MCAT Biology and Biochemistry: New for MCAT 2015 (2014)
Chapter 3. Biologically Important Molecules
The biological macromolecules are grouped into four classes of molecules that play important roles in cells and in organisms as a whole. All of them are polymers; strings of repeated units (monomers).
This chapter discusses the biomolecules from a biological perspective: what they are made of, how they are put together, and what their roles are in the body. These molecules are also discussed in MCAT Organic Chemistry Review (Chapter 7) from an organic chemistry perspective: nomenclature, chirality, etc.
3.1 PROTEIN BUILDING BLOCKS
Proteins are biological macromolecules that act as enzymes, hormones, receptors, channels, transporters, antibodies, and support structures inside and outside cells. Proteins are composed of twenty different amino acids linked together in polymers. The composition and sequence of amino acids in the polypeptide chain is what makes each protein unique and able to fulfill its special role in the cell. Here, we will start with amino acids, the building blocks of proteins, and work our way up to three-dimensional protein structure and function.
Amino Acid Structure and Nomenclature
Understanding the structure of amino acids is key to understanding both their chemistry and the chemistry of proteins. The generic formula for all twenty amino acids is shown below.
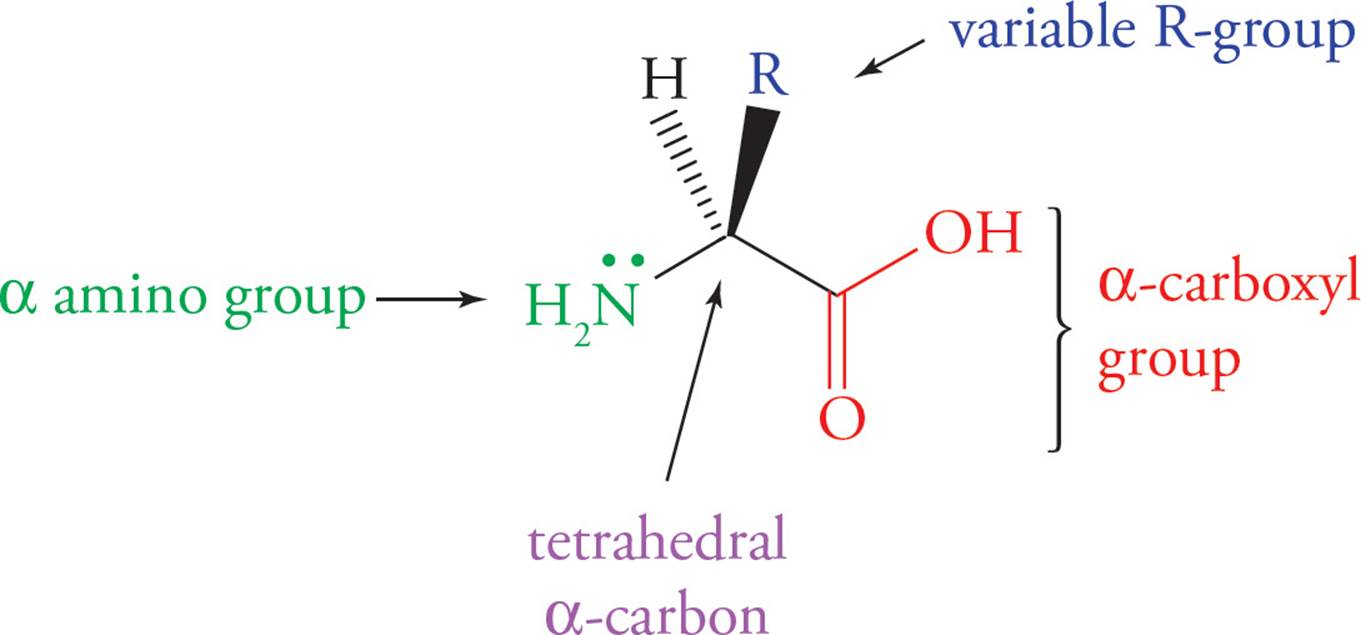
Figure 1 Generic Amino Acid Structure
All twenty amino acids share the same nitrogen-carbon-carbon backbone. The unique feature of each amino acid is its side chain (variable R-group), which gives it the physical and chemical properties that distinguish it from the other nineteen.
Classification of Amino Acids
Each of the twenty amino acids is unique because of its side chain. Each amino acid has a three-letter abbreviation and a one-letter abbreviation, which you do not need to memorize. Though they are all unique, many of them are similar in their chemical properties. It is not necessary to memorize all 20 side chains, but it is important to understand the chemical properties that characterize them. The important properties of the side chains include their varying shape, charge, ability to hydrogen bond, and ability to act as acids or bases. These side group properties are important in the structure of proteins.
We now consider the 20 amino acids, organizing them into broad categories:
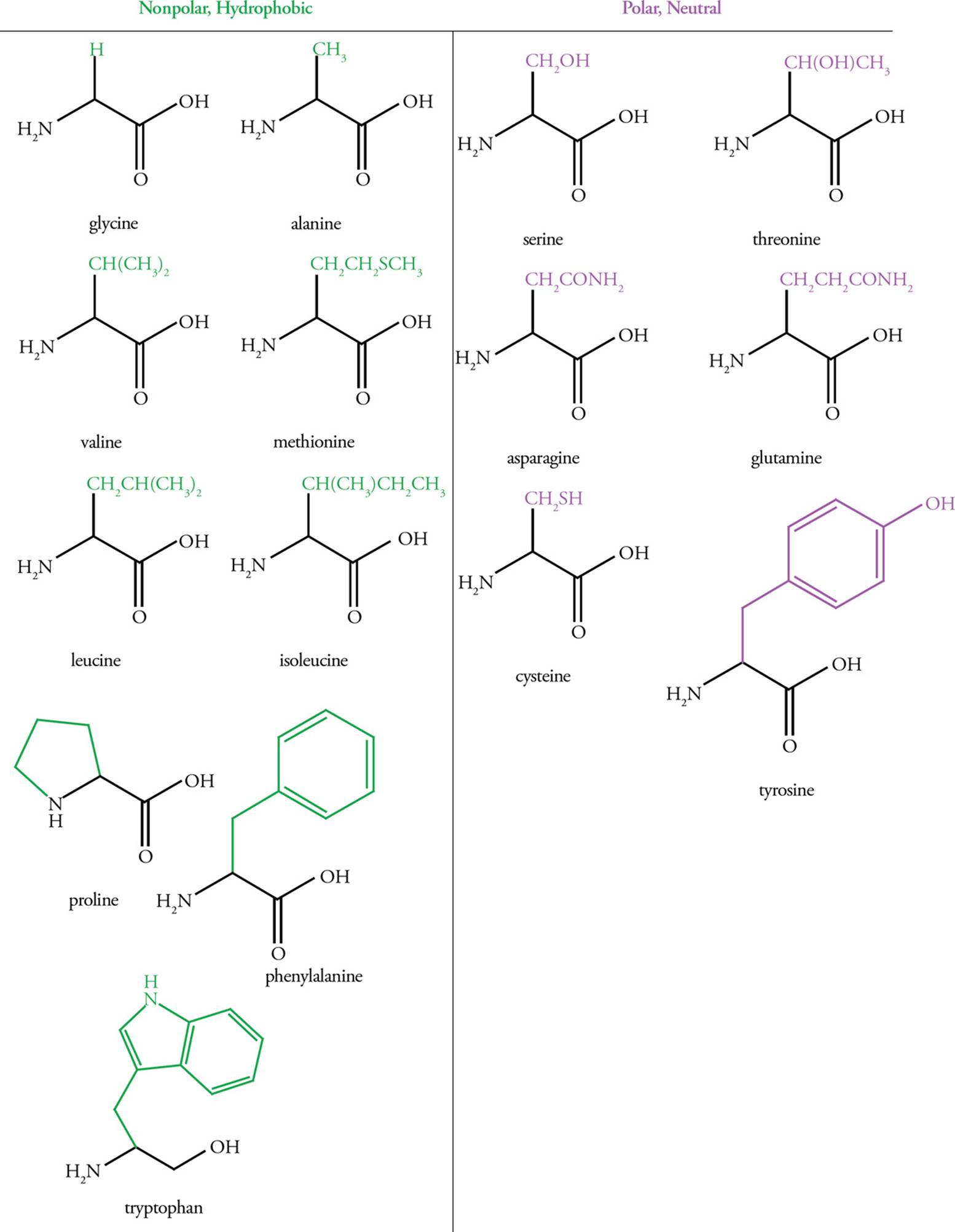
Hydrophobic (Nonpolar) Amino Acids
Hydrophobic amino acids have either aliphatic (straight-chain) or aromatic (ring structure) side chains. Amino acids with aliphatic side chains include glycine, alanine, valine, leucine, and isoleucine. Amino acids with aromatic side chains include phenylalanine, tyrosine, and tryptophan. Hydrophobic residues tend to associate with each other rather than with water, and therefore are found on the interior of folded globular proteins, away from water. The larger the hydrophobic group, the greater the hydrophobic force repelling it from water.
Polar Amino Acids
These amino acids are characterized by an R-group that is polar enough to form hydrogen bonds with water but not polar enough to act as an acid or base. This means they are hydrophilic and will interact with water whenever possible.
The hydroxyl groups of serine, threonine, and tyrosine residues are often modified by the attachment of a phosphate group by a regulatory enzyme called a kinase. The result is a change in structure due to the very hydrophilic phosphate group. This modification is an important means of regulating protein activity.
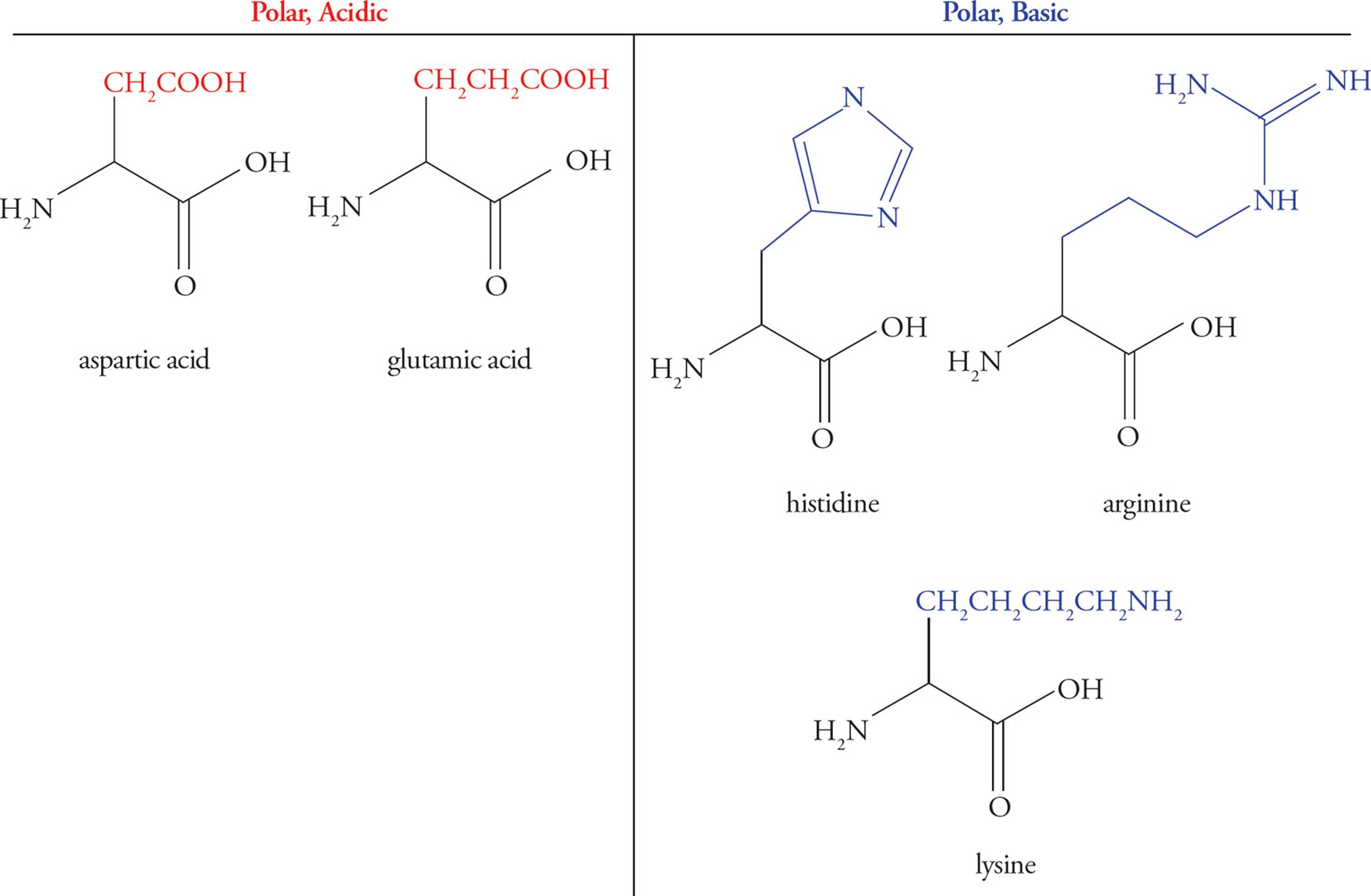
Acidic Amino Acids
Glutamic acid and aspartic acid are the only amino acids with carboxylic acid functional groups (pKa ≈ 4) in their side chains, thereby making the side chains acidic. Thus, there are three functional groups in these amino acids that may act as acids or bases—the two backbone groups and the R-group. You may hear the terms glutamate and aspartate—these simply refer to the anionic (unprotonated) form of the molecule.
Basic Amino Acids
Lysine, arginine, and histidine have basic R-group side chains. The pKas for the side chains in these amino acids are 10 for Lys, 12 for Arg, and 6.5 for His. Histidine is unique in having a side chain with a pKa so close to physiological pH. At pH 7.4 histidine may be either protonated or deprotonated—we put it in the basic category, but it often acts as an acid, too. This makes it a readily available proton acceptor or donor, explaining its prevalence at protein active sites (discussed below). A mnemonic is “His goes both ways.” This contrasts with amino acids containing –COOH or –NH2 side chains, which are always anionic (RCOO–) or cationic (RNH3+) at physiological pH. (By the way, histamine is a small molecule that has to do with allergic responses, itching, inflammation, and other processes. (You’ve heard of antihistamine drugs.) It is not an amino acid; don’t confuse it with histidine.)
Sulfur-Containing Amino Acids
Amino acids with sulfur-containing side chains include cysteine and methionine. Cysteine, which contains a thiol (also called a sulfhydryl—like an alcohol that has an S atom instead of an O atom), is actually fairly polar, and methionine, which contains a thioether (like an ether that has an S atom instead of an O atom) is fairly nonpolar.
Proline
Proline is unique among the amino acids in that its amino group is bound covalently to a part of the side chain, creating a secondary α-amino group and a distinctive ring structure. This unique feature of proline has important consequences for protein folding (see Section 3.2).
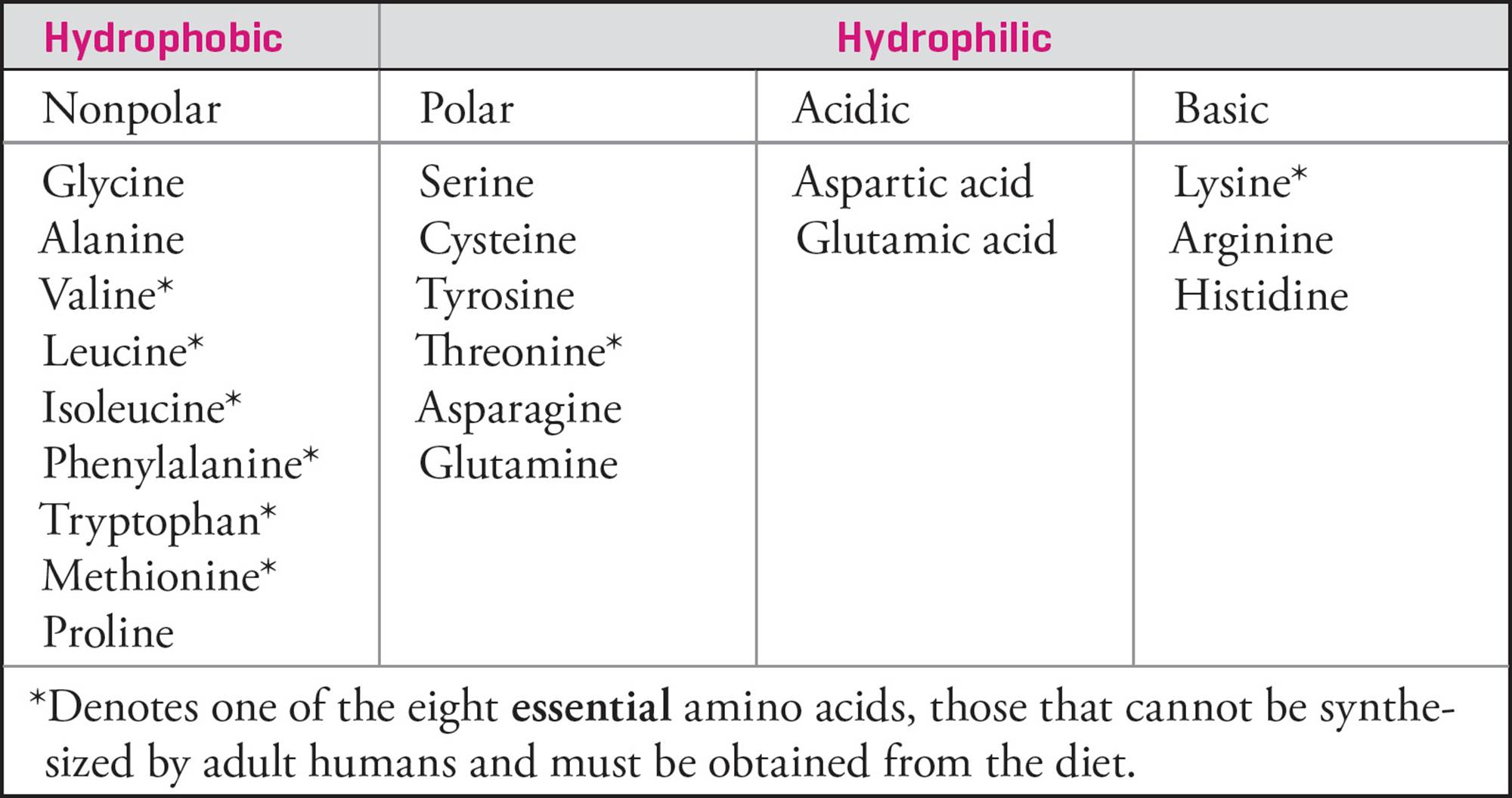
Table 1 Summary Table of Amino Acids
• Which of the following amino acids is most likely to be found on the exterior of a protein at pH 7.0?1
A) Leucine
B) Alanine
C) Serine
D) Isoleucine
3.2 PROTEIN STRUCTURE
There are two common types of covalent bonds between amino acids in proteins: the peptide bonds that link amino acids together into polypeptide chains and disulfide bridges between cysteine R-groups.
