5 Steps to a 5: AP Physics 2: Algebra-Based 2024 - Jacobs Greg 2023
STEP 4 Review the Knowledge You Need to Score High
15 Quantum, Atomic, and Nuclear Physics
IN THIS CHAPTER
Summary: Our modern understanding of physics shows us that time, space, and matter are not exactly as they appear. The nano-world of atoms and subatomic particles can seem strange and does not always follow the same rules as the world we live in. But the weird and wonderful world of the ultra-small are the building blocks that make up everything we know and experience in the big world we live in.

Key Ideas
![]() Time and length are not constant. Both depend on the speed the object is traveling. Only the speed of light in a vacuum is constant.
Time and length are not constant. Both depend on the speed the object is traveling. Only the speed of light in a vacuum is constant.
![]() Mass can be converted into energy and vice versa: E = mc2.
Mass can be converted into energy and vice versa: E = mc2.
![]() Massless particles can have mass-like properties such as momentum.
Massless particles can have mass-like properties such as momentum.
![]() The photoelectric-effect experiment proves that light has particle properties. The particle of light is called a photon. Its mass is zero and its energy is E = hf.
The photoelectric-effect experiment proves that light has particle properties. The particle of light is called a photon. Its mass is zero and its energy is E = hf.
![]() Subatomic particles can exhibit wave-like properties such as interference.
Subatomic particles can exhibit wave-like properties such as interference.
![]() Electrons in atoms can exist only in specific energy levels. To move up in energy, they must absorb a photon of light. To move down in energy, they must emit a photon.
Electrons in atoms can exist only in specific energy levels. To move up in energy, they must absorb a photon of light. To move down in energy, they must emit a photon.
![]() Conservation of mass/energy and conservation of momentum hold true in reactions involving subatomic particles, including photons.
Conservation of mass/energy and conservation of momentum hold true in reactions involving subatomic particles, including photons.
![]() The strong force holds the nucleus together. It is equivalent to the nuclear mass defect.
The strong force holds the nucleus together. It is equivalent to the nuclear mass defect.
![]() Some nuclei are radioactively unstable and decay into new elements.
Some nuclei are radioactively unstable and decay into new elements.
![]() The time it takes for one-half of a radioactive material to decay is called half-life.
The time it takes for one-half of a radioactive material to decay is called half-life.
![]() There are many types of decay: alpha, beta, gamma, positron, and neutron are a few.
There are many types of decay: alpha, beta, gamma, positron, and neutron are a few.
![]() In all nuclear reactions, four conservation laws are obeyed: conservation of mass/energy, conservation of charge, conservation of momentum, and conservation of nucleon number.
In all nuclear reactions, four conservation laws are obeyed: conservation of mass/energy, conservation of charge, conservation of momentum, and conservation of nucleon number.
Relevant Equations

What Is Modern Physics?
Modern physics takes us into the bizarre world of the uber fast and the nano small. This is the realm where the distinction between waves and particles gets fuzzy, where things without mass can have momentum, where elements spontaneously break apart, and where time and space are no longer constant.
Modern physics refers to all of the physics from about 1900 on, and that is when things really got weird. In fact, things got so bizarre that Albert Einstein refused to believe some of it, even though in many ways he was the guy who got the whole ball rolling when it came to modern physics. But quantum theory, a big part of modern physics, has proven over and over again to be correct, so much so, that practically all of our modern electronic toys are based upon it. So let’s dive into modern physics.
Space, Time, the Speed of Light, and E = mc2
Crazy-hair Einstein changed everything when he made these two simple postulates1 of special relativity:
1. All the laws of physics are the same in every uniformly moving frame of reference.
2. The speed of light in a vacuum is always measured to be c = 3 × 108 m/s no matter the motion of the source of the light, or the motion of the receiver of the light.
The first one makes sense. In a car, you don’t have to do anything special when you are drinking from a cup. But, if the driver steps on the gas or slams on the brakes, you might get wet. As long as you aren’t accelerating, everything is normal.
The second postulate has really weird consequences. In order for the speed of light to always equal c, time and space cannot be constant! The faster you go, the slower time moves and the shorter distances become in the direction you are going. The really odd thing is that you will not notice any of these changes, because of postulate 1. All the laws of physics will be the same to you no matter what speed you travel. All the effects of relativity seem to happen to the “other guy.” Confused yet? Here is an example:
I travel 10 years near the speed of light to an exoplanet. As you watch me through a telescope you see that my ship shrinks in the direction it is traveling, I also seem to be moving really slowly and my heart rate has nearly stopped. It’s as if time has stopped for me and you predict that I won’t even age on the 10-year trip. I, on the other hand, don’t notice anything strange happening to me. But, when I look outside, everything seems to be shrinking in the direction I am traveling. Looking back at you, I see that you are buzzing around really fast and aging like crazy. Since the distance around me has shrunk, I get to the exoplanet in no time at all. I have hardly aged a day! Cool! You, on the other hand, are 10 years older. Bummer.
That is really mind-bending because we never see this kind of stuff in everyday life. The effects of relativity don’t become easily visible until we start traveling a good fraction of the speed of light.
What do you have to know for the AP exam? Bottom line: you need to know that time and distances are not constant. If two people are moving at different speeds, they will not agree on the size of something or even when things have occurred. Only the velocity of light is constant. Moving faster shrinks distances and slows time, but the effects of relativity always are observed to happen outside of your own frame of reference. Everything in your frame of reference always seems “normal.”
Another gift from Einstein is the most famous equation of all time: E = mc2. It is a matter-shattering idea. Literally. It means that energy and mass are just two aspects of the same thing. Mass is like a solid form of energy and it can be converted into energy. (Lots of energy—c2 is a huge number!) Energy can be converted into mass. You heard that right. You can create matter out of energy. These things happen all the time in the nano-world. So let’s take a look at the super small.
Subatomic Particles
Let’s start with something familiar, the basic subatomic particles. One way to approach this is to look at things historically. Thousands of years ago, long before the idea of science, the accepted belief was that the universe was made of four basic things, earth, wind, fire, and water. Once humans got a little more scientific about looking at the world around themselves and started closely examining how matter behaves by breaking it down into smaller and smaller parts, it became accepted that matter was made of supposedly indivisible particles called atoms. Further experiments revealed the structure of an atom consists of protons and neutrons in the nucleus, with tiny electrons moving around the nucleus. Also discovered were little bundles of electromagnetic energy called photons.

There are even more subatomic particles. In the 1950s neutrinos were discovered. In the 1960s quarks, the building blocks of protons and neutrons, were discovered. Just recently the Higgs particle was found. Just make sure you know the Big 4 from the table above and know how to find them on your equation sheet. (Note: Neutrons are actually slightly more massive than protons, but this is the way they are listed on the AP constant sheet. If the numerical difference is important, the exact values will be provided in the problem.)
The Electron-Volt
The electron-volt, abbreviated eV, is a unit of energy that’s particularly useful for problems involving subatomic particles. One eV is equal to the amount of energy needed to change the potential of an electron by 1 volt. For example, imagine an electron nearby a positively charged particle, such that the electron’s potential is 4 V. If you were to push the electron away from the positively charged particle until its potential was 5 V, you would need to use 1 eV of energy.
![]()
The conversion to joules shows that an eV is an itty-bitty unit of energy. However, such things as electrons and protons are itty-bitty particles. So the electron-volt is actually a perfectly sized unit to use when talking about the energy of a subatomic particle.
Photons
Photons are light, or more precisely, light is made up of photons. A nice way to think of a photon is a bundle of electromagnetic waves or energy. The amount of energy in that bundle is directly related to the electromagnetic wave’s frequency:
![]()
where:
• E is the energy of the photon.
• h is Planck’s constant. Your AP Physics 2 reference table gives this to you in two forms: h = 6.63 × 10—34 J.s = 4.14 × 10—15 eV.s
• f is the frequency in hertz.
• c is the speed of light, c = 3.0 × 108 m/s.
• λ is the wavelength in meters.
Even though you have only E = hf on your reference table, it is very useful to use c = fλ to have the same equation in terms of wavelength, ![]() . It is especially useful since the AP people were kind enough to give you these constants to use:
. It is especially useful since the AP people were kind enough to give you these constants to use:
![]()
Be careful and notice that the second term is in nanometers, so your wavelength must be in nanometers.
