Make: The Annotated Build-It-Yourself Science Laboratory (2015)
Part II. Physics
Chapter 7. Meteorology (Weather)
Anemometer
Purpose: The anemometer is used to measure wind speed from any direction. The weather bureau uses one with three cups. As these cups turn, a dial connected electrically to the anemometer shows the wind speed to the weatherman inside the weather station.
Materials: Milk carton, four paper cups, medicine dropper or short piece of glass tubing, staples or paper clips, and a board with a large nail for the base.1
What to Do: Cut the four corner strips off a milk carton as shown with the dotted lines. Slip the folded ends of each pair of strips together. This should make two long strips. Lay one of the long strips on the table. Place the glass part of an eyedropper or a sealed end of a piece of glass tubing on the middle of the long strip. Lay the other long strip over the top of the eyedropper and fasten the two strips around the eyedropper with staples or paper clips. Bend the four arms out as shown. Cut a slot in each of the four paper cups and insert the end of one arm in each cup. Drive a spike through a piece of board and then drop the eyedropper over the end of the spike so that the cups will turn freely on the spike. This wooden base can be nailed to the top of a post outside. Color or mark one cup so that it is easy to see.
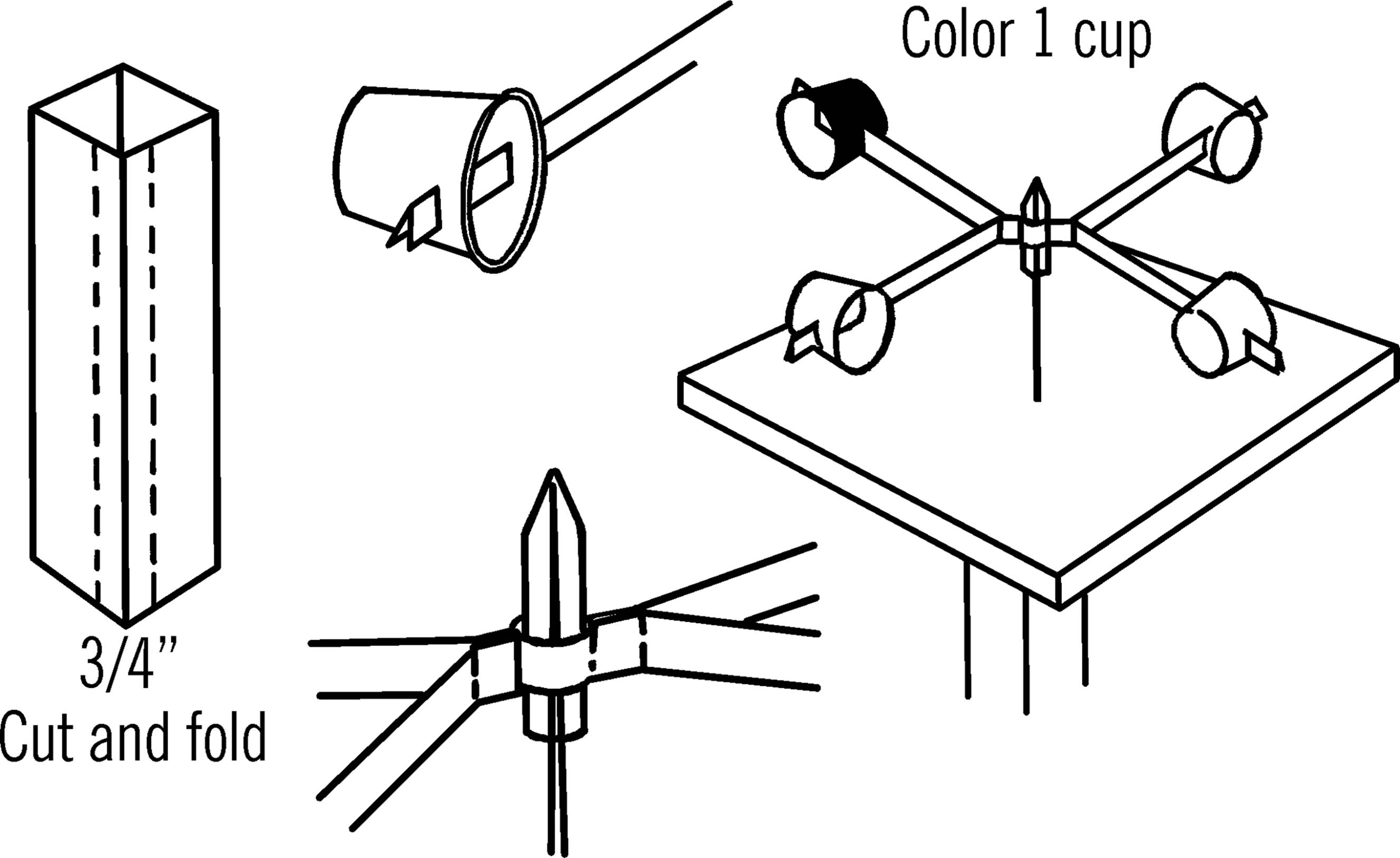
Operation of Equipment: In order to measure the wind speed, count the number of turns in 30 seconds and divide by five. This will give you the wind speed in miles per hour. Each anemometer is a little different. A more accurate way to calibrate the wind speed is to take a ride in a car on a calm day. Stick your anemometer out the car window2 and count how many times it turns in 30 seconds at, say, 5 miles an hour. Then count the number of turns when the speedometer shows ten miles an hour. You can do this for any speed and make an accurate table.
Can You Work Like a Scientist?
1. Can you think of a way you could connect up your anemometer to show the speed electrically on a dial?
2. What area near your home or school is the windiest? The calmest?
3. How does the wind vary for you and your friend who lives several miles away?3
4. What has wind speed to do with humidity (remember your hygrometer)?
5. What month of the year is the windiest? (Keep a chart for several months.)
6. How does your knowledge of wind speed help you predict weather?
7. With your wind-chill chart (“Wind Chill Table”) can you use your anemometer to help you decide what kind of clothes to wear on a given day?
Rain Gauge
Purpose: A rain gauge is used to measure the amount of rainfall over a period of time, for instance, 24 hours. The difficulty in measuring rainfall is that it seldom rains enough to be measured accurately with a ruler. Rainfall is usually measured in hundredths of an inch. This rain gauge magnifies the readings, so that the amount is measurable.
Materials: Baby bottle, milk carton, and a three-inch funnel.
What to Do: Cut a slot about ½ inch wide down the middle of one side of the carton. Fasten a piece of adhesive tape along this slot. This will serve as your measuring scale. Cut out part of the top of the carton so you can slip a baby bottle into it. Then a funnel is placed on top of the carton so that the rain, going into the funnel, will drain into the baby bottle.
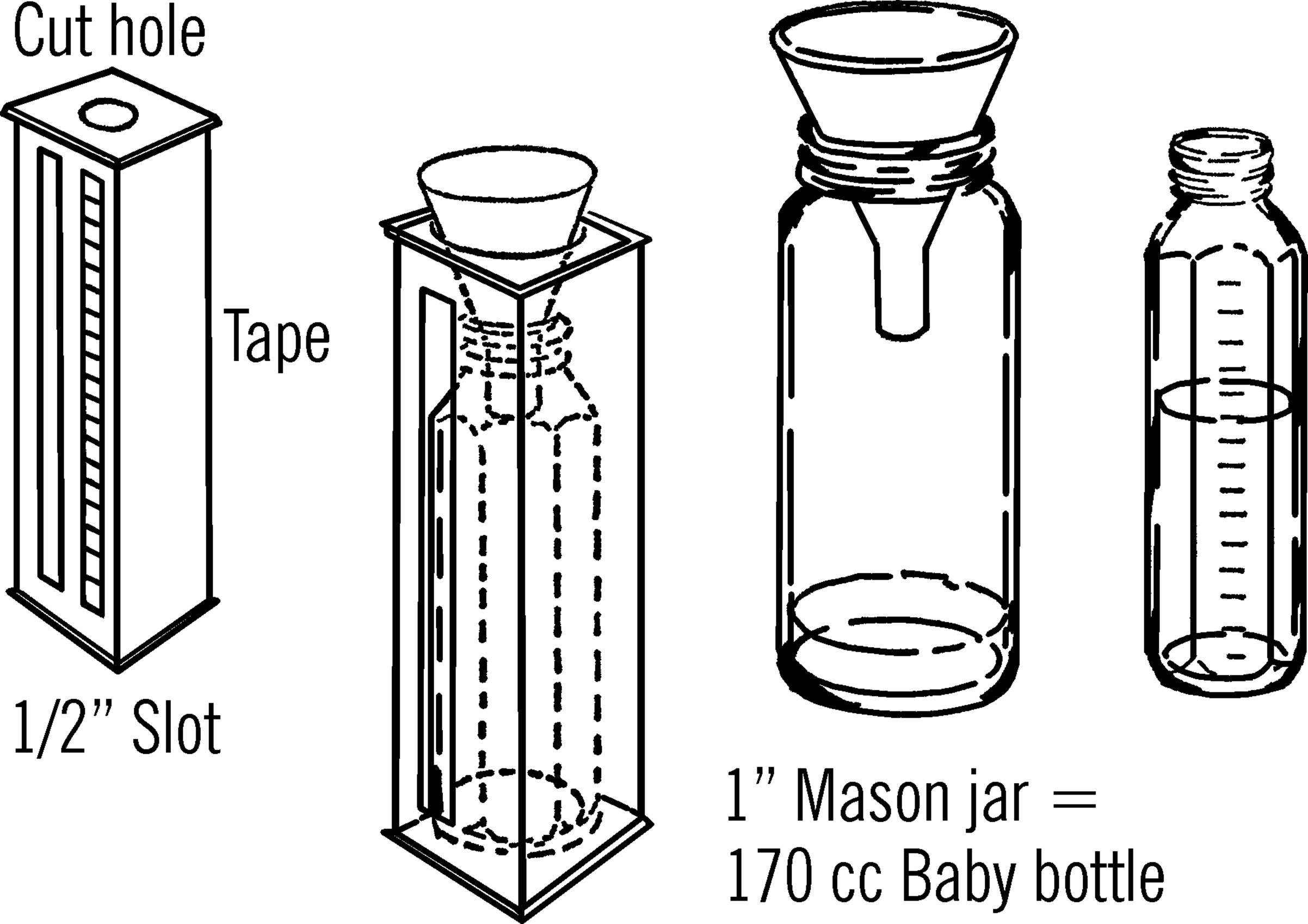
Operation of Equipment: The funnel is larger than the baby bottle in diameter, so an inch of rain going into the funnel will be much deeper than an inch in the baby bottle. The large funnel and the small baby bottle magnify the reading. In order to place accurate measurements on the scale, we must find out how high one inch of rain through the funnel will show in the baby bottle. To do this, we need a bottle whose opening is the same size as the funnel. Then one inch of water in this bottle would be equal to one inch of rainfall going through the funnel. We then pour this water into the baby bottle and mark the height as one inch. A wide-mouth mason jar has the same opening as a three-inch funnel. One inch of water in this jar when poured into the baby bottle comes up to 170 cc. Mark 1” on the tape at this spot. Divide the inch into tenths. These tenths can be redivided into ten parts so your rain gauge will measure hundredths of an inch.
Can You Work Like a Scientist?
1. How can you magnify your readings more?
2. Can you keep a chart of rainfall for the month and record the rainfall each day? Or what day does it rain the most?
3. Can you compare months with each other? In what month does it rain the most?
4. Does it rain as much at a friend’s house many miles away as it does at your house? Does it rain as much in the country as the city?
5. Do you know someone who lives in another state? Could you compare your readings with theirs?
6. In order to keep a daily record, would you have to record the rainfall at the same time each day?
7. How do your readings agree with those of the weather bureau?4 Could you both be right?
Wind Vane
Purpose: The wind vane detects the direction from which the wind is blowing close to the surface of the ground.
Materials: Paper milk carton, paper clips or staples, short piece of glass tubing or an eyedropper, spike, and wood for the base.
What to Do: Cut off a corner strip of the carton as shown. Shape the cardboard into an arrow. Be sure the tail is very large. Seal the end of a short piece of glass tubing or use a medicine dropper. Place the tubing between the double shaft of the arrow and keep it in position with staples or paper clips. The base to hold the wind vane can be a large nail (spike) driven through a board.
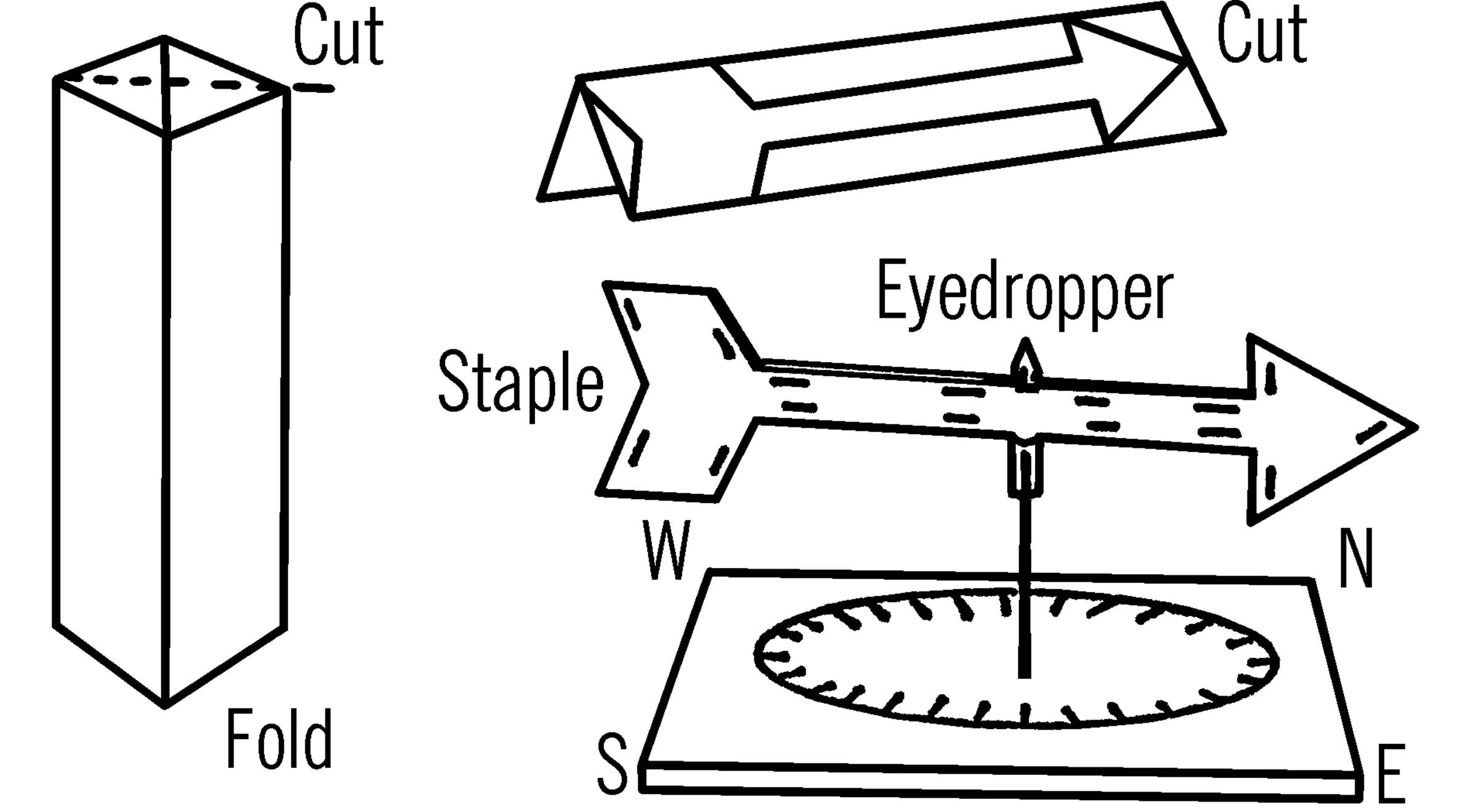
Operation of Equipment: You can use the wooden base with your wind vane almost any place. Just set the glass tubing or medicine dropper over the nail. This type of base can also be mounted on the top of a post by nailing down through the wood. A permanent base can be made from a coat hanger. However, this cannot be moved easily. If you do not wish to leave your weather instruments outside, you need only set up the stand. Then you can quickly set up your wind vane or anemometer to take a reading.
Can You Work Like a Scientist?
1. Can you make a simple wind vane with a pencil, thread, and a tack?
2. On the base you can write down the type of weather to expect if the wind is blowing from a certain direction. From what direction does most of the rain come?
3. Does your wind vane show the same direction as your school flag?
4. Why is the direction of the wind important to ships and planes? Can you think of an experiment that will show this?
5. Record the direction of the wind and the temperature over a period of time. What does the direction of the wind have to do with temperature?
Nephoscope
Purpose: The wind vane detects the direction of the wind near the ground. High above the earth the wind may be blowing in a different direction. The weather service uses weather balloons to find out this information. The clouds can serve as your weather balloons. The nephoscope helps you detect the slightest movement of the clouds.
Materials: Shaving mirror with stand (found in most variety stores for several dollars)5 and some gum labels6 or other paper.
What to Do: Write the directions on paper and glue onto the mirror as shown. Glue a small dot in the center of the mirror.