The Peptide Bond
Polypeptides are formed by linking amino acids together in peptide bonds. A peptide bond is formed between the carboxyl group of one amino acid and the α-amino group of another amino acid with the loss of water. The Figure below shows the formation of a dipeptide from the amino acids glycine and alanine.
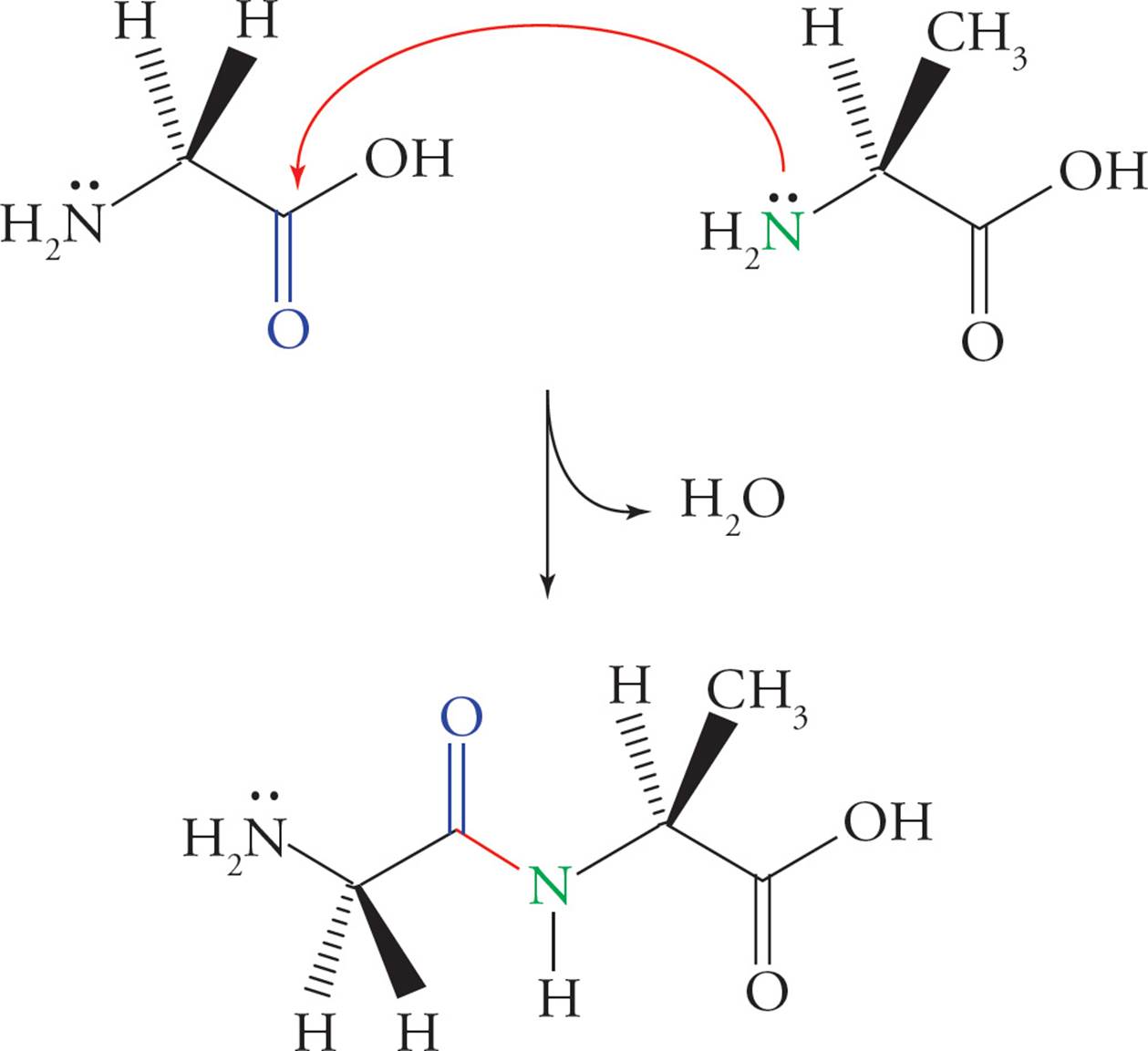
Figure 2 Peptide Bond (Amide Bond) Formation
In a polypeptide chain, the N–C–C–N–C–C pattern formed from the amino acids is known as the backbone of the polypeptide. An individual amino acid is termed a residue when it is part of a polypeptide chain. The amino terminus is the first end made during polypeptide synthesis, and the carboxy terminus is made last. Hence, by convention, the amino-terminal residue is also always written first.
• In the oligopeptide Phe-Glu-Gly-Ser-Ala, state the number of acid and base functional groups, which residue has a free α-amino group, and which residue has a free α-carboxyl group. (Refer to the beginning of the chapter for structures.)2
• Thermodynamics states that free energy must decrease for a reaction to proceed spontaneously and that such a reaction will spontaneously move toward equilibrium. The diagram below shows the free energy changes during peptide bond formation. At equilibrium, which is thermodynamically favored: the dipeptide or the individual amino acids?3
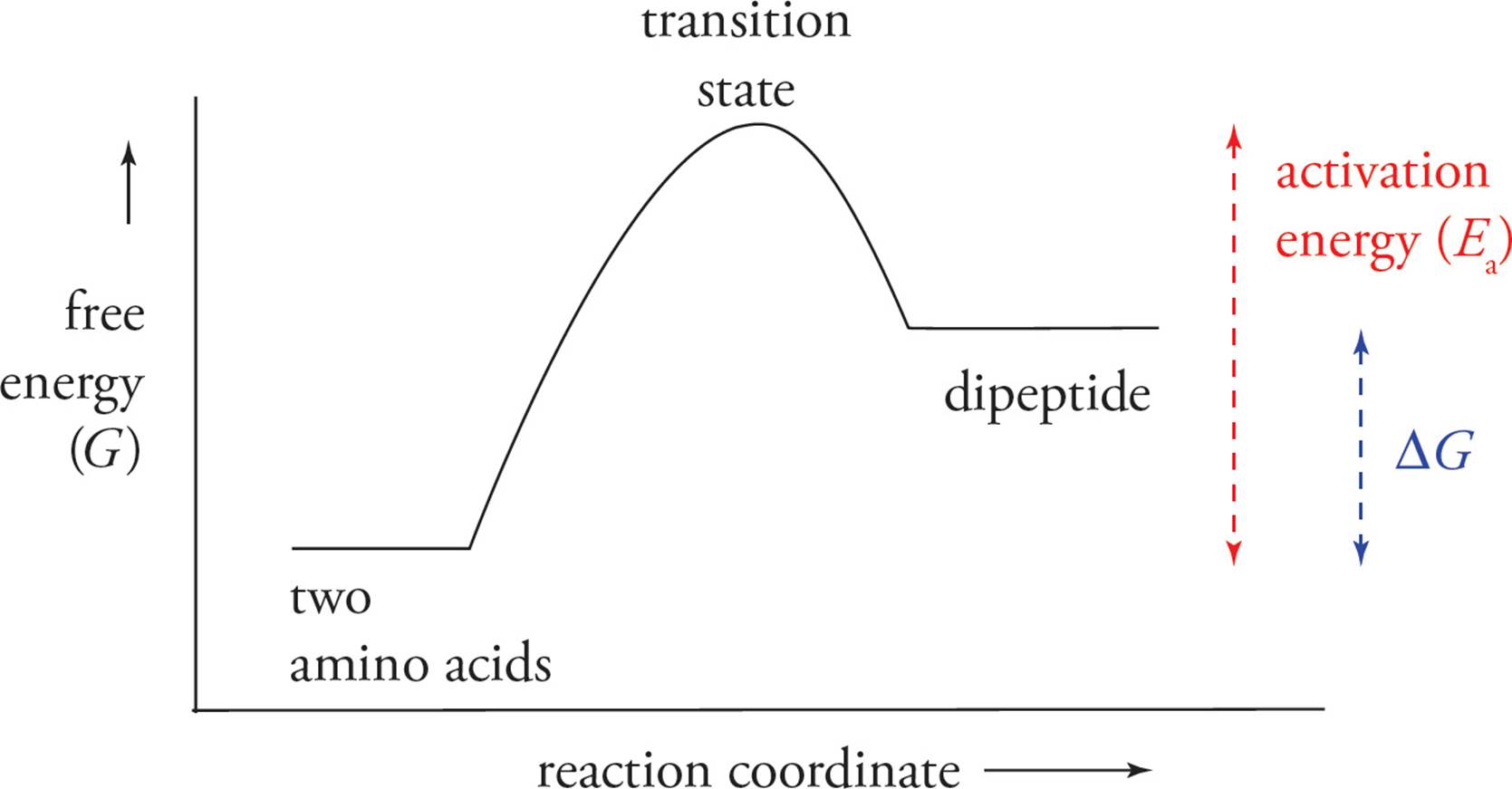
• In that case, how are peptide bonds formed and maintained inside cells?4
Hydrolysis of a protein by another protein is called proteolysis or proteolytic cleavage, and the protein that does the cutting is known as a proteolytic enzyme or protease. Proteolytic cleavage is a specific means of cleaving peptide bonds. Many enzymes only cleave the peptide bond adjacent to a specific amino acid. For example, the protease trypsin cleaves on the carboxyl side of the positively charged (basic) residues arginine and lysine, while chymotrypsin cleaves adjacent to hydrophobic residues such as phenylalanine. (Do not memorize these examples.)
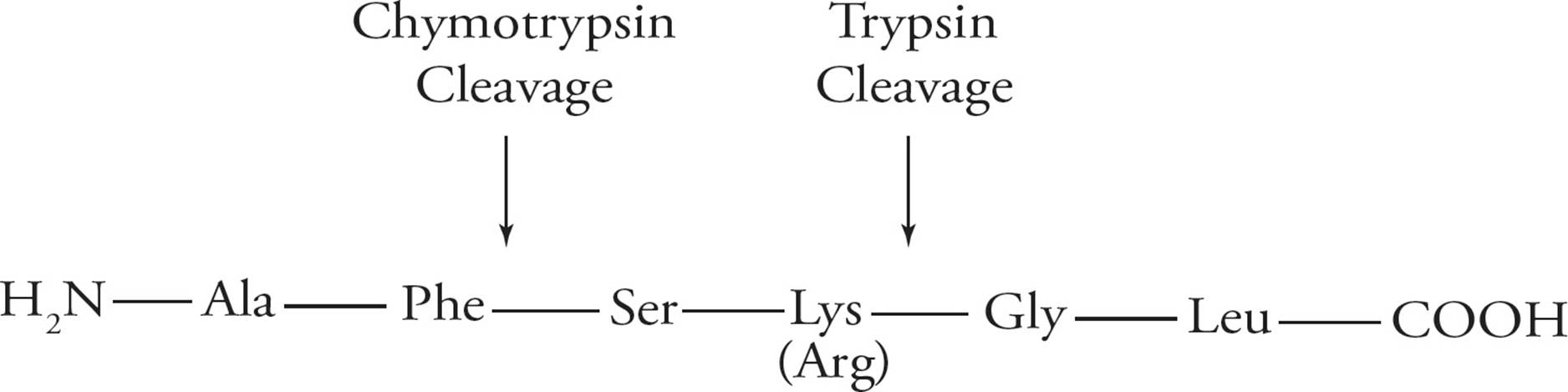
Figure 3 Specificity of Protease Cleavage
• Based on the above, if the following peptide is cleaved by trypsin, what amino acid will be on the new N-terminus and how many fragments will result: Ala-Gly-Glu-Lys-Phe-Phe-Lys?5
The Disulfide Bond
Cysteine is an amino acid with a reactive thiol (sulfhydryl, SH) in its side chain. The thiol of one cysteine can react with the thiol of another cysteine to produce a covalent sulfur-sulfur bond known as a disulfide bond, as illustrated below. The cysteines forming a disulfide bond may be located in the same or different polypeptide chain(s). The disulfide bridge plays an important role in stabilizing tertiary protein structure; this will be discussed in the section on protein folding. Once a cysteine residue becomes disulfide-bonded to another cysteine residue, it is called cystine instead of cysteine.
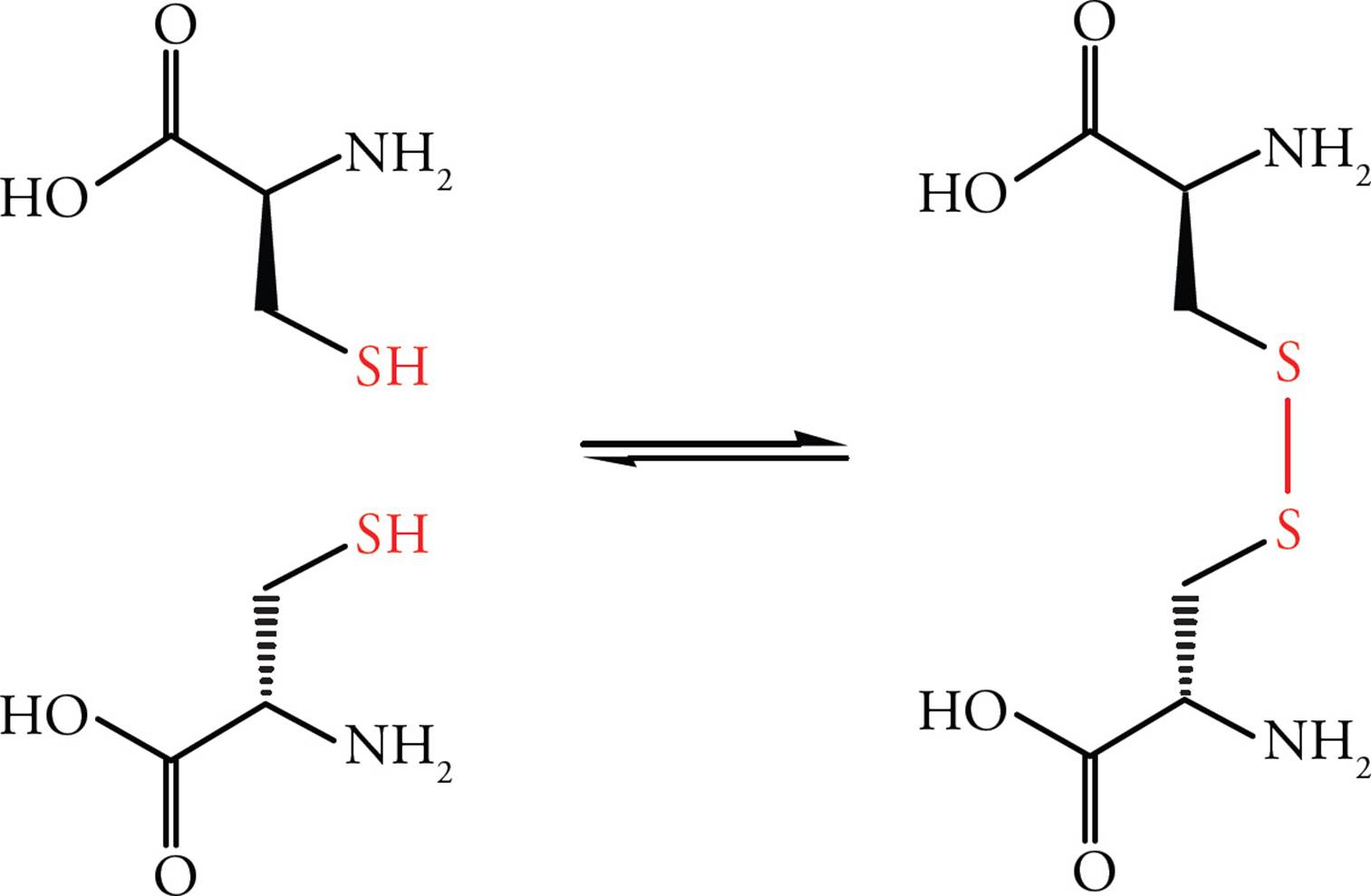
Figure 4 Formation of the Disulfide Bond
• Which is more oxidized, the sulfur in cysteine or the sulfur in cystine?6
• The inside of cells is known as a reducing environment because cells possess antioxidants (chemicals that prevent oxidation reactions). Where would disulfide bridges be more likely to be found, in extracellular proteins, under oxidizing conditions, or in the interior of cells, in a reducing environment?7
Protein Structure in Three Dimensions
Each protein folds into a unique three-dimensional structure that is required for that protein to function properly. Improperly folded, or denatured, proteins are non-functional. There are four levels of protein folding that contribute to their final three-dimensional structure. Each level of structure is dependent upon a particular type of bond, as discussed in the following sections.
