CHEMISTRY THE CENTRAL SCIENCE
16 ACID–BASE EQUILIBRIA
16.9 ACID–BASE PROPERTIES OF SALT SOLUTIONS
Even before you began this chapter, you were undoubtedly aware of many substances that are acidic, such as HNO3, HCl, and H2SO4, and others that are basic, such as NaOH and NH3. However, our discussion up to this point in the chapter has indicated that ions can also exhibit acidic or basic properties. For example, we calculated Ka for NH4+ and Kb for F– in Sample Exercise 16.17. Such behavior implies that salt solutions can be acidic or basic. Before proceeding with further discussions of acids and bases, let's examine the way dissolved salts can affect pH.
Because nearly all salts are strong electrolytes, we can assume that any salt dissolved in water is completely dissociated. Consequently, the acid–base properties of salt solutions are due to the behavior of the cations and anions. Many ions react with water to generate H+(aq) or OH–(aq) ions. This type of reaction is often called hydrolysis. The pH of an aqueous salt solution can be predicted qualitatively by considering the salt's cations and anions.
An Anion's Ability to React with Water
In general, an anion X– in solution can be considered the conjugate base of an acid. For example, Cl– is the conjugate base of HCl, and CH3COO– is the conjugate base of CH3COOH. Whether or not an anion reacts with water to produce hydroxide ions depends on the strength of the anion's conjugate acid. To identify the acid and assess its strength, we add a proton to the anion's formula. If the acid HX determined in this way is one of the seven strong acids listed on page 664, the anion has a negligible tendency to abstract protons from water and does not affect the pH of the solution. The presence of Cl– in an aqueous solution, for example, does not result in the production of any OH–and does not affect the pH. Thus, Cl– is always a spectator ion in acid–base chemistry.
If HX is not one of the seven strong acids, it is a weak acid. In this case, the conjugate base X– is a weak base and it reacts to a small extent with water to produce the weak acid and hydroxide ions:
![]()
The OH– ion generated in this way increases the pH of the solution, making it basic. Acetate ion, for example, being the conjugate base of a weak acid, reacts with water to produce acetic acid and hydroxide ions, thereby increasing the pH of the solution:
![]()
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What effect will NO3– ions have on the pH of a solution? What effect will CO32– ions have?
The situation is more complicated for salts containing anions that have ionizable protons, such as HSO3–. These salts are amphiprotic (Section 16.2), and how they behave in water is determined by the relative magnitudes of Ka and Kb for the ion, as shown in Sample Exercise 16.19. IfKa > Kb, the ion causes the solution to be acidic. If Kb > Ka, the solution is made basic by the ion.
A Cation's Ability to React with Water
Polyatomic cations containing one or more protons can be considered the conjugate acids of weak bases. The NH4+ ion, for example, is the conjugate acid of the weak base NH3. Thus, NH4+ is a weak acid and will donate a proton to water, producing hydronium ions and thereby lowering the pH:
![]()
Many metal ions react with water to decrease the pH of an aqueous solution. This effect is most pronounced for small, highly charged cations like Fe3+ and Al3+, as illustrated by the Ka values for metal cations in ![]() TABLE 16.6. A comparison of Fe2+ and Fe3+ values in the table illustrates how acidity increases as ionic charge increases.
TABLE 16.6. A comparison of Fe2+ and Fe3+ values in the table illustrates how acidity increases as ionic charge increases.
TABLE 16.6 • Acid-Dissociation Constants for Metal Cations in Aqueous Solution at 25 °C
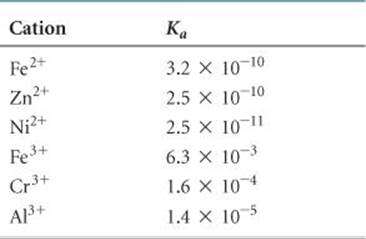
Notice that Ka values for the 3+ ions in Table 16.6 are comparable to the values for familiar weak acids, such as acetic acid (Ka = 1.8 × 10–5). The ions of alkali and alkaline earth metals, being relatively large and not highly charged, do not react with water and therefore do not affect pH. Note that these are the same cations found in the strong bases (Section 16.5). The different tendencies of four cations to lower the pH of a solution are illustrated in ![]() FIGURE 16.15.