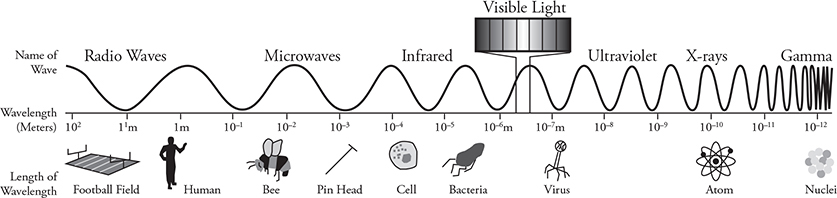
The figure below shows the entire electromagnetic spectrum of light from radio waves to gamma rays. Looking at the photon energy equation we can see that gamma rays have the most energy because they have the highest frequency and shortest wavelength. Radio waves have the least energy and the longest wavelengths and the lowest frequencies.
Photoelectric Effect
Before we begin, please make sure you understand this experiment. It is really important. This is the experiment that proves that light is a particle, even though we already know that it is a wave because light has wave properties like interference. This is the experiment that opened up the rabbit hole of wave-particle duality.
When light shines on a metal, you can get electrons to fly off the surface. Scientists call this the photoelectric effect. You might say to yourself, “What’s so important about that?” There are so many important processes in nature that are governed by the photoelectric effect you might be shocked. Photosynthesis, tanning, photographic film, solar panels, and cancer, for instance, are all related to the photoelectric effect.
To really understand the photoelectric effect, you have to understand the experiment that was used to study it. The next diagram shows the experimental setup. Don’t worry if it looks a little scary, it’ll make sense in a few minutes. In the experiment, two metal plates are sealed in a vacuum container. One of the plates is either made of, or coated with, the metal being studied. A variable potential source is connected across the two metal plates. (Think of the variable potential source as a dimmer switch in your home that varies the voltage across your lights, making them brighter or dimmer.) Incident light is directed at the plate with the metal being tested, and under the right conditions, electrons are emitted from that plate and travel to the other plate, and a current can be seen on the ammeter.
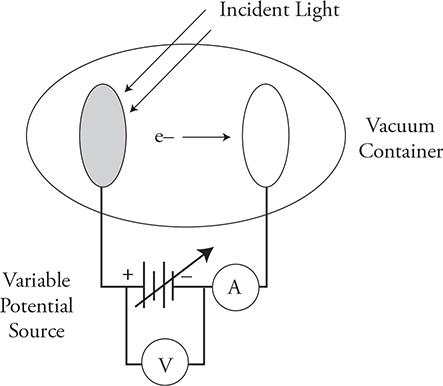
Notice that the variable potential source is oriented in such a way so that the electrons are traveling toward the negative plate and away from the positive plate. This means that the voltage across the plates is acting to stop the electrons from traveling from one side to the other. That voltage is called the stopping potential; it lets us calculate the maximum kinetic energy of the electrons. For example, let’s say we have light incident upon the left plate and electrons are flowing across to the plate on the right, this shows up as a current reading on our ammeter. We start turning up our voltage until all the electrons are stopped and our ammeter reads zero. For argument’s sake, I’ll say 2 volts stop all the current. The work done on the electron by the electric field is equal to the kinetic energy of the electron:
![]()
This tells us that the maximum kinetic energy of electrons is 2 eV. Remember, when you are getting your energy in electron-volts, your charge must be in elementary charges, and an electron is exactly one elementary charge.
So, the ammeter tells us the rate the electrons are being emitted and the voltmeter tells us the maximum kinetic energy of the electrons.
The easiest way to look at this is with an example.
Blue light with a wavelength of 443 nm is incident on a potassium surface, and electrons are emitted from the surface. A current is measured until a 0.5-V stopping potential is applied. What does this tell you about potassium?
Let’s first find the photon energy of the light incident on the surface:
![]()
Notice that since the wavelength was in nanometers, we should use the constant hc with nanometers as well. Next, let’s find the kinetic energy of the emitted electrons. It takes 0.5 volts to stop the electrons so:
![]()
Something does not make sense. Why, when a 2.8-eV photon hits potassium, does an electron get emitted with only 0.5 eV of kinetic energy? You know enough about conservation of energy to ask: “Where did the energy go?”
Let’s think about this—the electron is negative and the nucleus is positive, so they are attracted to each other. It takes work to “rip” that electron away from the nucleus. (For you chemistry students: it’s similar to ionization energy, the amount of energy required to strip a molecule of an outer electron.) Physicists call this amount of energy the work function, ϕ. It is based upon the properties of the element. Potassium has one work function, gold will have a different work function, and so on. To find the work function:
![]()
where:
• Kmax is the maximum kinetic energy of the emitted electron.
• hf is the energy of the incident photon, from E = hf.
• ϕ is the work function. The work function is the energy required to remove an electron from a specific element.
Plugging in our values:
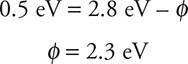
OK, so now you know it takes 2.3 eV to remove an electron from potassium.
So let’s change things. Suppose red light with photon energy of 1.9 eV is incident on potassium. What will happen?
It takes 2.3 eV to remove an electron, so nothing happens, no electron emission.
Let’s make that same red light brighter, what will happen then?
Making the red light brighter simply means there are more red 1.9 eV photons hitting the potassium. One important thing to keep in mind is that the photons cannot gang up; two can’t act together and add their energies. So no matter how bright the red light is made, there is no way a 1.9-eV photon will make potassium, with a 2.3-eV work function, emit an electron. So, blinding, skin-burning, bright-red light will never eject an electron from potassium. Never.
What if we make the original 443-nm blue light brighter, how will this change things?
The photon energy of the blue light is 2.8 eV, and the work function is 2.3 eV, so there will be electron emission, and the stopping potential will stay 0.5 eV. Making the light brighter means there are more 2.8-eV photons incident on the metal, so there will be more 0.5-eV electrons emitted, which would be seen as higher current measured by the ammeter.
Finally, let’s use violet light with photon energy of 3.1 eV. How does this change things?
That’s simple enough. The photons have 3.1 eV of energy and it takes 2.3 eV to remove the electron (the work function), so 3.1 eV — 2.3 eV = 0.8 eV, which is the maximum kinetic energy of the emitted electron.
A couple other quick points. You might be asked to evaluate a graph of kinetic energy of the emitted electron. There are a few things you need to remember about this graph, since it shows up on AP exams from time to time.
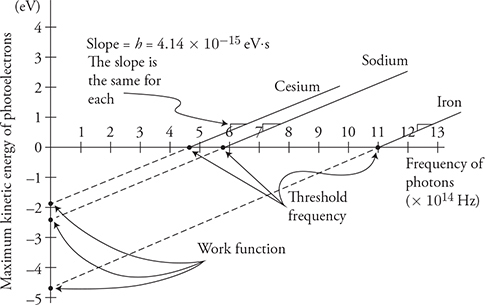
The next diagram shows an example of the plots you would get for two metals, potassium and iron. The first thing you might notice is that the lines are straight and parallel to each other. Let’s practice matching up our photoelectric equation with the equation of a line:
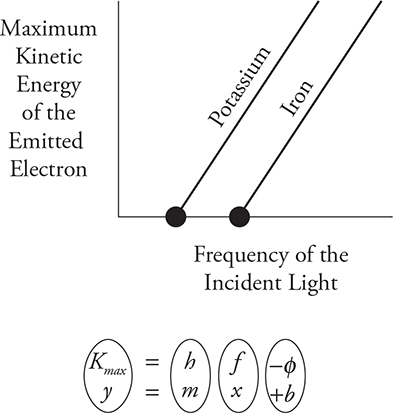
So, the slope of these lines always equals Planck’s constant and the intercept will equal the work function. Notice the large points where the lines intersect the x-axis. This point is called the threshold frequency. It is the minimum frequency required for electron emission. Potassium has a lower threshold frequency than iron because it takes less energy to remove an electron from potassium. Another way to find the threshold frequency is to use E = hf and enter the work function for your energy. Since the work function is the minimum energy needed to cause electron emission, the resulting frequency will be the lowest frequency to cause photo-electron emission.
Remember the importance of this experiment: The photoelectric effect shows us the particle behavior of light. We already discussed the double-slit experiment in Chapter 14, which demonstrates interference and the wave nature of light. So, we say that light exhibits the properties of both waves and particles. It has wave-particle duality. When we are working with energies down near the photon E = hf range, we see the particle nature of light. When light interacts with objects around the size of its wavelength and smaller, we will see its wave behavior.