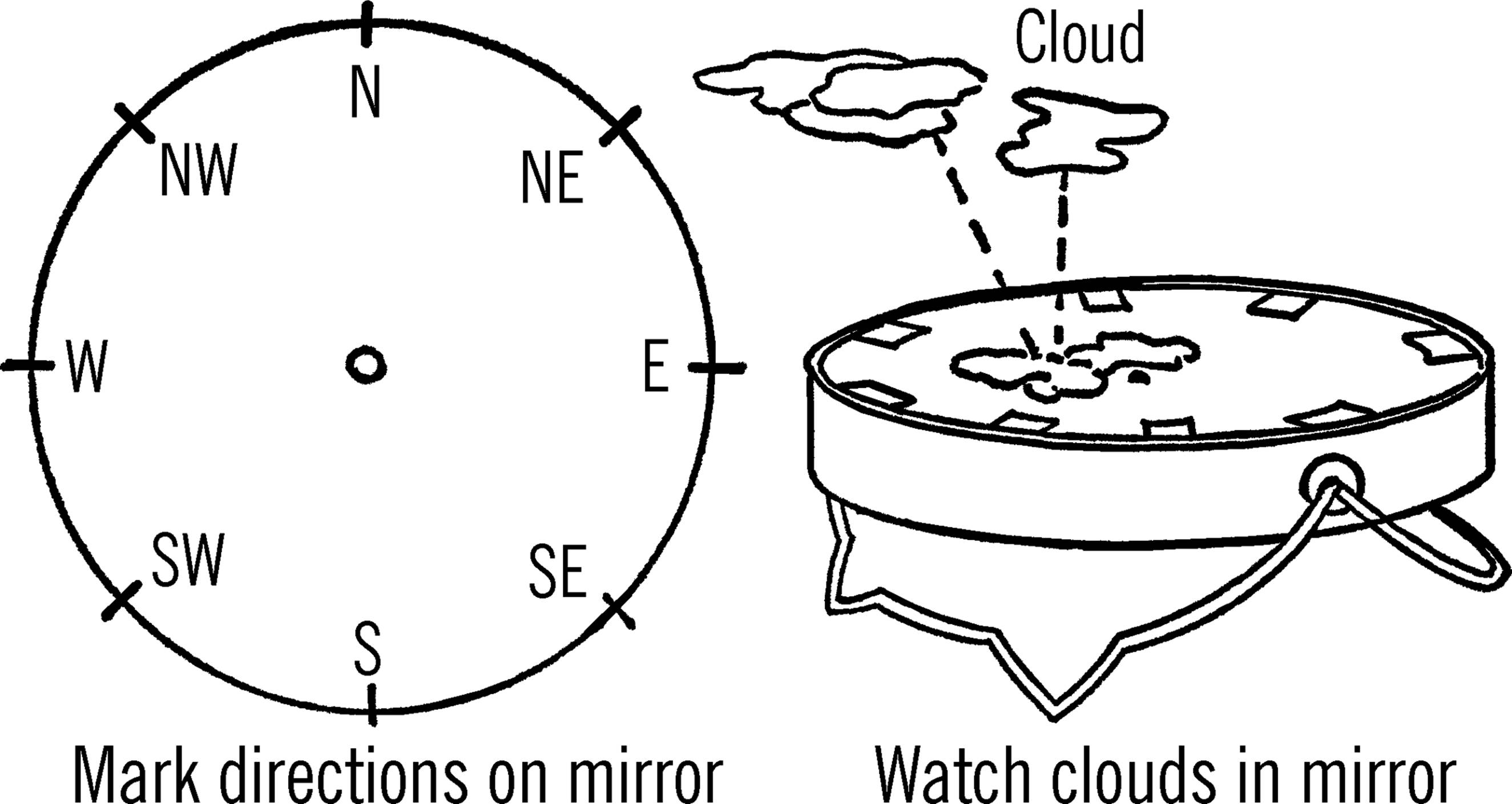
Operation of Equipment: Set your mirror up on its stand (as shown). Turn the mirror so that north on the mirror points to north. Check your directions with a compass. Locate the tip of a cloud overhead in your mirror. Center this tip with the center dot on your mirror. Follow the movement of the cloud in your mirror. The place where the cloud moves off your mirror glass is the direction toward which the cloud is moving. Since the mirror is so small, any slight movement is noticeable on the mirror.
Can You Work Like a Scientist?
1. Since most weather reports give the direction the wind is coming from, your nephoscope readings might confuse you. Could you switch directions on your nephoscope so it will give the direction the wind is coming from?
2. Record the direction of the winds for a period of time (several weeks at least). Also record weather conditions. Can you find any connection between weather conditions and the direction of the wind?
3. Could you use the nephoscope to give you the speed of the wind high above the ground? Could you make a nephoscope that could give you wind speed? What are the problems in making it accurate?
4. Record the winds high above the ground with your nephoscope. Record the winds close to the ground with your wind vane. How often are they the same? How often are they different?
5. Could you use your nephoscope on high-flying airplanes?
6. Could you use your nephoscope to observe the movement of the sun and the moon? Why do these move across the mirror? Don’t look directly at the reflection of the sun (see “Sunspot Viewer”).
7. Record both the wind direction and the temperature over a period of time. What effect on temperature does the direction of the wind have?
Beaufort Wind Scale
|
Wind speed |
Description |
Beaufort # |
|
< 1 MPH |
Smoke rises vertically |
0 |
|
1-3 MPH |
Smoke shows direction. Wind vane does not. |
1 |
|
4-7 MPH |
Vane moves. Wind felt on face. |
2 |
|
8-12 MPH |
Leaves and twigs in constant motion. |
3 |
|
13-18 MPH |
Dust and loose paper rise. Small branches move. |
4 |
|
19-24 MPH |
Small trees wave. Waves on small bodies of water. |
5 |
|
25-31 MPH |
Large branches move. Wires whistle |
6 |
|
32-38 MPH |
Whole tree in motion. Walking difficult. |
7 |
|
39-46 MPH |
Twigs break off. |
8 |
|
47-54 MPH |
Damage to chimneys and roofs |
9 |
|
55-63 MPH |
Trees uprooted. Considerable damage evident. |
10 |
|
64-75 MPH |
Widespread damage. Rarely experienced. |
11 |
|
>75 MPH |
Hurricane. |
12 |
Can You Work Like a Scientist?
1. How accurate is this scale? Check your observations with an anemometer.
2. Can you devise your own wind scale?
3. Make a wind tunnel with an air blower or fan for your source of wind. Test the Beaufort scale with your wind tunnel.
Wind Chill Table
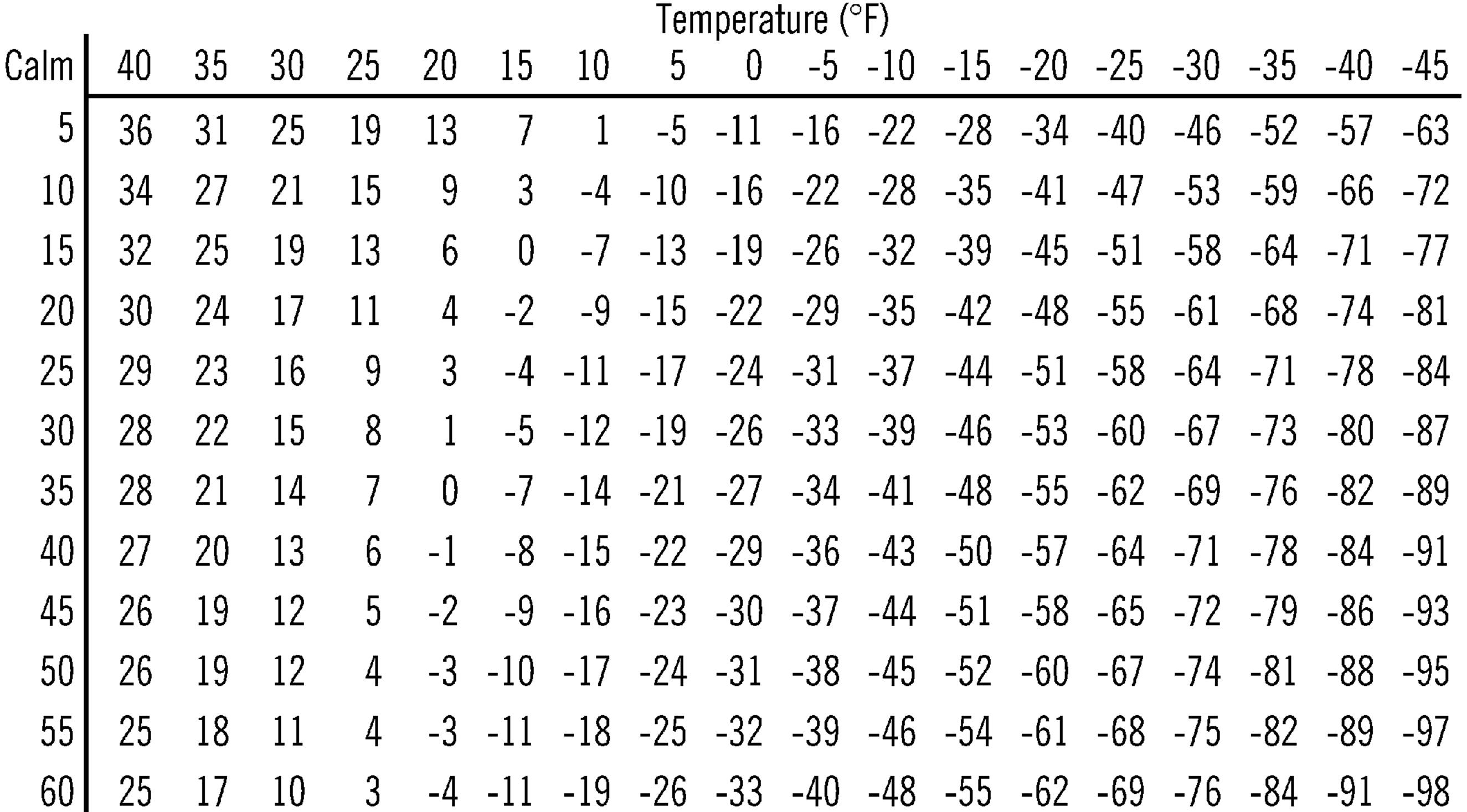
Figure 7-1. Wind Chill Table
Scientists, at work in the Arctic and Antarctic, have found that the faster the wind blows, the colder the skin gets. When wind speed rises, the exposed skin will become much colder than when there is no wind. Scientists call this wind chill. You can use the table following to find wind chill just as scientists do.
1. Find your present temperature and wind speed.
2. Find the number at the left of the chart (below) that is the same as, or closest to, your wind speed in miles per hour.
3. Read across the top row until you find the number that is closest to what your temperature is.
4. Read the number that is in the row and column matching your wind speed and temperature. This number is the equivalent temperature reading, or what the temperature would be equal to, if there were no wind.
Example: Suppose the temperature is 16 degrees. The wind speed is 28 miles an hour. Find the number closest to what your wind speed is in the wind speed column at the left. It would be 30. Read across the top row to find the number that would be nearest the 16 degrees. This would be 15. Follow down to the intersection between that row and column. The number you find is -5 degrees. Minus 5 degrees means 5 degrees below zero. This shows that with a temperature of 16 degrees and a wind of 28 miles an hour, the effect on all exposed skin would be the same as -5 degrees with no wind. We can say that the higher the wind speed, the more wind chill there will be. Wind chill compares the temperatures at various wind speeds to the temperature when no wind is blowing.
Balloon Barometer
Purpose: The barometer measures the pressure, or push, of the air molecules around us. Changes of pressure affect weather conditions, so the barometer is a tool to help forecast weather.
Materials: Mason jar, balloon, rubber band, and a broom straw7 or soda straw.
What to Do: Cut a balloon open as shown. Stretch the rubber over the opening of the mason jar. Fasten the balloon down with a rubber band. Fasten a broom or soda straw to the balloon with a piece of Scotch tape. The tape should be attached to the middle of the balloon so that when the center of the balloon is forced downward, the tip of the straw will go up.
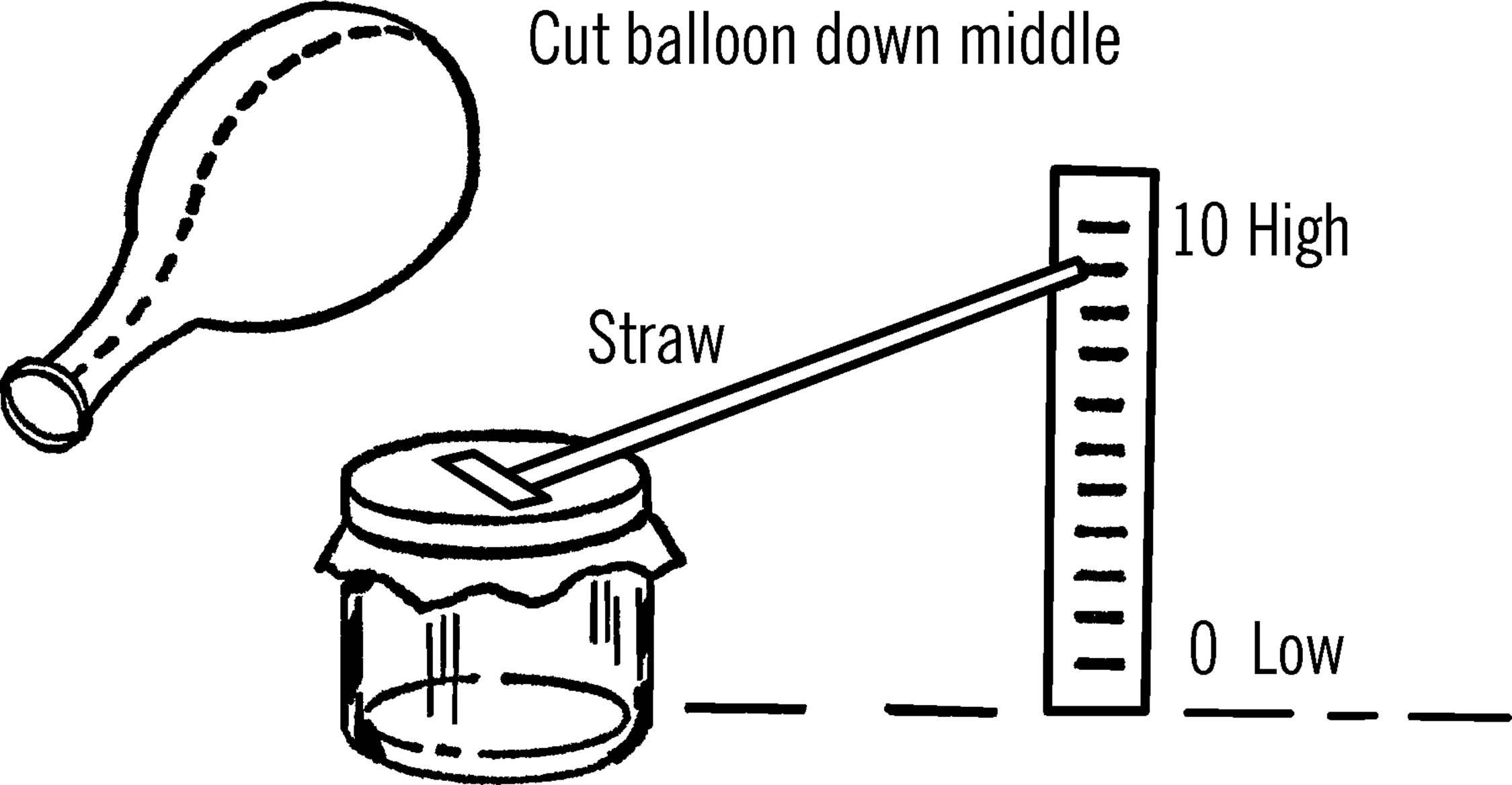
Operation of Equipment: Set the barometer on a table or window sill. Have the straw pointing to the wall or sill. Place a piece of tape on the wall or sill and make a mark each eighth of an inch. Number the mark the straw points to with the number “ten.” The bottom of the scale should be zero and the top of the scale about twenty. Make a chart and mark down where the straw points and the time of the reading. Check your barometer again after an hour or so. Again record the findings. If the barometer seems to be rising, the weather should be fair. If the readings are lower, we have low pressure, and you may expect cloudy weather and perhaps rain.
Can You Work Like a Scientist?
1. High pressure means the air is heavier. If the air pushes more on the center of the balloon, in which direction will the end of the straw move?
2. Low pressure means the air is lighter. If the air does not push as much on the center of the balloon, in which direction will the end of the straw pointer move?
3. Pressure is caused by the weight and movement of the air molecules. Would heavy air contain more or less molecules than light air (low pressure)?
4. High pressure is caused because the air molecules are all pushed together (packed or very dense). Low pressure is caused because the molecules move farther apart. When the air molecules are far enough apart, there is room for water molecules (vapor). Squeeze a sponge together. This represents molecules squeezed together (high pressure). Can you pick up much water with the sponge squeezed together? Now let the sponge spread out. Will the sponge hold more?
5. Take the barometer outside. Do you detect a change in pressure?
6. Check the-barometer at home when you wash8 clothes. Do you notice any difference?
7. Take the barometer into a warm room. Note any pressure changes.
8. Place the barometer in the refrigerator and leave for about an hour. Does cold air affect pressure? How? Can you explain why?
Aneroid Barometer
Purpose: The barometer detects changes in air pressure. An aneroid barometer is made from a metal container in which nearly all the air has been removed. The container is kept from collapsing by a spring coil inside the container.
Materials: The container can be made from a condensed milk can. It also can be made from a pint jar, a coiled spring, plywood, and a balloon. In either case, additional materials needed are a two-quart milk carton, a long needle or a piece of coat hanger wire, a penny, a broom straw, and some thread.