Denaturation is an important concept. It refers to the disruption of a protein’s shape without breaking peptide bonds. Proteins are denatured by urea (which disrupts hydrogen bonding interactions), by extremes of pH, by extremes of temperature, and by changes in salt concentration (tonicity).
Primary (1o) Structure: The Amino Acid Sequence
The simplest level of protein structure is the order of amino acids bonded to each other in the polypeptide chain. This linear ordering of amino acid residues is known as primary structure. Primary structure is the same as sequence. The bond which determines 1ο structure is the peptide bond, simply because this is the bond that links one amino acid to the next in a polypeptide.
Secondary (2o) Structure: Hydrogen Bonds Between Backbone Groups
Secondary structure refers to the initial folding of a polypeptide chain into shapes stabilized by hydrogen bonds between backbone NH and CO groups. Certain motifs of secondary structure are found in most proteins. The two most common are the α-helix and the β-pleated sheet.
All α-helices have the same well-defined dimensions that are depicted below with the R-groups omitted for clarity. The α-helices of proteins are always right handed, 5 angstroms in width, with each subsequent amino acid rising 1.5 angstroms. There are 3.6 amino acid residues per turn with the α-carboxyl oxygen of one amino acid residue hydrogen-bonded to the α-amino proton of an amino acid three residues away. (Don’t memorize these numbers, but do try to visualize what they mean.)
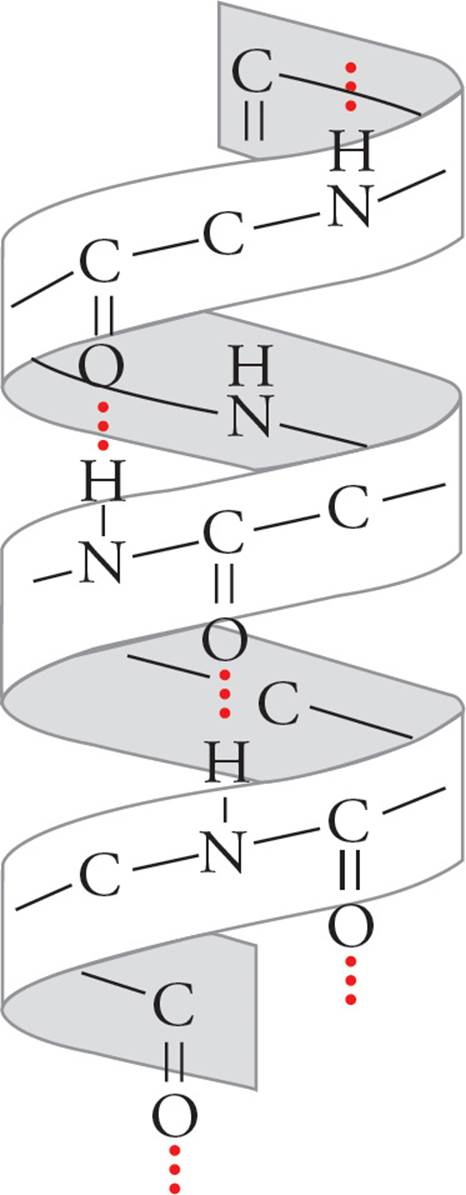
Figure 5 An α Helix
The unique structure of proline forces it to kink the polypeptide chain; hence proline residues never appear within the α-helix.
Proteins such as hormone receptors and ion channels are often found with α-helical transmembrane regions integrated into the hydrophobic membranes of cells. The α-helix is a favorable structure for a hydrophobic transmembrane region because all polar NH and CO groups in the backbone are hydrogen bonded to each other on the inside of the helix, and thus don’t interact with the hydrophobic membrane interior. α-Helical regions that span membranes also have hydrophobic R-groups, which radiate out from the helix, interacting with the hydrophobic interior of the membrane.
β-Pleated sheets are also stabilized by hydrogen bonding between NH and CO groups in the polypeptide backbone. In β-sheets, however, hydrogen bonding occurs between residues distant from each other in the chain or even on separate polypeptide chains. Also, the backbone of a β-sheet is extended, rather than coiled, with side groups directed above and below the plane of the β-sheet. There are two types of β-sheets, one with adjacent polypeptide strands running in the same direction (parallel β-pleated sheet) and another in which the polypeptide strands run in opposite directions (antiparallel β-pleated sheet).
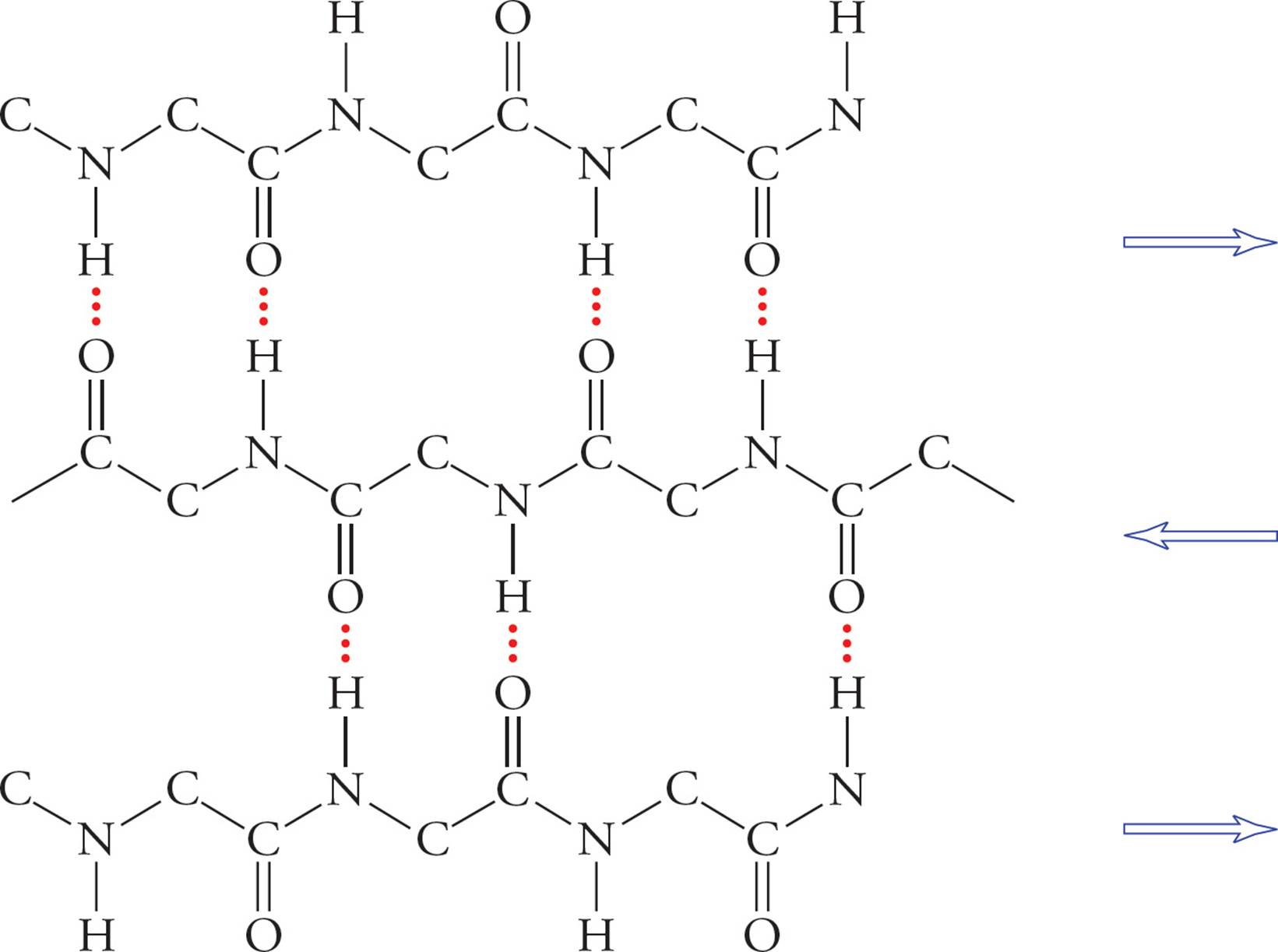
Figure 6 A β-Pleated Sheet
• If a single polypeptide folds once and forms a β-pleated sheet with itself, would this be a parallel or antiparallel β-pleated sheet?8
• What effect would a molecule that disrupts hydrogen bonding, e.g., urea, have on protein structure?9
Tertiary (3o) Structure: Hydrophobic/Hydrophilic Interactions
The next level of protein folding, tertiary structure, concerns interactions between amino acid residues located more distantly from each other in the polypeptide chain. The folding of secondary structures such as α-helices into higher order tertiary structures is driven by interactions of R-groups with each other and with the solvent (water). Hydrophobic R-groups tend to fold into the interior of the protein, away from the solvent, and hydrophilic R-groups tend to be exposed to water on the surface of the protein (shown for the generic globular protein).
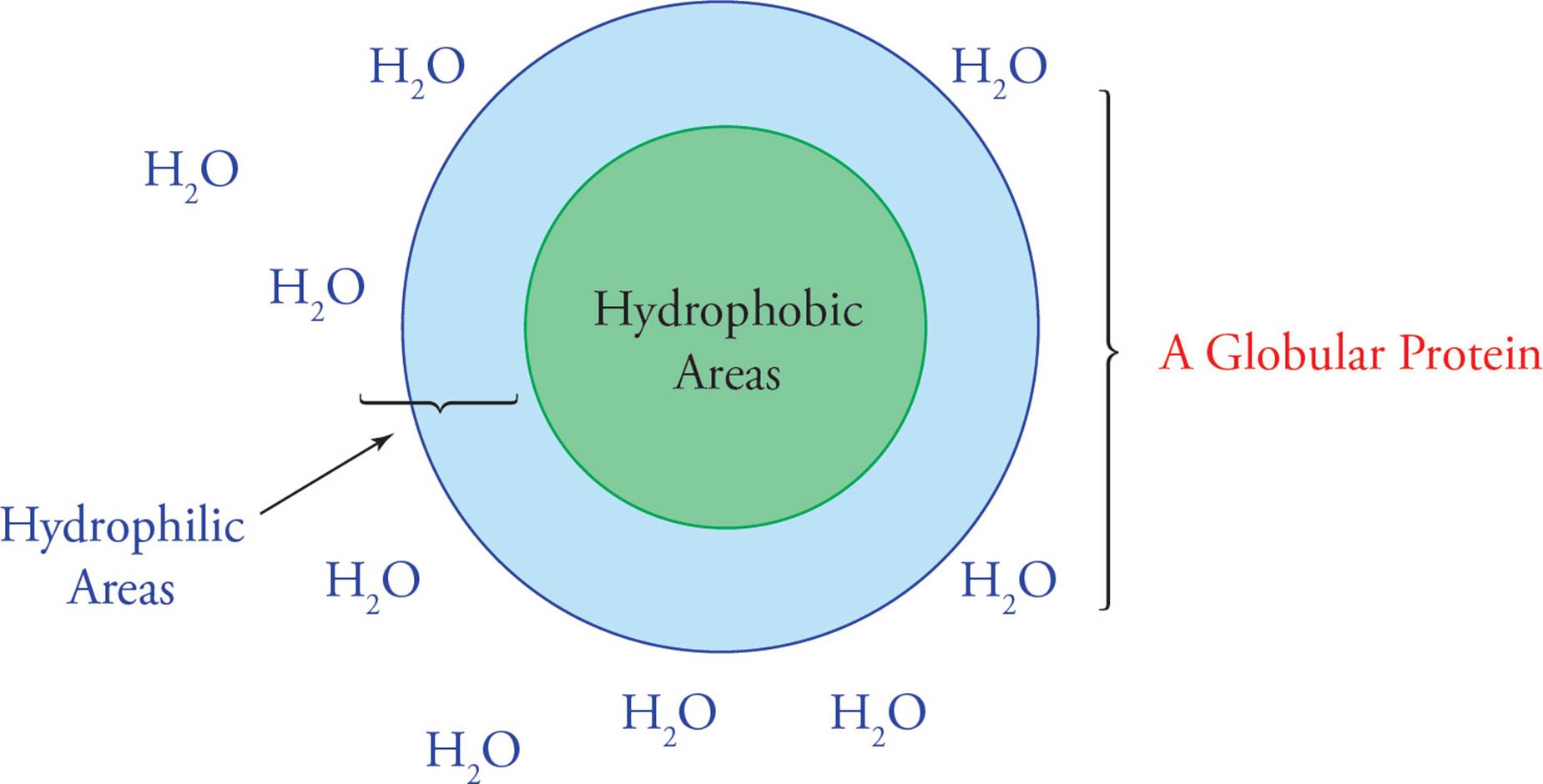
Figure 7 Folding of a Globular Protein in Aqueous Solution
Under the right conditions, the forces driving hydrophobic avoidance of water and hydrogen bonding will fold a polypeptide spontaneously into the correct conformation, the lowest energy conformation. In a classic experiment by Christian Anfinsen and coworkers, the effect of a denaturing agent (urea) and a reducing agent (β-mercaptoethanol) on the folding of a protein called ribonuclease were examined. In the following questions, you will reenact their thought processes. Figure out the answers before reading the footnotes.
• Ribonuclease has eight cysteines that form four disulfides bonds. What effect would a reducing agent have on its tertiary structure?10
• If the disulfides serve only to lock into place a tertiary protein structure that forms first on its own, then what effect would the reducing agent have on correct protein folding?11
• Would a protein end up folded normally if you (1) first put it in a reducing environment, (2) then denatured it by adding urea, (3) next removed the reducing agent, allowing disulfide bridges to reform, and (4) finally removed the denaturing agent?12
• What if you did the same experiment but in this order: 1, 2, 4, 3?13
The disulfide bridge is not a great example of 3° structure because it is a covalent bond, not a hydrophobic interaction. However, because the disulfide is formed after 2° structure and before 4° structure, it is usually considered part of 3° folding.
• Which of the following may be considered an example of tertiary protein structure?14
I. van der Waals interactions between two Phe R-groups located far apart on a polypeptide
II. Hydrogen bonds between backbone amino and carboxyl groups
III. Covalent disulfide bonds between cysteine residues located far apart on a polypeptide
Quaternary (4o) Structure: Various Bonds Between Separate Chains
The highest level of protein structure, quaternary structure, describes interactions between polypeptide subunits. A subunit is a single polypeptide chain that is part of a large complex containing many subunits (a multisubunitcomplex). The arrangement of subunits in a multisubunit complex is what we mean by quaternary structure. For example, mammalian RNA polymerase II contains twelve different subunits. The interactions between subunits are instrumental in protein function, as in the cooperative binding of oxygen by each of the four subunits of hemoglobin.
The forces stabilizing quaternary structure are generally the same as those involved in secondary and tertiary structure—non-covalent interactions (the hydrogen bond, and the van der Waals interaction). However, covalent bonds may also be involved in quaternary structure. For example, antibodies (immune system molecules) are large protein complexes with disulfide bonds holding the subunits together. It is key to understand, however, that there is one covalent bond that may not be involved in quaternary structure—the peptide bond—because this bond defines sequence (1° structure).
• What is the difference between a disulfide bridge involved in quaternary structure and one involved in tertiary structure?15
3.3 CARBOHYDRATES
Carbohydrates can be broken down to CO2 in a process called oxidation, which is also known as burning or combustion. Because this process releases large amounts of energy, carbohydrates generally serve as the principle energy source for cellular metabolism. Glucose in the form of the polymer cellulose is also the building block of wood and cotton. Understanding the nomenclature, structure, and chemistry of carbohydrates is essential to understanding cellular metabolism. This section will also help you understand key facts such as why we can eat potatoes and cotton candy but not wood and cotton T-shirts, and why milk makes some adults flatulent.
Monosaccharides and Disaccharides
A single carbohydrate molecule is called a monosaccharide (meaning “single sweet unit”), also known as a simple sugar. Monosaccharides have the general chemical formula CnH2nOn.
