SAT Physics Subject Test
Chapter 16 Modern Physics
THE BOHR MODEL OF THE ATOM
In the years immediately following Rutherford”s announcement of his nuclear model of the atom, a young physicist, Niels Bohr, added an important piece to the atomic puzzle.
For fifty years it had been known that atoms in a gas discharge tube emitted and absorbed light only at specific wavelengths. The light from a glowing gas, passed through a prism to disperse the beam into its component wavelengths, produced patterns of sharp lines called atomic spectra. The visible wavelengths that appeared in the emission spectrum of hydrogen had been summarized by a simple formula. But why do atoms emit (or absorb) radiation only at certain discrete wavelengths?
Bohr”s model of the atom explains. Using the simplest atom, hydrogen (which has one electron), Bohr postulated that the electron orbits the nucleus only at certain discrete radii. When the electron is in one of these special orbits, it does not lose energy (as the classical theory would predict). However, if the electron absorbs a certain amount of energy, it is excited to a higher orbit, one with a greater radius. After spending a short time in this excited state, it returns to a lower orbit, emitting a photon in the process. Since each allowed orbit—or energy level—has a specific radius (and corresponding specific energy), the photons emitted in each jump also have specific energies and wavelengths. We say that the electron”s energy levels are quantized.
The energy levels within the hydrogen atom are given by the formula
![]()
and for other one-electron atoms—ionized helium (Z = 2), doubly-ionized lithium (Z = 3), etc.—the energy levels are
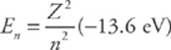
where Z is the number of protons in the atom”s nucleus.
When an excited electron drops from energy level n = j to a lower one, n = i, the transition causes a photon of energy to be emitted, and the energy of a photon is the difference between the two energy levels:
Eemitted photon = |∆E| = Ej − Ei
![]()
Questions 4-5
Refer to the diagram below.
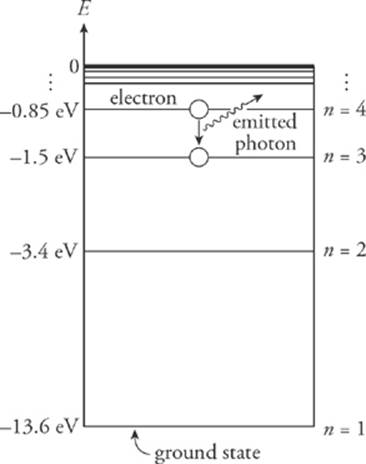
4. How much energy must a ground-state electron in a hydrogen atom absorb to be excited to the n = 4 energy level?
5. When the electron is in the n = 4 level, what energies are possible for the photon emitted when the electron drops to a lower energy level?
Here”s How to Crack It
4. For an electron to make the transition from E1 to E4, it must absorb energy in the amount E4 – E1 = (–0.85 eV) – (–13.6 eV) = 12.8 eV.
5. An electron in the n = 4 energy level can make several different transitions: It can drop to n = 3, n = 2, or all the way down to the ground state, n = 1.
So, there are three possible values for the energy of the emitted photon, E4→3, E4→2, or E4→1:
E4→3 = E4 − E3 = (−0.85 eV)−(−1.5 eV) = 0.65 eV
E4→2 = E4 − E2 = (−0.85 eV)−(−3.4 eV) = 2.55 eV
E4→1 = E4 − E1 = (−0.85 eV)−(−13.6 eV) = 12.8 eV
![]()