains two more ___________________ than
contains two more ___________________ than ![]() .
.
Answer: neutrons
![]()
![]() has a greater mass than
has a greater mass than ![]() because of the two extra neutrons. Which of the following atoms of antimony has the greater mass,
because of the two extra neutrons. Which of the following atoms of antimony has the greater mass, ![]() or
or ![]() ? _______________
? _______________
Answer: ![]() (because it has two more neutrons)
(because it has two more neutrons)
![]() Atoms of the same element having different masses are called isotopes. Elements as found in nature are usually mixtures of two or more isotopes. The atom
Atoms of the same element having different masses are called isotopes. Elements as found in nature are usually mixtures of two or more isotopes. The atom ![]() is one isotope of the element antimony;
is one isotope of the element antimony; ![]() is another isotope of antimony. The main difference between two isotopes of the same element is the number of (protons, neutrons, electrons) _______.
is another isotope of antimony. The main difference between two isotopes of the same element is the number of (protons, neutrons, electrons) _______.
Answer: neutrons
![]() Isotopes exist for every known element. The isotopes of the element neon were first discovered by two English scientists, J. J. Thomson and F. W. Aston. Thomson and Aston continued in their work to discover other isotopes through inventing the mass spectrograph (also called the mass spectrometer).
Isotopes exist for every known element. The isotopes of the element neon were first discovered by two English scientists, J. J. Thomson and F. W. Aston. Thomson and Aston continued in their work to discover other isotopes through inventing the mass spectrograph (also called the mass spectrometer).
In the mass spectrograph, atoms of different masses of the same element (mixtures of isotopes) are charged (no longer neutral) and accelerated by an electron beam toward a target, such as a photographic plate. A strong magnetic field bends the paths of the charged atoms. Atoms of greater mass have their paths bent to a lesser degree than atoms of lighter mass.
In the diagram of the mass spectrograph (pictured here) where do the lighter atoms strike, point A or point B? _______
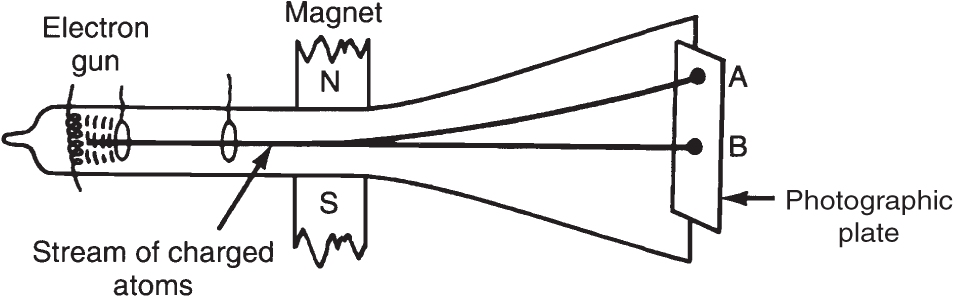
Answer: point A (because the path of the lighter atoms is bent to a greater degree)
![]() An analogy to the mass spectrograph would be to roll a bowling ball and a basketball at the same speed at a target while a stiff crosswind is blowing. The bowling ball is considerably heavier than a basketball. Look at the diagram below. Which ball would strike the target at point B? _______
An analogy to the mass spectrograph would be to roll a bowling ball and a basketball at the same speed at a target while a stiff crosswind is blowing. The bowling ball is considerably heavier than a basketball. Look at the diagram below. Which ball would strike the target at point B? _______
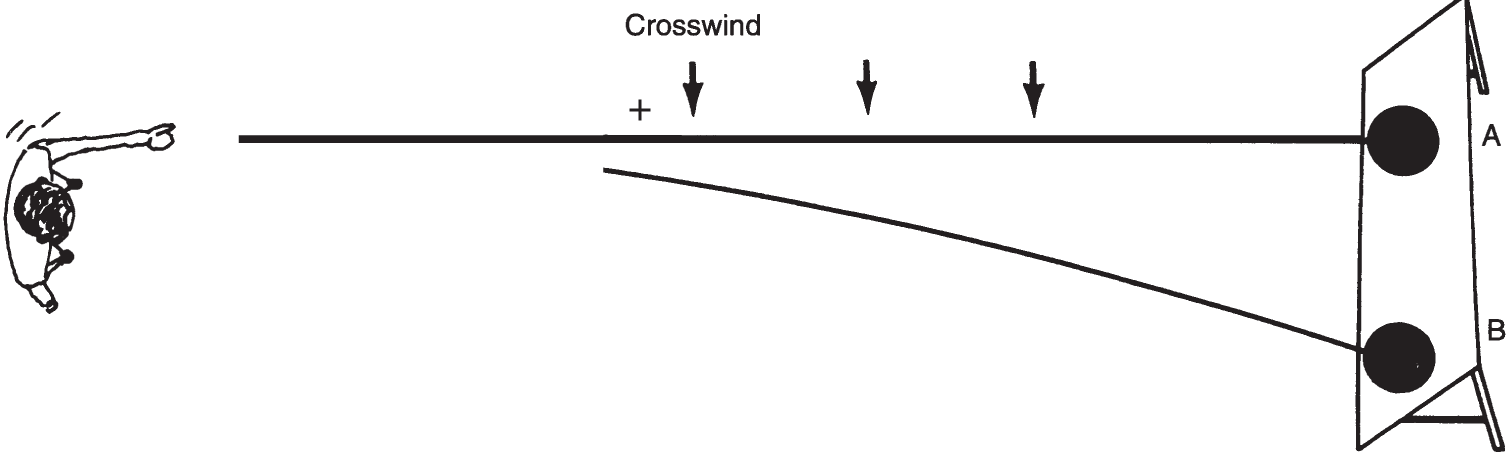
Answer: the basketball (The basketball is lighter; therefore, its path is more readily changed by the crosswind.)