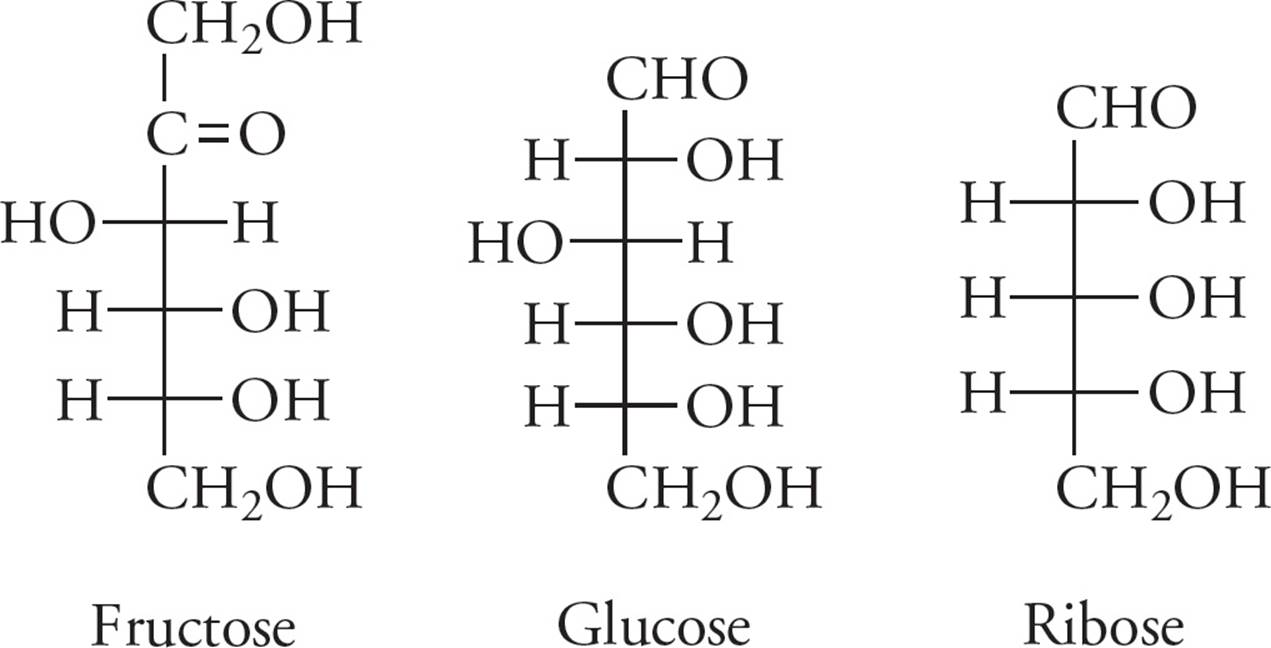
Figure 8 Some Metabolically Important Monosaccharides
Two monosaccharides bonded together form a disaccharide, a few form an oligosaccharide, and many form a polysaccharide. The bond between two sugar molecules is called a glycosidic linkage. This is a covalent bond, formed in a dehydration reaction that requires enzymatic catalysis.
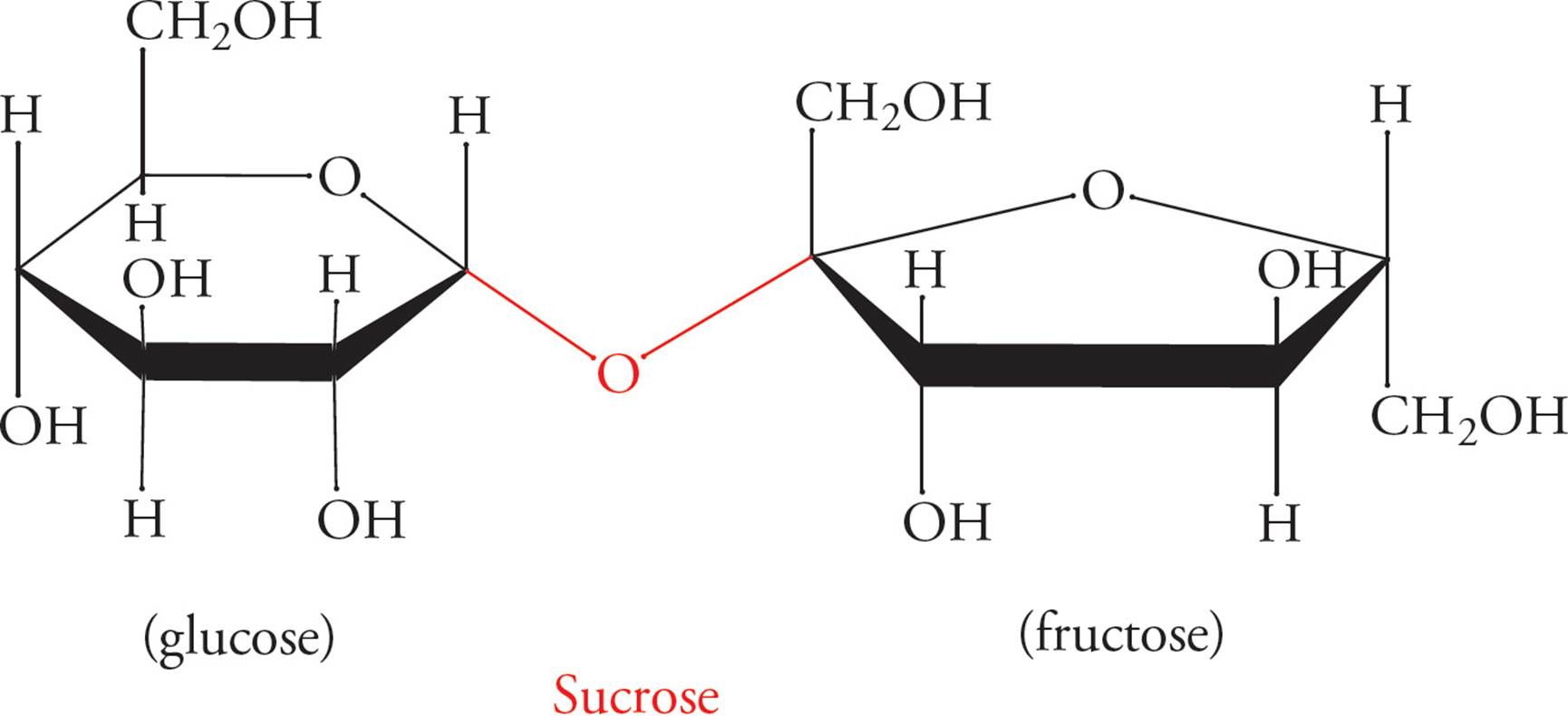
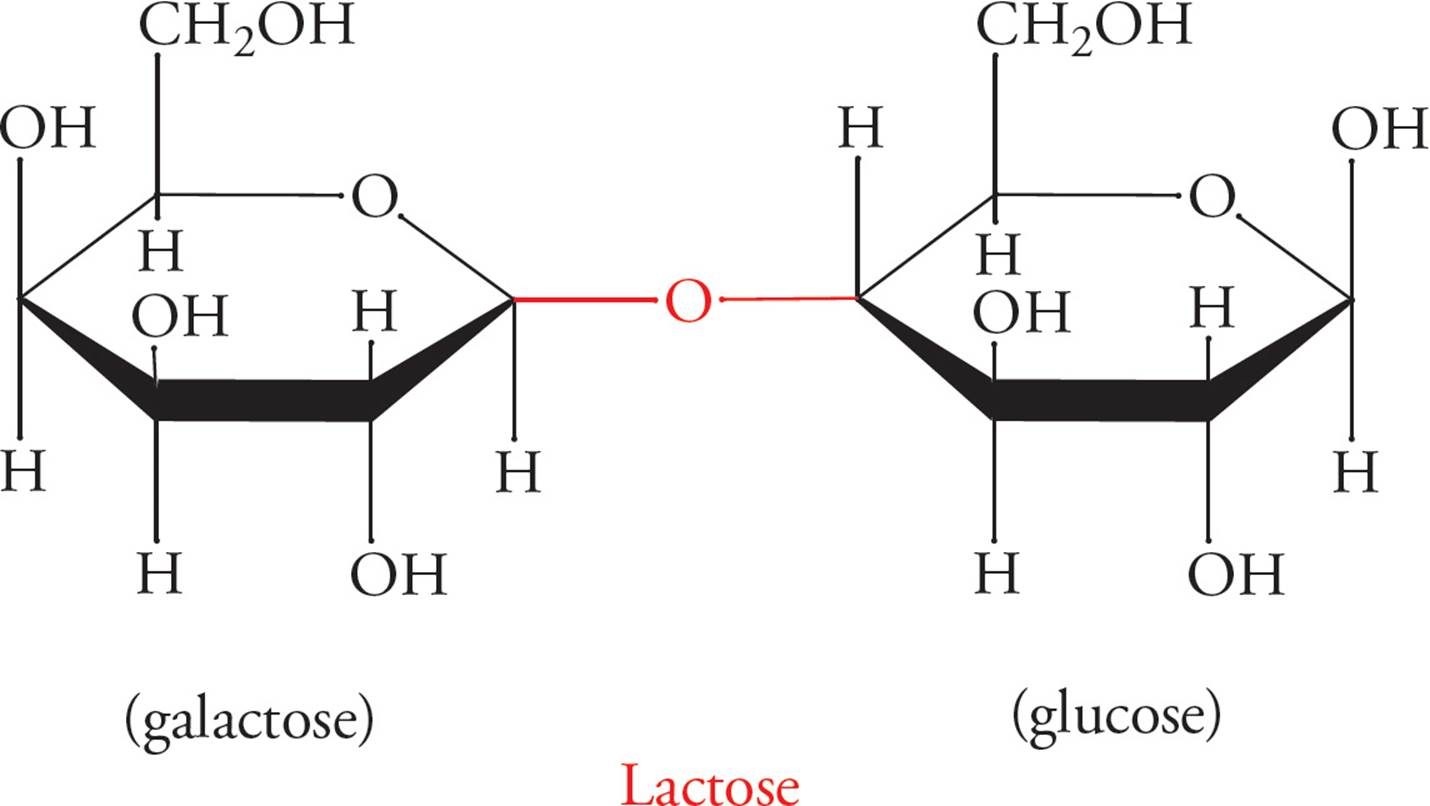
Figure 9 Disaccharides and the α- or β-Glycosidic Bond
Glycosidic linkages are named according to which carbon in each sugar comprises the linkage. The configuration (α or β) of the linkage is also specified. For example, lactose (milk sugar) is a disaccharide joined in a galactose-β-1,4-glucose linkage (above). Sucrose (Table sugar) is also shown above, with a glucose unit and a fructose unit.
• Does sucrose contain an α- or β-glycosidic linkage? 16
Some common disaccharides you might see on the MCAT are sucrose (Glc-α-1,2-Fru), lactose (Gal-β-1,4-Glc), maltose (Glc-α-1,4-Glc), and cellobiose (Glc-β-1,4-Glc). However, you should NOT try to memorize these linkages.
Polymers made from these disaccharides form important biological macromolecules. Glycogen serves as an energy storage carbohydrate in animals and is composed of thousands of glucose units joined in α‑1,4 linkages; α‑1,6 branches are also present. Starch is the same as glycogen (except that the branches are a little different), and serves the same purpose in plants. Cellulose is a polymer of cellobiose; but note that cellobiose does not exist freely in nature. It exists only in its polymerized, cellulose form. The β-glycosidic bonds allow the polymer to assume a long, straight, fibrous shape. Wood and cotton are made of cellulose.
Hydrolysis of Glycosidic Linkages
The hydrolysis of polysaccharides into monosaccharides is favored thermodynamically. Hydrolysis is essential in order for these sugars to enter metabolic pathways (e.g., glycolysis) and be used for energy by the cell. However, this hydrolysis does not occur at a significant rate without enzymatic catalysis. Different enzymes catalyze the hydrolysis of different linkages. The enzymes are named for the sugar they hydrolyze. For example, the enzyme that catalyzes the hydrolysis of maltose into two glucose monosaccharides is called maltase. Each enzyme is highly specific for its linkage.
This specificity is a great example of the significance of stereochemistry. Consider cellulose. A cotton T-shirt is pure sugar. The only reason we can’t digest it is that mammalian enzymes generally can’t break the β-glycosidic linkages found in cellulose. Cellulose is actually the energy source in grass and hay. Cows are mammals, and all mammals lack the enzymes necessary for cellulose breakdown. To live on grass, cows depend on bacteria that live in an extra stomach called a rumen to digest cellulose for them. If you’re really on the ball, you’re next question is: Humans are mammals, so how can we digest lactose, which has a β linkage? The answer is that we have a specific enzyme, lactase, which can digest lactose. This is an exception to the rule that mammalian enzymes cannot hydrolyze β-glycosidic linkages. People without lactase are lactose malabsorbers, and any lactose they eat ends up in the colon. There it may cause gas and diarrhea, if certain bacteria are present; people with this problem are said to be lactose intolerant. People produce lactase as children so that they can digest mother’s milk, but most adults naturally stop making this enzyme, and thus become lactose malabsorbers and sometimes intolerant.
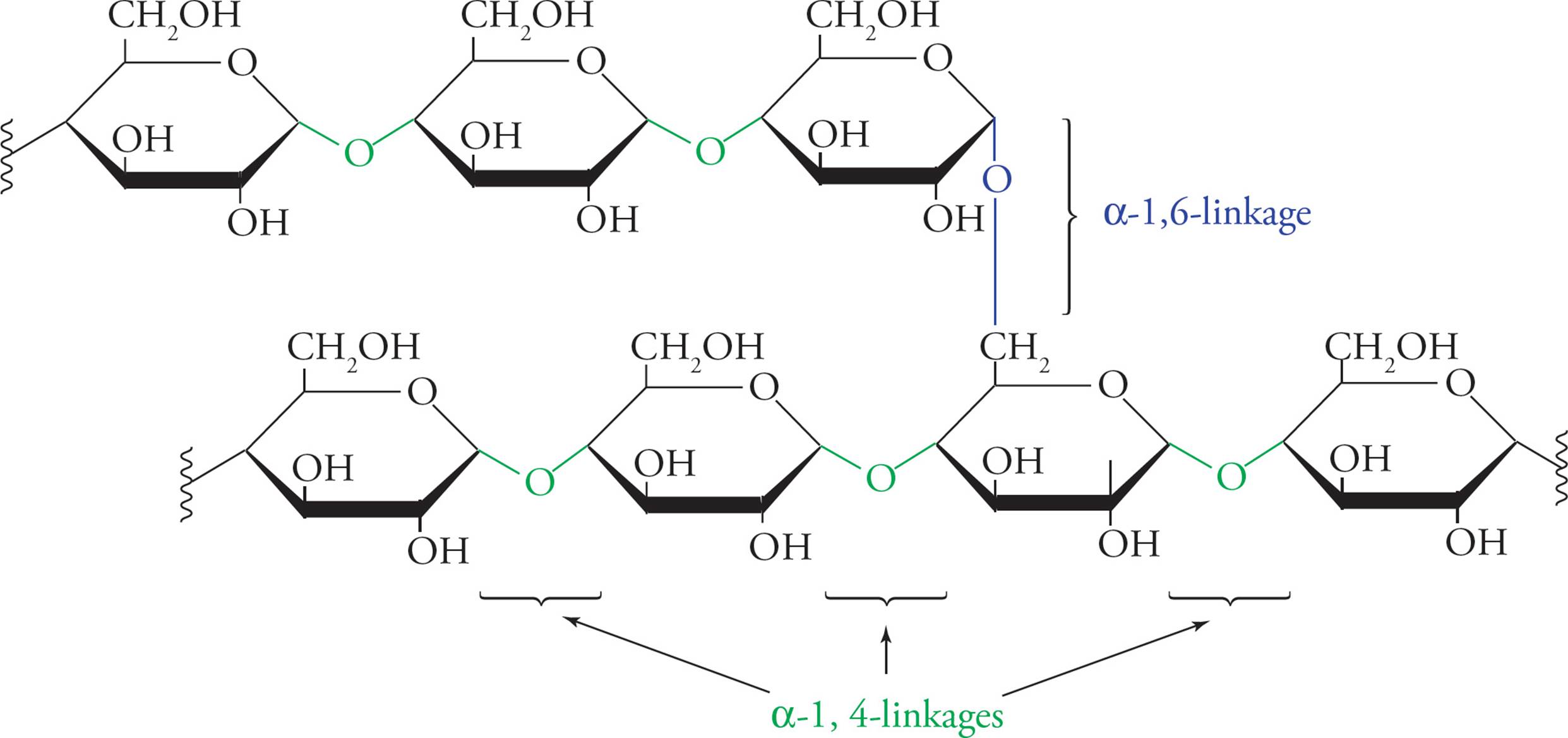
Figure 10 The Polysaccharide Glycogen
• Which requires net energy input: polysaccharide synthesis or hydrolysis? 17
• If the activation energy of polysaccharide hydrolysis were so low that no enzyme was required for the reaction to occur, would this make polysaccharides better for energy storage? 18
3.4 LIPIDS
Lipids are oily or fatty substances that play three physiological roles, summarized here and discussed below.
1) In adipose cells, triglycerides (fats) store energy.
2) In cellular membranes, phospholipids constitute a barrier between intracellular and extracellular environments.
3) Cholesterol is a special lipid that serves as the building block for the hydrophobic steroid hormones.