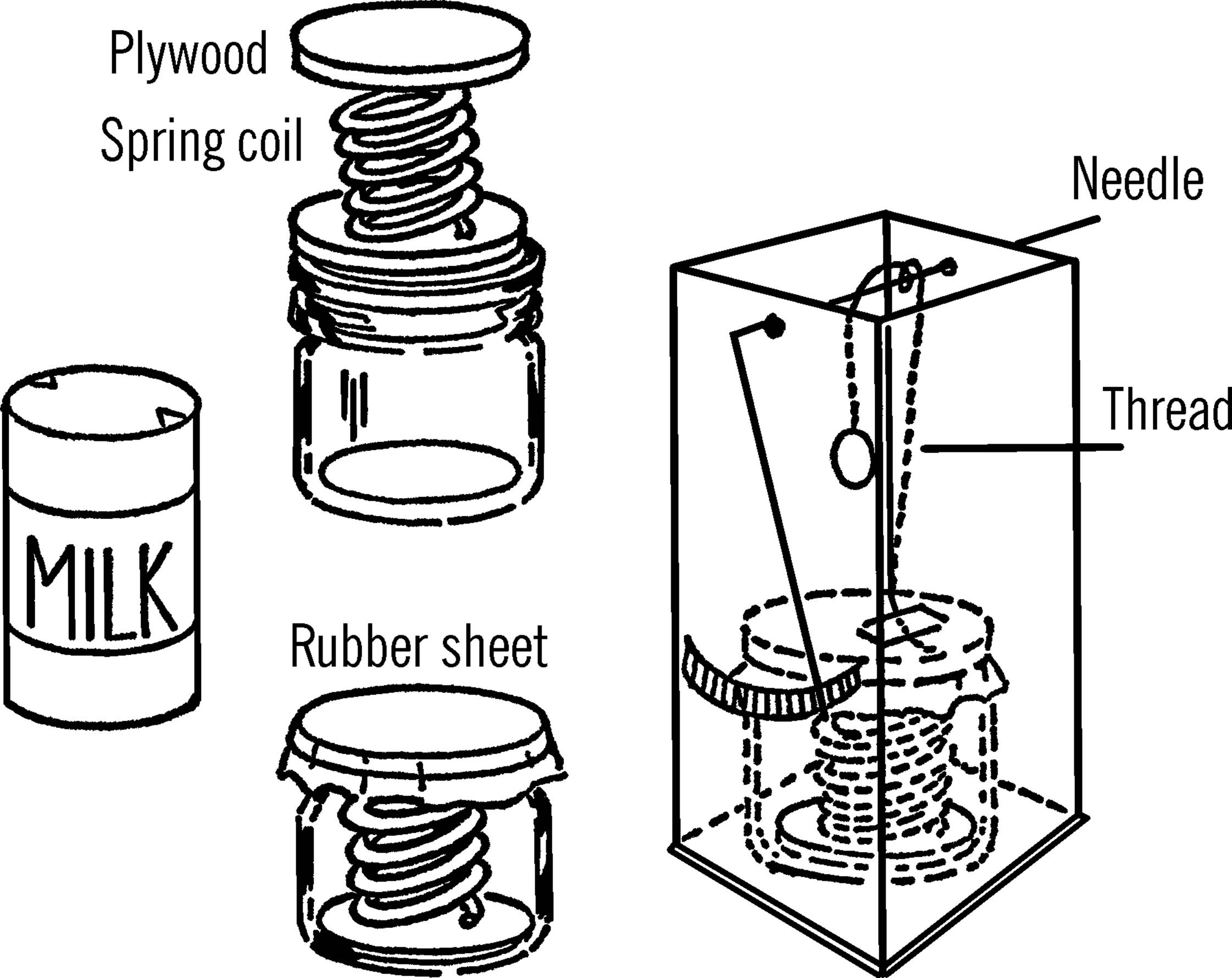
What to Do: If you use a milk can for your container, empty the milk can9 and wash it out as best you can through the two holes. Leave about one inch of water in the bottom. Solder one of the holes shut.10 Heat the can on a stove or over your alcohol burner. When the steam from the water in the can has removed most of the air, quickly remove the can from the source of heat and solder the other hole shut.
If you use a pint jar for your container, fasten one end of a coiled spring to a piece of plywood which is almost the same size as the bottom of the jar. Fasten a second piece of plywood to the other end of the spring. Place a little water in the jar. Insert and compress the spring and cover the jar with rubber from a balloon or rubber sheeting. Leave a small corner open for air to escape. Heat the bottle, and when most of the air has escaped, seal the jar by placing rubber bands over the rubber cover.
Cut off the top of the milk carton. Set the container in the bottom of the carton. Make two small holes near the top of the carton and insert the needle or coat hanger wire. This wire should turn easily. Attach a thread with tape to the top of the container inside the carton. Wrap the thread around the wire and hang a weight (penny) from the other end of the thread. With glue fasten a straw pointer to the wire or the eye of the needle. Make a scale as shown.
Modern Safety Practice
If soldering the condensed milk can, the can may become very hot. Take caution to avoid burns.
Operation of Equipment: When the pressure increases, it should push down on the top of the container. This should cause the needle or wire to turn slightly. This slight movement should make the tip of the straw pointer move a great distance.
Can You Work Like a Scientist?
One reading means little. Take readings every two hours and record your findings. Compare your findings with actual weather conditions the next day or so.
Hair Hygrometer
Purpose: A hygrometer measures the humidity or moisture in the air. 11
Materials: Two paper clips, milk carton, needle with a large eye, a human hair about ten inches long, glue or cement, broom straw, and a blank file card (any cardboard will do).
What to Do: Wash the hair in alcohol or soapy water to remove the oil. The oil coats the hair and keeps it from absorbing moisture. Rinse and allow the hair to dry completely. Cut two flaps near one end of the carton. Make holes in the two flaps as shown. Work the needle back and forth in the holes so that the needle turns easily. Cut a slot for a paper clip at the other end of the carton. Stick the end of the straw through the eye of the needle. Glue or cement the straw in place. Make a dial on your file card as shown. You can hold the file card in place with Scotch tape or tacks. Slip one end of the hair into the paper clip and place the clip in the slot. Do not touch the hair with your bare hand as you will get oil on it. Slip the needle in place and then wrap one turn of the hair around the needle. Slip the other end of the hair into the second paper clip. Hang this clip over the end of the carton.
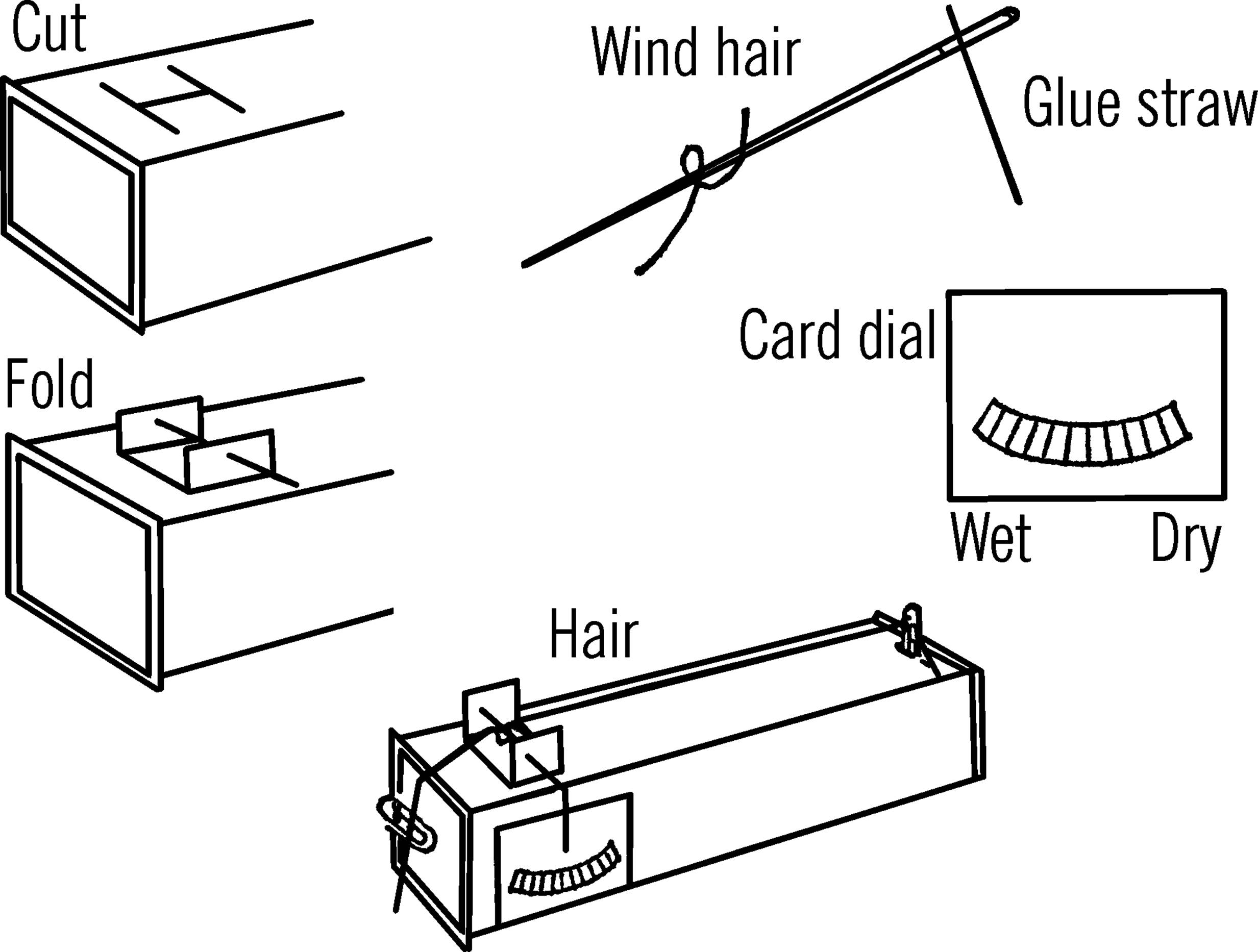
Operation of Equipment: When the hair is dry, it shrinks or gets shorter. When there is a lot of moisture in the air, the hair stretches or gets longer. You can’t see this change, but because a small movement of the hair moves the needle and the end of the straw a greater distance, you are magnifying the change in the length of the hair. In order to set your hair hygrometer to make accurate readings, place it in a bucket that contains a wet towel. Cover the bucket with another wet towel, thus making the air as moist as possible. After about 20 minutes, remove the hygrometer from the bucket and quickly set your dial on 10, or 100% humidity.
Can You Work Like a Scientist?
1. What is the humidity when you take a shower?
2. Does the humidity change when there are many people in a room?
3. What effect does humidity have on static electricity?
Wet- and Dry-bulb Hygrometer
Purpose: This instrument is used to measure both the temperature and the amount of moisture in the air.
Materials: Two thermometers, milk carton, two rubber bands, and a piece of clothesline rope about 5” long. The thermometers should read alike, and the bulb should not be covered. Thermometers with cardboard backs are ideal. The clothesline rope should be the kind with soft cotton fibers inside.
What to Do: Cut a hole about three inches from the bottom of the carton. Stick the clothesline rope through the hole so only about one inch sticks out. Slip this end over one of the thermometer bulbs. Fasten the thermometers to the outside of the carton with rubber bands. Pour about two inches of water into the carton.
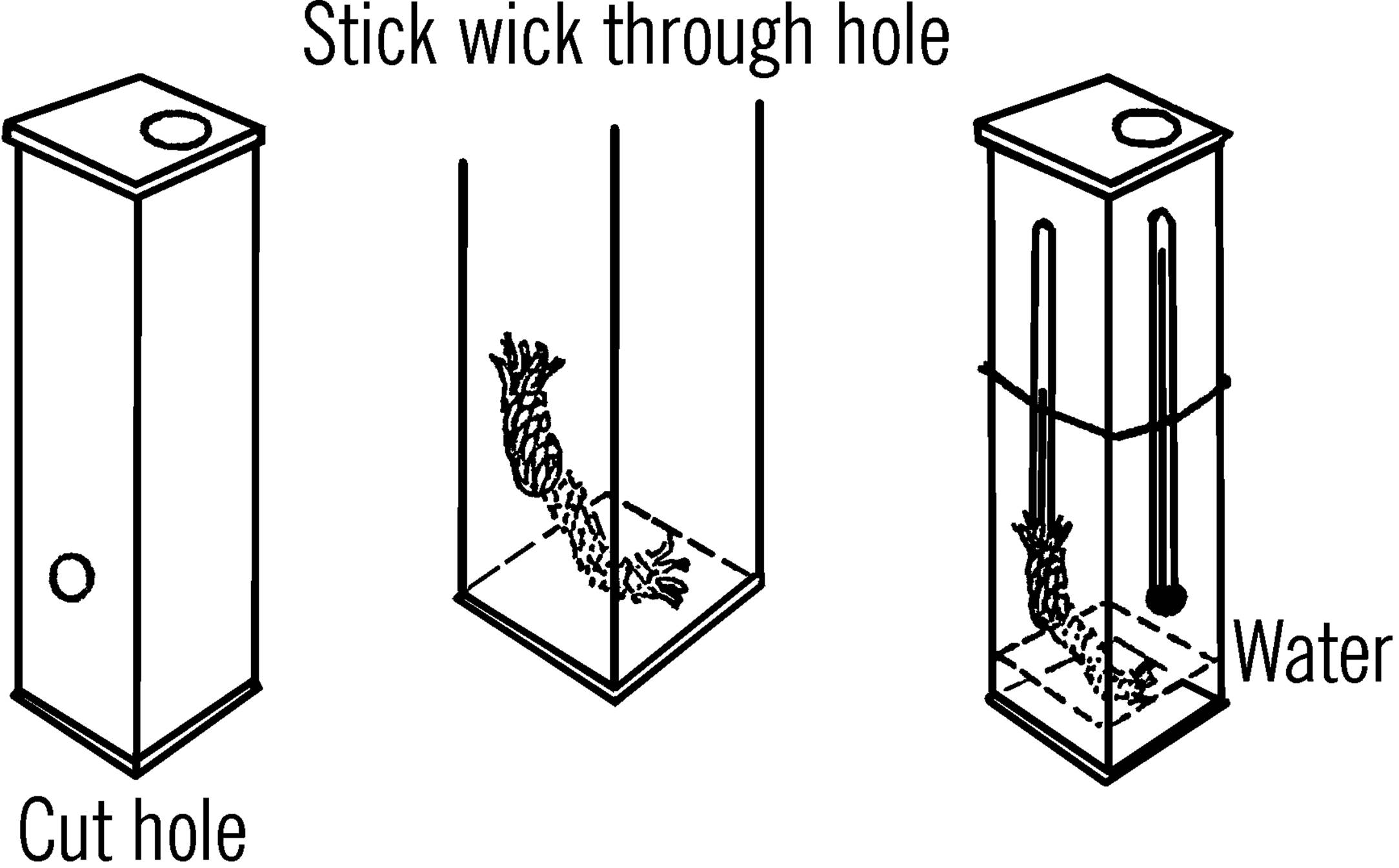
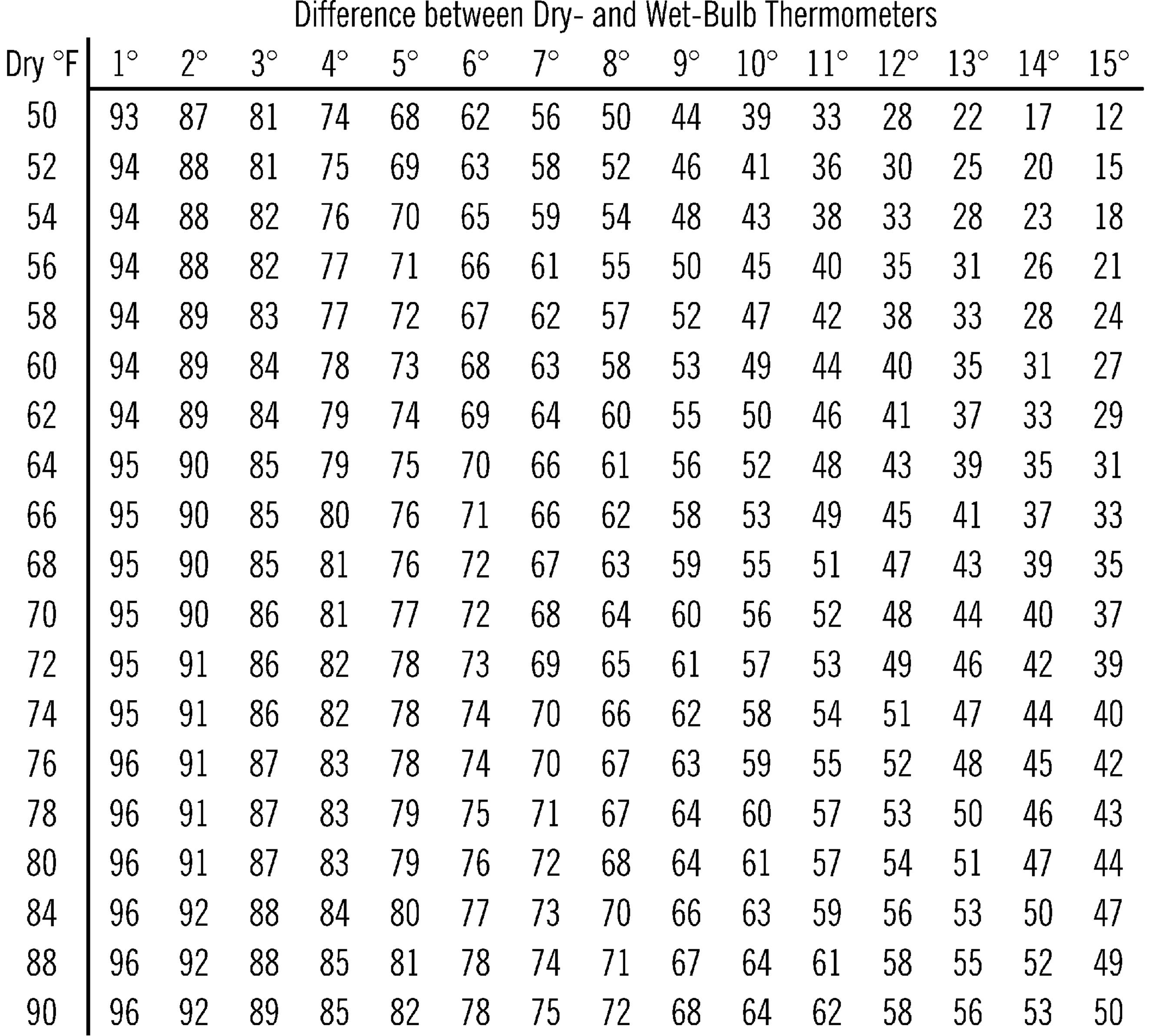
Figure 7-2. Table Showing Relative Humidity
Operation of Equipment: The fine hairlike strands in the cotton rope suck up the water. Soon the water will climb up the rope and dampen the bulb of the “wet” thermometer. In order to understand what happens, wet the back of your hand. Blow across it. The water on your hand evaporates, and your hand seems cooler. This is what happens to the wet bulb thermometer. The rate or speed of evaporation depends on how much water is in the air and the temperature of the air. To use this instrument, fan the wet bulb with a piece of cardboard. Check the temperature. Note the temperature of the dry thermometer and the difference in temperatures between the two thermometers. Use this information with your humidity chart to find the relative humidity (amount of moisture in the air compared to the amount that the air could hold at that temperature). The relative humidity is given as a percent.