So when we get down to the nano-world, some of our old physics models don’t work anymore. We have to invent a new model for how things behave. Electron behavior is another example of this. We will discuss electrons in a little bit.
Photon Momentum
We know that the energy of a photon is E = hf and that energy is related to mass: E = mc2. And now we know that photons have particle properties. Is it possible for photons to have particle properties like momentum? Yes! Here is a quick derivation:
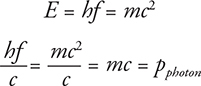
When a photon hits an atom, and that atom emits an electron as in the photoelectric effect, both energy and momentum are conserved in the process. This means that even though the photon does not have rest mass, it still has momentum. The momentum of a photon is:
![]()
where:
• p is the momentum of the photon in kg·m/s.
• E is the energy of the photon in joules.
So a photon of light can “collide” with an electron just like two cars collide, and the momentum will be conserved. Energy will also be conserved in the interaction. Let’s take a look at a couple of examples.
Examples
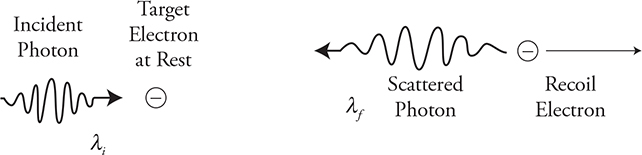
Example 1: A photon collides head-on with an electron. This is a one-dimensional interaction. The photon hits a stationary electron; afterward, the electron takes off with some of the photon’s original energy and momentum. Thus the photon lost both momentum and energy. Look back at our equation. To lose momentum and energy, the wavelength of the photon must have increased and the frequency decreased. The photon has changed “color.”
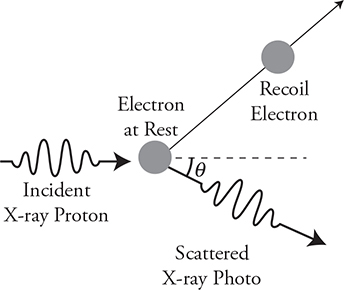
Example 2: The photon strikes an electron and the electron moves off at an angle. This is a two-dimensional momentum problem. Since the electron moves upward, the photon must move downward. The initial y-direction momentum of the system is zero; therefore, the final y-direction momentum must also equal zero. The initial and final x-momentums also remain constant.
De Broglie Wavelength and Wave Functions
You already know that a wave can act like a particle. Photon momentum and the photoelectric effect are examples of light acting like a particle or a photon. If that wasn’t weird enough, physicists (de Broglie in particular) started to contemplate: “If waves can act like particles, then maybe particles can act like waves.” This sounded ridiculous until it turned out to be true. To understand this, you need a good grasp of how the electromagnetic spectrum behaves.
• Long wavelengths of light, such as radio waves, demonstrate lots of wave behaviors like interference and diffraction. However, they have so little energy, they don’t exhibit much in the way of particle properties like photon momentum or the photoelectric effect.
• Short wavelengths of light, like gamma rays, demonstrate a huge amount of photon momentum and the photoelectric effect. However, their wavelengths are so short that it’s hard to measure wave properties like diffraction and interference for gamma ray photons.
Particles behave the same way. If the particle has a short wavelength, it should behave more like a particle, and if the particle has a longer wavelength, it should behave more like a wave. To find the wavelength for a particle, this is called the de Broglie wavelength, physicists took the equation for photon momentum and worked it in reverse:
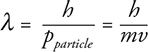
where:
• λ is the de Broglie wavelength in meters.
• h is Planck’s constant. (This time you have no choice, you must use h = 6.63 × 10—34 J·s.)
• m is the mass the particle in kilograms.
• v is the velocity of the particle in meters per second.
Let’s take an example. Find the de Broglie wavelength of a 0.15-kg baseball moving at 40 m/s:
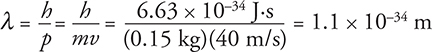
If this seems small, it certainly is. In fact it is smaller than gamma rays, which means it will behave like a particle. It won’t diffract around objects or show interference as you might expect from a wave. It will collide when it hits another particle.
To get more wavelike behavior from a particle, I need to increase its wavelength. To do that, I have to either decrease its mass or velocity, or both. Let’s find the de Broglie wavelength of one of the smallest particles, an electron moving at 3 × 106 m/s:
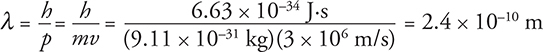
This might seem very small, but it is right in the wavelength range of an X-ray.
How can electrons, which are particles and have mass, show wave behavior? They can show interference through slits, just like interference patterns of light, in what is called electron crystal diffraction. The atoms in a crystal can line up to form slits. When X-rays are fired on a crystal, they undergo diffraction and interference, and an interference pattern can be seen. When electrons with the same wavelength are fired at the same crystal, a very similar interference pattern occurs, which means the electrons are undergoing diffraction and interference. Electron microscopes take advantage of the wave properties of electrons to form amazingly clear images of super-small objects.
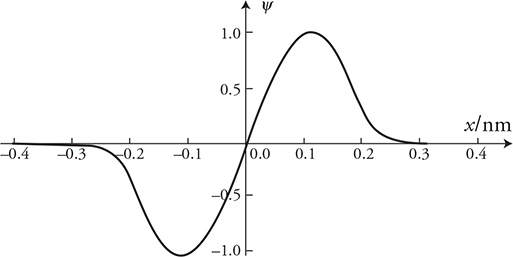
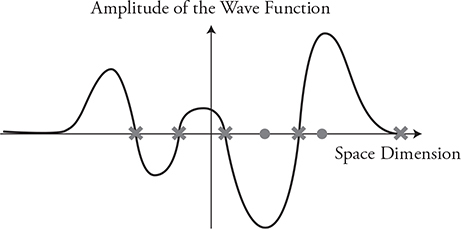
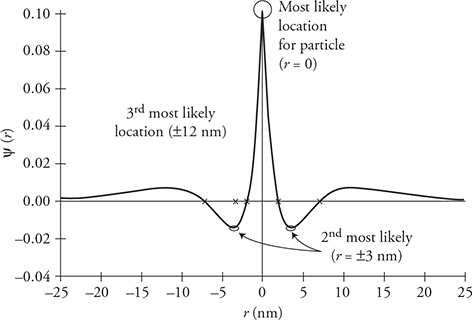
If particles have wave behaviors, what does their wave look like, and what does it mean? Particles will have a wave function, ψ, as a function of location. This can look like the example in the preceding figure. They represent the probability of finding the particle at a particular location. Here is how you read a wave function graph:
1. When the graph reads ψ = 0, there is no probability of ever finding the particle at that location. So in our graph, the particle will never be at location x = 0 and will never be found beyond ±0.3 nm.
2. The larger the amplitude (positive or negative), the more likely you will find the particle. In our graph we are most likely to find the particle at locations ±0.1 nm.
3. Is it possible to find the particle at −0.2 nm? Yes, but not as likely as finding it at ±0.1 nm.
The figure below is another example. You are most likely to find the particle at the locations marked with a circle. You won’t find the particle at the locations marked with an X.
Another example of the wavy behavior of electrons is in the electron energy shells in an atom. Electrons don’t orbit the nucleus in a circular “planetary” pattern. Because of their wave nature, they set up standing waves of constructive interference in whole wavelengths. See the following figure. They can exist only in orbital patterns where they have constructive interference with themselves. At different orbital radii, they will exhibit destructive interference with themselves and cancel themselves out. They simply cannot exist at destructive interference locations.
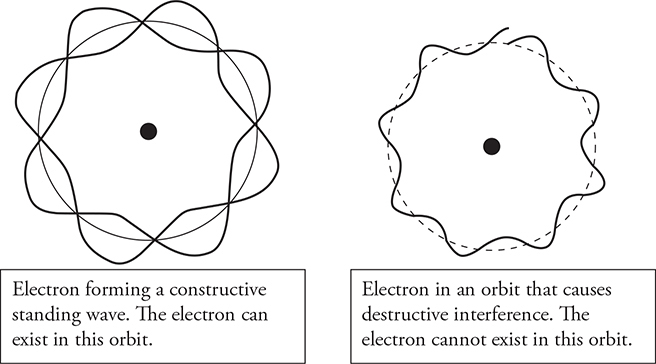
Let’s examine this orbital behavior closer.