![]() In the rolling balls analogy to the spectrograph, the bowling ball and basketball are analogous to isotopes of different mass. The strong crosswind is analogous to the _______________________. (Refer to the diagram of the spectrograph, if necessary.)
In the rolling balls analogy to the spectrograph, the bowling ball and basketball are analogous to isotopes of different mass. The strong crosswind is analogous to the _______________________. (Refer to the diagram of the spectrograph, if necessary.)
Answer: magnetic field (or magnet)
![]() Thomson and Aston invented an instrument that detects the presence and characteristics of isotopes. What is this instrument called? _____________________
Thomson and Aston invented an instrument that detects the presence and characteristics of isotopes. What is this instrument called? _____________________
Answer: mass spectrograph (or mass spectrometer)
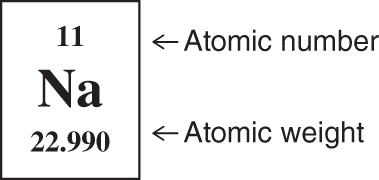
![]() The atomic weights of the elements are listed in the periodic table. The atomic weight of sodium, for example, is listed as 22.990. The atomic weight listed for sodium is actually the atomic weight of a mixture of isotopes,
The atomic weights of the elements are listed in the periodic table. The atomic weight of sodium, for example, is listed as 22.990. The atomic weight listed for sodium is actually the atomic weight of a mixture of isotopes, ![]() and
and ![]() . The proportion of these isotopes is generally constant wherever sodium is found.
. The proportion of these isotopes is generally constant wherever sodium is found.
The atomic weight of an element is the average weight of a mixture of two or more ________.
Answer: isotopes
![]() The periodic table lists the atomic weight of Al as _____________________.
The periodic table lists the atomic weight of Al as _____________________.
Answer: 26.982
![]() Atomic weights are based on the carbon 12 scale. That is, carbon 12 or
Atomic weights are based on the carbon 12 scale. That is, carbon 12 or ![]() , the most abundant isotope of carbon, is used as the standard unit in measuring atomic weights. By international standard, one atom of the
, the most abundant isotope of carbon, is used as the standard unit in measuring atomic weights. By international standard, one atom of the ![]() isotope has an atomic weight of exactly 12 atomic mass units, abbreviated amu. The atomic weight of the
isotope has an atomic weight of exactly 12 atomic mass units, abbreviated amu. The atomic weight of the ![]() isotope is exactly ________ amu.
isotope is exactly ________ amu.
Answer: 12
![]() All atomic weights can be expressed in atomic mass units. By international agreement, 12 amu would equal the mass of a single
All atomic weights can be expressed in atomic mass units. By international agreement, 12 amu would equal the mass of a single ![]() atom. One amu is equal to what fraction of a single
atom. One amu is equal to what fraction of a single ![]() atom? ________
atom? ________
Answer: ![]() the mass of a single
the mass of a single ![]() atom
atom
![]() Although the
Although the ![]() isotope weighs exactly 12.000 amu by definition, the atomic weight of C as listed on the periodic table is 12.011 amu. The atomic weight of carbon as listed on the periodic table is greater than that of the
isotope weighs exactly 12.000 amu by definition, the atomic weight of C as listed on the periodic table is 12.011 amu. The atomic weight of carbon as listed on the periodic table is greater than that of the ![]() isotope. Why? ______________
isotope. Why? ______________
Answer: The atomic weight of an element is the average weight of a mixture of two or more isotopes.
![]() While the element carbon as found in nature is made up largely of the
While the element carbon as found in nature is made up largely of the ![]() isotope (98.9%), a small quantity of
isotope (98.9%), a small quantity of ![]() isotope (1.1%) is mixed uniformly as part of the element. The
isotope (1.1%) is mixed uniformly as part of the element. The ![]() isotope has an atomic weight of 12.000 amu. The
isotope has an atomic weight of 12.000 amu. The ![]() isotope has an atomic weight of 13.003 amu. The resultant average atomic weight would be slightly (heavier, lighter) ________ than 12.000 amu.
isotope has an atomic weight of 13.003 amu. The resultant average atomic weight would be slightly (heavier, lighter) ________ than 12.000 amu.
Answer: heavier (In fact, the periodic table lists the atomic weight of carbon as 12.011 amu.)
![]() The atomic weights on the periodic table are the average atomic weights of the isotopic mixtures in the element. We can determine the average atomic weight of an element if we know the approximate mass of each isotope and the proportion of each isotope within the element.
The atomic weights on the periodic table are the average atomic weights of the isotopic mixtures in the element. We can determine the average atomic weight of an element if we know the approximate mass of each isotope and the proportion of each isotope within the element.
Here are the steps for calculating the average atomic weight of the element carbon. Look at the table below as you read through the steps.
Multiply the mass of the ![]() isotope by its decimal proportion (12.000 × 0.989).
isotope by its decimal proportion (12.000 × 0.989).
Multiply the mass of the ![]() isotope by its decimal proportion (13.003 × 0.011).
isotope by its decimal proportion (13.003 × 0.011).