Can You Work Like a Scientist?
1. Place a drop of water and a drop of alcohol on the back of your hand. Blow across each. Which feels cooler? Which one evaporates quicker? What has the rate of evaporation to do with cooling?
2. Note the readings on your thermometers when you use water. Then, instead of water, use alcohol. Could you make a humidity chart based on the evaporation of alcohol?
3. Try your gauge in the bathroom when someone is taking a shower. As the humidity increases, what happens to the mirror?
4. How is humidity related to temperature? What effect does wind have on your wet-and dry-bulb hygrometer?
What Determines Weather
Daily Weather Record12
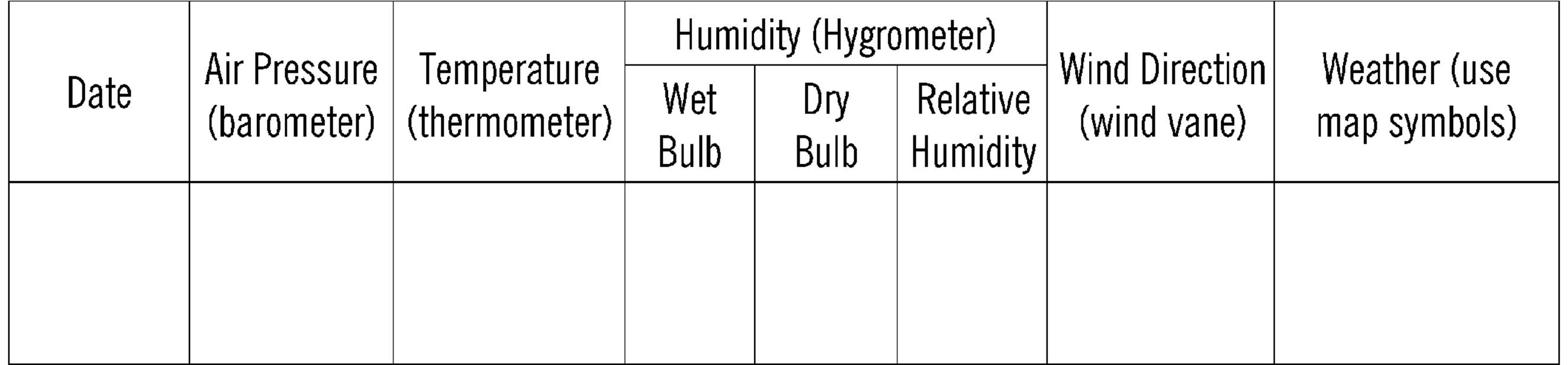
Figure 7-3. A Daily Weather Record Chart
Mercury Barometer
Purpose: A barometer is used to measure air pressure (the push exerted on an object by the weight and movement of all the air molecules above it). A mercury barometer is quite accurate because it is not affected by changes in temperature as are many other types of barometers.13
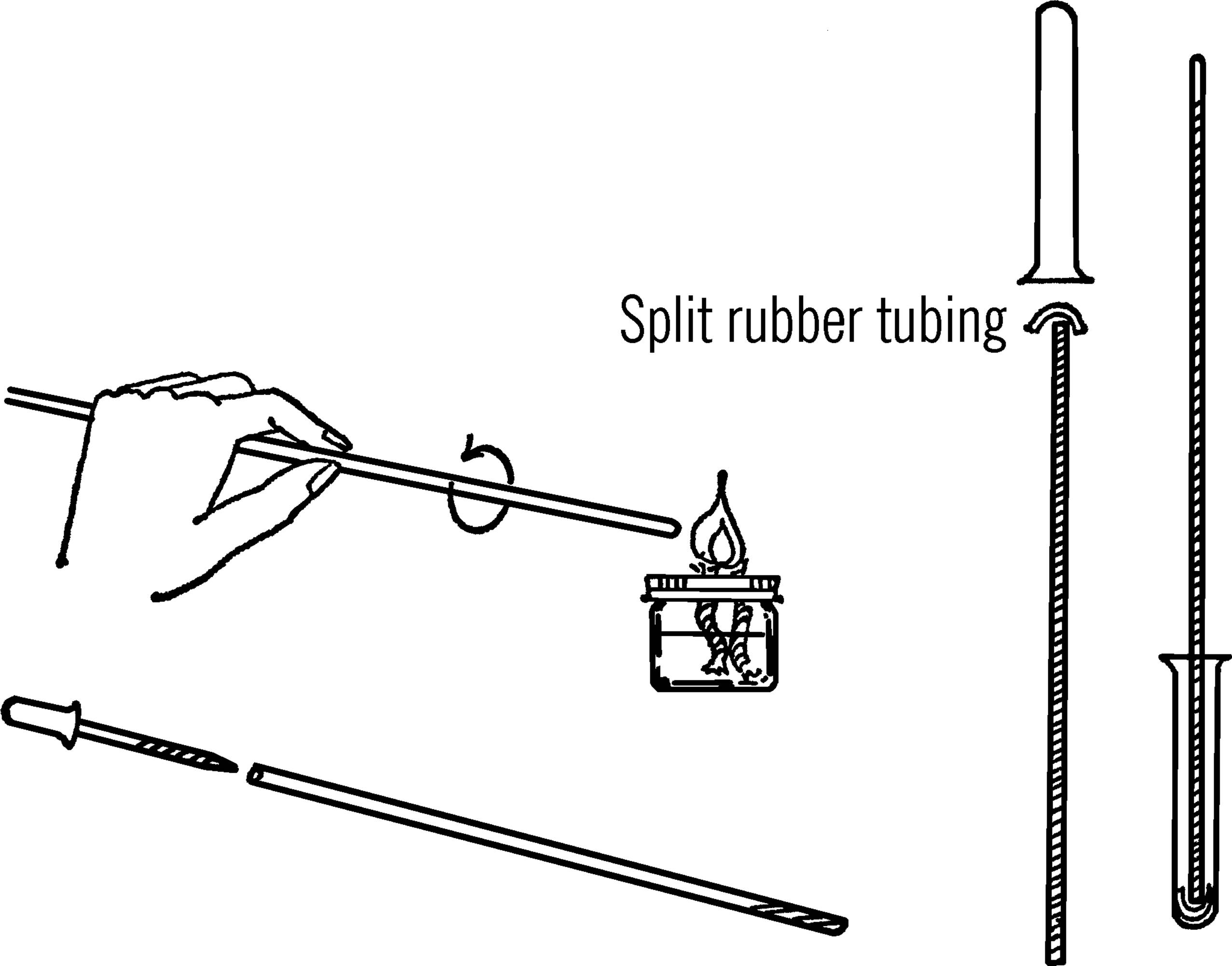
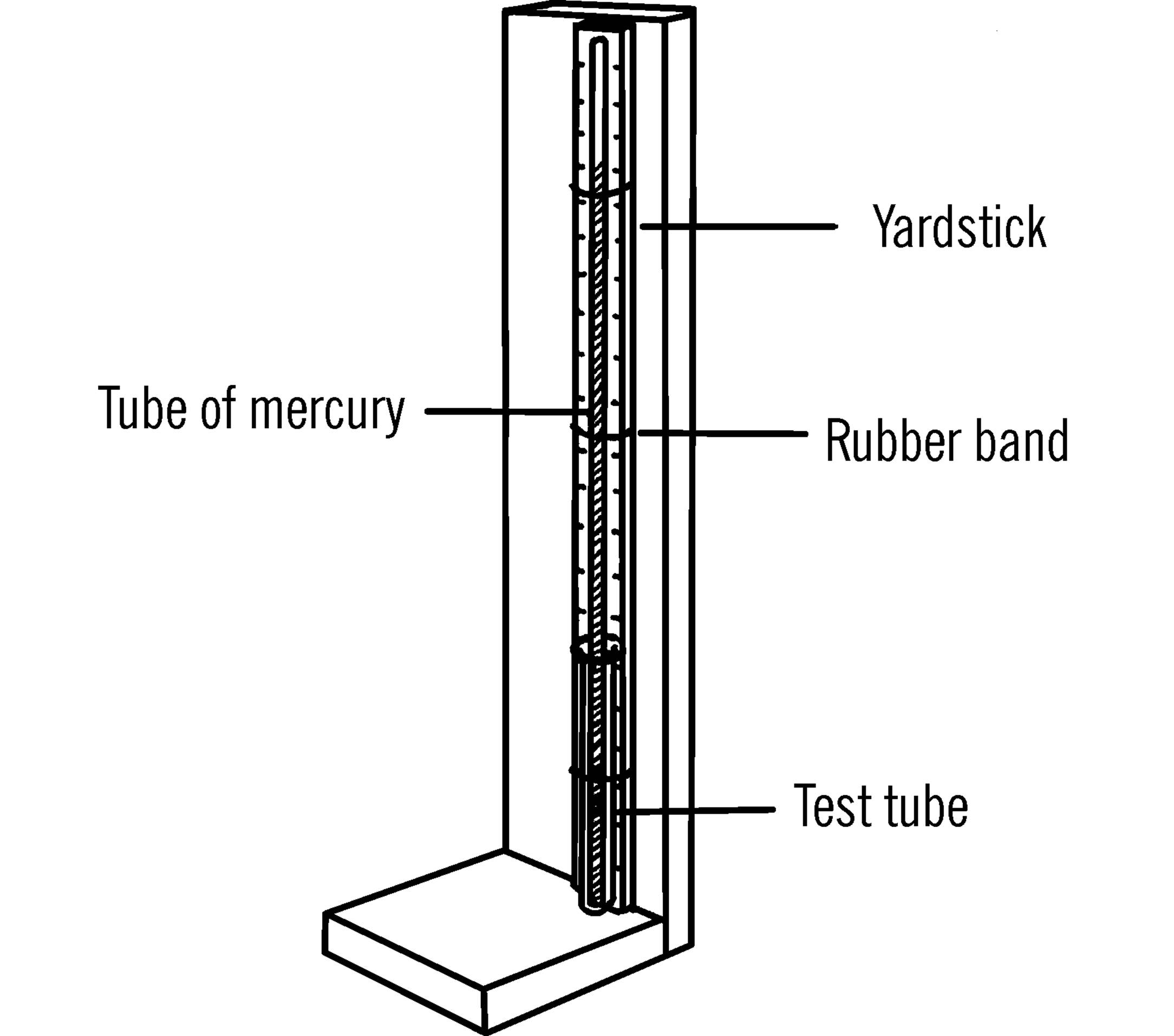
Materials: A piece of 6 mm glass tubing at least 32 inches long, six ounces of mercury,14 one-half-inch piece of rubber tubing, test tube, yardstick, rubber bands, wood to make the supporting stand as shown, and an eyedropper.
What to Do: Build the stand as shown. The stand should be tall enough to support the tube of mercury and the yardstick. Heat and seal the end of the glass tube in the flame of a gas burner. The double wick alcohol burner (“Broad Flame Alcohol Burner”) will produce enough heat to seal the tubing. Be careful to turn the tubing while you are heating it so that the glass will not crack. A slight leak will make the readings of your barometer inaccurate.
Use an eyedropper to fill the tube with mercury. The smaller the hole in your dropper, the easier it is to fill the long glass tubing with mercury.
Tip down the sealed end of the long glass tubing so that the mercury will run slowly down the tube. Use your eyedropper to add the mercury to the open end of the tube as shown. If the drops of mercury are small enough, the mercury will flow down to the bottom of the tube. If the drops are not small enough, you will have to shake them down to the bottom of the tube. Fill the long tube completely with mercury. This is to remove all the air.
Slice the short piece of rubber tubing lengthwise so that it will fit over the opened end of the tubing. Press the test tube down on the rubber tubing and turn the glass tubing over. Add a few drops of mercury to the bottom of the test tube and then release the pressure on the glass tubing. The glass tubing will rise slightly, and some mercury will drop down into the bottom of the test tube from the tubing. The level of the mercury in the glass tubing should be between 29 and 30 inches above the level of the mercury in the test tube. Fasten the test tube and the glass tubing to a yardstick or meter stick with rubber bands. The yardstick or meter stick is then attached to the wooden upright with small brads or screws.
Safety Tips
Mercury is very poisonous. Never touch it with any part of your skin. Mercury will enter the body through any cuts or opening in the skin.
Modern Safety Practice
Mercury is no longer considered safe to work with, even if you don’t touch it with your skin. See Note 42 in Appendix E about building a water barometer instead.
Operation of Equipment: Compare your reading with that of the weatherman or of a standard barometer. If the correct reading is 30.2 and your reading is 29.5, either raise the glass tubing slightly or lower the yardstick until the level of the mercury matches the correct mark on the measuring stick. Always tap your barometer before you take a reading. This will allow the column of mercury to move slightly up or down and settle at the correct level.
Can You Work Like a Scientist?
1. The atmosphere will hold up nearly 30 inches of mercury. The density of mercury is about 13 times that of water. How high a column of water will the pressure of the atmosphere support?
2. Can you find out the density of other liquids such as alcohol and then determine how high a column of the liquid you would need in order to make a barometer?
3. Could you make a water barometer similar to the mercury barometer by using plastic garden hose for your column?
4. Is your mercury barometer affected by altitude? Take readings at various floor levels in a tall building. Can you calibrate your barometer for different floor levels? This instrument might be helpful for an elevator operator.
5. Why do you need a vacuum at the top of the glass tubing? Make one barometer and let some air bubbles in through the mercury. Bring a heat source near the top of the barometer. Does the mercury level change? Try the same experiment after you have created a vacuum at the top of the tube by turning the tubing over and not allowing air bubbles to enter.
6. Does mercury expand (get larger) or contract when heated?
7. What material will mercury wet or stick to?
8. Can you make a large drop of mercury by pushing small drops of mercury together with a pencil? Try the same experiment with drops of water on a piece of wax paper.
9. Can you compare the readings of your mercury barometer with actual weather conditions over a period of a month or more?