Bohr Model of the Atom and Energy Levels in an Atom
Niels Bohr discovered that electrons existed only in specific orbital energy locations due to the constructive and destructive wave nature of electrons. For an electron to move from one of these energy levels to another, the atom had to either absorb or release a little bit of energy called a photon. To make it easier to understand, physicists came up with neat charts that help explain this.
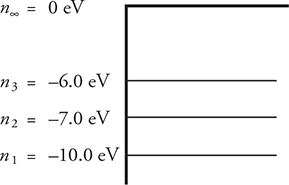
In the figure, you have an energy level diagram for a hypothetical atom—n1, n2, and n3 represent the first three energy levels of that atom. Another way to look at it is that, if the atom is in the n1 level, the path the electron takes around the atom is like a standing wave of one wavelength; n2, the path would be a standing wave of two wavelengths; and so on. n∞ means that the atom is ionized and the electron has had enough energy added to it to completely remove it from the atom. The numbers associated with each level tell you how much energy has to be added to the atom at that particular energy level to remove that electron. For example, in our atom, to completely remove the electron from the atom with the electron in the n1 level, 10 eV of energy must be added. The diagram shows n1 = −10 eV; the negative sign is telling you the atom needs 10 eV of energy before it can be ionized.
A nice way to think of this is that the nucleus is positive, the electron is negative, and it takes work to “pull” the electron away from the nucleus. If the electron is in a higher energy level, such as n2, it takes less energy, only 7 eV, to remove the electron. Bohr called this little bundle of energy a quanta; Einstein later called it a photon. You may be asked to find the energy needed to move the electron from a lower to a higher energy level, or how much energy is released if the electron moves from a higher to a lower energy level. Some hints to help you out:
• n1 is called the ground state. It is the lowest possible energy level for the electron.2
• Moving from a lower to a higher energy level, such as n1 to n2, tells you that the atom absorbed energy in the form of a photon of light.
• Moving from a higher to a lower energy level, such as n2 to n1, tells you that the atom released energy in the form of a photon of light.
• The electron can move only from one exact energy level to another. There are no intermediate levels, such as n1.7. If you think of this like a set of stairs, when you move up and down the stairs, you can land only exactly on each step. So you can be standing on the first step, second step, third step, and so on, but you cannot land on the 1.7 step. In the same way the electron can move up and down only directly from one energy level to another without ever being at an intermediate level.
• Note that I listed the energy levels as n1, n2, and n3. The AP exam has been known to list them as E1, E2, and E3. Or simply as 1, 2, and 3. Don’t let that trouble you! It all means the same thing.
• To find the energy of the absorbed or released photon, all you have to do is subtract the energies of the two energy levels:
![]()
Before moving on let’s take a look at some examples.
Examples
Example 1: What energy photon is absorbed or released in our hypothetical atom for the electron to move from n = 2 to n = 1?
No problem now that you understand what the energy level diagram means. Simply subtract the two energy levels:
![]()
Since the electron went to a lower energy level, you know that a 3-eV photon was released. Since it went from a higher to a lower energy level, that photon of light was released or emitted from the atom. Congratulations! You just solved a quantum physics problem!
Example 2: Our hypothetical atom in the ground state has a 3.5-eV photon incident upon it. Describe what happens.
We know that the electron can move only from one exact energy level to another. So for the electron to jump from n = 1 to n = 2, the atom must absorb exactly 3 eV, and to jump from n = 1 to n = 3, the atom must absorb 4 eV. Since there is no exact jump equal to 3.5 eV, the photon passes through the atom and does nothing.
One more thing to remember:
• When the atom releases energy going from a higher to a lower energy level, every possible combination of downward energy level jumps may happen.
For example:
If an atom is in the n = 4 level and is returning to the n = 1 level, how many different photons may be released?
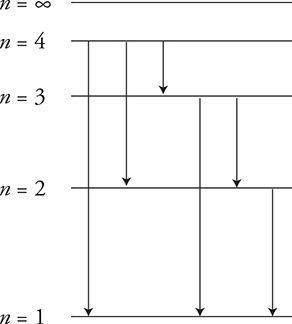
Electrons can jump directly from the n = 4 level to the n = 1 level, or jump from n = 4 to 3 to 2 to 1, releasing three photons. Jumping down from n = 4 to 2 to 1 is another possibility. So every possible jump down from every level can happen, as shown in the diagram above. There are six possible photons that could be emitted in this process.
You know that the electron can jump only from one energy level to another. But what happens if there’s more than enough energy to ionize the atom?
• If a photon is incident upon the atom with an energy equal to or greater than the energy needed to ionize the atom, the electron will be ejected from the atoms. The excess photon energy will be converted into kinetic energy of the released electron.
Let’s look at another example.
Example 3: Our hypothetical atom is again in the ground state and a 12-eV photon is incident on the atom. What happens?
It takes 10 eV to ionize the atom, so there’s more than enough energy to remove the electron. Assuming the photon gets completely absorbed, the electron will be released with a kinetic energy of 12 eV — 10 eV = 2 eV.
Three Types of Nuclear Decay Processes
When physicists first investigated nuclear decay in the early twentieth century, they didn’t quite know what they were seeing. The subatomic particles (protons, neutrons, electrons, and so forth) had not been definitively discovered and named. But, physicists did notice that certain kinds of particles emerged repeatedly from their experiments. They called these particles alpha, beta, and gamma particles.
Years later, physicists found out what these particles actually are:
• Alpha particle, α: two protons and two neutrons stuck together. This is the same thing as a helium nucleus.
• Beta particle, β: an electron or a positron3
• Gamma particle, γ: a gamma ray photon
Be sure you can recall the properties of these particles. A couple of observations: The alpha particle is by far the most massive; the gamma particle is both massless and chargeless.
Nuclear Notation
The two properties of a nucleus that are most important are its atomic number and its mass number. The atomic number, Z, tells how many protons are in the nucleus; this number determines what element we’re dealing with because each element has a unique atomic number. The atomic number also tells you the net positive elementary charge of the nucleus. The mass number, A, tells the total number of nuclear particles a nucleus contains. It is equal to the atomic number plus the number of neutrons in the nucleus. Like the name implies, the mass number tell you the approximate mass of the nucleus in atomic mass units “u”. Isotopes of an element have the same atomic number, but different mass numbers.
A nucleus is usually represented using the following notation:
![]()
where “symbol” is the symbol for the element we’re dealing with. For example, ![]() represents helium with two protons and two neutrons. A different isotope of helium might be
represents helium with two protons and two neutrons. A different isotope of helium might be ![]() , which contains three neutrons.
, which contains three neutrons.
This is not a chemistry exam, you don’t have to memorize the periodic table, nor do you have to remember what element is number 74.4 But you do have to recognize the significance of the atomic number and the mass number. Both numbers are conserved in nuclear reactions. This is explained in detail in the following sections.
Alpha Decay

Alpha decay happens when a nucleus emits an alpha particle. Since an alpha particle has two neutrons and two protons, then the daughter nucleus (the nucleus left over after the decay) must have two fewer protons and two fewer neutrons than it had initially.
Example: ![]() undergoes alpha decay. Give the atomic number and mass number of the nucleus produced by the alpha decay.
undergoes alpha decay. Give the atomic number and mass number of the nucleus produced by the alpha decay.
The answer is found by simple arithmetic. The atomic number decreases by two, to 90. The mass number decreases by four, to 234. (The element formed is thorium, but you don’t have to know that.) This alpha decay can be represented by the equation below:
![]()
Beta Decay

In beta decay, a nucleus emits either a positron (β+ decay) or an electron (β— decay). Because beta particles have very little mass and do not reside in the nucleus, the total mass number of the nucleus will stay the same after beta decay. The total charge must also stay the same—charge is a conserved quantity. So, consider an example of neon (Ne) undergoing β+ decay:
![]()
Here e+ indicates the positron. The mass number stayed the same. But look at the total charge present. Before the decay, the neon nucleus has a charge of +10. After the decay, the total net charge must still be +10, and it is: +9 for the protons in the fluorine (F), and +1 for the positron. Effectively, then, in β+ decay, a proton turns into a neutron and emits a positron.
For β− decay, a neutron turns into a proton, as in the decay process important for carbon dating:
![]()
The mass number of the daughter nucleus didn’t change. But the total charge of the carbon nucleus was initially +6. Thus, a total charge of +6 has to exist after the decay, as well. This is accounted for by the electron (charge −1) and the nitrogen (charge +7).
In beta decay, a neutrino or an antineutrino also must be emitted to carry off some extra energy. But that doesn’t affect the products of the decay, just the kinetic energy of the products.