The cardinal characteristic of the lipid is its hydrophobicity. Hydrophobic means water-fearing. It is important to understand the significance of this. Since water is very polar, polar substances dissolve well in water; these are known as water-loving, or hydrophilic substances. Carbon-carbon bonds and carbon-hydrogen bonds are nonpolar. Hence, substances that contain only carbon and hydrogen will not dissolve well in water. Some examples: Table sugar dissolves well in water, but cooking oil floats in a layer above water or forms many tiny oil droplets when mixed with water. Cotton T-shirts become wet when exposed to water because they are made of glucose polymerized into cellulose, but a nylon jacket does not become wet because it is composed of atoms covalently bound together in a nonpolar fashion. A synonym for hydrophobic is lipophilic (which means lipid-loving); a synonym for hydrophilic is lipophobic. We return to these concepts below.
Fatty Acid Structure
Fatty acids are composed of long unsubstituted alkanes that end in a carboxylic acid. The chain is typically 14 to 18 carbons long, and because they are synthesized two carbons at a time from acetate, only even-numbered fatty acids are made in human cells. A fatty acid with no carbon-carbon double bonds is said to be saturated with hydrogen because every carbon atom in the chain is covalently bound to the maximum number of hydrogens. Unsaturated fatty acids have one or more double bonds in the tail. These double bonds are almost always (Z) (or cis).
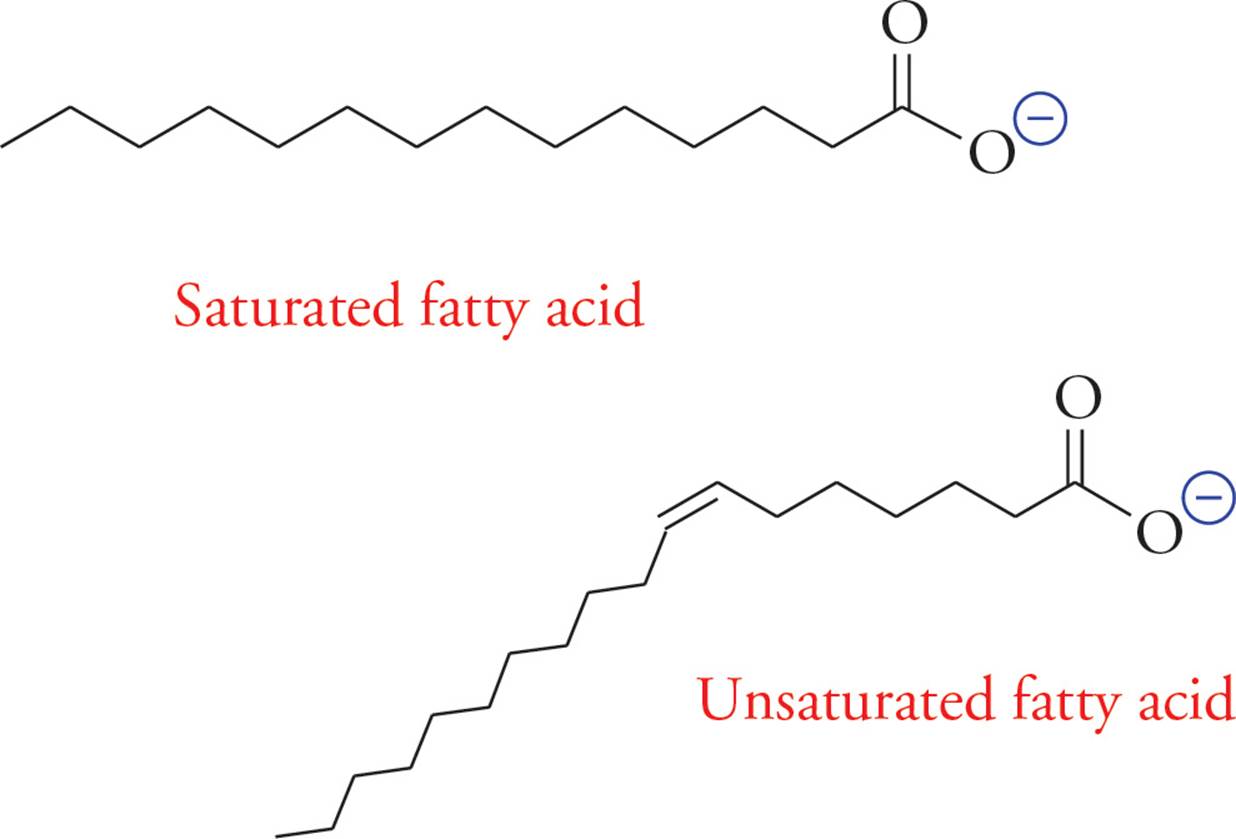
• How does the shape of an unsaturated fatty acid differ from that of a saturated fatty acid?19
• If fatty acids are mixed into water, how are they likely to associate with each other?20
The drawing on the next page illustrates how free fatty acids interact in an aqueous solution; they form a structure called a micelle. The force that drives the tails into the center of the micelle is called the hydrophobic interaction. The hydrophobic interaction is a complex phenomenon. In general, it results from the fact that water molecules must form an orderly solvation shell around each hydrophobic substance. The reason is that H2O has a dipole that “likes” to be able to share its charges with other polar molecules. A solvation shell allows for the most water-water interaction and the least water-lipid interaction. The problem is that forming a solvation shell is an increase in order and thus a decrease in entropy (∆S < 0), which is unfavorable according to the second law of thermodynamics. In the case of the fatty acid micelle, water forms a shell around the spherical micelle with the result being that water interacts with polar carboxylic acid head groups while hydrophobic lipid tails hide inside the sphere.
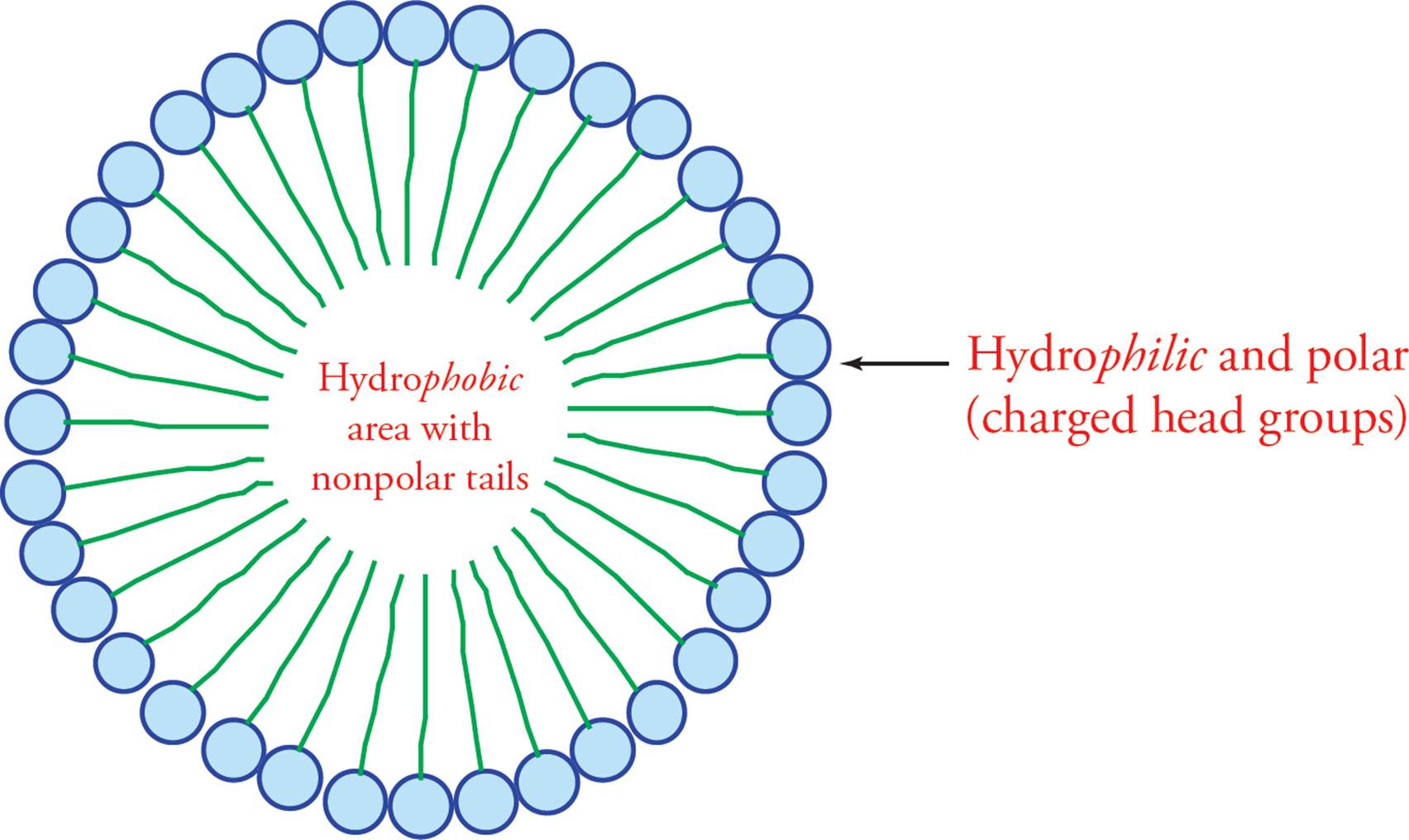
Figure 11 A Fatty Acid Micelle
• How does soap help to remove grease from your hands?21
Triacylglycerols (TG)
The storage form of the fatty acid is fat. The technical name for fat is triacylglycerol or triglyceride (shown below). The triglyceride is composed of three fatty acids esterified to a glycerol molecule. Glycerol is a three-carbon triol with the formula HOCH2–CHOH–CH2OH. As you can see, it has three hydroxyl groups that can be esterified to fatty acids. It is necessary to store fatty acids in the relatively inert form of fat because free fatty acids are reactive chemicals.
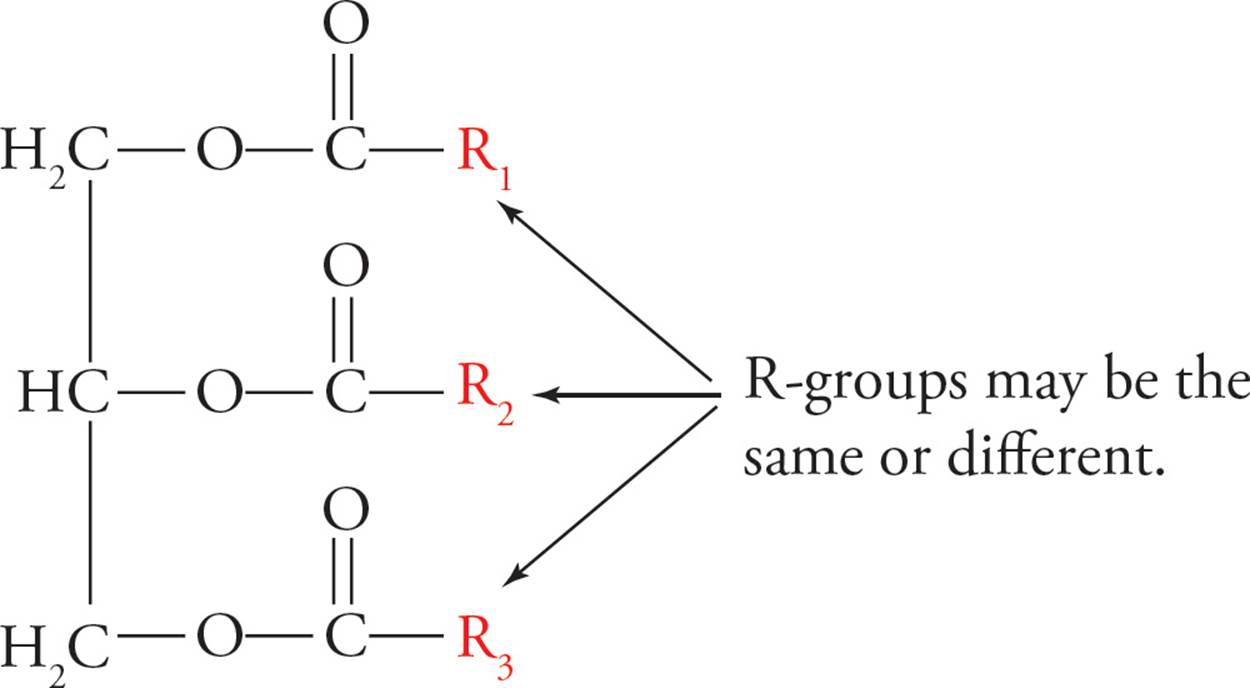
Figure 12 A Triglyceride (Fat)
The triacylglycerol undergoes reactions typical of esters, such as base-catalyzed hydrolysis. Soaps are the sodium salts of fatty acids (RCOO-Na+). They are amphipathic, which means they have both hydrophilic and hydrophobic regions. Soap is economically produced by base-catalyzed hydrolysis of triglycerides from animal fat into fatty acid salts (soaps). This reaction is called saponification and is illustrated below.
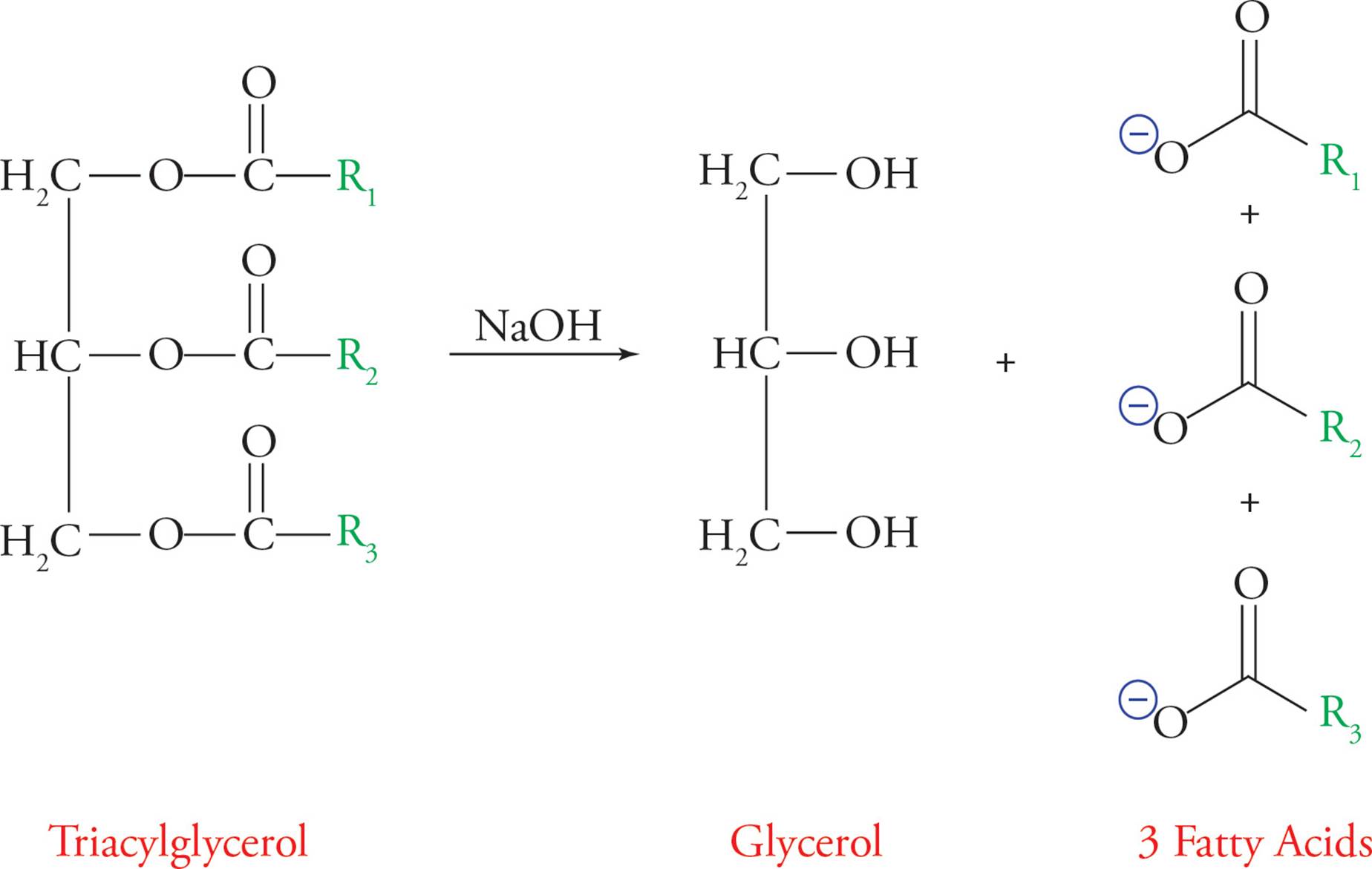
Figure 13 Saponification
Lipases are enzymes that hydrolyze fats. Triacylglycerols are stored in fat cells as an energy source. Fats are more efficient energy storage molecules than carbohydrates for two reasons: packing and energy content.
1) Packing: Their hydrophobicity allows fats to pack together much more closely than carbohydrates. Carbohydrates carry a great amount of water-of-solvation (water molecules hydrogen bonded to their hydroxyl groups). In other words, the amount of carbon per unit area or unit weight is much greater in a fat droplet than in dissolved sugar. If we could store sugars in a dry powdery form in our bodies, this problem would be obviated.