Add the results to find the average atomic weight of the element C.
Element |
Isotope |
Mass of isotope |
Proportion in element |
Mass × proportion |
Sum |
||||
C |
|
12.000 |
× |
0.989 |
= |
11.868 |
+ |
= |
|
|
13.003 |
× |
0.011 |
= |
0.143 |
Now you do the final calculation step, adding the two results. Fill in the blank in the “Sum” column. Round off this answer and all others to the nearest hundredth (two places to the right of the decimal) unless otherwise indicated.
Answer: The calculated atomic weight of C is 12.011, rounded off to 12.01 amu.
![]() Now calculate the atomic weight for fluorine.
Now calculate the atomic weight for fluorine.
Element |
Isotope |
Mass of isotope |
Proportion in element |
Mass × proportion |
Sum |
||||
F |
|
19.000 |
× |
99.7% or 0.997 |
= |
+ |
= |
||
|
18.000 |
× |
0.3% or 0.003 |
= |
Answer:
Mass × proportion |
Sum |
||
18.943 |
+ |
= |
18.997 |
0.054 |
The calculated atomic weight of F is rounded off to 19.00 amu.
![]() Sodium has two isotopes,
Sodium has two isotopes, ![]() and
and ![]() . The isotope
. The isotope ![]() has an atomic mass of approximately 23.000 amu, and its proportion in the element is 99.2%. The isotope
has an atomic mass of approximately 23.000 amu, and its proportion in the element is 99.2%. The isotope ![]() has a mass of approximately 22.000 amu and a proportion within the element of 0.8%. Determine the atomic weight of sodium. (Remember, 0.8% = 0.008 and 99.2% = 0.992.) Use a separate sheet of paper to set up a table as we have done in the past few frames. Here again are the column headings you will need.
has a mass of approximately 22.000 amu and a proportion within the element of 0.8%. Determine the atomic weight of sodium. (Remember, 0.8% = 0.008 and 99.2% = 0.992.) Use a separate sheet of paper to set up a table as we have done in the past few frames. Here again are the column headings you will need.
Element |
Isotope |
Mass ofisotope |
Proportionin element |
Mass × proportion |
Sum |
Answer:
Element |
Isotope |
Mass of isotope |
Proportion in element |
Mass × proportion |
Sum |
||||
Na |
|
23.000 |
× |
0.992 |
= |
22.816 |
+ |
= |
22.992 |
|
22.000 |
× |
0.008 |
= |
0.176 |
The calculated atomic weight of Na is 22.99 amu.
![]() The element cobalt has an isotope
The element cobalt has an isotope ![]() that has an approximate mass of 60.00 and constitutes 48.0% of the element. Another isotope,
that has an approximate mass of 60.00 and constitutes 48.0% of the element. Another isotope, ![]() , has an approximate mass of 58.00 and constitutes 52.0% of the element. Calculate the atomic weight of Co using the given data. Use a separate sheet of paper to set up a table of calculations.
, has an approximate mass of 58.00 and constitutes 52.0% of the element. Calculate the atomic weight of Co using the given data. Use a separate sheet of paper to set up a table of calculations.
Answer:
Element |
Isotope |
Mass of isotope |
Proportion in element |
Mass × proportion |
Sum |
||||
Co |
|
60.00 |
× |
0.480 |
= |
28.80 |
+ |
= |
58.96 |
|
58.00 |
× |
0.520 |
= |
30.16 |
The calculated atomic weight of Co is 58.96 amu.
![]() The percentage proportions of all the isotopes within an element must add up to a total of ________ %. The decimal proportions of all the isotopes within an element must add up to ________ (Hint: See above example, 0.480 + 0.520.)
The percentage proportions of all the isotopes within an element must add up to a total of ________ %. The decimal proportions of all the isotopes within an element must add up to ________ (Hint: See above example, 0.480 + 0.520.)
Answer: 100; 1
![]() We've been calculating atomic weight given the mass and proportion of isotopes. We can also determine the proportion of each individual isotope within an element if we know the atomic weight of the element. Set up a table like the ones you have been using and fill in the information given below. Use letters such as A and B to represent unknown proportions.
We've been calculating atomic weight given the mass and proportion of isotopes. We can also determine the proportion of each individual isotope within an element if we know the atomic weight of the element. Set up a table like the ones you have been using and fill in the information given below. Use letters such as A and B to represent unknown proportions.
The element Cr, which has an overall atomic weight of 51.996 amu, has two isotopes: ![]() with atomic mass of 52.000 amu and
with atomic mass of 52.000 amu and ![]() with an atomic mass of 51.000 amu. Just fill in the “givens” and “unknowns” for now; don't try to solve the problem yet.
with an atomic mass of 51.000 amu. Just fill in the “givens” and “unknowns” for now; don't try to solve the problem yet.