FIGURE 16.15.

![]() FIGURE 16.15 Effect of cations on solution pH. The pH values of 1.0 M solutions of four nitrate salts are estimated using acid–base indicators.
FIGURE 16.15 Effect of cations on solution pH. The pH values of 1.0 M solutions of four nitrate salts are estimated using acid–base indicators.
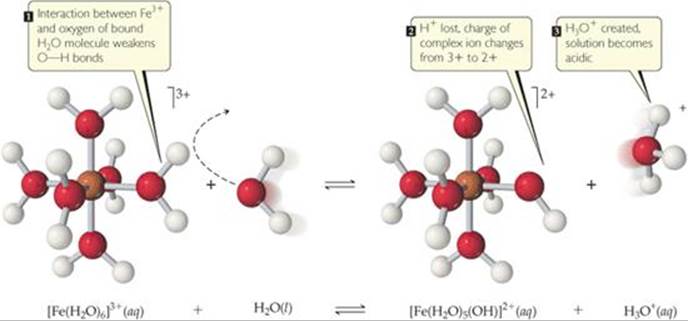
![]() FIGURE 16.16 A hydrated Fe3+ ion acts as an acid by donating an H+ to a free H2O molecule, forming H3O+.
FIGURE 16.16 A hydrated Fe3+ ion acts as an acid by donating an H+ to a free H2O molecule, forming H3O+.
The mechanism by which metal ions produce acidic solutions is shown in ![]() FIGURE 16.16. Because metal ions are positively charged, they attract the unshared electron pairs of water molecules and become hydrated.
FIGURE 16.16. Because metal ions are positively charged, they attract the unshared electron pairs of water molecules and become hydrated. ![]() (Section 13.1) The larger the charge on the metal ion, the stronger the interaction between the ion and the oxygen of its hydrating water molecules. As the strength of this interaction increases, the O—H bonds in the hydrating water molecules become weaker. This facilitates transfer of protons from the hydration water molecules to solvent water molecules.
(Section 13.1) The larger the charge on the metal ion, the stronger the interaction between the ion and the oxygen of its hydrating water molecules. As the strength of this interaction increases, the O—H bonds in the hydrating water molecules become weaker. This facilitates transfer of protons from the hydration water molecules to solvent water molecules.
Combined Effect of Cation and Anion in Solution
To determine whether a salt forms an acidic, a basic, or a neutral solution when dissolved in water, we must consider the action of both cation and anion. There are four possible combinations.
1. If the salt contains an anion that does not react with water and a cation that does not react with water, we expect the pH to be neutral. Such is the case when the anion is a conjugate base of a strong acid and the cation is either from group 1A or one of the heavier members of group 2A (Ca2+, Sr2+, and Ba2+). Examples: NaCl, Ba(NO3)2, RbClO4.
2. If the salt contains an anion that reacts with water to produce hydroxide ions and a cation that does not react with water, we expect the pH to be basic. Such is the case when the anion is the conjugate base of a weak acid and the cation is either from group 1A or one of the heavier members of group 2A (Ca2+, Sr2+, and Ba2+). Examples: NaClO, RbF, BaSO3.
3. If the salt contains a cation that reacts with water to produce hydronium ions and an anion that does not react with water, we expect the pH to be acidic. Such is the case when the cation is a conjugate acid of a weak base or a small cation with a charge of 2+ or greater. Examples:NH4NO3, AlCl3, Fe(NO3)3.
4. If the salt contains an anion and a cation both capable of reacting with water, both hydroxide ions and hydronium ions are produced. Whether the solution is basic, neutral, or acidic depends on the relative abilities of the ions to react with water. Examples: NH4ClO, Al(CH3COO)3, CrF3.