Gamma Decay

A gamma particle is a photon, a particle of light. It has no mass and no charge. So a nucleus undergoing gamma decay does not change its outward appearance:
![]()
However, the photon carries away some energy and momentum, so the nucleus recoils.
Here is a decay we have not discussed—neutron decay.
Fill in the blank in the following diagram with what is missing.
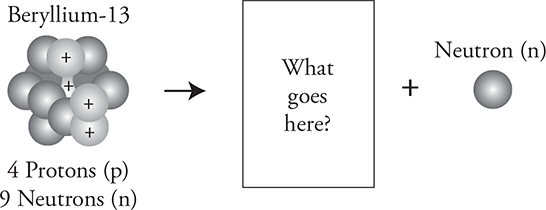
No worries. A neutron, which has a mass number of 1 and an atomic number of zero because it has no charge, has been ejected. Therefore, what remains behind must have 4 protons and 8 neutrons. So, it has to be a different isotope of beryllium: ![]() .
.
E = mc2 and Conservation During Nuclear Reactions

We can imagine that the nuclei in the last few examples are at rest before emitting alpha, beta, or gamma particles. But when the nuclei decay, the alpha, beta, or gamma particles come whizzing out at an appreciable fraction of light speed.5 So, the daughter nucleus must recoil after decay in order to conserve momentum.
But now consider energy conservation. Before decay, no kinetic energy existed. After the decay, both the daughter nucleus and the emitted particle have gobs of kinetic energy.6 Where did this energy come from? Amazingly, it comes from mass.
The total mass present before the decay is very slightly greater than the total mass present in both the nucleus and the decay particle after the decay. That slight difference in mass is called the mass defect, often labeled Δm. This mass is destroyed and converted into kinetic energy.
How much kinetic energy? Use Einstein’s famous equation to find out. Multiply the mass defect by the speed of light squared:
![]()
And that is how much energy is produced.
Let’s review before moving on. Things that are conserved during nuclear reactions:
1. Conservation of mass/energy—In every naturally occurring nuclear reaction, mass of the original nucleus is lost to other forms of energy such as photons or kinetic energy, and the final mass is less than the original mass. Less mass means more nuclear stability.
2. Conservation of momentum—When a nucleus undergoes decay and shoots off a particle, there is a recoil just like when a bullet is fired from a gun.
3. Conservation of charge—The sum of the atomic numbers before and the sum of the atomic numbers after the nuclear reaction are equal.
4. Conservation of nucleons—The sum of the neutrons and protons (atomic mass number) before and after must be the same.
Half-Life
Radioactive isotopes decay at different rates. The decay rate is called half-life, and it is the time it takes for half of the radioactive atoms to decay (transmute) into a new nucleus. Uranium-238 has a half-life of 4.5 billion years. Some elements have a half-life of less than a second! Don’t blink . . . or it will already be transmuted into something new.
Which material is more dangerous? One with a long half-life like uranium or one with a short half-life?
A short half-life = fast decay rate = lots of radiation in the short term and then it is gone. So, short half-life materials are likely to kill you quick and then disappear /transmute.
A long half-life = slow decay rate = small radiation emissions = little danger to you. But the material will be around for a long time.
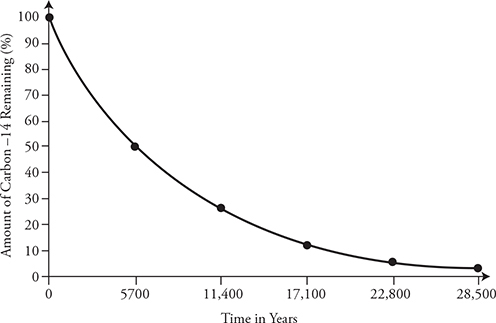
Carbon-14 has a half-life of 5700 years. Look at the graph above. Starting at time = 0, we have 100% of the original carbon-14. After 5700 years, only 50% is left. After two half-lives, only 25% is left (that’s ½ times ½). After 28,500 years, or five half-lives, there is only about 3% of the original material left. Five half-lives is ½ × ½ × ½ × ½ × ½ = 1⁄32 = 3.125%.
Decay rates appear to be independent of external conditions like temperature, electric or magnetic fields, and even chemical reactions. How do scientists measure half-life? Well, they can’t actually count the atoms because they are tiny, and we can’t see atoms. So, scientists measure the radioactive activity with a Geiger counter. A Geiger counter literally counts the number of decay products like alpha and beta particles that are emitted by the radioactive material.

Look at the preceding activity count graph. What is the half-life of this material? Look to see how long it takes the count to be cut in half. The time to go from 8 to 4 counts is about 80 seconds. This is experimental data, which always has some errors or uncertainty, so let’s check with another data point. The time it takes to go from 4 to 2 counts is about 100 s. So, the half-life is approximately 90 seconds.
Other Nuclear Reactions
Decay is not the only type of nuclear reaction. There are lots of different kinds. The following are a few that you may run across on the exam.
Fission
Fission occurs when a heavy nucleus is split into two relatively large chunks. Fission reactions are begun by shooting a neutron into the nucleus, which initiates the reaction. Large amounts of energy are created as mass converts to energy in the reaction. This is the reaction in nuclear power plants and nuclear weapons. Below is an example of a plutonium fission reaction. Can you figure out what goes where the question mark is? (See the Answers section that follows.)
![]()
Fusion
Fusion occurs when two light nuclei combine to make a new heavier and more stable nucleus. Large amounts of energy are released in the reaction. This is what happens in the sun. Here is an example of fusion. Notice how one of the products of the reaction is larger than any of the originals. Where did all the energy come from, and what does all that energy do? (See the Answers section that follows.)
![]()
Induced Reaction
Sometimes scientists bombard a nucleus with high-speed particles to see what will happen. This is called an induced reaction because you are causing a reaction to happen. Rutherford was the first to do this in 1919 when he bombarded a stable isotope of nitrogen with a particle to induce a transmutation into oxygen and a proton. Here is the Rutherford reaction. What did he bombard the nitrogen with, and where do you think he got this particle from? (See the Answers section that follows.)
![]()
Antimatter and Annihilation
Every “normal” particle has an antimatter twin. For instance, an electron has an antimatter twin called a positron. Positrons are exactly like electrons in every way except they have a positive charge. A curious behavior is that when matter and antimatter meet, they annihilate each other. All the mass is converted into energy in the form of photons. For example: an electron and positron greet each other and shake hands. Oh no, annihilation! The electron and positron disappear and turn into photon energy. How much energy will be converted into photon energy? Both turn into energy. Both have the same mass:
![]()
Question: Why does the electron—positron annihilation not just produce one photon? Think about all the conservation laws we have to abide by. (See the Answers section that follows.)
Answers
Fission: 3 neutrons
Fusion: The energy produced in the reaction comes from a loss of mass E = mc2. As for what all that energy does . . . it creates photons of light energy that are emitted. This is why the sun glows and keeps us toasty warm.
Induced Reaction: He bombarded it with an alpha particle or helium nucleus. Rutherford got his alpha particles from the alpha decay of other radioactive isotopes, though he could have stripped helium gas of its electrons as well.
Antimatter and Annihilation: Due to conservation of momentum, two photons of the same energy traveling in the opposite direction have to be created so that the final momentum is zero, just like the initial momentum.
Mass Defect, Binding Energy, and the Strong Nuclear Force
Carbon-12 is made up of 6 protons and 6 neutrons. We know the mass of a proton. We know the mass of a neutron. So, you would think that if we added the mass of 6 protons to the mass of 6 neutrons, we would get the mass of a carbon-12 nucleus. Remarkably, the mass of the carbon-12 is a little less than we expect! Scientists call this a mass defect, Δm.
What has happened to the lost mass? The small amount of missing mass has been converted into energy (E = Δmc2) that holds the nucleus together. The mass defect has become the nuclear binding energy and is equivalent to the strong nuclear force holding the nucleus together. If you wanted to break the carbon-12 apart into separate protons and neutrons, you would need to supply enough energy to the nucleus to re-create the mass that is missing ![]() .
.
Because physicists so often need to convert matter into energy and back again, they took Einstein’s equation and a bunch of conversions to give you the following relationship:
![]()
Where u is called the unified atomic mass unit. Remember, that MeV is a mega-electron-volt. Don’t worry so much about the c2, so long as you know that 1 u is 931 MeV when converted to energy.
Let’s take a look at an example.