Answer:
Element |
Isotope |
Mass of isotope |
Proportion in element |
Mass × proportion |
Sum |
||||
Cr |
|
52.000 |
× |
A |
= |
52.000 × A |
+ |
= |
51.996 |
|
51.000 |
× |
B |
= |
51.000 × B |
![]() To solve this equation with two unknowns, you must form a second equation showing the relationship between A and B. You have already learned the answer to the following question in frame 20. Add the decimal proportions. A + B = _____________
To solve this equation with two unknowns, you must form a second equation showing the relationship between A and B. You have already learned the answer to the following question in frame 20. Add the decimal proportions. A + B = _____________
Answer: 1
![]() Modify the equation in frame 22 so that just B remains on the left side of the equation. B = _______
Modify the equation in frame 22 so that just B remains on the left side of the equation. B = _______
Answer: 1 − A
![]() Here is the equation you need for calculating the sum of the isotopes' mass × proportion: (52.000A) + (51.000B) = 51.996. Substitute the expression (1 − A) for B in the equation _______
Here is the equation you need for calculating the sum of the isotopes' mass × proportion: (52.000A) + (51.000B) = 51.996. Substitute the expression (1 − A) for B in the equation _______
Answer: ![]()
![]() Solve the equation derived in frame 24 to determine the value of A (the proportion of
Solve the equation derived in frame 24 to determine the value of A (the proportion of ![]() in an average mixture of chromium) to the nearest thousandth. Use a separate sheet of paper for your calculations.
in an average mixture of chromium) to the nearest thousandth. Use a separate sheet of paper for your calculations.
Answer:
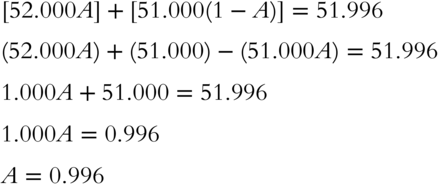
![]() Since B = 1 − A, what is the value of B (nearest thousandth)? B = _______
Since B = 1 − A, what is the value of B (nearest thousandth)? B = _______
Answer:
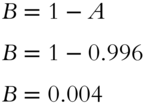
![]() Here is a similar problem. In the next few frames, you will determine the proportion of the
Here is a similar problem. In the next few frames, you will determine the proportion of the ![]() isotope in an average mixture of chlorine, which is made up of both
isotope in an average mixture of chlorine, which is made up of both ![]() and
and ![]() . Here is our table of givens and unknowns.
. Here is our table of givens and unknowns.
Element |
Isotope |
Mass of isotope |
Proportion in element |
Mass × proportion |
Atomic weight |
||||
Cl |
|
34.97 |
× |
A |
= |
34.97 × A |
+ |
= |
35.45 |
|
36.97 |
× |
B |
= |
36.97 × B |
The values of A and B added together must equal _______.
Answer: 1
![]() Since A + B = 1, then B = _______
Since A + B = 1, then B = _______
Answer: 1 − A
![]() The proportion 1 − A has been substituted for B in the following table.
The proportion 1 − A has been substituted for B in the following table.
Element |
Isotope |
Mass of isotope |
Proportion in element |
Mass × proportion |
Atomic weight |
||||
Cl |
|
34.97 |
× |
A |
= |
34.97 × A |
+ |
= |
35.45 |
|
36.97 |
× |
(1 − A) |
= |
36.97 × (1 − A) |
Using the table above, determine the proportion of ![]() within the element chlorine (find the value of A). Use a separate sheet of paper for your set of equations.
within the element chlorine (find the value of A). Use a separate sheet of paper for your set of equations.
Answer:
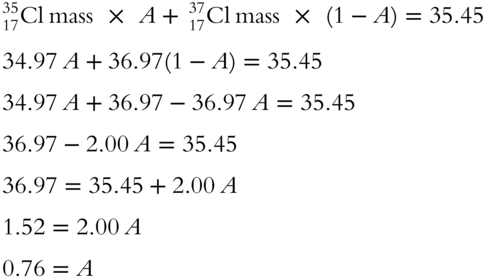
![]() Determine the proportion of
Determine the proportion of ![]() in Cl (find B).
in Cl (find B).
Answer: The proportion of ![]() in Cl has been given the value of B in the table.
in Cl has been given the value of B in the table.
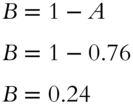
![]() Neon has two isotopes:
Neon has two isotopes: ![]() and
and ![]() . The approximate mass of
. The approximate mass of ![]() is 22.000 amu and the mass of
is 22.000 amu and the mass of ![]() is approximately 20.000 amu. The atomic weight of neon is 20.179 amu. Determine the proportion (to the nearest thousandth) of the
is approximately 20.000 amu. The atomic weight of neon is 20.179 amu. Determine the proportion (to the nearest thousandth) of the ![]() isotope within the element. (Let the proportion of
isotope within the element. (Let the proportion of ![]() within Ne be equal to B.) Use a separate sheet of paper for your table of calculations and set of equations.
within Ne be equal to B.) Use a separate sheet of paper for your table of calculations and set of equations.
Answer:
Element |
Isotope |
Mass of isotope |
Proportion in element |
Mass × proportion |
Atomic weight |
||||
Ne |
|
22.000 |
× |
A = |
= |
(22.000 × A) |
+ |
= |
20.179 |
|
20.000 |
× |
B = |
= |
(20.000 × B) |

![]()
![]() What is the proportion (to the nearest thousandth) of the other isotope,
What is the proportion (to the nearest thousandth) of the other isotope, ![]() , within the element Ne? _______
, within the element Ne? _______
Answer: The proportion of ![]() is equal to the value of B in the table.
is equal to the value of B in the table.
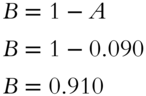
![]() So far, we have considered all atomic weights in terms of atomic mass units (amu).
So far, we have considered all atomic weights in terms of atomic mass units (amu).