2) Energy content: All packing considerations aside, fat molecules store much more energy than carbohydrates. In other words, regardless of what you dissolve it in, a fat has more energy carbon-for-carbon than a carbohydrate. The reason is that fats are much more reduced. Remember that energy metabolism begins with the oxidation of foodstuffs to release energy. Since carbohydrates are more oxidized to start with, oxidizing them releases less energy. Animals use fat to store most of their energy, storing only a small amount as carbohydrates (glycogen). Plants such as potatoes commonly store a large percentage of their energy as carbohydrates (starch).
Introduction to Lipid Bilayer Membranes
Membrane lipids are phospholipids derived from diacylglycerol phosphate or DG-P. For example, phosphatidyl choline is a phospholipid formed by the esterification of a choline molecule [HO(CH2)2N+(CH3)3] to the phosphate group of DG-P. Phospholipids are detergents, substances that efficiently solubilize oils while remaining highly water-soluble. Detergents are like soaps, but stronger.
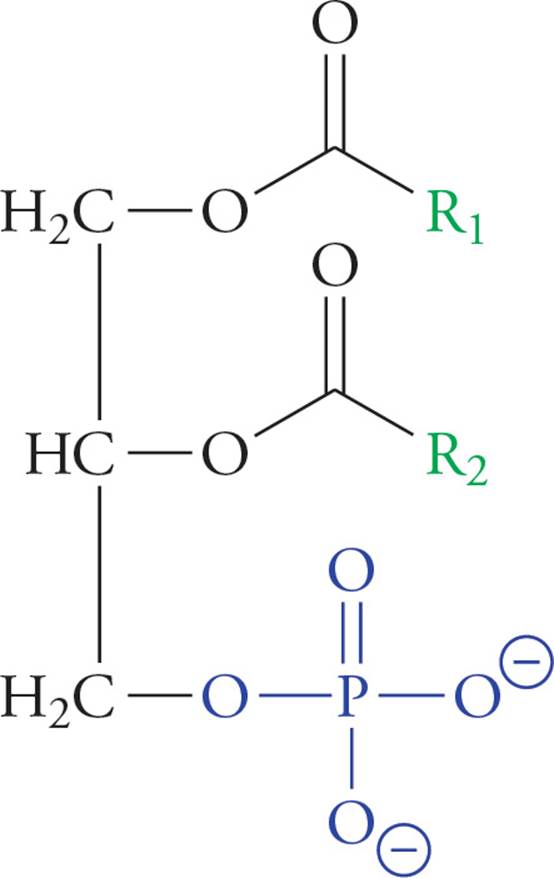
Figure 14 A Phosphoglyceride (Diacylglycerol Phosphate, or DGP)
We saw above how fatty acids spontaneously form micelles. Phospholipids also minimize their interactions with water by forming an orderly structure—in this case, it is a lipid bilayer (below). Hydrophobic interactions drive the formation of the bilayer, and once formed, it is stabilized by van der Waals forces between the long tails.
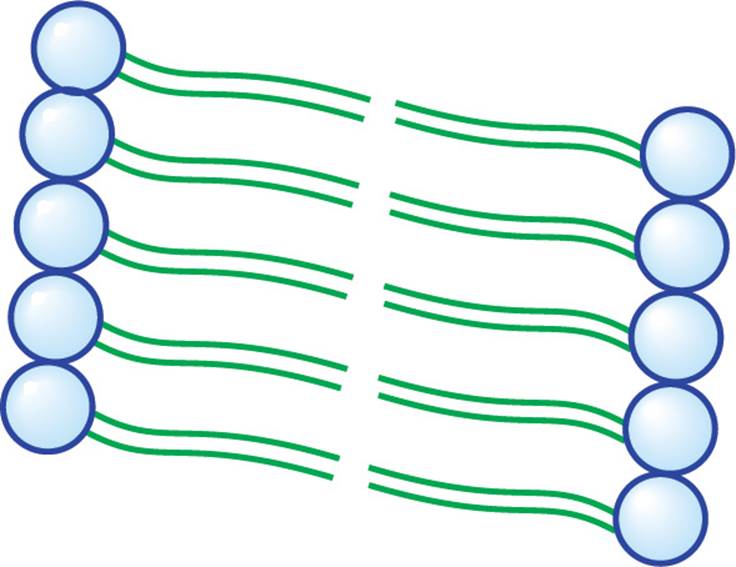
Figure 15 A Small Section of a Lipid Bilayer Membrane
• Would a saturated or an unsaturated fatty acid residue have more van der Waals interactions with neighboring alkyl chains in a bilayer membrane?22
A more precise way to give the answer to the question above is to say that double bonds (unsaturation) in phospholipid fatty acids tend to increase membrane fluidity. Unsaturation prevents the membrane from solidifying by disrupting the orderly packing of the hydrophobic lipid tails. The right amount of fluidity is essential for function. Decreasing the length of fatty acid tails also increases fluidity. The steroid cholesterol (discussed a bit later) is a third important modulator of membrane fluidity. At low temperatures, it increases fluidity in the same way as kinks in fatty acid tails; hence, it is known as membrane antifreeze. At high temperatures, however, cholesterol attenuates (reduces) membrane fluidity. Don’t ponder this paradox too long; just remember that cholesterol keeps fluidity at an optimum level. Remember, the structural determinants of membrane fluidity are: degree of saturation, tail length, and amount of cholesterol.
The lipid bilayer acts like a plastic bag surrounding the cell in the sense that it separates the interior of the cell from the exterior. However, the cell membrane is much more complex than a plastic bag. Since the plasma bilayer membrane surrounding cells is impermeable to charged particles such as Na+, protein gateways such as ion channels are required for ions to enter or exit cells. Proteins that are integrated into membranes also transmit signals from the outside of the cell into the interior. For example, certain hormones (peptides) cannot pass through the cell membrane due to their charged nature; instead, protein receptors in the cell membrane bind these hormones and transmit a signal into the cell in a second messenger cascade (see Chapter 7 for more details on the plasa membrane).
Terpenes
A terpene is a member of a broad class of compounds built from isoprene units (C5H8) with a general formula (C5H8)n.
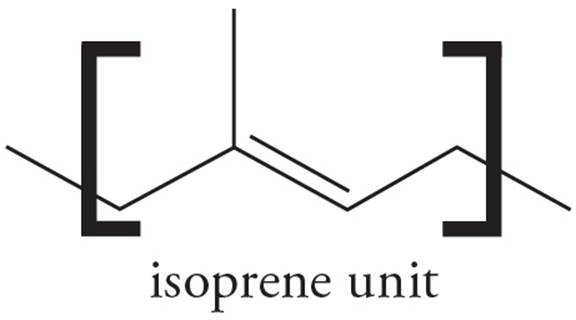
Terpenes may be linear or cyclic, and are classified by the number of isoprene units they contain. For example, monoterpenes consist of two isoprene units, sesquiterpenes consist of three, and diterpenes contain four.
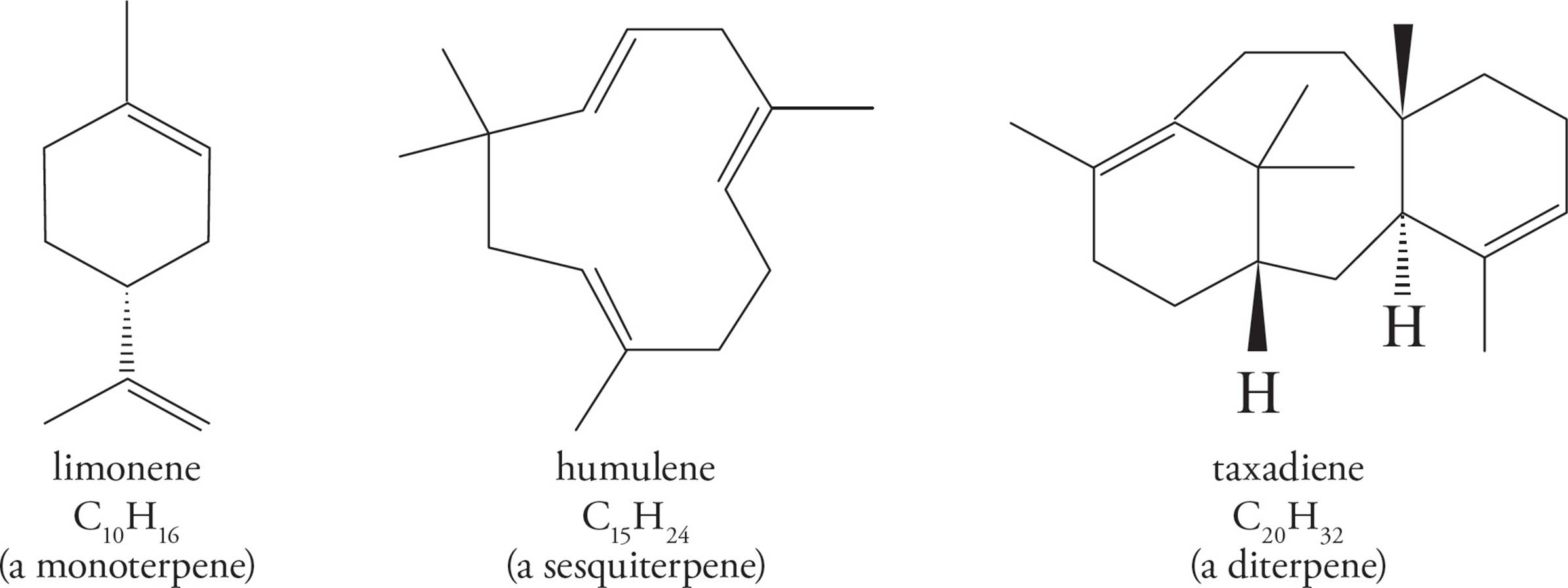
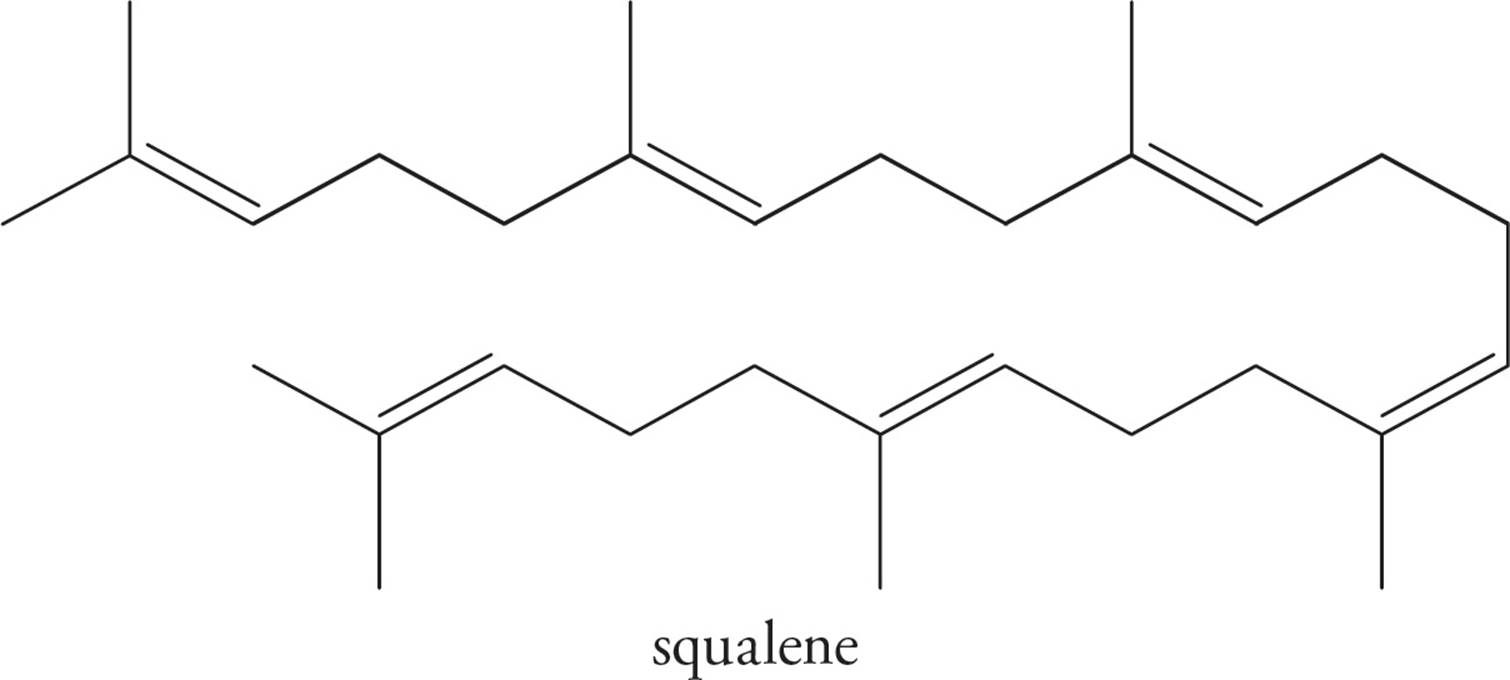
Squalene is a triterpene (made of six isoprene units), and is a particularly important compound as it is biosynthetically utilized in the manufacture of steroids. Squalene is also a component of earwax.
Whereas a terpene is formally a simple hydrocarbon, there are a number of natural and synthetically derived species that are built from an isoprene skeleton and functionalized with other elements (O, N, S, etc.). These functionalized-terpenes are known as terpenoids. Vitamin A (C20H30O) is an example of a terpenoid.
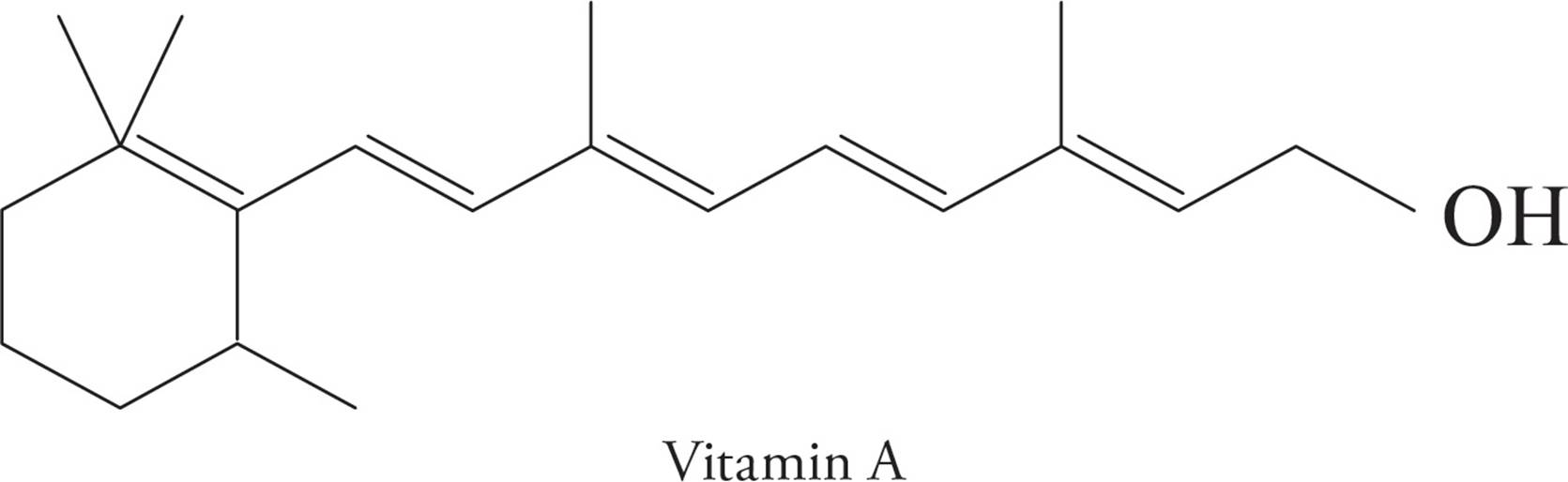
Steroids
Steroids are included here because of their hydrophobicity, and, hence, similarity to fats. Their structure is otherwise unique. All steroids have the basic tetracyclic ring system (see below), based on the structure of cholesterol, a polycyclic amphipath. (Polycyclic means several rings, and amphipathic means displaying both hydrophilic and hydrophobic characteristics.)
As discussed above, the steroid cholesterol is an important component of the lipid bilayer. It is both obtained from the diet and synthesized in the liver. It is carried in the blood packaged with fats and proteins into lipoproteins. One type of lipoprotein has been implicated as the cause of atherosclerotic vascular disease, which refers to the build-up of cholesterol “plaques” on the inside of blood vessels.
