CHEMISTRY THE CENTRAL SCIENCE
2 ATOMS, MOLECULES, AND IONS
2.8 NAMING INORGANIC COMPOUNDS
The names and chemical formulas of compounds are essential vocabulary in chemistry. The system used in naming substances is called chemical nomenclature, from the Latin words nomen (name) and calare (to call).
There are more than 50 million known chemical substances. Naming them all would be a hopelessly complicated task if each had a name independent of all others. Many important substances that have been known for a long time, such as water (H2O) and ammonia (NH3), do have traditional names (called common names). For most substances, however, we rely on a set of rules that leads to an informative and unique name for each substance, a name based on the composition of the substance.
The rules for chemical nomenclature are based on the division of substances into categories. The major division is between organic and inorganic compounds. Organic compounds contain carbon and hydrogen, often in combination with oxygen, nitrogen, or other elements. All others are inorganic compounds. Early chemists associated organic compounds with plants and animals and inorganic compounds with the nonliving portion of our world. Although this distinction is no longer pertinent, the classification between organic and inorganic compounds continues to be useful. In this section we consider the basic rules for naming three categories of inorganic compounds: ionic compounds, molecular compounds, and acids.
Names and Formulas of Ionic Compounds
Recall from Section 2.7 that ionic compounds usually consist of metal ions combined with nonmetal ions. The metals form the cations, and the nonmetals form the anions.
1. Cations
a. Cations formed from metal atoms have the same name as the metal:
![]()
b. If a metal can form cations with different charges, the positive charge is indicated by a Roman numeral in parentheses following the name of the metal:

Ions of the same element that have different charges have different properties, such as different colors (![]() FIGURE 2.23).
FIGURE 2.23).

![]() Figure 2.23 Different ions of the same element have different properties. Both substances shown are compounds of iron. The substance on the left is Fe3O4, which contains Fe2+ and Fe3+ ions. The substance on the right is Fe2O3, which contains Fe3+ ions.
Figure 2.23 Different ions of the same element have different properties. Both substances shown are compounds of iron. The substance on the left is Fe3O4, which contains Fe2+ and Fe3+ ions. The substance on the right is Fe2O3, which contains Fe3+ ions.
Most metals that form cations with different charges are transition metals, elements that occur in the middle of the periodic table, from group 3B to group 2B. The metals that form only one cation (only one possible charge) are those of group 1A and group 2A, as well as Al3+(group 3A) and two transition-metal ions: Ag+ (group 1B) and Zn2+ (group 2B). Charges are not expressed when naming these ions. However, if there is any doubt in your mind whether a metal forms more than one cation, use a Roman numeral to indicate the charge. It is never wrong to do so, even though it may be unnecessary.
An older method still widely used for distinguishing between differently charged ions of a metal uses the endings -ous and -ic added to the root of the element's Latin name:

Although we will only rarely use these older names in this text, you might encounter them elsewhere.
c. Cations formed from nonmetal atoms have names that end in -ium:
![]()
These two ions are the only ions of this kind that we will encounter frequently in the text.
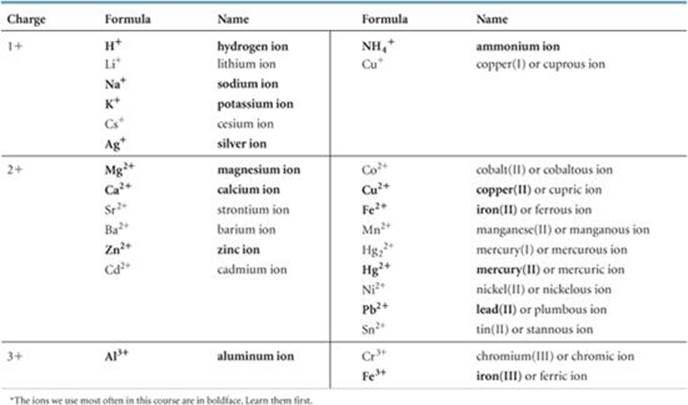
The names and formulas of some common cations are shown in ![]() TABLE 2.4 and on the back inside cover of the text. The ions on the left side in Table 2.4 are the monatomic ions that do not have more than one possible charge. Those on the right side are either polyatomic cations or cations with more than one possible charge. The Hg22+ ion is unusual because, even though it is a metal ion, it is not monatomic. It is called the mercury(I) ion because it can be thought of as two Hg+ ions bound together. The cations that you will encounter most frequently are shown in boldface. You should learn these cations first.
TABLE 2.4 and on the back inside cover of the text. The ions on the left side in Table 2.4 are the monatomic ions that do not have more than one possible charge. Those on the right side are either polyatomic cations or cations with more than one possible charge. The Hg22+ ion is unusual because, even though it is a metal ion, it is not monatomic. It is called the mercury(I) ion because it can be thought of as two Hg+ ions bound together. The cations that you will encounter most frequently are shown in boldface. You should learn these cations first.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
a. Why is CrO named using a Roman numeral, chromium(II) oxide, whereas CaO is named without a Roman numeral, calcium oxide?
b. What does the -ium ending on the name ammonium ion tell you about the composition of the ion?
TABLE 2.4 • Common Cations*
2. Anions
a. The names of monatomic anions are formed by replacing the ending of the name of the element with -ide:
![]()
A few polyatomic anions also have names ending in -ide:
![]()
b. Polyatomic anions containing oxygen have names ending in either -ate or -ite and are called oxyanions. The -ate is used for the most common or representative oxyanion of an element, and -ite is used for an oxyanion that has the same charge but one O atom fewer:

Prefixes are used when the series of oxyanions of an element extends to four members, as with the halogens. The prefix per- indicates one more O atom than the oxyanion ending in -ate; hypo- indicates one O atom fewer than the oxyanion ending in -ite:

These rules are summarized in ![]() FIGURE 2.24.
FIGURE 2.24.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What information is conveyed by the endings -ide, -ate, and -ite in the name of an anion?
![]() FIGURE 2.25 can help you remember the charge and number of oxygen atoms in the various oxyanions. Notice that C and N, both period 2 elements, have only three O atoms each, whereas the period 3 elements P, S, and Cl have four O atoms each. Beginning at the lower right inFigure 2.25, note that ionic charge increases from right to left, from 1– for CIO4– to 3– for PO43–. In the second period the charges also increase from right to left, from 1– for NO3– to 2– for CO32–. Notice also that although each of the anions in Figure 2.25 ends in -ate, the ClO4- ion also hasa per- prefix.
FIGURE 2.25 can help you remember the charge and number of oxygen atoms in the various oxyanions. Notice that C and N, both period 2 elements, have only three O atoms each, whereas the period 3 elements P, S, and Cl have four O atoms each. Beginning at the lower right inFigure 2.25, note that ionic charge increases from right to left, from 1– for CIO4– to 3– for PO43–. In the second period the charges also increase from right to left, from 1– for NO3– to 2– for CO32–. Notice also that although each of the anions in Figure 2.25 ends in -ate, the ClO4- ion also hasa per- prefix.
![]() GO FIGURE
GO FIGURE
Name the anion obtained by removing one oxygen atom from the perbromate ion, BrO4–.
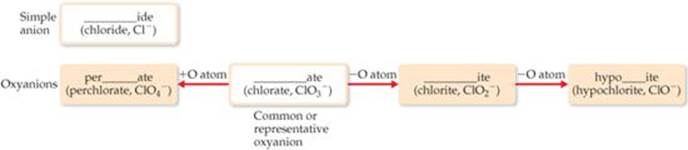
![]() Figure 2.24 Procedure for naming anions. The first part of the element's name, such as “chlor” for chlorine or “sulf” for sulfur, goes in the blank.
Figure 2.24 Procedure for naming anions. The first part of the element's name, such as “chlor” for chlorine or “sulf” for sulfur, goes in the blank.
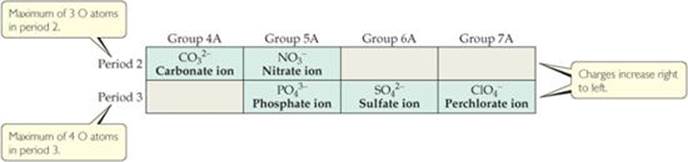
![]() Figure 2.25 Common oxyanions. The composition and charges of common oxyanions are related to their location in the periodic table.
Figure 2.25 Common oxyanions. The composition and charges of common oxyanions are related to their location in the periodic table.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Predict the formulas for the borate ion and silicate ion, assuming they contain a single B and Si atom, respectively, and follow the trends shown in Figure 2.25.
SAMPLE EXERCISE 2.11 Determining the Formula of an Oxyanion from Its Name
Based on the formula for the sulfate ion, predict the formula for (a) the selenate ion and (b) the selenite ion. (Sulfur and selenium are both in group 6A and form analogous oxyanions.)
SOLUTION
(a) The sulfate ion is SO42–. The analogous selenate ion is therefore SeO42–.
(b) The ending -ite indicates an oxyanion with the same charge but one O atom fewer than the corresponding oxyanion that ends in -ate. Thus, the formula for the selenite ion is SeO32–.
PRACTICE EXERCISE
The formula for the bromate ion is analogous to that for the chlorate ion. Write the formula for the hypobromite and bromite ions.
Answer: BrO– and BrO2–
c. Anions derived by adding H+ to an oxyanion are named by adding as a prefix the word hydrogen or dihydrogen, as appropriate:

Notice that each H+ added reduces the negative charge of the parent anion by one. An older method for naming some of these ions uses the prefix bi-. Thus, the HCO3– ion is commonly called the bicarbonate ion, and HSO4– is sometimes called the bisulfate ion.
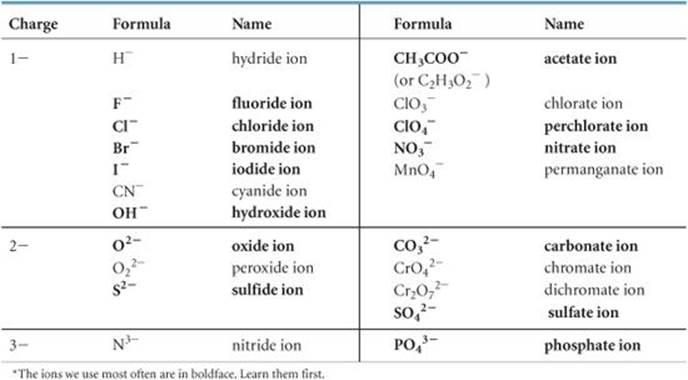
The names and formulas of the common anions are listed in ![]() TABLE 2.5 and on the back inside cover of the text. Those anions whose names end in -ide are listed on the left portion of Table 2.5, and those whose names end in -ate are listed on the right. The most common of these ions are shown in boldface. You should learn names and formulas of these anions first. The formulas of the ions whose names end with -ite can be derived from those ending in -ate by removing an O atom. Notice the location of the monatomic ions in the periodic table. Those of group 7A always have a 1– charge (F–, Cl–, Br–, and I–), and those of group 6A have a 2– charge (O2– and S2–).
TABLE 2.5 and on the back inside cover of the text. Those anions whose names end in -ide are listed on the left portion of Table 2.5, and those whose names end in -ate are listed on the right. The most common of these ions are shown in boldface. You should learn names and formulas of these anions first. The formulas of the ions whose names end with -ite can be derived from those ending in -ate by removing an O atom. Notice the location of the monatomic ions in the periodic table. Those of group 7A always have a 1– charge (F–, Cl–, Br–, and I–), and those of group 6A have a 2– charge (O2– and S2–).
3. Ionic Compounds
Names of ionic compounds consist of the cation name followed by the anion name:

In the chemical formulas for aluminum nitrate and copper(II) perchlorate, parentheses followed by the appropriate subscript are used because the compounds contain two or more polyatomic ions.
SAMPLE EXERCISE 2.12 Determining the Names of Ionic Compounds from Their Formulas
Name the ionic compounds (a) K2SO4, (b) Ba(OH)2, (c) FeCl3.
SOLUTION
In naming ionic compounds, it is important to recognize polyatomic ions and to determine the charge of cations with variable charge.
(a) The cation is K+, the potassium ion, and the anion is SO42–, the sulfate ion, making the name potassium sulfate. (If you thought the compound contained S2– and O2– ions, you failed to recognize the polyatomic sulfate ion.)
(b) The cation is Ba2+, the barium ion, and the anion is OH–, the hydroxide ion: barium hydroxide.
(c) You must determine the charge of Fe in this compound because an iron atom can form more than one cation. Because the compound contains three chloride ions, Cl–, the cation must be Fe3+, the iron(III), or ferric, ion. Thus, the compound is iron(III) chloride or ferric chloride.
PRACTICE EXERCISE
Name the ionic compounds (a) NH4Br, (b) Cr2O3, (c) Co(NO3)2.
Answers: (a) ammonium bromide, (b) chromium(III) oxide, (c) cobalt(II) nitrate
TABLE 2.5 • Common Anions*
SAMPLE EXERCISE 2.13 Determining the Formulas of Ionic Compounds from Their Names
Write the chemical formulas for (a) potassium sulfide, (b) calcium hydrogen carbonate, (c) nickel(II) perchlorate.
SOLUTION
In going from the name of an ionic compound to its chemical formula, you must know the charges of the ions to determine the subscripts.
(a) The potassium ion is K+, and the sulfide ion is S2–. Because ionic compounds are electrically neutral, two K+ ions are required to balance the charge of one S2– ion, giving K2S for the empirical formula.
(b) The calcium ion is Ca2+. The carbonate ion is CO32–, so the hydrogen carbonate ion is HCO3–. Two HCO3– ions are needed to balance the positive charge of Ca2+, giving Ca(HCO3)2.
(c) The nickel(II) ion is Ni2+. The perchlorate ion is ClO4–. Two ClO4– ions are required to balance the charge on one Ni2+ ion, giving Ni(ClO4)2.
PRACTICE EXERCISE
Give the chemical formulas for (a) magnesium sulfate, (b) silver sulfide, (c) lead(II) nitrate.
Answers: (a) MgSO4, (b) Ag2S, (c) Pb(NO3)2
Names and Formulas of Acids
Acids are an important class of hydrogen-containing compounds, and they are named in a special way. For our present purposes, an acid is a substance whose molecules yield hydrogen ions (H+) when dissolved in water. When we encounter the chemical formula for an acid at this stage of the course, it will be written with H as the first element, as in HCl and H2SO4.
An acid is composed of an anion connected to enough H+ ions to neutralize, or balance, the anion's charge. Thus, the SO42– ion requires two H+ ions, forming H2SO4. The name of an acid is related to the name of its anion, as summarized in ![]() FIGURE 2.26.
FIGURE 2.26.
1. Acids containing anions whose names end in -ide are named by changing the -ide ending to -ic, adding the prefix hydro- to this anion name, and then following with the word acid:

2. Acids containing anions whose names end in -ate or -ite are named by changing -ate to -ic and -ite to -ous and then adding the word acid. Prefixes in the anion name are retained in the name of the acid:
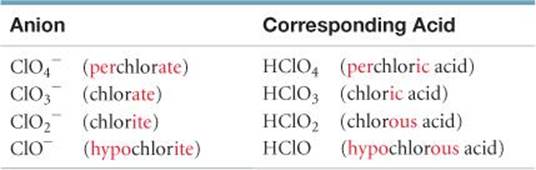
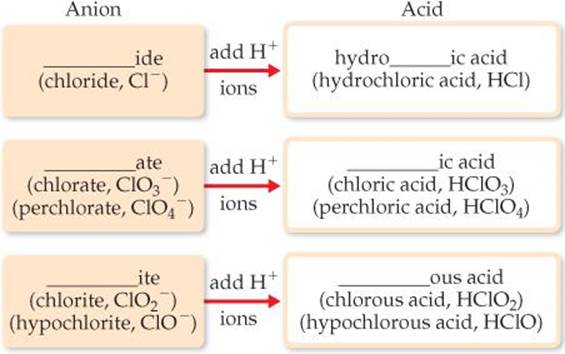
![]() Figure 2.26 How anion names and acid names relate. The prefixes per- and hypo- are retained in going from the anion to the acid.
Figure 2.26 How anion names and acid names relate. The prefixes per- and hypo- are retained in going from the anion to the acid.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Name the acid obtained by adding H+ to the iodate ion, IO3–.
SAMPLE EXERCISE 2.14 Relating the Names and Formulas of Acids
Name the acids (a) HCN, (b) HNO3, (c) H2SO4, (d) H2SO3.
SOLUTION
(a) The anion from which this acid is derived is CN–, the cyanide ion. Because this ion has an -ide ending, the acid is given a hydro- prefix and an -ic ending: hydrocyanic acid. Only water solutions of HCN are referred to as hydrocyanic acid. The pure compound, which is a gas under normal conditions, is called hydrogen cyanide. Both hydrocyanic acid and hydrogen cyanide are extremely toxic.
(b) Because NO3– is the nitrate ion, HNO3 is called nitric acid (the -ate ending of the anion is replaced with an -ic ending in naming the acid).
(c) Because SO42– is the sulfate ion, H2SO4 is called sulfuric acid.
(d) Because SO32– is the sulfite ion, H2SO3 is sulfurous acid (the -ite ending of the anion is replaced with an -ous ending).
PRACTICE EXERCISE
Give the chemical formulas for (a) hydrobromic acid, (b) carbonic acid.
Answers: (a) HBr, (b) H2CO3
Names and Formulas of Binary Molecular Compounds
The procedures used for naming binary (two-element) molecular compounds are similar to those used for naming ionic compounds:
1. The name of the element farther to the left in the periodic table (closest to the metals) is usually written first. An exception occurs when the compound contains oxygen and chlorine, bromine, or iodine (any halogen except fluorine), in which case oxygen is written last.
2. If both elements are in the same group, the lower one is named first.
3. The name of the second element is given an -ide ending.
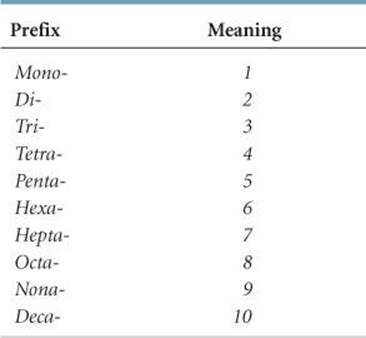
4. Greek prefixes (![]() TABLE 2.6) are used to indicate the number of atoms of each element. The prefix mono- is never used with the first element. When the prefix ends in a or o and the name of the second element begins with a vowel, the a or o of the prefix is often dropped.
TABLE 2.6) are used to indicate the number of atoms of each element. The prefix mono- is never used with the first element. When the prefix ends in a or o and the name of the second element begins with a vowel, the a or o of the prefix is often dropped.
The following examples illustrate these rules:

TABLE 2.6 • Prefixes Used in Naming Binary Compounds Formed between Nonmetals
Rule 4 is necessary because we cannot predict formulas for most molecular substances the way we can for ionic compounds. Molecular compounds that contain hydrogen and one other element are an important exception, however. These compounds can be treated as if they were neutral substances containing H+ ions and anions. Thus, you can predict that the substance named hydrogen chloride has the formula HCl, containing one H+ to balance the charge of one Cl–. (The name hydrogen chloride is used only for the pure compound; water solutions of HCl are called hydrochloric acid.) Similarly, the formula for hydrogen sulfide is H2S because two H+ are needed to balance the charge on S2–.
SAMPLE EXERCISE 2.15 Relating the Names and Formulas of Binary Molecular Compounds
Name the compounds (a) SO2, (b) PCl5, (c) Cl2O3.
SOLUTION
The compounds consist entirely of nonmetals, so they are molecular rather than ionic. Using the prefixes in Table 2.6, we have (a) sulfur dioxide, (b) phosphorus pentachloride, and (c) dichlorine trioxide.
PRACTICE EXERCISE
Give the chemical formulas for (a) silicon tetrabromide, (b) disulfur dichloride.
Answers: (a) SiBr4, (b) S2Cl2