Example
Given the following masses, what is the mass defect and binding energy for the helium atom?
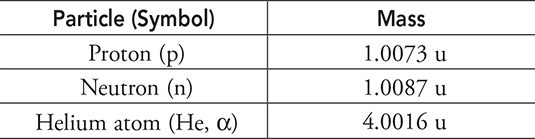
Let’s first find the mass defect. It’s really simple to do, just subtract the mass of the atom from the mass of its separate particles.
The helium atom is made of 2 protons plus 2 neutrons, so:
![]()
Now subtract the mass of the atom from its separate parts:
![]()
This is the mass defect. It is the amount of matter that’s converted to binding energy and released from the atom when its component nucleons7 are put together.
Now we want to convert that amount of matter into energy. You could convert the mass into kilograms, use E = mc2 to change the mass into energy, and then convert the energy from joules into mega-electron-volts, but it is so much easier to use the conversion listed above and on the reference table:
![]()
So there is 28.3 MeV of energy that would have to be added back to the helium nucleus to tear it apart into four separate nucleons.
The mass of a nucleus is less than the mass of its constituent parts. The bigger the mass defect, the harder it will be to break the nucleus apart, and the more stable the nucleus will be. In essence, the mass defect is a measure of how large a nuclear force holding the nucleus together is. Remember that the nucleus is populated with protons that repel each other. The force holding it all together must be very strong. Thus the name given to this binding force is: the strong nuclear force. (May the force be with you!)
❯ Practice Problems
Multiple Choice
1. Which of the following lists types of electromagnetic radiation in order from least to greatest energy per photon?
(A) ultraviolet, infrared, red, green, violet
(B) red, green, violet, infrared, ultraviolet
(C) infrared, red, green, violet, ultraviolet
(D) ultraviolet, violet, green, red, infrared
Questions 2 and 3
In a nuclear reactor, uranium fissions into krypton and barium via the reaction
![]()
2. What are the mass number A and atomic number Z of the resulting krypton nucleus?

3. How much mass is converted into the kinetic energy of the resulting nuclei?
(A) 1 amu
(B) 2 amu
(C) zero
(D) much less than 1 amu
4. ![]() decays via β+ emission. Which of the following is the resulting nucleus?
decays via β+ emission. Which of the following is the resulting nucleus?
(A) ![]()
(B) ![]()
(C) ![]()
(D) ![]()
5. A physicist has two samples of radioactive material with these properties:
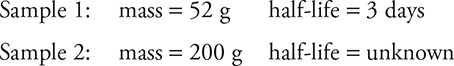
After 12 days, both samples have approximately the same mass of radioactive material remaining. The half-life of sample 2 is most nearly which of the following?
(A) 6 days
(B) 4 days
(C) 3 days
(D) 2 days
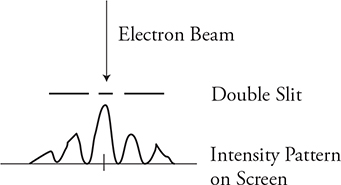
6. An electron beam is shot through a double slit, which produces the intensity pattern shown in the figure. Which of the following diagrams best represents the intensity pattern resulting when the velocity of the electron beam is reduced?
(A) 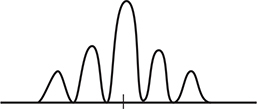
(B) ![]()
(C) 
(D) 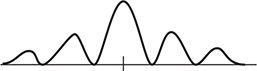
7. A uniform ultraviolet light source shines on two metal plates causing electrons to be emitted from each plate. Plate A emits twice as many electrons as plate B. However, the electrons emitted from plate B have a higher maximum velocity. Which of the following describes a plausible explanation for the differences in electron emission? (Select two answers.)
(A) Plate A must have a larger work function than plate B.
(B) The higher velocity electrons in case B would be produced by placing plate B closer to the light source, where plate B would receive more intense ultraviolet light from the source.
(C) More electrons would be produced from plate A if it were larger in area than plate B.
(D) Due to the unpredictability inherent in the wave nature of electrons, it is possible for more electrons with less energy to be emitted from one plate while fewer electrons are emitted with more energy from a second plate, as long as the total energy of the electrons from both plates equals the absorbed energy of the ultraviolet light.
8. In which of the cases listed below would it be important to consider the particle nature of electromagnetic radiation? (Select two answers.)
(A) Ultraviolet light of various intensities shining on a sodium sample produce ejected electrons of uniform maximum kinetic energy.
(B) Red laser light passing through two adjacent narrow slits produces a pattern of light and dark red lines on a white screen.
(C) A car driver can eliminate reflected road glare from the sun by wearing polarized glasses.
(D) NASA engineers have developed a method for propelling spaceships utilizing large sails that reflect sunlight.
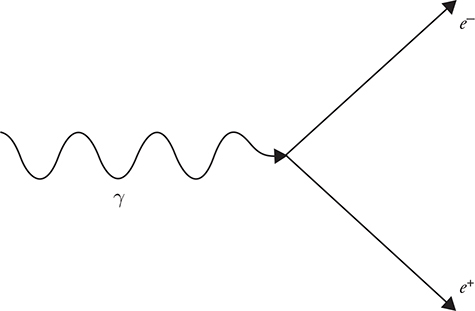
9. Under the right conditions, a photon of light can be converted into a positron and electron, as shown in the figure and represented in the following equation: γ → e+ + e-. Which of the following correctly explain why this interaction is possible? (Select two answers.)
(A) The positron is the antimatter particle of the electron, so the mass of the two particles cancel.
(B) The signs of the positron and the electron cancel.
(C) The momentum of the two particles sum to that of the photon.
(D) The kinetic energy of the two particles sum to that of the photon.

10. The figure shows energy levels for a gas. Electrons in the ground state are excited by a continuous range of electrical energy from 100 eV to 115 eV. After the electrical input is turned off, the gas emits light in discrete frequencies. Which of the following correctly indicates the electron transition from the gas that would produce the highest frequency light?
(A) n = 5 to n = 4
(B) n = 4 to n = 3
(C) n = 3 to n = 2
(D) n = 5 to n = 2
11. ![]() Which of the following correctly expresses the energy of the released gamma ray in the reaction represented by the equation shown?
Which of the following correctly expresses the energy of the released gamma ray in the reaction represented by the equation shown?
(A) mC + mH — mN
(B) (mC + mH — mN)c2
(C) (mC + mH — mN)c2 — hfγ
(D) (mC — mH — mN)c2
Free Response
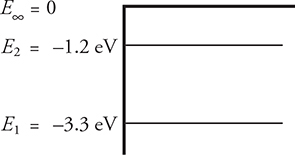
12. A hypothetical atom has two energy levels, as shown above.
(A) What wavelengths of electromagnetic radiation can be absorbed by this atom? Indicate which of these wavelengths, if any, represents visible light.
(B) Now, monochromatic 180-nm ultraviolet radiation is incident on the atom, ejecting an electron from the ground state. What will be
(i) the ejected electron’s kinetic energy
(ii) the ejected electron’s speed
(iii) the incident photon’s speed
For parts (C) and (D), imagine that the 180-nm radiation ejected an electron that, instead of being in the ground state, was initially in the —1.2 eV state.
(C) Would the speed of the ejected electron increase, decrease, or stay the same? Justify your answer briefly.
(D) Would the speed of the incident photon increase, decrease, or stay the same? Justify your answer briefly.
13. Explain why ultraviolet (UV) light can discharge a negatively charged electroscope but not a positively charged electroscope.
14. Sodium has a work function of 2.4 eV. Iron has a work function of 4.7 eV. Cesium has a threshold frequency of 4.7 × 1014 Hz.
(A) Calculate the threshold frequency of sodium and iron. Calculate the work function of cesium.
(B) Plot photoelectron energy as a function of photon frequency for the three elements. Label the slope.
(C) Ultraviolet light (200 nm) shines on cesium. What is the maximum energy of ejected electrons?
15. Which of the following behaviors of light support the wave model of light, and which support the particle model of light? Justify your response in each case.
(A) Light that passes through a double slit produces an alternating pattern of intensity.
(B) X-rays can be used to ionize gas.
(C) X-rays can be directed at crystals to produce interference patterns.
(D) Photons of light can impart momentum to electrons in a collision.
(E) Long wavelength radio waves can bend around hills and buildings.
(F) Light from distant stars is redshifted.
(G) Infrared (IR) light does not generate electricity in a solar cell no matter how intense the light is.