Chemical Weather Glass
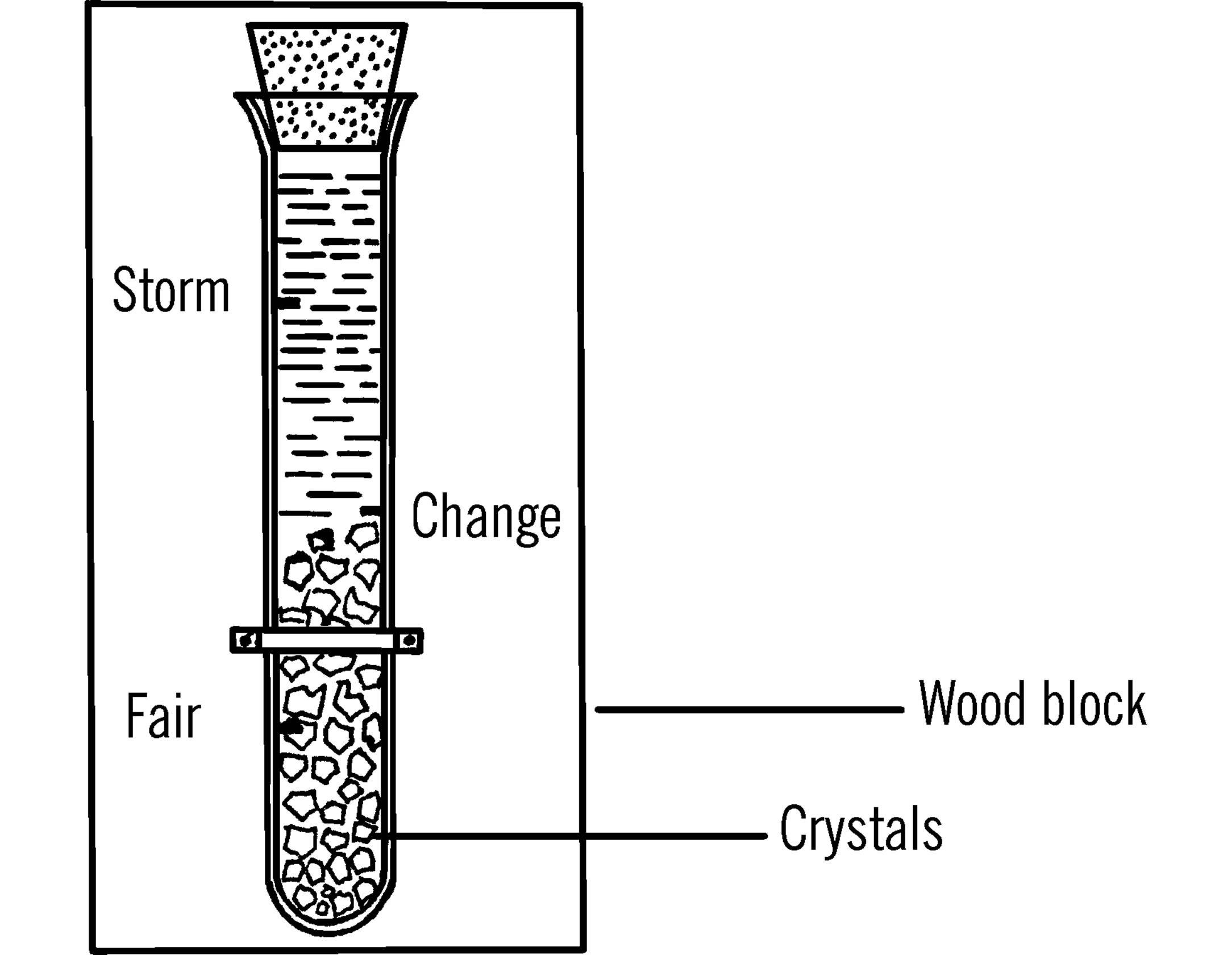
Purpose: The chemical weather glass is another instrument that helps you predict the weather.15
Materials: Test tube, about two ounces of water, two ounces of pure alcohol, ½ dram of potassium nitrate, ½ dram of ammonium chloride, and two drams of camphor. A dram is about an eighth of an ounce. (A baby bottle is marked off in ounces.)16
What to Do: Mix the chemicals in the test tube. Fill the test tube up to about one inch from the top. Seal with a cork. Mount the test tube on a wood block with either rubber bands or strips of tin for brackets. Make a scale on the wood block as shown. The mark at the bottom should be about 1¼” from the bottom of the test tube. Measure up another inch and a quarter and label the mark CHANGE. Measure another inch and a quarter up and label the mark STORM.
Operation of Equipment: Can you predict the weather from the following chart?
|
Tube full of crystals |
Storm |
|
Dim liquid |
Storm |
|
Crystals rising and nearing change mark |
Weather change (probably a storm) |
|
Crystals falling below the change mark |
Weather change (probably better weather) |
|
Crystals near the bottom |
Fair weather (heavy frost in the winter and high humidity in the summer) |
|
Clear liquid |
Fair weather |
Modern Safety Practice
Note 17 in Appendix E discusses safety when working with chemicals.
Can You Work Like a Scientist?
1. How does the chemical weather glass work?
2. Why should crystals form when a storm is coming?
3. What affects the weather glass, humidity or pressure? Can you test your guess (hypothesis)?
4. Keep a weather chart. How accurate is the chemical weather glass?
Air Current Detector
Purpose: To detect slight movements of air, such as warm or cold air currents that are constantly moving.
Materials: Pencil with eraser, soda straw, pin, paper clip, sheet of paper about 2½×4 inches, and a piece of Scotch tape.
Helpful Hints for Building: Scotch tape a piece of paper on one end of the straw as shown. Turn the end of the straw so that the paper is horizontal (like the top of a table). Pinch the other end of the straw and slip a paper clip on it about one inch from the end. The paper clip should be vertical (straight up and down). Now stick the pin through the side of the straw and into the eraser of a pencil. The paper tail and the paper clip should just balance. Move the paper clip until the balance is as perfect as possible.
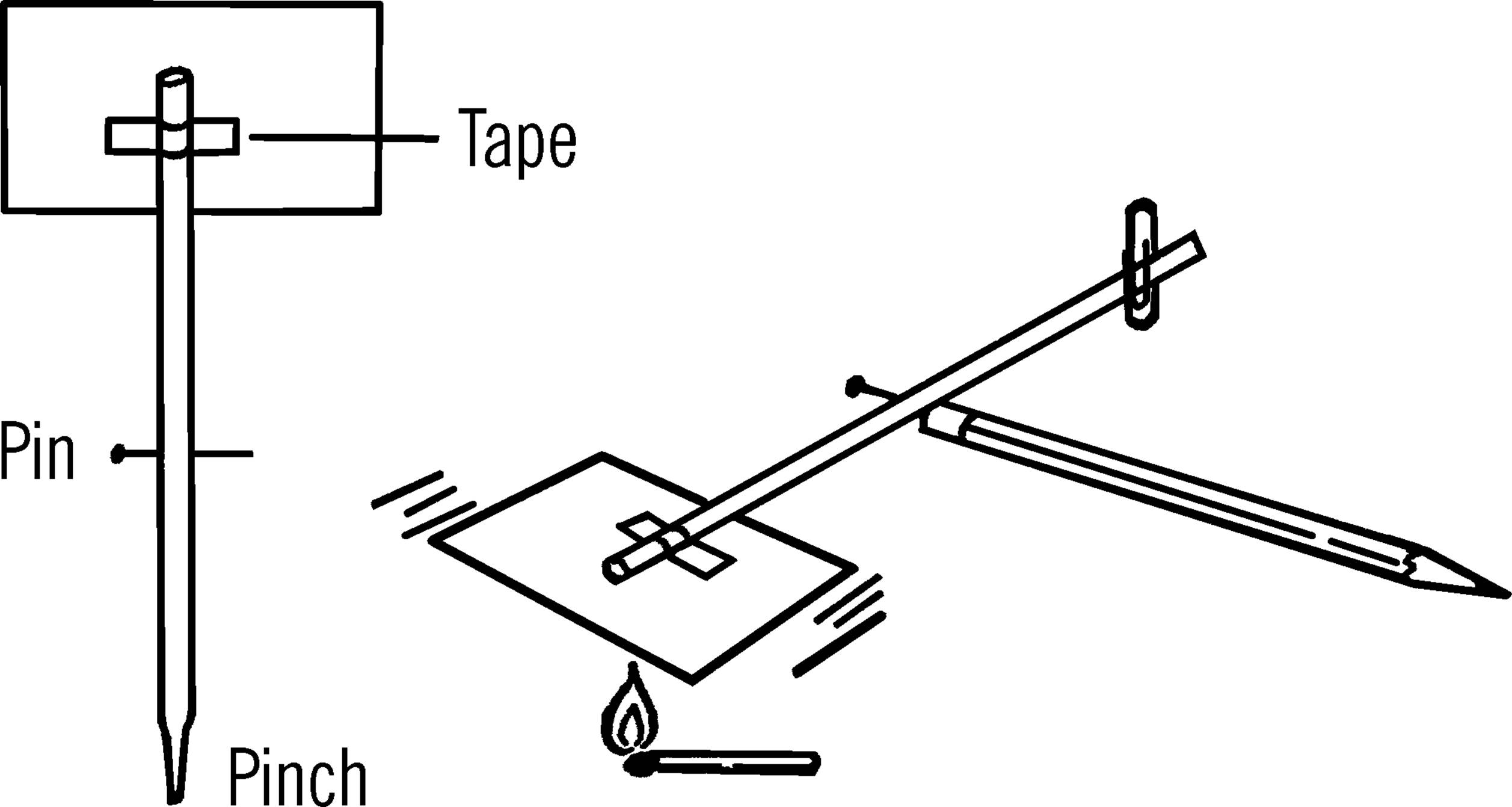
Operation of Equipment: Hold a match under the paper tail of the warm air current detector. Be sure the match is not close enough to burn the tail. The detector should move if it is properly balanced. Faint currents of air push against the tail of the detector and upset the balance. The tail will either go up or down depending on the direction of the air current.
Modern Safety Practice
Use common sense and standard safety practice (Note 3 in Appendix E) around flames (safety glasses, available fire extinguisher, good ventilation, etc.) when using the match. Be careful not to set the air current detector (or anything else!) on fire.
Can You Work Like a Scientist?
1. Does the detector tail always go up, or does it sometimes go down?
2. What kind of air makes the tail go up? What currents make it go down?
3. Hold the detector near a cold windowpane. Can you explain what happens?
4. Hold the detector above a light bulb or a candle. How far away can you get from the bulb and still have the detector move?
5. Open the refrigerator door a few inches. Hold the detector near the bottom of the door. Can you explain what happens? What about the top of the door?
6. Place a sheet of white paper and a sheet of black paper on a window sill that gets lots of direct sunshine. Move the detector slowly over the black paper and then over the white. Can you explain what happens?
7. From question 6, can you explain air currents over a field of dark soil and over a light-colored concrete road?
8. Hold your detector over your bare forearm. If your detector is finely balanced, the detector will rise. Why? Where did the heat come from?
9. Try your detector over cold water and then over warm water. Can you explain the difference?
10.Can you use your detector to plot the warm and cold air currents in your room?
11.Can you make the detector more sensitive by increasing the size of the tail? Would the detector be more sensitive if you used something longer than a straw?
Air Thermometer
Purpose: A thermometer is used to measure temperature, or the heat energy of air molecules. The one you are about to make is the same kind that Galileo, an Italian scientist, made over 350 years ago.17
Materials: Light bulb or baby bottle, one-hole rubber stopper, glass tubing about 1½’ long, and a pan holding water.
What to Do: You can make either of two kinds of air thermometers. The easiest to make is the one with the baby bottle or bulb on the bottom. If you use an old light bulb, remove the inside of the bulb as explained in the light bulb chemistry flask section (“Light Bulb Chemistry Flask”). Fill the bulb or bottle about half full of cold water. Put some food coloring or ink in the water. Insert the glass tubing through the stopper and all the way into the bulb or baby bottle. Insert the stopper as shown. Place a cardboard scale on the back of the tubing and hold it in place with Scotch tape. You can calibrate the scale later by comparing your readings with that of a regular thermometer. As the water in the bulb warms up to room temperature, it warms the air in the bulb. The air molecules move faster and push harder on the water. This pushes the water up the tube.
A second type of air thermometer has an inverted bulb. Heat the bulb so that some of the air will leave the bulb. Quickly insert the rubber stopper in the bulb or bottle, and stick the other end of the tubing in a pan or jar containing colored water. As the air in the bulb cools, the molecules don’t move as fast, and the water pushes part way up the tube. Fix a stand to hold the bulb or bottle and again attach cardboard for a scale.
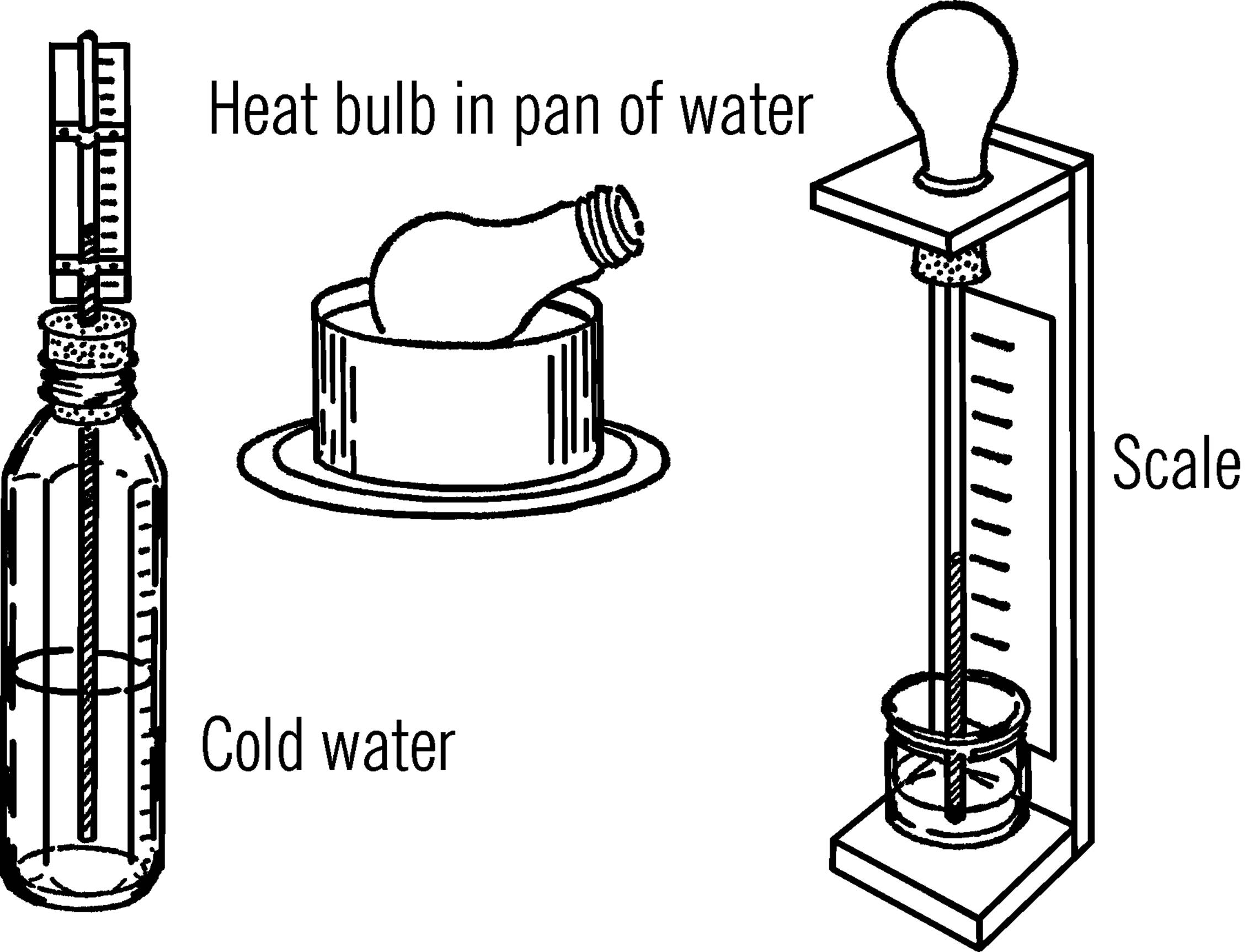
Operation of Equipment: After the air or water has reached room temperature, make a mark on the scale for the height of the liquid. Use another thermometer to give you the correct reading and mark it on the scale. Try your thermometer with different room and outdoor temperatures. Again calibrate with readings from a standard thermometer.
Can You Work Like a Scientist?
1. Does air pressure affect your readings? Which thermometer would be the most accurate?18
2. Try your thermometer directly in the sun. Is it accurate under direct sunlight? Check with a standard thermometer.
3. How high up a tube can you make the water climb by heating the air?
Cloud Jar
Purpose: The cloud jar is used to produce artificial clouds in the classroom in order to study the causes of clouds and do experimentation with clouds.
Materials: Gallon jug, one-hole rubber stopper (#6½),19 tire or hand pump, alcohol20 or ditto fluid.
What to Do: Pour a little alcohol into the bottom of the jug (you may use water if alcohol is not available). Insert the stopper into the jar. Pump air into the jar. Remove the stopper quickly.21
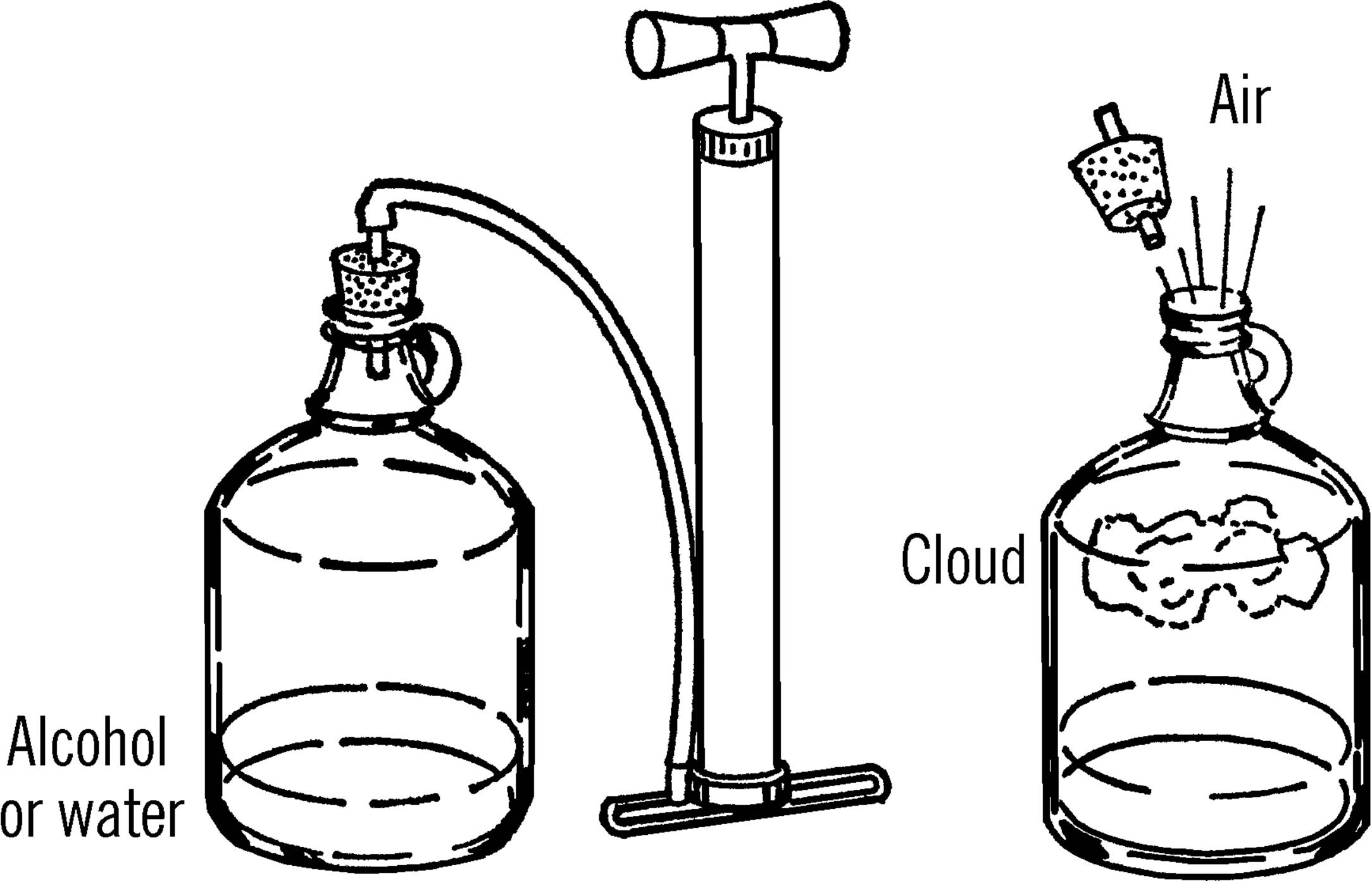
Operation of Equipment: When you remove the stopper quickly, a cloud should form.22
Modern Safety Practice
Wear safety glasses, just in case the glass should break. If using alcohol, follow procedures for working with chemicals, Note 17 in Appendix E, and keep away from sparks or flames.