An atomic weight expressed in amu represents the average weight of how many atom(s) of an element? _______
Carbon has an atomic weight of 12.011 amu, which represents the average weight of how many atom(s) of carbon? _______
Answer: (a) one (An atomic weight expressed in amu represents the average weight of one single atom of an element.); (b) one
GRAM ATOMIC WEIGHT
![]() Since it is impossible to measure the weight of one atom with a laboratory balance, another unit for expressing atomic weight must be used. Atomic weight can be expressed in grams as well as amu. An atomic weight expressed in grams (called a gram atomic weight) contains 6.022 × 1023 atoms. This number, called Avogadro's number, will be encountered often in this book.
Since it is impossible to measure the weight of one atom with a laboratory balance, another unit for expressing atomic weight must be used. Atomic weight can be expressed in grams as well as amu. An atomic weight expressed in grams (called a gram atomic weight) contains 6.022 × 1023 atoms. This number, called Avogadro's number, will be encountered often in this book.
1. If the atomic weight of carbon is expressed as 12.011 amu, it represents the average weight of how many atom(s)? _______
2. If the atomic weight of carbon is expressed as 12.011 grams, it represents the average weight of how many atom(s)? _______
Answer: (a) one; (b) 6.022 × 1023
![]() Using information in frame 34, answer the following question. One gram is how many times heavier than 1 amu? _______
Using information in frame 34, answer the following question. One gram is how many times heavier than 1 amu? _______
Answer: 6.022 × 1023 (One gram is equivalent to 6.022 × 1023 amu.)
![]() One ton is equivalent to 2000 pounds. One pound represents
One ton is equivalent to 2000 pounds. One pound represents ![]() of a ton. 6.022 × 1023 amu is equivalent to 1 gram. One amu represents what fraction of a gram? ______
of a ton. 6.022 × 1023 amu is equivalent to 1 gram. One amu represents what fraction of a gram? ______
Answer:
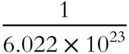
![]() Avogadro's number (6.022 × 1023) is written in exponential notation (sometimes called scientific notation). It actually represents a very large number: 602,200,000,000,000,000,000,000. Exponential notation will be used throughout this book, as in most other chemistry texts. You may already be familiar with exponential notation and with multiplying and dividing numbers with exponents. If so, you may skip to frame 38. If you need a quick refresher of exponential notation, we have summarized the basic rules in the examples below.
Avogadro's number (6.022 × 1023) is written in exponential notation (sometimes called scientific notation). It actually represents a very large number: 602,200,000,000,000,000,000,000. Exponential notation will be used throughout this book, as in most other chemistry texts. You may already be familiar with exponential notation and with multiplying and dividing numbers with exponents. If so, you may skip to frame 38. If you need a quick refresher of exponential notation, we have summarized the basic rules in the examples below.
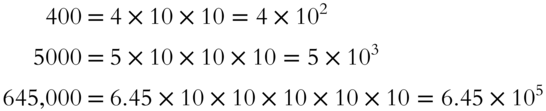
The exponent indicates the number of places that the decimal must be moved. The number 645,000 has the decimal moved five places to the left ![]() . The result is 6.45 × 105.
. The result is 6.45 × 105.
We can also use exponential notation as in the examples below.
![]()
![]()
In these two cases, we moved the decimal to the right, with the result that the exponent is negative.
![]() (Move the decimal five places to the right to arrive at the notation 4 × 10−5.)
(Move the decimal five places to the right to arrive at the notation 4 × 10−5.)
![]() (Move the decimal two places to the right to arrive at the notation 7.3 × 10−2.)
(Move the decimal two places to the right to arrive at the notation 7.3 × 10−2.)
For 0.073, we could have moved the decimal three places to the right, with a result of 73 × 10−3. We could also move the decimal one place to the right, with a result of 0.73 × 10−1. Normally, in chemistry calculations we write a single digit of value 1 through 9 to the left of the decimal.
To multiply two numbers with exponential notation, multiply the decimal portions of the numbers and add the exponents.
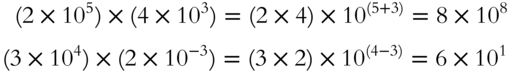
To divide two numbers with exponential notation, divide the decimal portions of the numbers and subtract the exponent in the denominator from the exponent in the numerator.
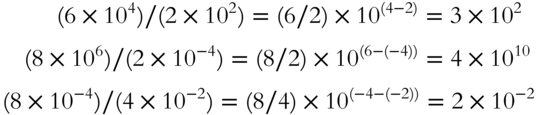
Try these problems.
1. 42,000 = 4.2 × _____________
2. 0.00465 = 4.65 × _____________
3. (7.0 × 10−5) × (4.0 × 108) = _____________
4. 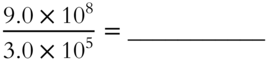
5. 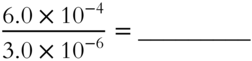
Answer:
1. 4.2 × 104
2. 4.65 × 10−3
3. (7.0 × 4.0) × 10(−5 + 8) = 28 × 103 = 2.8 × 104
4. 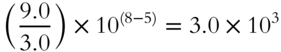
5. 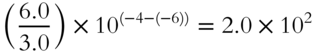
![]() Look at the following examples of fractions that are converted to exponential notation. Then fill in the correct answer for the equivalence of amu to grams by using the same process of eliminating the fraction and expressing the equivalence in proper exponential notation.
Look at the following examples of fractions that are converted to exponential notation. Then fill in the correct answer for the equivalence of amu to grams by using the same process of eliminating the fraction and expressing the equivalence in proper exponential notation.