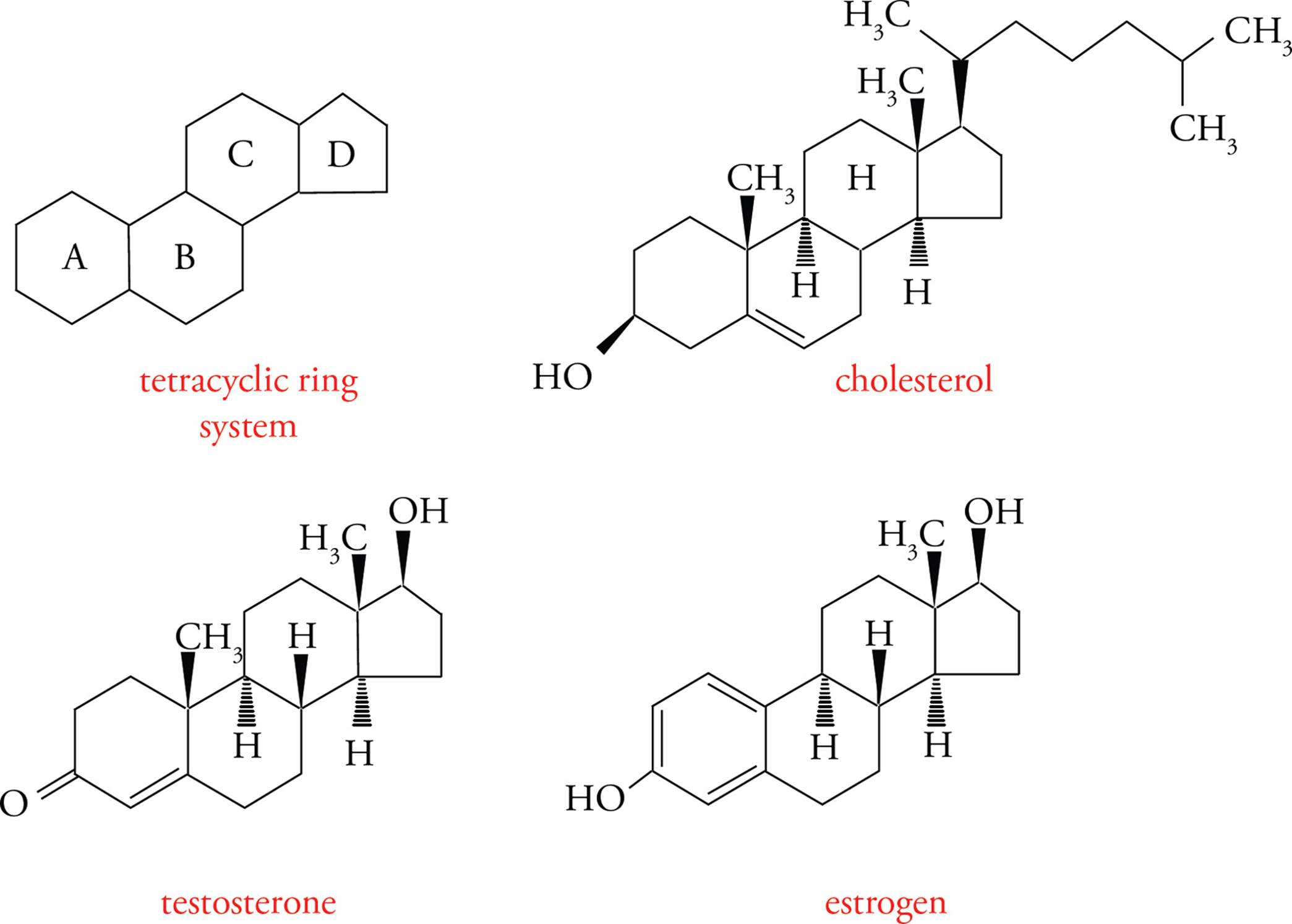
Figure 16 Cholesterol-Derived Hormones
Steroid hormones are made from cholesterol. Two examples are testosterone (an androgen or male sex hormone) and estradiol (an estrogen or female sex hormone). There are no receptors for steroid hormones on the surface of cells; because steroids are highly hydrophobic, they can diffuse right through the lipid bilayer membrane into the cytoplasm. The receptors for steroid hormones are located within cells rather than on the cell surface. This is an important point! You must be aware of the contrast between peptide hormones, such as insulin, which exert their effects by binding to receptors at the cell-surface, and steroid hormones, such as estrogen, which diffuse into cells to find their receptors.
3.5 PHOSPHORUS-CONTAINING COMPOUNDS
Phosphoric acid is an inorganic acid (it does not contain carbon) with the potential to donate three protons. The Kas for the three acid dissociation equilibria are 2.1, 7.2, and 12.4. Therefore, at physiological pH, phosphoric acid is significantly dissociated, existing largely in anionic form.
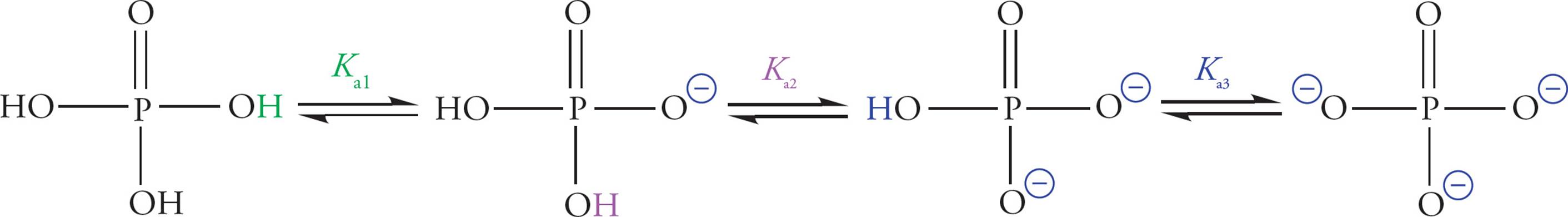
Figure 17 Phosphoric Acid Dissociation
Phosphate is also known as orthophosphate. Two orthophosphates bound together via an anhydride linkage form pyrophosphate. The P–O–P bond in pyrophosphate is an example of a high-energy phosphate bond. This name is derived from the fact that the hydrolysis of pyrophosphate is thermodynamically extremely favorable. The ∆G° for the hydrolysis of pyrophosphate is about −7 kcal/mol. This means that it is a very favorable reaction. The actual ∆G° in the cell is about −12 kcal/mol, which is even more favorable.
There are three reasons that phosphate anhydride bonds store so much energy:
1) When phosphates are linked together, their negative charges repel each other strongly.
2) Orthophosphate has more resonance forms and thus a lower free energy than linked phosphates.
3) Orthophosphate has a more favorable interaction with the biological solvent (water) than linked phosphates.
The details are not crucial. What is essential is that you fix the image in your mind of linked phosphates acting like compressed springs, just waiting to fly open and provide energy for an enzyme to catalyze a reaction.
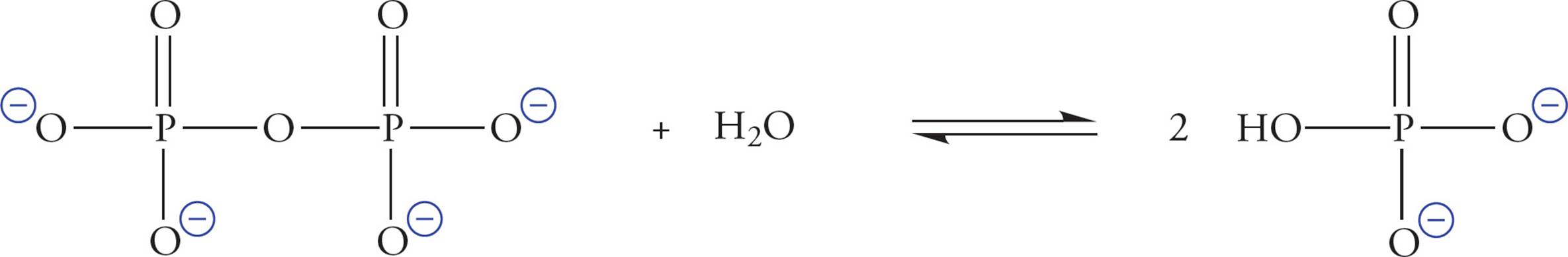
Figure 18 The Hydrolysis of Pyrophosphate
Nucleotides
Nucleotides are the building blocks of nucleic acids (RNA and DNA). Each nucleotide contains a ribose (or deoxyribose) sugar group; a purine or pyrimidine base joined to carbon number one of the ribose ring; and one, two, or three phosphate units joined to carbon five of the ribose ring. The nucleotide adenosine triphosphate (ATP) plays a central role in cellular metabolism in addition to being an RNA precursor. Significantly more information about the structure and function of the nucleic acids RNA and DNA will be provided in Chapter 5.
ATP is the universal short-term energy storage molecule. Energy extracted from the oxidation of foodstuffs is immediately stored in the phosphoanhydride bonds of ATP. This energy will later be used to power cellular processes; it may also be used to synthesize glucose or fats, which are longer-term energy storage molecules. This applies to all living organisms, from bacteria to humans. Even some viruses carry ATP with them outside the host cell, though viruses cannot make their own ATP.
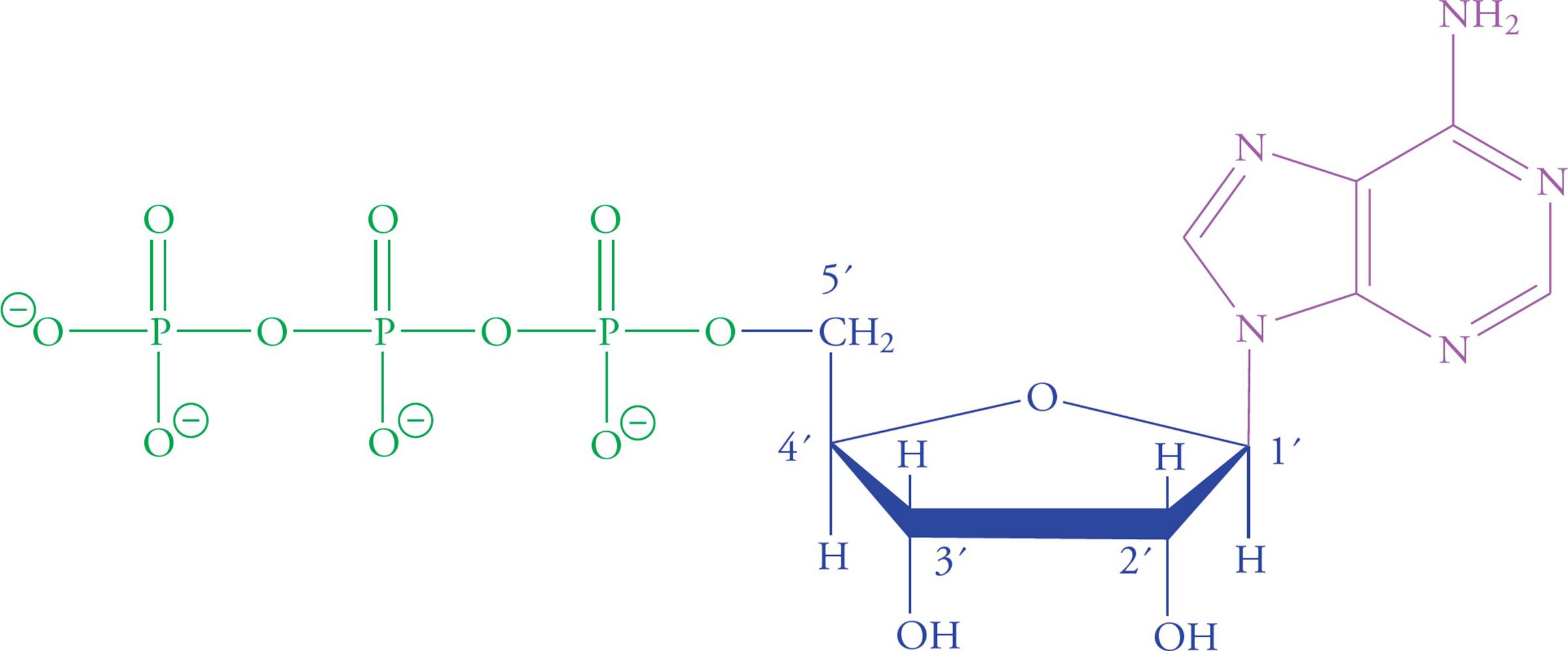
Figure 19 Adenosine Triphosphate (ATP)
Chapter 3 Summary
• Amino acids (AAs) consist of a tetrahedral a-carbon connected to an amino group, a carboxyl group, and a variable R group, which determines the AA’s properties.
• Proteins consist of amino acids linked by peptide bonds, which are very stable. The primary structure of a protein consists of its amino acid sequence.
• The secondary structure of proteins (α-helices and β-sheets) is formed through hydrogen bonding interactions between atoms in the backbone of the molecule.
• The most stable tertiary protein structure generally places polar AA’s on the exterior and nonpolar AA’s on the interior of the protein. This minimizes interactions between nonpolar AA’s and water, while optimizing interactions between side chains inside the protein.
• Proteins have a variety of functions in the body including (but not limited to) enzymes, structural roles, hormones, receptors, channels, antibodies, transporters, etc.
• The monomer for a carbohydrate is a monosaccharide (simple sugar), with the molecular formula CnH2nOn. The common monosaccharides are glucose, fructose, galactose, ribose, and deoxyribose.
• Two monosaccharides joined with a glycosidic linkage form a disaccharide. The common disaccharides are maltose, sucrose, and lactose. Mammals can digest a glycosidic linkages, but generally not blinkages.
• Polysaccharides consist of many monosaccharides linked together. Glycogen (animals) and starch (plants) are storage units for glucose and can be broken down for energy. Cellulose is also a glucose polymer, but the beta linkage prevents digestion. It forms wood and cotton.
• Lipids are found in several forms in the body, including triglycerides, phospholipids, cholesterol and steroids, and terpenes. Triglycerides and phospholipids are linear, while cholesterol and steroids have a ring structure.
• Lipids are hydrophobic. Triglycerides are used for energy storage, phospholipids form membranes, and cholesterol is the precursor to the steroid hormones.
• The building blocks of nucleic acids (DNA and RNA) are nucleotides, which are comprised of a pentose sugar, a purine or pyrimidine base, and 2-3 phosphate units.
CHAPTER 3 FREESTANDING PRACTICE QUESTIONS
1. Why is ATP known as a “high energy” structure at neutral pH?
A) It exhibits a large decrease in free energy when it undergoes hydrolytic reactions.
B) The phosphate ion released from ATP hydrolysis is very reactive.
C) It causes cellular processes to proceed at faster rates.
D) Adenine is the best energy storage molecule of all the nitrogenous bases.
2. Which of the following best describes the secondary structure of a protein?
A) Various folded polypeptide chains joining together to form a larger unit
B) The amino acid sequence of the chain
C) The polypeptide chain folding upon itself due to hydrophobic/hydrophilic interactions
D) Peptide bonds hydrogen-bonding to one another to create a sheet-like structure
3. Phenylketonuria (PKU) is an autosomal recessive disorder that results from a deficiency of the enzyme phenylalanine hydroxylase. This enzyme normally converts phenylalanine into tyrosine. PKU results in intellectual disability, growth retardation, fair skin, eczema and a distinct musty body odor. Which of the following is most likely true?
A) Treatment should include a decrease in tyrosine in the diet.
B) The musty body odor is likely caused by a disorder in aromatic amino acid metabolism.
C) Patients with PKU should increase the amount of phenylalanine in their diet.
D) PKU can be acquired by consuming too much aspartame (an artificial sweetener that contains high levels of phenylalanine).
4. A genetic regulator is found to contain a lysine residue that is important for its binding to DNA. If a mutation were to occur such that a different amino acid replaces the lysine at that location, which of the following resulting amino acids would likely be the least harmful to its ability to bind DNA?
A) Glycine
B) Glutamate
C) Aspartate
D) Arginine
5. Increasing the amount of cholesterol in a plasma membrane would lead to an increase in:
A) permeability.
B) atherosclerotic plaques.
C) melting temperature.
D) freezing temperature.
6. A human space explorer crash-lands on a planet where the native inhabitants are entirely unable to digest glycogen, but are able to digest cellulose. Consequently, they make their clothing out of glycogen-based material. The starving space explorer eats one of the native inhabitants’ shirts and the natives are amazed. Based on this information, which of the following is/are true?
I. The explorer can digest α-glycosidic linkages.
II. The native inhabitants can digest α-glycosidic linkages.
III. The native inhabitants can digest starch.
A) I only
B) I and III only
C) II and III only
D) I, II, and III
CHAPTER 3 PRACTICE PASSAGE
Photosynthesis is the process plants use to derive energy from sunlight and is associated with a cell’s chloroplasts. The energy is used to produce carbohydrates from carbon dioxide and water. Photosynthesis involves light and dark phases. Figure 1 represents two initial steps associated with the light phase.
The light phase supplies the dark phase with NADPH and a high-energy substrate.
A researcher attempted to produce a photosynthetic system outside the living organism according to the following protocols:
• Chloroplasts were extracted from green leaves and ruptured, and their membranes were thereby exposed, then a solution of hexachloroplatinate ions carrying a charge of −2 was added.
• The structure of the composite was analyzed, and the amount of oxygen produced by the system was measured.
The researcher concluded that the ions were bound to the membrane’s Photosystem 1 site by the attraction of opposite charges. The resulting composite is shown in Figure 2. It was found that the hexachloroplatinate-membrane composite was photosynthetically active.