Can You Work Like a Scientist?
1. Is the alcohol just in the bottom of the jar? After you pour the alcohol in the jug, smell the opening to the bottle.23 Where does this smell come from? Can you see any other alcohol besides that in the bottom of the jar? How long does it take for the alcohol to evaporate?
2. If you plug the hole in the bottle, will all the alcohol completely evaporate? Why?
3. When you pump air into the bottle, do you increase the air pressure? When you compress air, do you increase the temperature? Will warm air hold more vapor (bits of water or alcohol) than cold air?
4. When you put extra air into the bottle, does it hold more vapor?
5. When you pull out the cork, what happens to the extra air in the bottle? Does this raise or lower the temperature in the bottle? When air expands, does the temperature increase or decrease?
6. Does cooler air hold as much moisture?
7. What happens if air is suddenly cooled?
8. After you have formed your cloud, insert the rubber stopper and pump air into the jug. What happens to the cloud? Why?
9. If you don’t have a pump, try blowing into the bottle. Can you think of a way to make a permanent cloud jar that will make clouds and destroy them as you wish?
10.Put some water in a bucket. Drop in some pieces of dry ice.24 Does a cloud form? Can you explain what happens?
11.Put some dry ice in a can. Pour some alcohol on the ice. Let the can set for 15 minutes. Does snow form on the can? What causes snow? (Be careful-don’t touch dry ice or a mixture of dry ice and alcohol.)
Air Barometer
Purpose: The air barometer is used as a tool to measure the pressure of the air and thus predict future weather. The barometer is affected somewhat by temperature, so keep it in a place where the temperature doesn’t change very much.
Materials: Small 15-watt light bulb, #1 rubber stopper (one-hole), glass tubing about 9” long (5 mm.), a jar, and wood for a stand.25
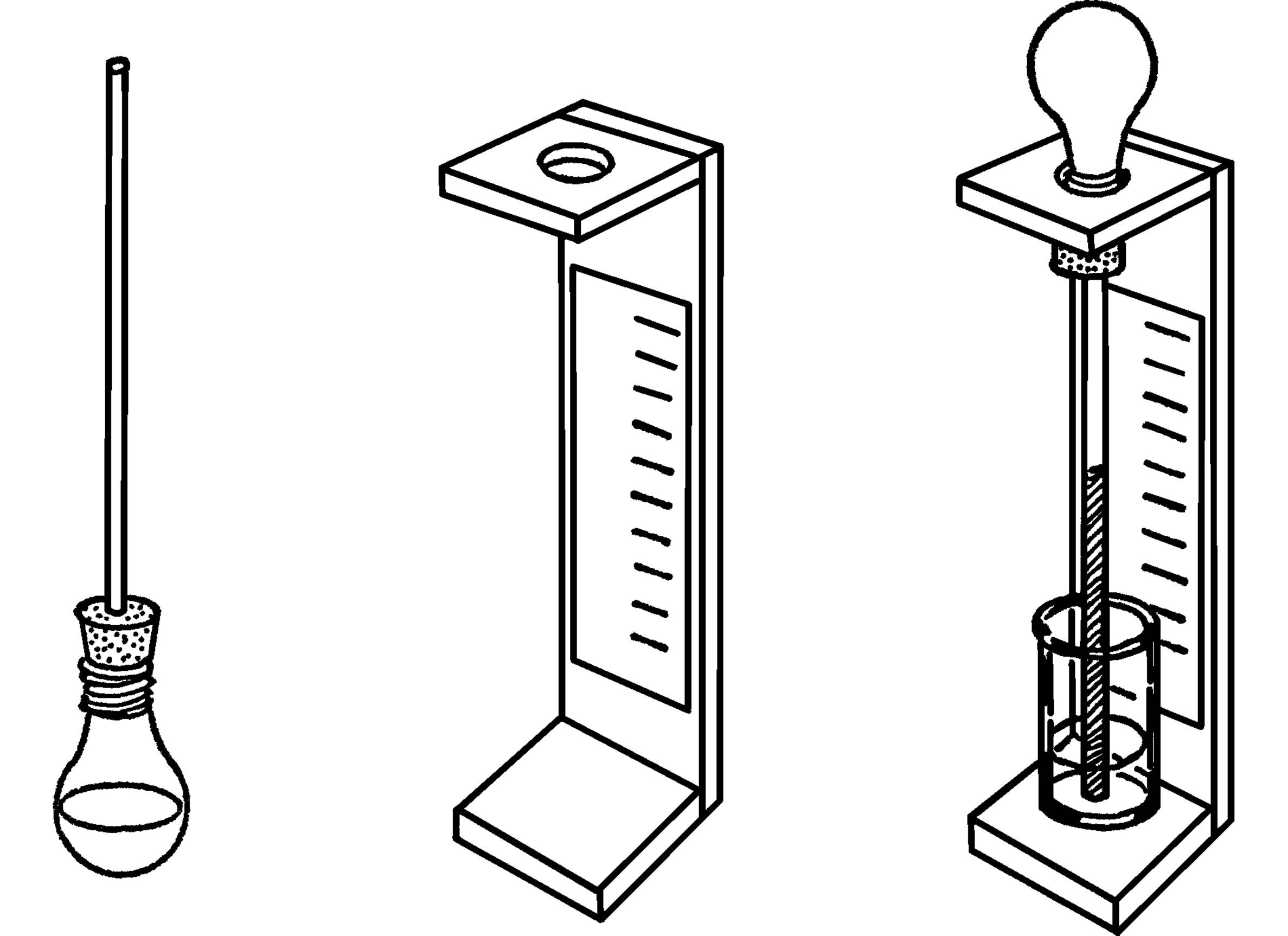
What to Do: You can blow a small ball on the end of your glass tubing (see “Large Pipette-Glass Blowing”), or you can use the light bulb and stopper. If you use the light bulb, remove the inside (see “Light Bulb Chemistry Flask”). Insert a #1 one-hole stopper as shown. Fill the bulb about one-fourth full of water. You can color the water with food coloring or ink. Insert the glass tubing into the stopper. Use soapy water on the glass tubing so the tubing will slip easily into the stopper. Turn the bulb and tube over so the small end is in a glass full of water. Attach a small strip of cardboard to the tubing with Scotch tape. Mark your scale by using a regular barometer to take sample readings. If one is not available, make division marks on the card and number from 1 to 10.
Operation of Equipment: When the air pressure increases, it pushes down on the water in the glass and forces the water up the tube. When the air pressure decreases, the pressure in the bulb is greater and forces the water down the tube.26
Modern Safety Practice
This project potentially involves glassblowing. Follow the glass working and fire safety guidelines in Note 2, Note 3, and Note 4 of Appendix E.
Can You Work Like a Scientist?
1. Place your hands on the bulb. What happens? Is this caused by a pressure change?
2. Keep a chart of the readings on your barometer over a period of time. Check your barometer every three hours. Also keep a record of the weather. Does your barometer help to forecast the weather? Remember, your readings forecast changes to occur in the weather one or two days ahead.
3. Place your barometer in a room containing lots of water vapor in the air. What happens? Why?
4. How can air be lighter when it contains water vapor? The bits of air (molecules) are pushed so closely together when the air pressure is high that there is little room for water vapor. When the pressure decreases, the bits of air move farther apart and can hold water vapor. Water vapor molecules weigh about half as much as the average air molecule. Can you demonstrate this using a sponge? When the sponge is squeezed together, this is high pressure. When the sponge is spread out we have low pressure. Which holds the most water?
Convection Current Box
Purpose: This box is used to study air currents caused by convection. When gases such as air are heated, the molecules move more rapidly. They spread out over a larger area and are not as tightly bunched together (dense). This warm air then contains fewer molecules and is lighter then the surrounding cooler air. The warm air, being lighter, rises, and the cooler air moves in to take the place of the rising warm air. Heat being moved or transferred because of unequal heating is known as convection.
Materials: Plywood or cardboard box, two quart jars, candle, and a roll of paper.
What to Do: Make the box as shown. Cut two holes just smaller than the diameter of the two mason jars. Cover the open side of the box with Saran Wrap or a piece of glass. Place a candle in one of the holes as shown.
Cut off the bottom of the two jars with your bottle cutter (see “Bottle Cutter”). Place the jars on the box as shown.
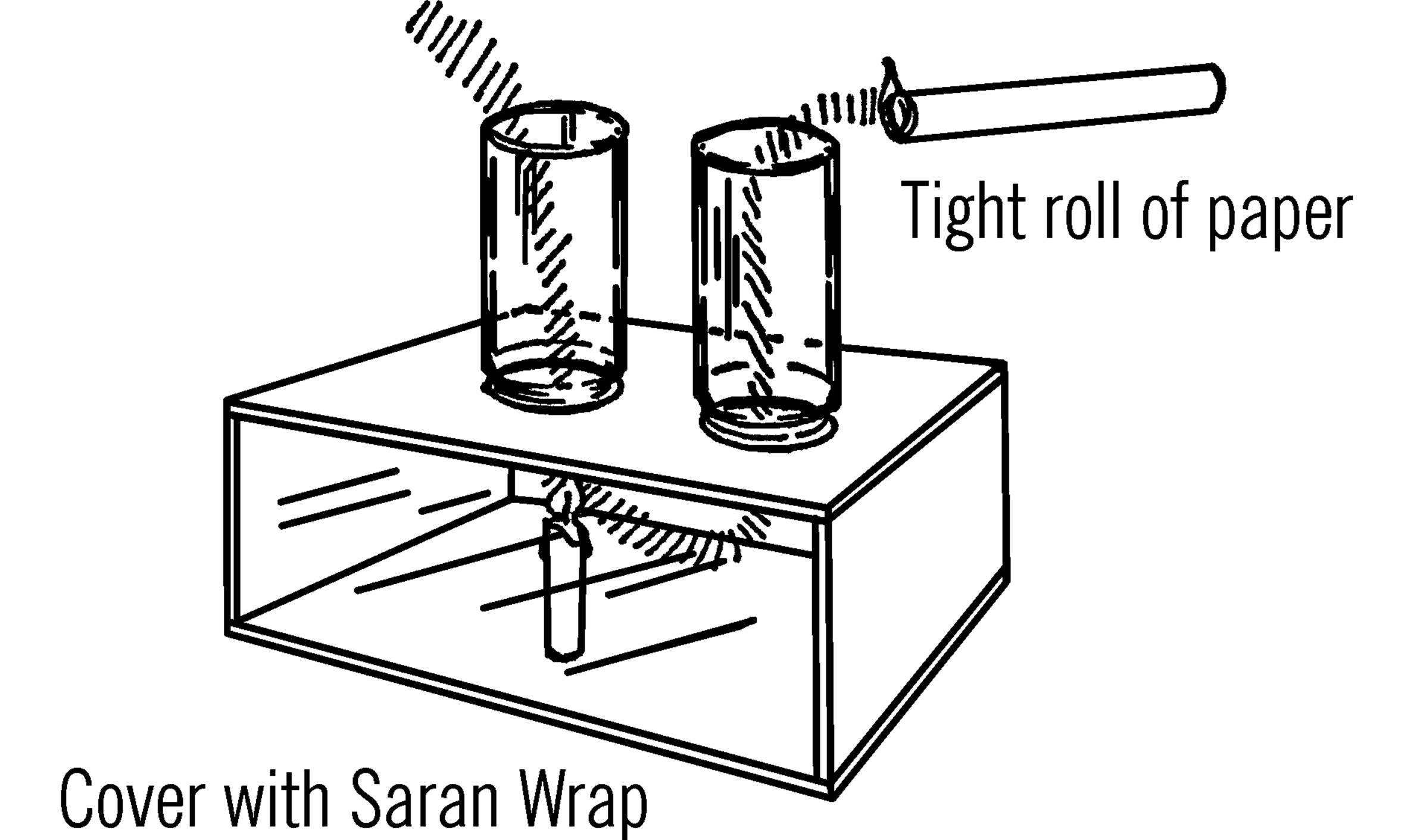
Operation of Equipment: Light the candle. Set the jars in place. Roll up some newspaper into a tight roll. Light the newspaper. The smoke should be sucked down the one jar chimney and up out the other one.
Modern Safety Practice
When lighting things on fire, use care, safety glasses, a non-flammable surface, and an area with good ventilation. And, have a fire extinguisher handy, just in case.
Read Note 3 in Appendix E for additional guidance on safety around fire.
Can You Work Like a Scientist?
1. Place a thermometer on the top of each jar. What is the difference in temperature of the two columns of air?
2. What is the effect on the strength of the convection current if the amount of heat given off by the candle is increased?
3. What effect do convection currents have on our weather?
4. What could cause the convection currents that affect our weather? What causes the unequal heating on the surface of the Earth?
5. Can you prove there are convection currents in your house or classroom?
6. Do convection currents occur in liquids as well as gases? Set up an experiment in an aquarium or a large pan of water. Use ink to trace the path of the water currents.27
Sunshine Recorder
Purpose: A sunshine recorder28 is used to measure and record periods of bright sunshine throughout the day.
Materials: Large burned-out light bulb, tin can, cardboard (2” × 10”), wood for base and support, and a rubber stopper to fit the light bulb.
What to Do: Remove the insides of a large light globe (see “Light Bulb Chemistry Flask”) such as is used in an auditorium or gymnasium. The clear light globe is ideal.29 Fill the light globe with water and insert a rubber stopper or cork.
Drill a hole in the wooden base so that the neck of the light globe fits in the hole. This sphere focuses the rays of the sun onto the cardboard scale.
Examine a globe of the Earth or a map to find out at what degree of latitude your city is located.30 Cut a triangular support board so that the sharp angle near the base is equal to the degree of latitude.
Cut a tin strip from a can that is large enough to hold the cardboard strip. The tin strip should be carefully formed so that it follows the curve of the light bulb. A cardboard is fastened on the tin strip so it can be easily removed and replaced by another piece of cardboard.
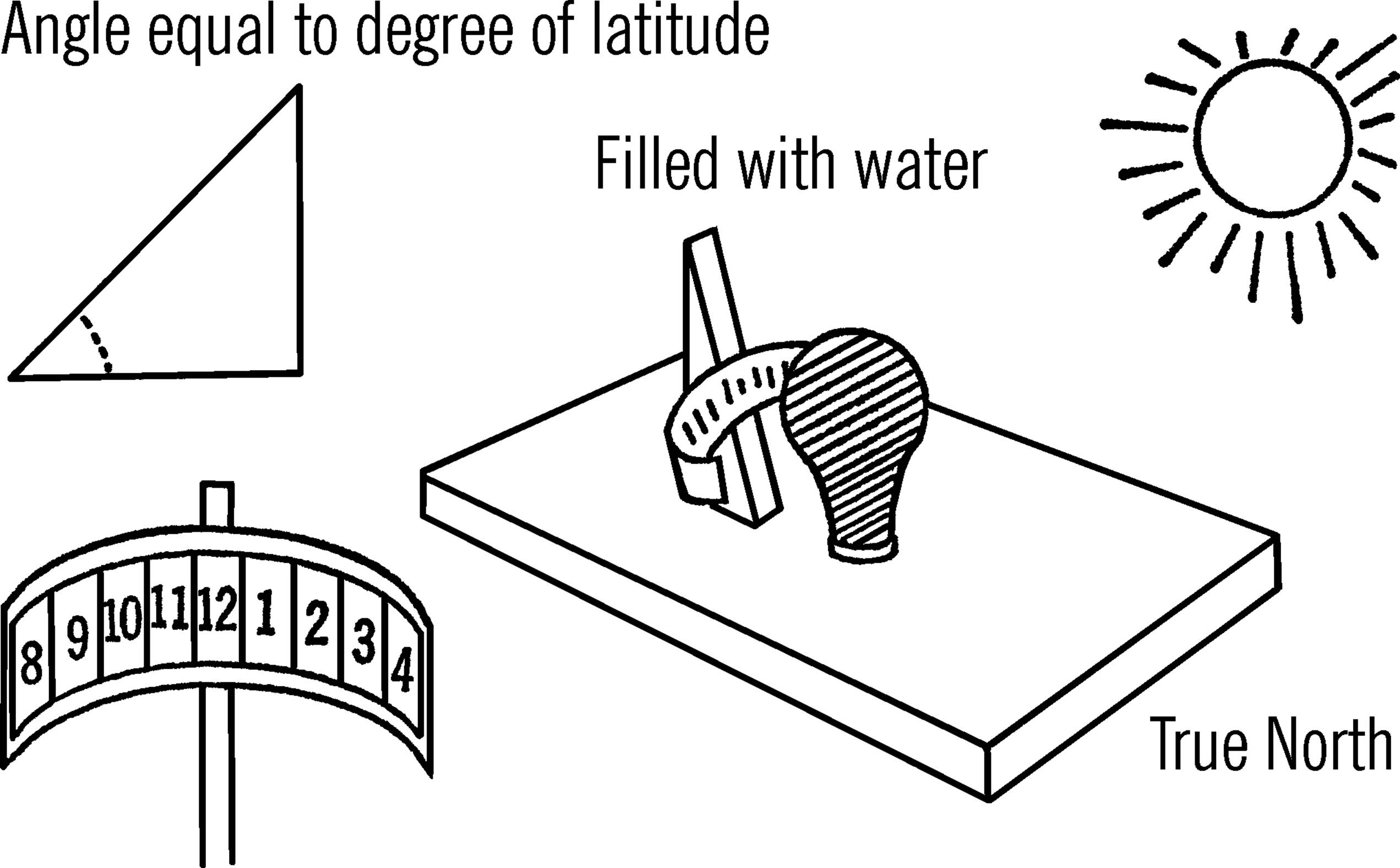
Operation of Equipment: On a sunny day place your sunshine recorder so it faces true north. Move your support and tin strip until the sun’s rays focus on the cardboard. The support is then screwed to the base. The tin strip is then adjusted and fastened to the support.