· 1 pound equals 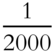 ton
ton
· 1 pound equals 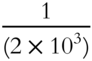 ton
ton
· 1 pound equals ![]() ton
ton
· 1 pound equals 0.5 × 10−3 ton
· 1 pound equals 5.0 × 10−4 ton
· 1 amu equals 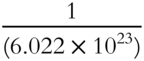 grams
grams
· 1 amu equals _________________________________
Answer: 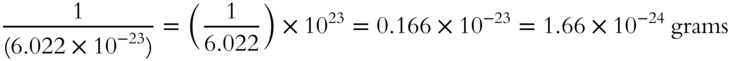
![]() You should memorize Avogadro's number (6.022 × 1023) and its meaning.
You should memorize Avogadro's number (6.022 × 1023) and its meaning.
1. How many atoms are in an atomic weight expressed in grams?_______________
2. One gram is equivalent to the weight of how many amu?__________________
Answer: (a) 6.022 × 1023 atoms per gram atomic weight; (b) 6.022 × 1023 amu per gram
![]() The average atomic weight of neon is 20.180 according to the periodic table (rounded off to the nearest thousandth).
The average atomic weight of neon is 20.180 according to the periodic table (rounded off to the nearest thousandth).
1. The average weight of one neon atom is how many amu? _______
2. The average weight of 6.022 × 1023 neon atoms is how many grams? ______________
Answer: (a) 20.180; (b) 20.180
![]() There are 6.022 × 1023 atoms in 1 gram atomic weight (atomic weight expressed in grams). The element neon has a gram atomic weight of 20.180 g. How many atoms are contained in 10.090 grams of neon? _______ (Hint: 10.090 grams of neon is half of the gram atomic weight of neon.)
There are 6.022 × 1023 atoms in 1 gram atomic weight (atomic weight expressed in grams). The element neon has a gram atomic weight of 20.180 g. How many atoms are contained in 10.090 grams of neon? _______ (Hint: 10.090 grams of neon is half of the gram atomic weight of neon.)
Answer: Since 10.090 grams is half of a gram atomic weight, the number of atoms is half of 6.022 × 1023 atoms, or 3.011 × 1023 atoms.
UNIT FACTOR ANALYSIS (FACTOR LABEL ANALYSIS/DIMENSIONAL ANALYSIS)
![]() If the problem in frame 41 had been more difficult, we would have used the unit factor method (also called factor label analysis and dimensional analysis in some texts) for solving problems, a mathematical procedure used in most chemistry textbooks for its convenience in calculations. We would have set up the problem in the manner shown below.
If the problem in frame 41 had been more difficult, we would have used the unit factor method (also called factor label analysis and dimensional analysis in some texts) for solving problems, a mathematical procedure used in most chemistry textbooks for its convenience in calculations. We would have set up the problem in the manner shown below.
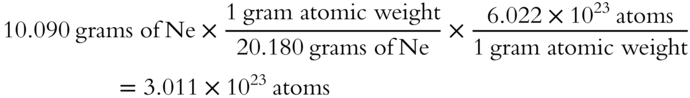
All of the unit names except “atoms” cancel out during multiplication.

Just the numbers and the name “atoms” are left after cancelation of unit names.
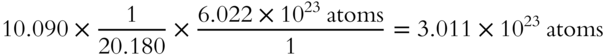
The unit factor method involves multiplying the given value by one or more conversion factors. In this problem, we multiplied the 10.0895 grams of Ne by the conversion factors of
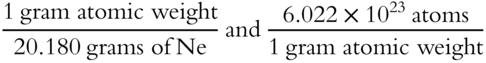
The conversion factors come from definitions. For example, 1 gram atomic weight is equal to 6.022 × 1023 atoms, and 20.180 grams of Ne are equal to 1 gram atomic weight of Ne. The conversion factors are arranged so that the unit names will cancel out. They convert the units of the given values to those of the answer being sought and will give us the correct numerical answer.
Here is another example using the unit factor method. The necessary definitions are: 1 meter = 39.37 inches, and 1 yard = 36 inches.
The first conversion factor can be 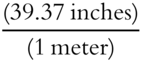 or
or 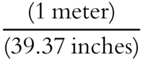 .
.
The second conversion factor can be 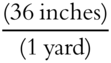 or
or 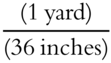 .
.
Suppose we wish to determine the number of meters in 2.12 yards. We arrange the conversion factors so that the answer will be in meters and all other unit names will cancel.
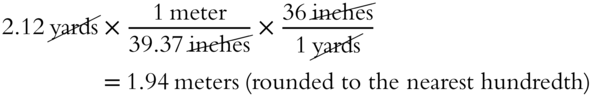
Use these conversion factors to determine the number of yards in 3.55 meters (nearest hundredth).
Answer: 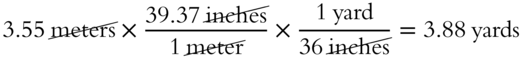
![]() The gram atomic weight of silicon is 28.09 grams. Using the unit factor method shown in frame 42, calculate the number of atoms in 2.00 grams of silicon (Si).
The gram atomic weight of silicon is 28.09 grams. Using the unit factor method shown in frame 42, calculate the number of atoms in 2.00 grams of silicon (Si).