16. A photon moving to the right collides with a stationary electron, as shown in the figure. The photon recoils to the left along the same path.
(A) Describe any changes to the photon. Justify your claims.
(B) Describe any changes to the electron. Justify your claims.
17. Visible light shines on an unknown gas. The gas is found to absorb light of a 400-nm wavelength. When the light is turned off, the gas is seen to emit both 400-nm and 600-nm wavelengths of light.
(A) Draw an energy level diagram for this gas. Label the energy levels in electron volts.
(B) Is there another wavelength emission that is not in the visible spectrum? Justify your answer with your diagram.

18. A gun fires electrons at a barrier with two small openings. A screen registers electron impact locations as shown in the figure.
(A) What does this prove about the nature of electrons?
(B) What happens to the pattern when the voltage used to accelerate the electrons is increased? Justify your answer with an equation.
(C) What happens to the pattern if the distance between the openings is decreased? Justify your answer with an equation.
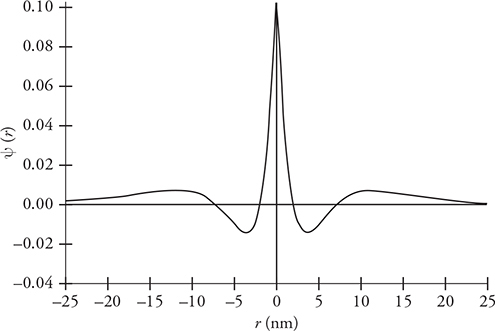
19. The figure represents the wave function of an unknown particle.
(A) On the figure, circle the locations where the particle is most likely to be found. Rank the locations from most likely to least likely.
(B) Place an X on the horizontal axis where the particle will never be found. If there are no locations where the particle will never be found, explain why this is.
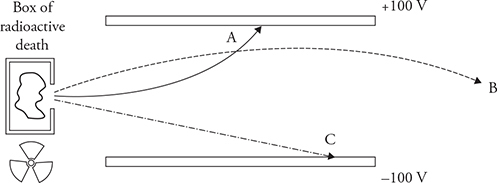
20. Scientists in the early 1900s discovered that some materials (radioactive substances) emitted strange, unknown rays, originally called Becquerel rays. In an effort to understand the nature of these rays, scientists sent them through electric fields and observed the results. The figure shows the path of three rays that exit a box of radioactive material.
(A) Determine the direction of the electric field through which the rays are being sent. Justify your claim.
(B) What can be learned about rays A, B, and C from this experiment? Explain.
(C) What could each of the rays be? Justify your answer.
21. Derive a mathematical equation for the mass defect (Δm) of a helium nucleus. Justify your equation.
22. Decide if the following nuclear reactions are possible. If not, explain why not.
(A) ![]()
(B) ![]()
23. Complete the following reactions and classify as either decay (specify the type), fission, or fusion.
(A) ![]()
(B) ![]()
(C) ![]()
(D) ![]()
(E) ![]()

24. (A) Use the figure to estimate the half-life of the radioactive sample.
(B) Explain what the term half-life means. Be sure to include reactants and products in your discussion.
❯ Solutions to Practice Problems
1. (D) The radiation with the highest frequency (or shortest wavelength) has the highest energy per photon by E = hf. In the visible spectrum, red has the longest wavelength and violet has the shortest. Outside the visible spectrum, infrared radiation has a longer wavelength than anything visible, and ultraviolet has a shorter wavelength than anything visible. So, infrared has the smallest energy per photon, and so on up the spectrum to ultraviolet with the most energy per photon.
2. (A) The total number of protons + neutrons is conserved. Before the reaction, we have one free neutron plus 235 protons and neutrons in the uranium, for a total of 236 amu. After the reaction, we have 141 amu in the barium plus 3 free neutrons for a total of 144 amu . . . leaving 92 amu for the krypton.
Charge is also conserved. Before the reaction, we have a total charge of +92 from the protons in the uranium. After the reaction, we have 56 protons in the barium. Since a neutron carries no charge, the krypton must account for the remaining 36 protons.
3. (D) Einstein’s famous equation is written ΔE = Δmc2, because it is only the lost mass that is converted into energy. Since we still have a total of 236 amu after the reaction, an entire amu of mass was not converted to energy. Still, the daughter particles have kinetic energy because they move. That energy came from a very small mass difference, probably about a million times less than one amu.
4. (C) In β+ emission, a positron is ejected from the nucleus. This does not change the total number of protons + neutrons, so the atomic mass A is still 15. However, charge must be conserved. The original O nucleus had a charge of +8, so the products of the decay must combine to a charge of +8. These products are a positron, charge +1, and the daughter nucleus, which must have charge +7.
5. (D) Twelve days is four half-lives for sample 1. In four half-lives, only 1⁄16 (3.25 g) of the original 52-g sample will remain. For sample 2 to reach 3.25 grams in the same time, it must have a shorter half-life. Answer choice D is the only choice shorter than 3 days. However, to confirm the answer, 3.25 grams is approximately 1⁄64 of the original 200 grams. That is six half-lives of decay, or a half-life of two days.
6. (D) Reducing the velocity also reduces the momentum of the electron. This will increase the wavelength of the electron ![]() . Increasing the wavelength also increases the pattern spacing (d sin θ = mλ).
. Increasing the wavelength also increases the pattern spacing (d sin θ = mλ).
7. (A) and (C) All the photons of the uniform UV light have the same energy, which is entirely imparted to the electrons on a one-to-one basis. Thus, plate A must have electrons that are harder to remove (higher work function). Placing the light source closer to the plate or having a larger surface area would account for more electrons being emitted from plate A. Moving the light closer to the plate does not increase the energy of the individual photons and will not increase the energy of the ejected electrons.
8. (A) and (D) The behavior of ejected electrons from a surface due to electromagnetic radiation (the photoelectric effect) was the experiment that first proved light waves have particle properties. Light waves reflecting off “solar sails” to propel spacecraft can only be understood by modeling light as a photon particle that collides with the sail, imparting momentum to the sail during the collision.
9. (B) and (C) Conservation of charge is satisfied because the net charge before and after is zero. Conservation of momentum tells us that the initial momentum of the gamma ray must be equal to the net momentum of the two particles. The gamma ray has x-direction momentum and no y-direction momentum. This appears to be satisfied in the figure. The answer choice D is incorrect; we have to use conservation of mass/energy because most of the gamma ray energy has been converted into mass, not just kinetic energy of the two particles.
10. (C) Adding the excitation energy of 100 eV −115 eV to the ground state energy, we get the electron energy range of (−22.4 eV) - (−7.4 eV). This will take the electrons of the gas from the ground state only to the n = 3 and 4 energy levels. The highest frequency photon occurs when the electron jumps down in energy by the largest amount: E = hf. Therefore, the transition from n = 3 to n = 2 is the answer. Answer choices A and D are not possible because the electrons were not excited to the n = 5 level.
11. (B) The energy of the gamma ray comes from the mass that is lost in the reaction. Adding the mass of the reactants, subtracting the mass of the products, and converting into energy via E = mc2 gives us the mass defect that was converted into the energy of the gamma ray. This is an application of conservation of mass/energy.
12. (A) ΔE = hc/λ, so hc/ΔE = λ. hc = 1240 eV·nm, as found on the equation sheet. ΔE represents the difference in energy levels. An electron in the ground state can make either of two jumps: it could absorb a 2.1-eV photon to get to the middle energy level, or it could absorb a 3.3 eV photon to escape the atom. An electron in the middle state could absorb a 1.2-eV photon to escape the atom. That makes three different energies. Convert these energies to wavelengths using ΔE = hc/λ, so hc/ΔE = 1. hc = 1240 eV·nm, as found on the equation sheet; ΔE represents the energy of the absorbed photon, listed above. These photons thus have wavelengths of 590 nm for the E1 to E2 transition; 380 nm or less for the E1 to E∞ transition; and 1030 nm or less for the E2 to E∞ transition. Only the 590 nm wavelength is visible because the visible spectrum is from about 400—700 nm.
(B) (i) Find the energy of the incident photon using ΔE = hc/λ, getting 6.9 eV. This is the total energy absorbed by the electron, but 3.3 eV of this is used to escape the atom. The remaining 3.6 eV is kinetic energy.