You can mark the position of the sun’s rays each hour on the piece of tin. The longer the tin and cardboard the longer the period of time you can record the duration of sunshine. Usually you will find a period of six to eight hours adequate and easy to check.
The rays of the sun focus on the cardboard strip and burn a line on the cardboard. The longer the period of sunshine, the wider the line going across the cardboard. You can measure somewhat the intensity of the sunlight by the degree of burning. Each mark on the cardboard can be subdivided so that the sunlight can be measured in tenths and even hundredths of an hour.
Each day you must change the cardboard or move it so that a fresh path can be burned across your chart.
Modern Safety Practice
When used correctly, this instrument will generally darken the cardboard, burning the amount of sunlight into the cardboard without causing a flame that spreads. Nonetheless, take precautions as though the cardboard could catch fire. It may be wise to use a fireproof base and locate it on a safe concrete area that can’t burn. You may also want to keep a close eye on it during its first use, just in case.
Can You Work Like a Scientist?
1. Why do you use a round lens such as the large light bulb instead of a regular lens?
2. What month of the year do you have the most days of sunshine?
3. How does the amount of sunshine and the duration of sunshine each day affect the growth rate of various plants, seeds, and trees?
Radiometer
Purpose: A radiometer is an instrument that can be used to measure indirectly the amount and intensity (strength) of sunlight. It has many other uses as a measuring device.
Materials: You can purchase a ready-made radiometer for about ten dollars from various sources including scientific supply companies. If you wish to make one, the materials needed are: pint mason jar, cork, long needle, glass eyedropper, 2” square of very thin cardboard, jar lid adapter, and a one-hole rubber stopper.
What to Do: Seal the small end of the eyedropper in the flame of an alcohol burner. After the dropper cools, cut a one-inch piece off the end with a file. Punch a hole in the center of the white cardboard square so that the cardboard will fit snugly over the tip of the eyedropper. Remove the cardboard and make the cuts shown with the dotted lines. Bend the cardboard tips down along the solid lines to form vanes. Blacken the back side of each vane by holding the cardboard carefully over a candle. Mark one white side with an “x.”
Stick the large end of the needle into the cork and place the cork in the mason jar. Set the eyedropper tip and cardboard vanes over the needle. Fasten the jar lid adapter (see “Tripod and Adjustable Rings”) to the mason jar with a jar ring. Connect a vacuum pump hose to the rubber stopper and remove as much air from the jar as possible. Seal the opening to the stopper.
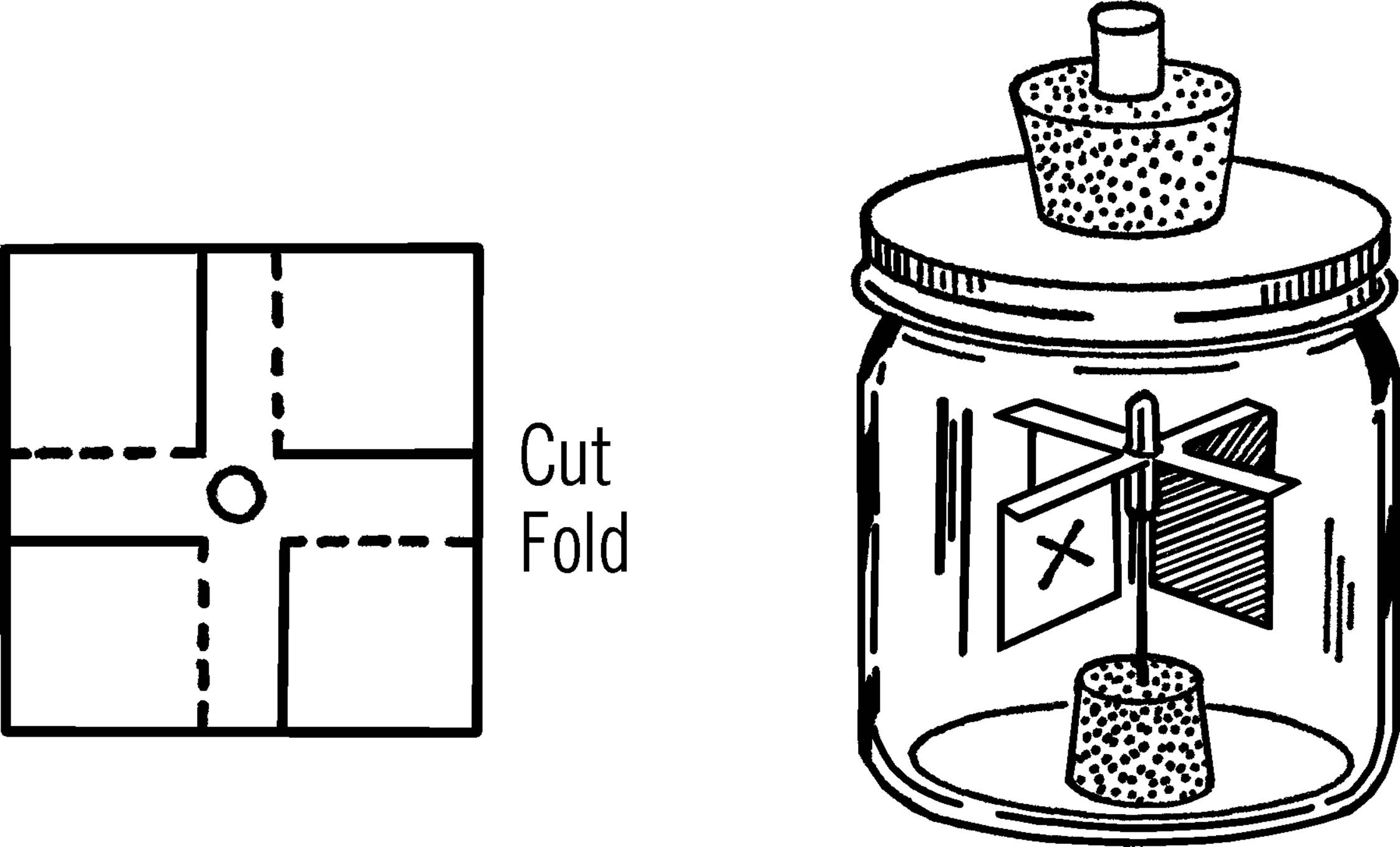
Operation of Equipment: Since most of the air has been removed from the inside of the radiometer, the paddle wheel or vanes are free to move. Normally the paddles would strike air molecules and be slowed up or stopped. With most air molecules removed, the slightest energy will start the paddles turning.
Can You Work Like a Scientist?
1. Will the energy from a light cause the paddles to turn?
2. Will heat energy (a match) cause the paddles to turn?
3. Does the paddle wheel always turn in the same direction? Does the energy seem to push the black or the white sides? Light and heat energy seems to travel in bundles somewhat like bullets. This energy striking the black side is absorbed or taken in. The energy striking the white side is repelled or reflected. Can you see why there is more push on the black side?
4. Do the paddles seem to turn faster when the light increases? Could you use this paddle wheel to measure the amount and strength of sunlight?
5. If you change electrical energy to light, can you measure the electrical energy by using the radiometer?
6. Can you measure the strength of heat or light by the rate the paddle wheel turns? How does the strength decrease as you increase the distance from the radiometer to the light or heat source?
Weather Balloon
Purpose: The weather balloon is used to carry various weather instruments high up into the atmosphere in order to record weather conditions at various altitudes.
Materials: Plastic cleaner bag.31
What to Do: The plastic bag should be filled with a light gas. Helium is non-explosive and can be secured at industries which store and distribute bottled gas.32 Hydrogen is easier to secure in large amounts but is somewhat explosive, especially if mixed with oxygen.
You can generate your own hydrogen, as mentioned in the chemistry section under hydrogen gas generator. Aluminum foil and hydrochloric acid produce the greatest amount of gas in the shortest time. Care should be taken that the bottle used to mix the acid and the foil can stand a great amount of heat, since much heat is given off during the reaction.
After the bag is filled with gas, the bag is tied. Light-weight instruments such as maximum and minimum thermometers can be attached to the plastic bag.33
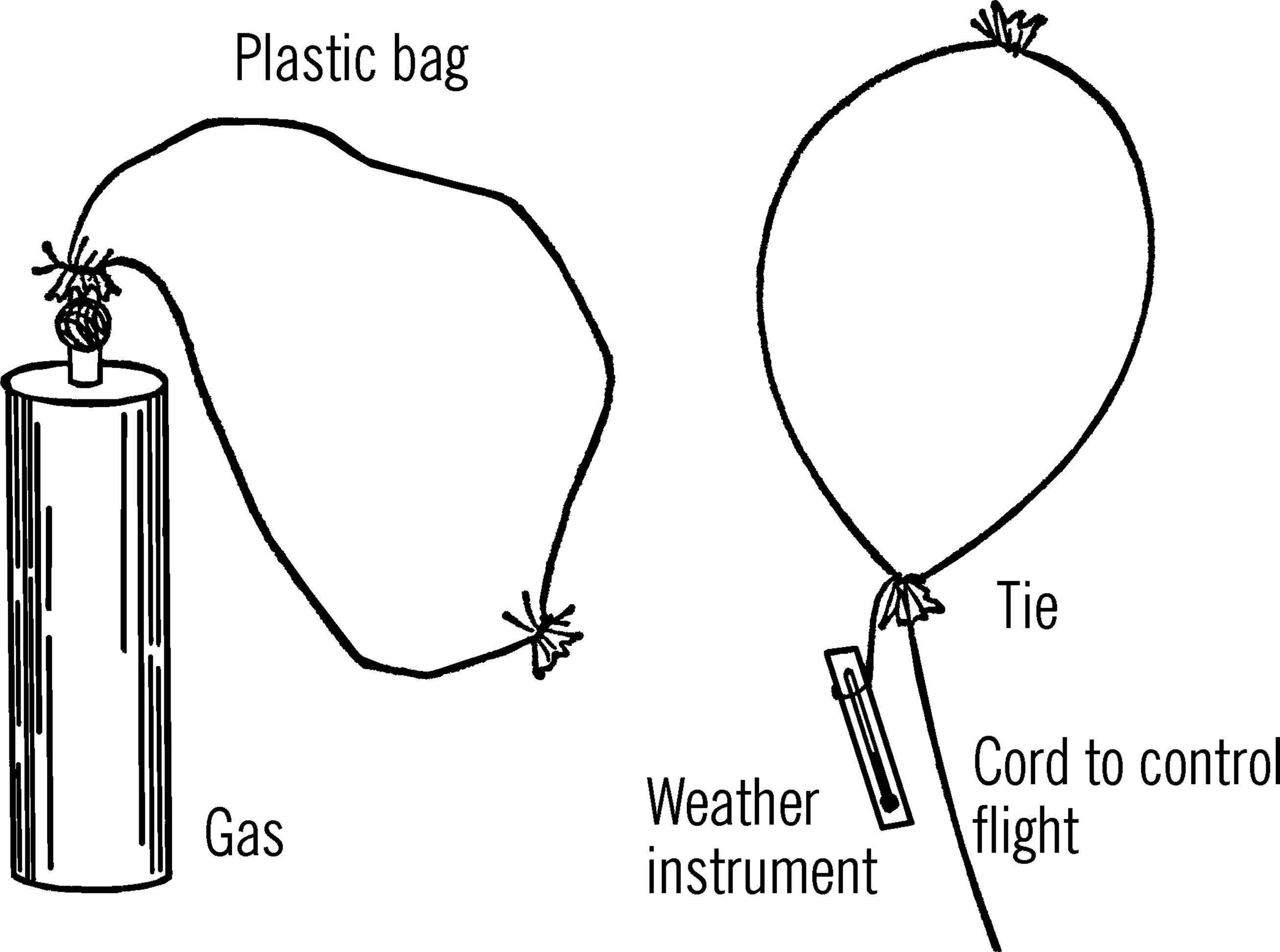
Operation of Equipment: A strong line or cord can be attached to the balloon. When the balloon is released, the cord should be eased through the fingers. Every ten feet can be marked on the cord so altitude can be determined. The plastic balloon can be recovered by reeling in the cord.34
Modern Safety Practice
1. A bag filled with hydrogen can make a very impressive explosion, which could cause injuries. If you use hydrogen, take extreme care to keep it away from possible sources of heat or flame.
2. Helium gas presents no explosion hazard, but does present a danger of asphyxiation. Never fill balloons or otherwise open a tank of helium without excellent ventilation. Helium can displace air in the room. Humans (and other living things) require the oxygen in air in order to breathe.
Can You Work Like a Scientist?
1. Can you measure the lifting force of your balloon by adding weights to the balloon until it is just held to the ground?
2. What effect does altitude have on the temperature?
3. What are the problems in trying to record weather data from a balloon?
4. How does the weather bureau secure its information?
5. How high can you get your balloon to rise?
6. Does your plastic balloon increase or decrease in size as it rises up into the atmosphere?
7. What effect does altitude have on air pressure? Can you use your balloon to prove this?
8. Can you devise a way of parachuting instruments or materials from your weather balloon?
9. Will your balloon rise if you fill it with hot air instead of with the gases mentioned?
10.What can you tell about upper air currents from the movement of your weather balloon?