Answer:
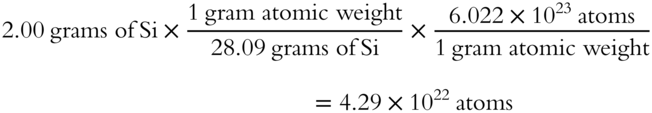
![]() The atomic weight of silicon is 28.09. Determine the average weight (in amu) of one silicon atom. _______ amu
The atomic weight of silicon is 28.09. Determine the average weight (in amu) of one silicon atom. _______ amu
Answer: 28.09 (same as the atomic weight).
![]() There is a big difference between an atomic weight expressed in amu and an atomic weight expressed in grams. You have already determined that one silicon atom weighs 28.09 amu. Now calculate the weight of one silicon atom in grams.
There is a big difference between an atomic weight expressed in amu and an atomic weight expressed in grams. You have already determined that one silicon atom weighs 28.09 amu. Now calculate the weight of one silicon atom in grams.
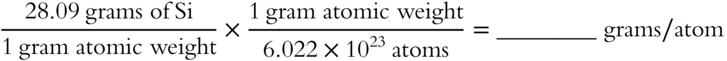
Answer: 4.66 × 10−23
![]() Determine the mass in grams of one boron (B) atom. (The gram atomic weight of boron is 10.81 grams.)
Determine the mass in grams of one boron (B) atom. (The gram atomic weight of boron is 10.81 grams.)
Answer:
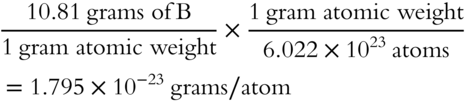
![]() If one boron atom weighs 1.795 × 10−23 grams, what would 1,000,000 atoms of boron weigh? (1,000,000 = 1 × 106)
If one boron atom weighs 1.795 × 10−23 grams, what would 1,000,000 atoms of boron weigh? (1,000,000 = 1 × 106)
Answer: 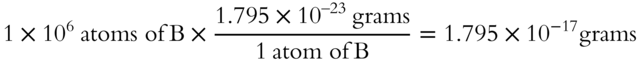
![]() The atomic weight of gold is 196.97. If you were offered an atomic weight of gold for only one dollar, would you buy it? Why or why not?
The atomic weight of gold is 196.97. If you were offered an atomic weight of gold for only one dollar, would you buy it? Why or why not?
Answer: We wouldn't. The atomic weight of gold would be expressed as 196.97 amu, so you are buying one atom of gold for a dollar, definitely no bargain. (However, a gram atomic weight of gold weighs 196.97 grams, which would definitely be a bargain for one dollar.)
![]() How much would a billion billion atoms (1 × 1018 atoms) of magnesium weigh? The gram atomic weight of magnesium is 24.305 grams. (Hint: Find the weight of one atom of magnesium and multiply the result by 1 × 1018.)
How much would a billion billion atoms (1 × 1018 atoms) of magnesium weigh? The gram atomic weight of magnesium is 24.305 grams. (Hint: Find the weight of one atom of magnesium and multiply the result by 1 × 1018.)
Answer: Here are two methods for solving this problem. The first method is one long step.
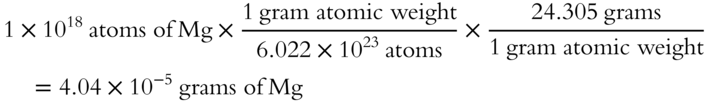
The second method involves two shorter steps.

What are the most important concepts in this chapter?
· The weights assigned to atoms are relative weights. That is, all atoms are compared to the weight of a single ![]() atom.
atom.
· All atoms of the same element do not have the same weight (elements exist as isotopes).
· The isotopes of an element are not present in nature in equal amounts.
· The atomic weight of an element may be determined experimentally.
· Atoms have very small weights. A chemist deals with large numbers of atoms (on the order of 6 × 1023) in the laboratory.
In Chapter 3 you will see that atomic weight is one of several periodic properties. You will encounter atomic weights throughout the remainder of this book, especially in Chapters 4 and 7, so you should have their meaning and significance clearly in mind before proceeding.
SELF-TEST
This self-test is designed to show how well you have mastered this chapter's objectives. Correct answers and review instructions follow the test. Calculate answers to the nearest hundredth unless otherwise indicated.
1. Isotopes of the element neon were first discovered by two English scientists, ______ and _____.
2. What is a mass spectrometer used for? _______
3. In a mass spectrometer's magnetic field which atoms are affected to a greater degree? _______ (lighter or heavier mass)
4. The atoms ![]() and
and ![]() are called _______ of chlorine.
are called _______ of chlorine.
5. How many protons, neutrons, and electrons are found in each of the chlorine isotopes in question 4?
6. How many neutrons are in the following carbon isotopes?
carbon-12 _______, carbon-13 ________, carbon-14 _______
7. What are the atomic weights of the following elements?
chromium (Cr) _______, potassium (K) _______________, aluminum (Al) _______
8. What are the atomic weights of the following elements?
osmium (Os) _______, calcium (Ca) ______________, gallium (Ga) _______
9. What elements have the following atomic weights?
4.00 _________
183.84 ________
244 _______
10. You are given 0.100 gram atomic weight of gold (Au). The atomic weight of gold is 196.97 grams to the nearest hundredth.
1. How many atoms would you have? _______
2. How many grams? _______
11. A sample of iron has a total of 9.77 × 1021 atoms of iron. Calculate the mass, in grams, of this sample.
12. Calculate the number of platinum atoms in a 15.5-gram sample of platinum. The atomic weight of platinum is 195.08 amu to the nearest hundredth.