(ii) To find speed, set kinetic energy equal to ½mv2. However, to use this formula, the kinetic energy must be in standard units of joules. Convert 3.6 eV to joules by multiplying by 1.6 × 10−19 J/eV (this conversion is on the constant sheet), getting a kinetic energy of 5.8 × 10−19 J. Now solve the kinetic energy equation for velocity, getting a speed of 1.1 × 106 m/s. This is reasonable—fast, but not faster than the speed of light.
(iii) A photon is a particle of light. Unless it is in an optically dense material (which the photons here are not), the speed of a photon is always 3.0 × 108 m/s.
(C) The electron absorbs the same amount of energy from the incident photons. However, now it takes only 1.2 eV to get out of the atom, leaving 5.7 eV for kinetic energy. With a larger kinetic energy, the ejected electron’s speed is greater than before.
(D) The speed of the photon is still the speed of light, 3.0 × 108 m/s.
13. Negativly charged objecs have more electrons than protons. Ultraviolet photons have enough energy to dislodge electrons from a object, which can eliminate the excess of electrons from the negatively charged object. This will neutralize the object’s charge. On the other hand, positively charged objects have more protons than electrons. The only way to discharge a positively charged object is to add electrons to the object or remove protons from the object. A UV photon cannot do either of these.
14. (A) At the threshold frequency, the kinetic energy of the photoelectrons is zero:
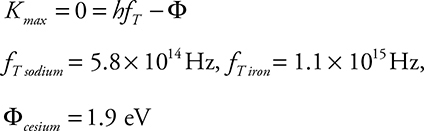
(B)
(C) 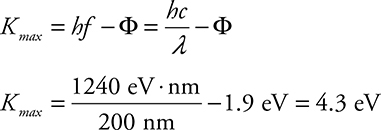
15. (A) Wave property: Only waves exhibit interference.
(B) Particle property: Light behaves like a particle by colliding with an electron, thus imparting momentum to the electron and knocking it from the atom.
(C) Wave property: Interference is a wave property.
(D) Particle property: Collisions and momentum are particle properties.
(E) Wave property: Diffraction, the bending of waves around boundaries, is a wave property.
(F) Wave property: The Doppler effect is a property of waves.
(G) Particle property: Solar cells, or photovoltaic cells, are a practical application of the photoelectric effect, which demonstrates the particle nature of light.
16. (A) The X-ray will lose energy and momentum. Conservation of momentum must be obeyed in the collision. Since the electron gained momentum in the collision, the photon must lose momentum. Also, the electron gains kinetic energy in the collision. Therefore, the photon must lose energy. Losing momentum and energy means the frequency of the X-ray has decreased: E = hf = pc.
(B) The electron gains both kinetic energy and momentum to the right from the collision with the X-ray.
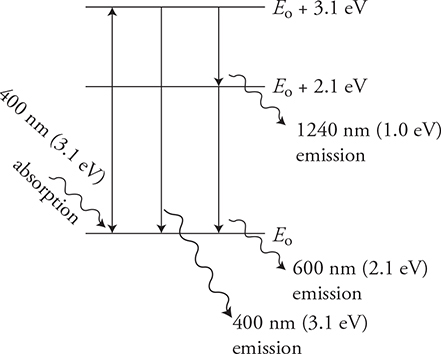
17. (A) The problem does not tell us the starting position of the electron or its initial energy. So, let’s just label it at E0. Using the equation 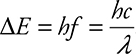 , we can calculate the change in energy of the electrons in the atom associated with the photons:
, we can calculate the change in energy of the electrons in the atom associated with the photons:
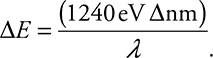
The wavelengths of 400 nm and 600 nm give us energies of 3.1 eV and 2.1 eV, respectively. See figure.
(B) Yes! The figure shows that when the electron is in the E0 + 3.1 eV energy level, it can fall to the E0 + 2.1 eV energy level. This will produce a 1240 nm/1.0 eV photon that is not in the visible spectrum, so it will not be seen.
18. (A) This is an interference pattern. Therefore, electrons are exhibiting wave properties (wave-particle duality).
(B) When the accelerating voltage increases, the electron velocity increases: 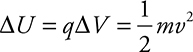 When electron velocity increases, the de Broglie wavelength of the electron decreases:
When electron velocity increases, the de Broglie wavelength of the electron decreases: 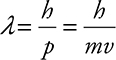
When wavelength decreases, the pattern gets tighter. The angle of the maxima decreases: dsin θ = mλ.
(C) The interference pattern will become more spread out as the distance d decreases: 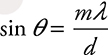 .
.
19. (A) and (B) See figure.
20. (A) This is a capacitor. The top plate has a higher electric potential. The electric field is directed straight down toward the bottom plate. The electric field is uniform in strength and direction between the plates except for at the edges of the capacitor.
(B) Ray C must not have a charge because it is not affected by the electric field. Ray B curves in the direction of the electric field. Therefore, it must be receiving a force from the field and must be positively charged. Ray A must be negative as it receives a force in the opposite direction of the electric field and curves upward.
(C) Ray C could be a neutron or a gamma ray because neither has an electric charge. Ray B could be a proton or an alpha particle. Ray A could be an electron. Ray A curves more tightly, which could be accounted for by an electron having less mass and therefore more acceleration than an alpha particle or a proton as in the case of ray B. However, we do not know the speed of the particles exiting the box. So it is impossible to draw any definitive conclusions by comparing the curve radius of ray A and B.
21. Δm = (2mneutron + 2mproton) — mHe. The mass defect will be the difference in mass between the helium nucleus and the masses of all the parts that are used to construct the nucleus (two neutrons and two protons).
22. (A) Not possible! The number of nucleons is not conserved.
(B) Not possible! Conservation of charge is violated.
23. (A) ![]()
(B) ![]()
(C) ![]()
(D) ![]()
(E) ![]()
24. (A) Approximately 3.5 years.
(B) Half-life is the time it takes for one half of a radioactive sample to decay into a new element. After two half-lives, a quarter of the original radioactive substance will remain. This means that three quarters of the original material has transmuted into a new element.
❯ Rapid Review
• Time and space (length) are not constants. They depend on the speed of the object or person making the observations.
• Mass and energy are two aspects of the same thing and can be converted back and forth.
• The photoelectric effect experiment proved that light has particle properties.
• Photons are massless particles of light. Their energy depends on their frequency. Higher frequency = higher energy.
• Photons have momentum and can interact with particles, creating collisions similar to objects colliding. When photons lose momentum and energy, their frequency decreases and their wavelength gets longer.
• Particles can display wave properties. The wave function of a particle is interpreted to be the probability of finding the particle at a location.
• Atoms contain a nucleus, made of protons and neutrons, and one or more electrons that orbit that nucleus. Protons, neutrons, and electrons all have mass. By contrast, photons are subatomic particles without mass.
• The electron-volt is a unit of energy that’s convenient to use when solving atomic physics problems.
• The electrons that surround an atom can have only certain, specific amounts of energy because of the wave nature of electrons. To go from a low-energy level to a high-energy level, an electron absorbs a photon. To go from a high-energy level to a low-energy level, an electron emits a photon.
• If an electron absorbs a photon that has a higher energy than the electron’s work function, the electron will be expelled from the atom.
• Moving particles have a characteristic wavelength, found by the de Broglie equation.
• Nuclei can undergo several types of decay. In alpha decay, a nucleus emits an alpha particle, which consists of two protons and two neutrons. In beta decay, a nucleus emits either a positron or an electron. In gamma decay, a nucleus emits a photon.
• When solving nuclear problems, remember to conserve nucleon number, nuclear charge, mass/energy, and momentum.
• During nuclear decay, mass is converted to energy. The relationship between the mass defect and the gained energy is found by Einstein’s famous formula, E = (Δm)c2.
• Half-life is the time it takes one-half of the radioactive material to decay.
1Postulate is just a fancy word for a statement that is assumed to be true based on reasoning.
2Yes, we did say in Chapter 14 that accelerating charges give off electromagnetic radiation. This is one way quantum physics differs from classical physics. In quantum physics, it is possible for the electron to accelerate in an orbit and not emit energy.
3The positron is the antimatter equivalent of an electron. It has the same mass as an electron, and the same amount of charge, but the charge is positive. Since there are two different types of beta particles, we often write them as β+ for the positron and β− for the electron.
4Tungsten: symbol “W.” Used in lightbulb filaments.
5100% of the speed of light in the case of a gamma particle!
6No, the energy doesn’t cancel out, because the nucleus and the particle move in opposite directions; energy is a scalar, so direction is irrelevant.
7Remember that a nucleon is a proton or neutron.