SAMPLE EXERCISE 16.18 Determining Whether Salt Solutions Are Acidic, Basic, or Neutral
Determine whether aqueous solutions of each of these salts are acidic, basic, or neutral: (a) Ba(CH3COO)2, (b) NH4Cl, (c) CH3NH3Br, (d) KNO3, (e) Al(ClO4)3.
SOLUTION
Analyze We are given the chemical formulas of five ionic compounds (salts) and asked whether their aqueous solutions will be acidic, basic, or neutral.
Plan We can determine whether a solution of a salt is acidic, basic, or neutral by identifying the ions in solution and by assessing how each ion will affect the pH.
Solve
(a) This solution contains barium ions and acetate ions. The cation is an ion of a heavy alkaline earth metal and will therefore not affect the pH. The anion, CH3COO–, is the conjugate base of the weak acid CH3COOH and will hydrolyze to produce OH– ions, thereby making the solution basic (combination 2).
(b) In this solution, NH4+ is the conjugate acid of a weak base (NH3) and is therefore acidic. Cl– is the conjugate base of a strong acid (HCl) and therefore has no influence on the pH of the solution. Because the solution contains an ion that is acidic (NH4+) and one that has no influence on pH (Cl–), the solution of NH4Cl will be acidic (combination 3).
(c) Here CH3NH3+ is the conjugate acid of a weak base (CH3NH2, an amine) and is therefore acidic, and Br– is the conjugate base of a strong acid (HBr) and therefore pH neutral. Because the solution contains one ion that is acidic and one that has no influence on pH, the solution of CH3NH3Br will be acidic (combination 3).
(d) This solution contains the K+ ion, which is a cation of group 1A, and the NO3– ion, which is the conjugate base of the strong acid HNO3. Neither of the ions will react with water to any appreciable extent, making the solution neutral (combination 1).
(e) This solution contains Al3+ and ClO4– ions. Cations, such as Al3+, that have a charge of 3+ or higher are acidic. The ClO4– ion is the conjugate base of a strong acid (HClO4) and therefore does not affect pH. Thus, the solution of Al(ClO4)3 will be acidic (combination 3).
PRACTICE EXERCISE
Indicate which salt in each of the following pairs forms the more acidic (or less basic) 0.010 M solution: (a) NaNO3 or Fe(NO3)3, (b) KBr or KBrO, (c) CH3NH3Cl or BaCl2, (d) NH4NO2 or NH4NO3.
Answers: (a) Fe(NO3)3, (b) KBr, (c) CH3NH3Cl, (d) NH4NO3
SAMPLE EXERCISE 16.19 Predicting Whether the Solution of an Amphiprotic Anion Is Acidic or Basic
Predict whether the salt Na2HPO4 forms an acidic solution or a basic solution when dissolved in water.
SOLUTION
Analyze We are asked to predict whether a solution of Na2HPO4 is acidic or basic. This substance is an ionic compound composed of Na+ and HPO42– ions.
Plan We need to evaluate each ion, predicting whether it is acidic or basic. Because Na+ is a cation of group 1A, it has no influence on pH. Thus, our analysis of whether the solution is acidic or basic must focus on the behavior of the HPO42– ion. We need to consider the fact that HPO42–can act as either an acid or a base:
![]()
![]()
Of these two reactions, the one with the larger equilibrium constant determines whether the solution is acidic or basic.
Solve The value of Ka for Equation 16.45 is 4.2 × 10–13 (Table 16.3). For Equation 16.46, we must calculate Kb for the base HPO42– from the value of Ka for its conjugate acid, H2PO4–, and the relationship Ka× Kb = Kw (Equation 16.40). Using the value Ka(H2PO4–) = 6.2 × 10–8 from Table 16.3, we have
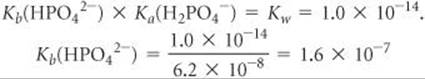
This Kb value is more than 105 times larger than Ka for HPO42–; thus, the reaction in Equation 16.46 predominates over that in Equation 16.45, and the solution is basic.
PRACTICE EXERCISE
Predict whether the dipotassium salt of citric acid (K2HC6H5O7) forms an acidic or basic solution in water (see Table 16.3 for data).
Answer: acidic