Problems to Investigate in the Study of Meteorology (Weather)
(P)-Primary
(I)-Intermediate
(U)-Upper
1. Are there different kinds of clouds in the air? What are the names of these various clouds? (P)
2. What are clouds made of? Can you make a cloud? (P)
3. Where does rain come from? What causes rain? (P)
4. How are clouds formed? Are all clouds the same? (P)
5. What causes snow? Can you produce snow? (I)
6. What happens to snow when it melts? How much snow is necessary to make an inch of rainfall? (I)
7. Why is the sky different colors at different times? (I)
8. What is fog? Can you make fog? Blow across dry ice. (P)
9. Is soil always the same temperature? How about water and air? (P)
10.How often do weather conditions change? What causes these changes? (P)
11.Do winds warm or cool the earth? Why? (P)
12.From which direction does the wind blow most frequently where you live? (I)
13.What percentage or part of the time does the wind blow from the different directions around your home or school? (I)
14.How do the seasons of the year vary around the world? Why? Can you duplicate these conditions? (U)
15.Do all clouds form at the same heights in the sky? What kinds of clouds form at the different levels? Can you measure these heights? (U)
16.What is above the clouds? Can you produce this condition in a vacuum jar? (U)
17.What happens to the water that forms puddles on the ground? Where does the water go? Can you measure the rate of evaporation of different mud puddles? (P)
18.How does the water get into lakes, rivers, and ponds? Can you make a study of local bodies of water? (I)
19.Does the level of lakes, rivers, and ponds remain the same throughout the year? Can you measure the level of a body of water for a period of time? (I)
20.Is any water ever lost during the water cycle? Can you set up a water cycle? (P)
21.What conditions affect the speed of the water cycle? (I)
22.Why are some things damp in the early morning? Where does the water come from? (I)
23.Does dew make all objects equally damp? (I)
24.How much dew is formed on one square foot of surface throughout the year? (I)
25.Does the amount of dew vary with the season? (I)
26.What causes the windshield of your car to fog up on the inside on certain mornings? (I)
27.What causes the windshield of your car to fog up on the outside on certain mornings? (I)
28.What causes ice to form on the outside of your windshield? On the inside? (I)
29.Why is the outside of a milk carton sometimes wet? (P)
30.What causes drawers to stick at certain times of the year? (I)
31.Does the wind high up in the atmosphere blow in the same direction as the wind close to the ground? Make observations over a period of time. (I)
32.Does the wind far above the surface of the ground blow faster or slower than the wind close to the surface? (I)
33.What causes wind? Can you produce wind current by natural means? (I)
34.How fast can the wind blow? Measure with an anemometer over a period of time. (P)
35.What effect does the speed of the wind have on plant growth? (I)
36.What causes seasons? Can you demonstrate this in the classroom? (I)
37.What caused the Earth’s axis to tilt? (I)
38.What effect would the straightening of the Earth’s axis have on the Earth? (U)
39.Are both poles of the Earth illuminated at the same time? When does this occur? (U)
40.Can you record the length of the days and the length of the nights for a period of time? When is a day over and a night begun? (P)
41.Are the length of days and nights the same all over the world? Are they the same in your state? In your city? (I)
42.What must the temperature of the air be in order for frost to form? (I)
43.Does the amount of humidity have any effect on the formation of frost? (I)
44.Does the amount of humidity have any effect on the rusting of metals? (I)
45.Are frost and dew made of the same percentages of hydrogen and oxygen as regular water? (I)
46.Do frost and dew contain minerals? (I)
47.What periods of the year have days and nights of equal length? Is this true all over the world? Use a universal sundial to determine this. (I)
48.What effect does pressure have on the amount of rainfall in your area? (U)
49.How does air pressure vary from day to day in your area? (I)
50.Does air pressure vary in different parts of the city? (I)
51.Is warm air heavier or lighter than cold air? How much? (I)
52.Is the air warmer nearer the floor or the ceiling of your home? (P)
53.Does the temperature difference between the floor and the ceiling of your house remain the same throughout the year? Is this difference the same in all parts of your house? (U)
54.What conditions are necessary for thunder and lightning in your area? (I)
55.What kinds of clouds are present when thunder and lightning occur? (I)
56.Can you cause precipitation (rain) using any temperature of air? (U)
57.Is the rate of evaporation of water affected by temperature? (I)
58.Is this rate affected by humidity? By wind speed? (I)
59.What affects the rate of evaporation most: temperature, humidity, or wind speed? (U)
60.Does rapid evaporation cause heating or cooling? (P)
61.What effect does this rate of evaporation have on the temperature change? (P)
62.Does weather have an effect on the amount of curl in your hair? (I)
63.Does the amount of humidity have an effect on how fast your hair dries out? (I)
64.Can you measure the force of raindrops? Does this force vary? (U)
65.How fast does a raindrop travel? (U)
66.What is the shape of a raindrop? (P)
67.Into what layers is the atmosphere usually divided? Can you produce conditions similar to each layer? (U)
68.Does the temperature of the atmosphere increase or decrease as altitude increases? Is there any exception to this? (I)
69.What causes meteors? Can you find material on Earth similar to meteors? (U)
70.What is a meteorite? Why do meteors burn up? Can you produce similar friction conditions with a grinding stone? (I)
71.How hot do meteors get when they enter our atmosphere? Is there any kind of life that can live at this temperature? (U)
72.How can we predict meteor showers? (I)
73.What velocity must a meteor be traveling when it strikes the Earth? (I)
74.Do all solar bodies have atmospheres? Why? (I)
75.Do land surfaces or water surfaces retain heat the longest? (I)
76.Does salt water retain heat for a longer time than an equal amount of fresh water? (I)
77.Does the temperature of the Earth vary with depth? (I)
78.Does the temperature of water vary with depth? (I)
79.During what part of the day does the Earth receive heat? During what part of the day does the Earth start to lose heat? (I)
80.Does air pressure increase or decrease with an increase in altitude? (I)
81.Does air density increase or decrease with an increase in altitude? (I)
82.What effect does increased temperature have on air density? (U)
83.Do water vapor molecules weigh more or less than dry air molecules? (U)
84.How much moisture can the air hold at different temperatures? How does increased temperature affect the amount of moisture necessary to saturate the air? (U)
85.At what temperature can the air retain the most moisture? (U)
86.Can you separate a sample of the atmosphere into its parts? What is the percentage of each part in the total sample? (I)
87.Is the make-up of the atmosphere constant, or does it vary from place to place? (U)
88.Does the composition of the atmosphere vary with increased altitude? (U)
89.What holds the atmosphere to the Earth? What kind of atmosphere could the moon retain? (I)
90.Is heat given off by land and water? How does this heat energy travel? (I)
91.Does the sun heat the air directly? How? (U)
92.What is heat radiation? What factors affect the amount of heat radiated? (I)
93.What is conduction? What factors affect the amount of heat conducted? (I)
94.What is convectional mixing in the atmosphere? How can heat travel by convection? (U)
95.How does pressure rise in the atmosphere increase the temperature of the atmosphere? (U)
96.How does the atmosphere retard the heat loss? (U)
97.How can impurities in the air affect the amount of radiant energy from the sun that is transmitted to the Earth? (U)
98.Would changes in the composition of the atmosphere affect the amount of radiant energy transmitted to the Earth from the sun? (U)
99.How accurate are snow forecasts? (I)
100. How accurate are rain forecasts in your area? How about sun forecasts? (P)
101. How accurate are studies of cloud formations as a basis for predicting weather? (I)
102. What parts of the world could benefit from cloud seeding? What conditions are necessary for cloud seeding? (U)
103. Would weather change if the amount of heat were constant through the Earth? (I)
104. Can you compute what fraction of the sun’s energy is reflected by snow? What part is absorbed? (U)
105. Can you compute what fraction of the sun’s energy reaches the Earth? (U)
106. Does a cloud reflect sunlight? (I)
107. How do different surfaces affect the amount of sunlight reflected and absorbed? (I)
108. What kind of rays pass through glass? Why is it warmer inside a greenhouse? (I)
109. Does the Earth cool faster on cloudy or clear evenings? (I)
110. What land and water surfaces cause local winds in your area? Can you produce a wind in your classroom? (I)
111. Can you collect the amounts of water in the air at different temperatures? (I)
112. How much water is given off by the leaves of various trees and plants? What type of plant gives off the most water per square inch of leaf surface? (I)
113. What type of plant gives off the least amount per square inch? (I)
114. What is the highest temperature at which your breath can form a miniature cloud? (I)
115. What causes early-morning sea or lake breezes? (I)
116. Why do clouds often form near a mountain peak? (I)
117. What is the difference between the temperature in direct sunlight and the temperature in complete shade? Is this difference constant or does it vary? (P)
118. What do different cloud formations mean in your area? (I)
119. How does the amount of moisture in the air affect a spider’s web? (I)
120. Why doesn’t rain or snow faIl constantly from all clouds? (U)
121. How does the size of a cloud droplet compare with the size of a raindrop? (U)
122. How does a cloud droplet grow? (I)
123. How cold must a cloud be before snow crystals will form? (U)
124. What causes a halo around the moon? Does it help you to predict weather? (I)
125. What causes freezing rain? What causes ice pellets? (I)
126. How does a hailstone grow? (U)
127. What causes snow crystals? Are snow crystals all alike? (U)
128. What causes freezing rain? Can you produce this condition? (I)
129. Does dew form on clear or cloudy nights? (P)
130. How do frost crystals compare with snow crystals? (I)
131. How do different kinds of soil affect the amount of run-off water that results from a thunderstorm or cloudburst? (I)
132. What is the height of the water table on your property during the year? (I)
133. What is the weight of snow as compared to an equal amount of water? (P)
134. What effect has temperature on the rate at which snow melts? (P)
135. What causes water to seep through the ground? What affects this seepage? (I)
136. How does dry ice seeding cause rain? (U)
137. What gases found in air will sustain life? (I)
138. What effect does ozone have on the radiation from the sun? (U)
139. What is the composition of the atmosphere 500 or more miles above the surface of the Earth? (I)
140. What effect does the amount of carbon dioxide in the atmosphere have on the heat energy from the sun? (U)
141. How high up in the sky can you send a hydrogen balloon? (I)
142. Can you study upper wind currents with hydrogen balloons? (I)
143. Which balloon will rise quicker and higher, one filled with hydrogen or one filled with helium? Can you measure the difference? (U)
144. What effect has ionized air on radio waves? (U)
145. Does the temperature increase or decrease as a gas expands? (I)
146. Does the temperature increase or decrease as a gas contracts or is compressed? (I)
147. Can you make an air conditioner? (I)
148. Why does air cool as it rises? (I)
149. Why is it warmer on the leeward side of a mountain? Set up a class experiment. (I)
150. How do Chinook winds warm temperature? (I)
151. What causes the jet streams? (U)
152. What causes vapor trails after high-flying jets? (I)
153. What causes the temperature range in the ionosphere? (U)
154. What causes auroras? (U)
155. What is the summer solstice? The winter solstice? What is the equinox? (I)
156. Why is the summer warmer than the winter? (I)
157. Why do direct light rays of sunlight give off more energy than rays coming in at a slant? (I)
158. If the longest day of the year in the Northern Hemisphere is in June, why is the hottest month of the year usually August? (I)
159. Why is the warmest part of the day in the afternoon? (I)
160. How does the temperature change during the day? (P)
161. What time of day is usually the warmest? (P)
162. How fast does the Earth rotate at the various latitudes? (I)
163. How does the rotation of the Earth affect the pattern of winds in the Northern and Southern Hemispheres? (Use a phonograph for the experiment.) (U)
164. What causes the “trade winds” and the “prevailing westerlies?” (I)
165. What causes the polar easterlies? (I)
166. Are the wind patterns the same in the Southern Hemisphere? (I)
167. What causes the “equatorial doldrums” and the “horse latitudes?” (I)
168. What causes high- and low-pressure areas? (I)
169. Does wind flow from low-pressure regions to high-pressure regions? (I)
170. Why do the winds near the ground blow in a different direction from those high above the Earth? (I)
171. How are air masses formed? How do air masses affect our weather? (U)
172. What is the difference in winter and summer temperatures of the ocean? (I)
173. What causes warm and cold fronts? (I)
174. Does the speed of cold fronts vary from winter to summer? (U)
175. What causes squall lines? (I)
176. Can you predict the sequences of a cold front? How accurately? (I)
177. Can you predict the sequences of a warm front? (I)
178. What causes thunderstorms? Can you tell how far away such a storm is by the sound of the thunder? (I)
179. Why does cold air precede a thunderstorm? (U)
180. What causes thunder? (P)
181. What causes lightning? (P)
182. Does lightning originate from the cloud or from the ground? (U)
183. How do lightning rods protect people? (I)
184. What causes tornadoes? (I)
185. How can a tornado cause a house or barn to explode? (I)
186. Why do most hurricanes which affect the U.S. occur in the summer? (I)
187. Can you forecast weather from reports and maps issued by the weather bureau? (U)
188. How does an anemometer measure the speed of the wind? (P)
189. How does a rain gauge work? Can you measure the rainfall over a period of time and compare the amount with that of the weather bureau? (P)
190. Can you design an instrument to measure the duration of sunshine each day? (I)
191. Can you design a new method for making a wind vane? How does the direction of the wind in your area affect the type of weather you will have in the near future? (I)
192. Can you compare the accuracy of a mercury barometer with that of an aneroid barometer? Can you construct these two types? (I)
193. Can you design and build an automatic recording weather station? Be sure to record your findings over a period of time. (U)
194. What is the freezing temperature of mercury? Of alcohol? Of salt water? (I)
195. Can you construct an accurate thermometer? (P)
196. How does a maximum thermometer work? A minimum thermometer? (I)
197. Is a sling psychrometer more accurate than a wet- and dry-bulb thermometer for measuring humidity? Can you construct a sling psychrometer? (I)
198. How much does a human hair contract and expand when exposed to changing humidity? (U)
199. How does the human hair compare with the hair from other animals as far as contracting and expanding during changing humidity? (U)
200. Can you experiment with other materials and compare their expansion and contraction with changes in humidity and also temperature? (I)
201. Can you make an automatic recording rain gauge? How does it compare with your regular rain gauge? (U)
202. Can you make an aerovane wind speed and direction indicator? Can you make one so it will automatically record both speed and direction of the wind? Be sure to use your instrument for some project. (U)
203. Can you check the accuracy of the Beaufort wind scale? (I)
204. Can you devise ways of measuring the ceiling height of clouds? (U)
205. What is an inclinometer? Can you make a similar instrument and use it? (U)
206. Can you make a theodolite and then use a pilot balloon to keep track of winds aloft? Be sure to record your findings. (U)
207. Can you devise a radiosonde receiver and transmitter that will work at a limited height? (Be sure you attach a cord so you won’t lose your instruments.) (U)
208. Does it rain more in the city or in the nearby country? (I)
209. What are local problems in forecasting weather in your area? (I)
210. Can you determine the average temperature in your area for a period of time? (P)
211. What kinds of crops will grow in the temperature ranges you have determined? (I)
212. Can you determine the average hours of sunshine during different periods of the year? (I)
213. What effect will this average have on different crops? (I)
214. How many killing frosts do you have in your area during a year? (I)
215. How much snowfall do you have in a year? Compare this with your readings in other localities. (I)
216. How many clear days do you have in a year? (P)
217. How do animals and insects adjust to temperature changes in your area? (P)
218. Can you predict weather by observing the movements or activities of various animals and insects? (I)
219. What is the maximum amount of water a cubic foot of air can hold at various temperatures? (U)
220. What effect does the relative humidity reading have on bodily health and comfort? (I)
221. What effect does relative humidity have on fire danger? Can you experiment with the kindling temperature necessary for fire at different humidity readings? (U)
222. Can you determine the dew point of the air over a period of time? What effect does temperature have on the dew point? (I)
223. How accurate are old-time weather signs and sayings? (P)
224. How accurate is the almanac in predicting weather in your area? (I)
225. What is the background count due to cosmic rays in your area? Use a Geiger counter to determine this. (U)
226. Can you determine the fallout rate of radiation in your area due to the testing of nuclear weapons?35 (U)
227. Can you determine from weather maps the percentage of times wind currents would carry radiation from a target area to various locations in your state? (U)
228. Why would a nuclear bomb suck up dirt into its mushroom? (I)
229. Does the temperature of the air have any effect on the rate radiation travels? (U)
230. Does radiation affect both living and non-living materials? (U)
231. Are students more restless during cloudy weather? (I)
232. What effect do changing temperatures have on mental ability? (I)
233. What effect does temperature have on the lubricating qualities of various oils? (U)
234. What is the air pollution rate in your area? How does this rate vary with industrial activity? (U)
235. What effect has a polluted atmosphere on the health of various animals, such as white rats? (U)
236. What industries around your area contribute to the pollution problems? What materials are released? (U)
237. What causes “smog?” (I)
238. What effect does ionized air have on behavior? (U)
239. Do negative ions relieve hay fever and other air-borne allergies? (U)
240. What is the value of various insulating materials? (I)
241. Why is it possible for Eskimos to keep warm in ice huts? (I)
242. Can you make an estimate of the heat loss through the windows and walls of your house? (U)
243. How can a mercury barometer be used to determine altitude? (I)
244. Why do breezes at the shore blow in one direction during the day and in the opposite direction at night? (I)
1 Could you design a version of this instrument that could be 3D printed instead?
2 In the modern era, we are normally trained not to stick appendages out of a car window, because it’s an excellent method of creating a rapid unscheduled amputation. Use appropriate care.
3 Related question: How detailed are the wind speed maps available online?
4 Check online precipitation maps.
5 Check your local dollar store. The mirror surface itself needs to be flat (not curved for magnification).
6 I.e., self-adhesive labels.
7 I.e., a bristle from a straw broom.
8 The 1960s version of this book had “your mother washes”(!) here.
9 Use a pointy piercing can opener, not one that cuts the lid off.
10 See Note 41 in Appendix E about soldering to a can.
11 This particular hygrometer is interesting but more challenging to build and get working than the “Wet- and Dry-bulb Hygrometer”.
12 You can use Figure 7-11 as a “template” to make your own daily weather chart. What other things have you learned to measure that might have some bearing on predicting the weather?
13 Mercury may be affected less by changes in temperature than other types, but it is not unaffected. If mercury did not change volume/density with temperature, would mercury thermometers work?
14 For better or worse, that’s no longer so easy to get. See Modern Safety Practice for this project.
15 This is a traditional weather forecasting device also known as “FitzRoy’s Storm Glass.” But does it actually work? See Note 43 in Appendix E.
16 See Appendix A for sourcing information.
17 This apparatus is useful to build for the experience and understanding, but it is not very useful as a thermometer. (Why?) Use a modern thermometer (glass or digital) when you need to monitor temperatures.
18 Hint: Compare this apparatus with the one in “Air Barometer”.
19 A 2-liter soda bottle with a #3 stopper is a good substitute.
20 See Note 6 in Appendix E about alcohols.
21 You will likely need to hold the stopper in place by hand; it will want to pop out on its own. If your pump has a gauge, aim for about 20 PSI of pressure.
22 How is this different from the “Diffusion Cloud Chamber”?
23 This is improper procedure. Waft the air near the opening to the bottle towards your face, using your hand. Never smell chemicals directly.
24 See Note 14 in Appendix E about working with dry ice.
25 For an alternative design, see the “weather glass,” Note 42 in Appendix E.
26 How is this apparatus different from that of the “Air Thermometer”? What does that tell you about how the two instruments behave?
27 Small aquariums are normally made from soda lime (window) glass, which does not handle changes in heat very gracefully. (By contrast, Pyrex, or borosilicate glass, is frequently made into glass stovetop cookware.) Thus, putting an open flame directly under an aquarium could crack the glass, resulting in the release of its water. Can you think of a way to heat part of the bottom, but that would be safe for the glass?
28 This particular type of sunshine recorder is known as a Campbell–Stokes recorder. Commercial versions are typically made with a glass sphere and metal support.
29 That is, a “globe” style (near-spherical) incandescent light bulb with a clear—not frosted or silvered—glass envelope. Globe bulbs are sometimes called “vanity” bulbs. The “G25” size bulb is a good choice.
30 Alternately, use an online geocoder service and enter your address.
31 A lightweight, clear plastic trash can liner bag is an excellent choice. Would there be any advantage or disadvantage to using a black bag?
32 Helium can now be obtained easily from party stores, to fill balloons.
33 A maximum and minimum thermometer is a special type of thermometer (mercury or digital) that records the maximum and minimum temperatures experienced.
34 See Note 44 in Appendix E about cords and tethered balloons.
35 Low levels are plausibly still present from historical nuclear tests, in some areas.