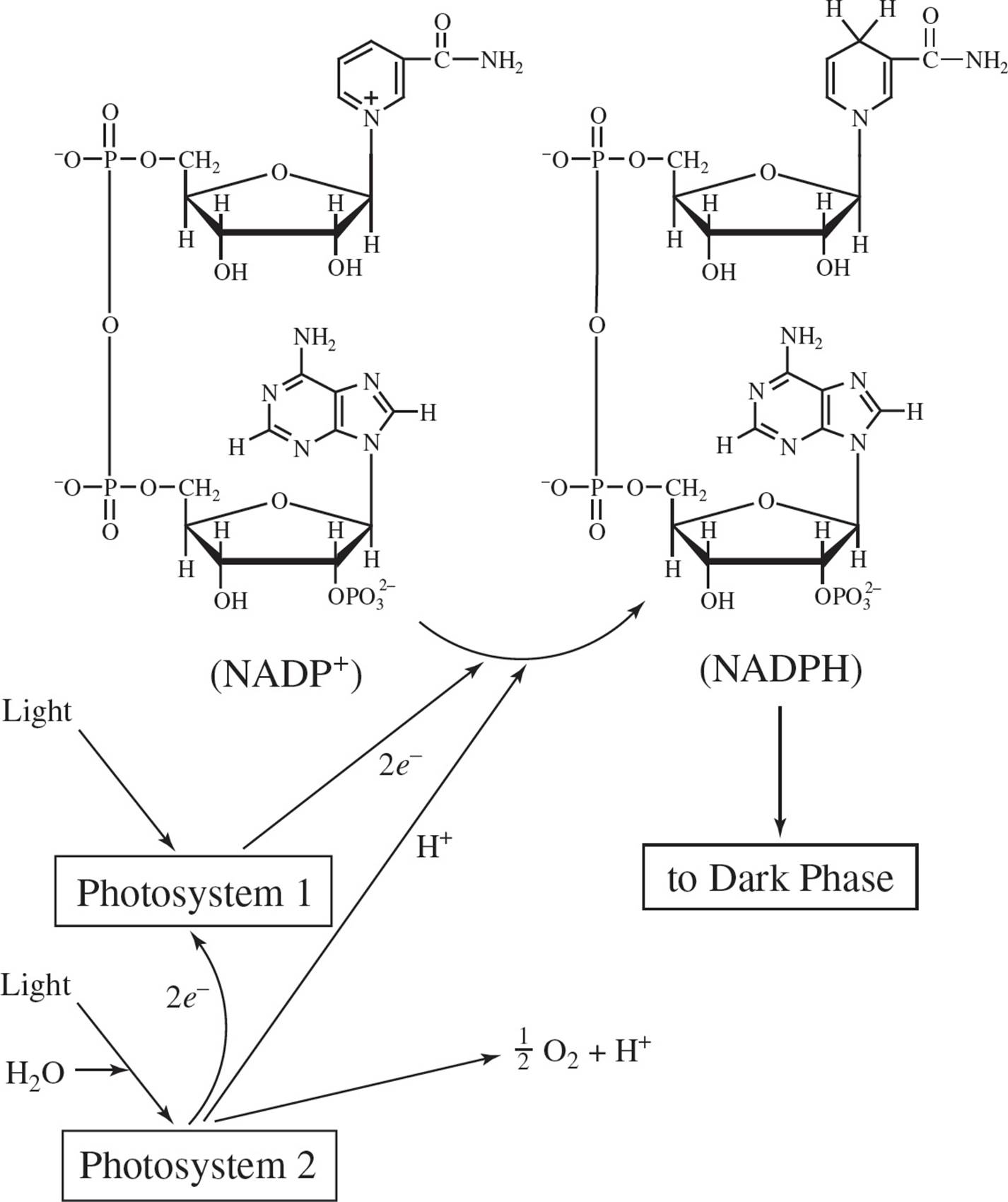
Figure 1
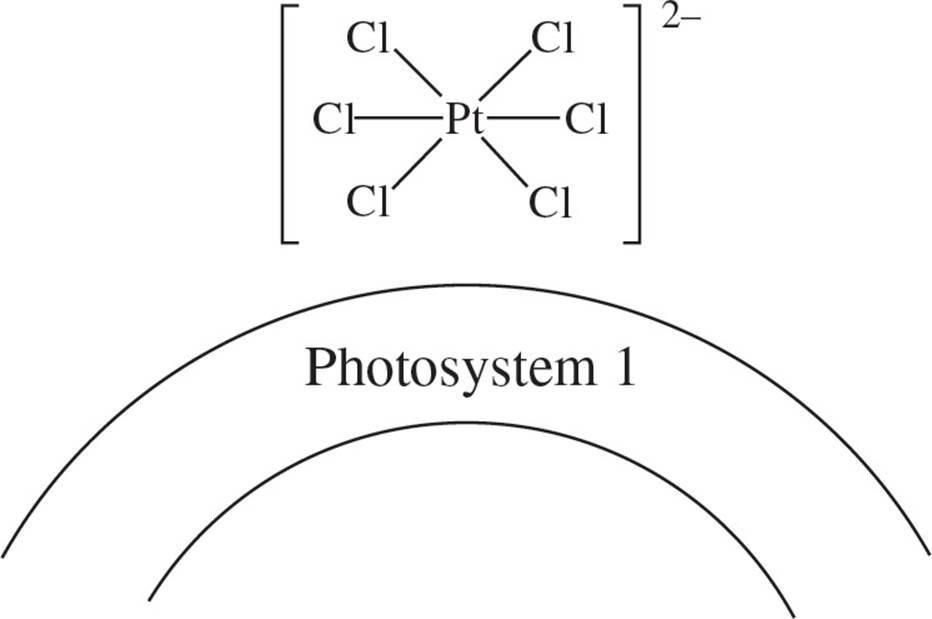
Figure 2
1. In concluding that the hexachloroplatinate ions were bound to Photosystem 1 due to the attraction of opposite charges, the researchers apparently assumed that the structure of the membrane was:
A) determined solely by hydrophobic bonding.
B) positively charged.
C) covalently bound to the platinate.
D) negatively charged.
2. Figure 1 indicates that:
A) photoactivation of the chloroplast membrane results in the reduction of the anhydride-containing molecule NADP+.
B) electrons are lost from Photosystem 1 through the conversion of NADPH to NADP+, and are replaced by electrons from Photosystem 2.
C) there is a net gain of electrons by the system.
D) electrons are lost from Photosystem 1 through the conversion of NADP+ to NADPH, but are not replaced by electrons from Photosystem 2.
3. In addition to NADPH, the photosynthetic light phase must supply the dark phase with another molecule which stores energy for biosynthesis. Among the following, the molecule would most likely be:
A) ADP.
B) CO2.
C) inorganic phosphate.
D) ATP.
4. If NADP+ is fully hydrolyzed to its component bases, phosphates, and sugars, what type of monosaccharide would result?
A) A three-carbon triose
B) A hexose
C) A pentose
D) An α-D-glucose
5. If in a given cell the photosynthetic dark phase were artificially arrested while the light phase proceeded, the cell would most likely experience:
A) decreased levels of NADPH.
B) increased levels of NADPH.
C) increased levels of carbohydrate.
D) increased photoactivation of the chloroplast.
6. To determine the primary structure of the protein portion of Photosystem 1, a series of cleavage reactions was undertaken. To break apart the protein, the most logical action to take would be to:
A) decarboxylate free carboxyl groups.
B) hydrolyze peptide bonds.
C) repolymerize peptide bonds.
D) hydrolyze amide branch points.
7. A researcher examined a sample of the principal substance produced by the photosynthetic dark phase and concluded that he was working with a racemic mixture of glucose isomers. Which of the following experimental findings would be inconsistent with such a conclusion?
A) The sample is composed of carbon, hydrogen, and oxygen only.
B) The sample consists of an aldohexose.
C) The sample rotates the plane of polarized light to the left.
D) The sample is optically inactive.
SOLUTIONS TO CHAPTER 3 FREESTANDING PRACTICE QUESTIONS
1. A Choice A is the best because it directly addresses the energetics of ATP hydrolysis. Choice B discusses the reactivity of the released phosphate ion and not the structure of ATP itself, so it can be eliminated. Choice C can be eliminated because it describes the rate of cellular processes not the energy of ATP. Choice D can be eliminated because the structure of adenine is not related to why ATP is a good energy storage molecule.
2. D The secondary structure of proteins is the initial folding of the polypeptide chain into a-helices or b-pleated sheets. Choice A describes the formation of a quaternary protein, choice B can be eliminated because it describes the primary protein structure, and choice C can be eliminated because it describes the tertiary protein structure.
3. B A defect in phenylalanine hydroxylase (or the THB cofactor) would result in a build-up of phenylalanine. This would lead to an excess of phenylalanine byproducts such as phenylacetate, phenyllactate and phenylpyruvate, and a decrease in tyrosine. Therefore, patients with PKU should increase the amount of tyrosine in their diet (it becomes an essential amino acid in this condition; choice A is wrong), as well as eliminate phenylalanine from their diet (choice C is wrong). PKU is a genetically acquired disorder (autosomal recessive), as mentioned in the passage, and thus it is not acquired by consuming too much phenylalanine (choice D is wrong). It is true that phenylalanine and its derivatives are aromatic amino acids and that the high levels of these compounds lead to the distinct musty body odor (choice B is correct). Process of elimination (POE) is probably the best method to use in answering this question since it is unclear (without prior knowledge of PKU) what the underlying mechanism of the body odor would be.
4. D While knowing the structures of the different amino acids is unlikely to be important for the MCAT, knowing which of the amino acids are basic (histidine, arginine, lysine) and which are acidic (glutamate, aspartate) is likely to be relevant. In this case, since lysine is basic (and therefore best at binding the negatively charged DNA), one can assume that a mutation resulting in another basic amino acid would cause the least change in its ability to bind DNA. Therefore, a mutation from lysine to arginine would cause the least harm (choice D is correct). A mutation from lysine to glutamate or aspartate (both acidic) would likely cause the most harm to its ability to bind DNA (choices B and C are wrong). Glycine is a neutral amino acid (choice A is wrong).
5. C Plasma membranes can be up to 50% composed of sterols. Sterols help stabilize the membrane at both spectrums of the temperature. At low temperatures, they increase fluidity because the ring structure of cholesterol does not allow for tight phospholipid tail packing. This decreases the temperature at which the membrane would freeze (choice D is wrong). At high temperatures, cholesterol decreases membrane fluidity (the OH group of cholesterol prevents phospholipid dispersion) and permeability (by filling in the “holes” between the fatty acid tails, choice A is wrong), thus increasing the temperature at which membranes would melt (choice C is correct). The formation of atherosclerotic plaques, while related to cholesterol, is due to high levels of blood cholesterol, not membrane cholesterol (choice B is wrong).
6. A Item I is true: humans can digest α-glycosidic linkages, such as those found in glycogen. If the natives’ shirts are made of glycogen, our space explorer should have no trouble consuming and digesting them (choice C can be eliminated). Item II Is false: cellulose contains β-glycosidic linkages. If the natives can digest cellulose, but not glycogen, then they cannot digest α-glycosidic linkages (choice D can be eliminated). Item III is false: starch also contains a α-glycosidic linkages. If the natives cannot digest glycogen, then they likely cannot digest starch either (choice B can be eliminated and choice A is true).
SOLUTIONS TO CHAPTER 3 PRACTICE PASSAGE
1. B The passage states that the ion is attracted to Photosystem 1 by the attraction of opposite charges (positively-charged photosystem and negatively-charged hexachloroplatinate ion).
2. A The main result of the light phase, as depicted in Figure 1, is the reduction of NADP+ to make NADPH (choice A). Choice B is wrong since NADP+ is converted into NADPH, not vice versa. Choice C is incorrect since in any system, mass and charge are conserved. Electrons move from one molecule to another, but they are not created or destroyed in a chemical reaction. Choice D is eliminated since Figure 1 depicts electrons moving from Photosystem 2 to Photosystem 1.
3. D The passage states that the light reactions supply the dark reactions with a “high energy substrate”. The most likely candidate among the choices is ATP.
4. C NADPH contains ribose, a pentose.
5. B The light phase makes NADPH, and the dark phase consumes it. In the absence of the dark phase, NADPH will continue to be produced, but none will be consumed, making NADPH levels rise (choice B). Choice C is wrong since the dark phase is responsible for biosynthesis, such as carbohydrate production, so this will decrease, not increase. Choice D can be eliminated since the amount of light and photoactivation should remain the same.
6. B Proteins are composed of amino acid residues which are joined together by peptide bonds during the translation process. To split the protein into smaller pieces, proteases and chemical reagents act to hydrolyze the peptide bond, reversing the biosynthetic process.
7. C A racemic mixture is one which contains equal quantities of two stereoisomers that rotate plane-polarized light in opposite directions. Since there are equal quantities of both, racemic mixtures are optically inactive. Thus, choice C, which states that the sample rotates light, is inconsistent with the conclusion that the sample is racemic and is the correct answer choice. All other choices are consistent with the conclusion that the sample is a racemic mixture of glucose. Carbohydrates, of which glucose is one, are made of only carbon, hydrogen and oxygen (choice A is consistent and can be eliminated), glucose, with six carbons and a carbonyl group on the 6thcarbon, is an aldohexose (choice B is consistent and can be eliminated), and racemic mixtures do not rotate light (choice D is consistent and can be eliminated).
1 Leucine, alanine, and isoleucine are all hydrophobic residues more likely to be found on the interior than the exterior of proteins. Serine (choice C), which has a hydroxyl group that can hydrogen bond with water, is the correct answer.
2 As stated above, the amino end is always written first. Hence, the oligopeptide begins with an exposed Phe amino group and ends with an exposed Ala carboxyl; all the other backbone groups are hitched together in peptide bonds. Out of all the R-groups, there is only one acidic or basic functional group, the acidic glutamate R-group. This R-group plus the two terminal backbone groups gives a total of three acid/base functional groups.
3 The dipeptide has a higher free energy, so its existence is less favorable. In other words, existence of the chain is less favorable than existence of the isolated amino acids.
4 During protein synthesis, stored energy is used to force peptide bonds to form. Once the bond is formed, even though its destruction is thermodynamically favorable, it remains stable because the activation energy for the hydrolysis reaction is so high. In other words, hydrolysis is thermodynamically favorable but kinetically slow.
5 Trypsin will cleave on the carboxyl side of the Lys residue, with Phe on the N-terminus of the new Phe-Phe-Lys fragment. There will be two fragments after trypsin cleavage: Phe-Phe-Lys and Ala-Gly-Glu-Lys.
6 The sulfur in cysteine is bonded to a hydrogen and a carbon; the sulfur in cystine is bonded to a sulfur and a carbon. Hence, the sulfur in cystine is more oxidized.
7 In a reducing environment, the S-S group is reduced to two SH groups. Disulfide bridges are found only in extracellular polypeptides, where they will not be reduced. Examples of protein complexes held together by disulfide bridges include antibodies and the hormone insulin.
8 It would be antiparallel because one participant in the β-pleated sheet would have a C to N direction, while the other would be running N to C.
9 Putting a protein in a urea solution will disrupt H-bonding, thus disrupting secondary structure by unfolding α-helices and β-sheets. It would not affect primary structure, which depends on the much more stable peptide bond. Disruption of 2°, 3°, or 4° structure without breaking peptide bonds is denaturation.
10 The disulfide bridges would be broken. Tertiary structure would be less stable.
11 The shape should not be disrupted if breaking disulfides is the only disturbance. It’s just that the shape would be less sturdy—like a concrete wall without the rebar.
12 No. If you allow disulfide bridges to form while the protein is still denatured, it will become locked into an abnormal shape.
13 You should end up with the correct structure. In step one, you break the reinforcing disulfide bridges. In step two, you denature the protein completely by disrupting H-bonds. In step four, you allow the H-bonds to reform; as stated in the text, normally the correct tertiary structure will form spontaneously if you leave the polypeptide alone. In step three, you reform the disulfide bridges, thus locking the structure into its correct form.
14 This is a simple question provided to clarify the classification of the disulfide bridge. Item I is a good example of 3° structure. Item II is describes 2°, not 3°, structure. Item III describes the disulfide, which is considered to be tertiary because of when it is formed, despite the fact that it is a covalent bond.
15 Quaternary disulfides are bonds that form between chains that aren’t linked by peptide bonds. Tertiary disulfides are bonds that form between residues in the same polypeptide.
16 The anomeric carbon of glucose is pointing down, which means the linkage is α-1,2. So, sucrose is Glc-α-1,2-Fru.
17 Because hydrolysis of polysaccharides is thermodynamically favored, energy input is required to drive the reaction toward polysaccharide synthesis.
18 No, because then polysaccharides would hydrolyze spontaneously (they’d be unstable). The high activation energy of polysaccharide hydrolysis allows us to use enzymes as gatekeepers—when we need energy from glucose, we open the gate of glycogen hydrolysis.
19 An unsaturated fatty acid is bent, or “kinked,” at the cis double bond.
20 The long hydrophobic chains will interact with each other to minimize contact with water, exposing the charged carboxyl group to the aqueous environment.
21 Grease is hydrophobic. It does not wash off easily in water because it is not soluble in water. Scrubbing your hands with soap causes micelles to form around the grease particles.
22 The bent shape of the unsaturated fatty acid means that it doesn’t fit in as well and has less contact with neighboring groups to form van der Waals interactions. Phospholipids composed of saturated fatty acids make the membrane more solid.