13. The atomic weight of iron is 55.85 amu to the nearest hundredth and it has isotopes with approximate masses of 55.000 amu and 56.000 amu. What is the proportion of the 5626Fe isotope? (The other isotope is 5526Fe.)
14. The atomic weight of sodium is 22.99 amu to the nearest hundredth. A sample has isotopes with approximate masses of 20.998 and 23.991 amu. What is the proportion of the 24Na isotope? (The other isotope is 21Na.)
15. If you could cash it in, which would you rather have, 1 × 1040 amu of silver or 150 grams? (Hint: The problem is to determine which value has the greater number of atoms.)
ANSWERS
Compare your answers to the self-test with those given below. If you answer all questions correctly, you are ready to proceed to the next chapter. If you miss any, review the frames indicated in parentheses following the answers. If you miss several questions, you should probably reread the chapter carefully.
1. J.J. Thomson and F. W. Aston (frame 6)
2. to detect the presence of isotopes of each element (frame 6)
3. lighter atoms are affected to a greater degree (frame 6)
4. isotopes (frames 5, 6)
5. Chlorine-35 has 17 protons, 18 neutrons, and 17 electrons. Chlorine-37 has 17 protons, 20 neutrons, and 17 electrons (frames 1—5).
6. Carbon-12 has six neutrons, carbon-13 has seven neutrons, and carbon-14 has eight neutrons (frames 1—5).
7. Cr = 51.996, K = 39.0983, Al = 26.98154 (frame 10)
8. Os = 190.23, Ca = 40.078, Ga = 69.723 (frame 10)
9. He (4.00), W (183.84), Pu (244) (frame 10)
10.
1. 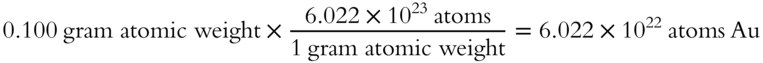
2. 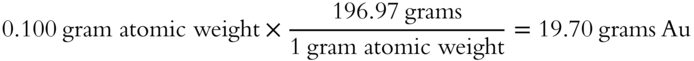
(frames 34, 39—43)
11. 0.906 g Fe (frames 42—49)
12. 4.78 × 1022 atoms Pt
13. Following the examples in the tables in frames 27-31, let A represent the proportion of 5626Fe isotope, and let 1 − A represent the proportion 5526Fe isotope.
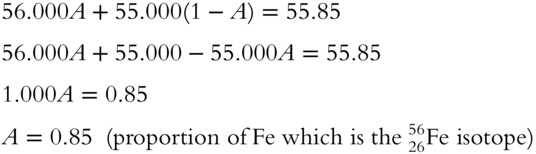
(frames 21—32)
14. Let A represent proportion: 24Na isotope, and let 1 − A represent proportion 21Na isotope.
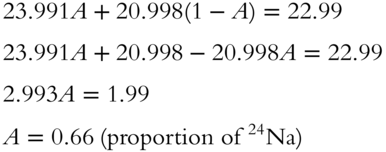
(frames 21—32)
15. Ag = 107.87 amu/atom, so our two equivalences are:

You would probably rather cash in the 1 × 1040 amu.
(frames 47—49)
KILOGRAM'S CHANGE
Have you ever considered how important it is to have standards of measure? Without them how would we know how much mass an object possesses or how much time it takes for a chemical reaction to occur? There are many standards of measure for all kinds of measurements whether it be for mass, length, volume, time, etc.
Around the world the standard unit for mass is the kilogram. For more than 100 years the kilogram was defined by the mass of a platinum-iridium alloy that was housed at the International Bureau of Weights and Measures in Paris, France. The kilogram mass has served as the base unit of mass in the International System of Units (SI) from 1889 until present day.
 A 1-kilogram platinum-iridium alloy.
A 1-kilogram platinum-iridium alloy.
The international unit for mass, the kilogram, is used in chemistry and physics for a variety of applications. For example, if a chemist needs to know how many kilograms are in a sample of carbon s/he can obtain that through the atomic mass of carbon from the periodic table and then convert to kilograms.
Also, we can convert kilograms to another unit of measure. For instance, the mass of a 1983 penny is 2.5 × 10−3 kilograms, but we can convert this amount into grams.
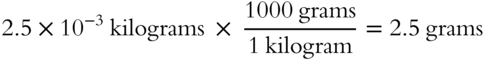
The kilogram is not only involved in converting between moles and kilograms, or kilograms to another unit of measure. The unit of mass used to express atomic and molecular weights is equal to one-twelfth of the mass of an atom of carbon-12, which is approximately 1.66 × 10−27 kilograms.
In addition to the kilogram there are 17 derived units that are defined in relation to the kilogram. In other words the kilogram is part of their unit of measure. Some of these include the Newton (N), Pascal (Pa), Joule (J), volt (V), Ohm (Ω), and Sievert (Sv). If we look at forces and Newton's second law of motion we see that force (F) = ma. Force is measured in Newtons, where m is the mass in kilograms and a is the acceleration of the object. One Newton is the equivalent of one kilogram multiplied by a meter per second squared.
![]()
As you can imagine, how a kilogram is defined affects every derived unit mentioned. As of 20 May 2019 the International Committee for Weights and Measures approved a proposed redefinition of the kilogram. This redefinition, although small in its change, defines the kilogram in terms of the second and the meter. With the change in the kilogram's definition we can obtain even greater accuracy with the results obtained from a variety of experiments.