SAT Physics Subject Test
Chapter 15 Thermal Physics
THE IDEAL GAS LAW
Three physical properties—pressure (P), volume (V), and temperature (T)—are commonly used to describe a gas. Ideal gases exhibit certain properties: the volume of the gas molecules is negligible compared with that of the container that holds them, they experience no electrical forces, and undergo elastic collisions. These three variables are related by the equation
PV = nRT
where n is the number of moles of gas and R is a constant (8.31 J/mol·K) called the universal gas constant. This equation is known as the ideal gas law.
This equation tells us that for a fixed volume of gas, an increase in P gives a proportional increase in T. The pressure increases when the gas molecules strike the walls of their container with more force, which occurs if they move more rapidly.
Also, the equation
Kavg = ![]() kBT
kBT
tells us that the average translational kinetic energy of the gas molecules is directly proportional to the absolute temperature of the sample.
From this we can find root-mean-square (a kind of average) speed of the gas molecule.
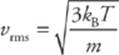
Because kB = R/NA and mNA = M (the mass of one mole of the molecules—the molar mass), the equation for vrms is also often written as follows:
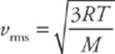
Notice that these last two equations can only determine vrms. It”s important to realize that the molecules in the container have a wide range of speeds; some are much slower and others are much faster than vrms. The root-mean-square speed is important because it gives us a type of average speed that”s easy to calculate from the temperature of the gas.
![]()
8. What happens to the rms speed of the molecules in a sample of helium gas if the temperature is increased from –73°C to 527°C ?
Here”s How to Crack It
When we use the ideal gas law or the formulas for vrms, we have to use absolute temperatures—that is, temperatures expressed in kelvins. The conversion between degrees Celsius and kelvins is T°C + 273 = T, so –73°C = 200 K and 527°C = 800 K. Therefore, the absolute temperature of the gas is increased by a factor of 4 (from 200 K to 800 K). Since vrms is proportional to ![]() , if T increases by a factor of 4, then vrms increases by a factor of
, if T increases by a factor of 4, then vrms increases by a factor of ![]() = 2.
= 2.
![]()
![]()
9. What happens to the pressure of a sample of helium gas if the temperature is increased from 200 K to 800 K, with no change in volume?
Here”s How to Crack It
The ideal gas laws, PV = nRT, tells us that if V remains constant, then P is proportional to T. So, if T increases by a factor of 4, then so will P.
![]()
![]()
10. What happens to the pressure of a sample of helium gas if the volume is reduced from 6 liters to 3 liters, with no change in temperature?
Here”s How to Crack It
The ideal gas law, PV = nRT, tell us that if T remains constant, then P is inversely proportional to V. So, if V decreases by a factor of 2, then P will increase by a factor of 2.
![]()