CHEMISTRY THE CENTRAL SCIENCE
22 CHEMISTRY OF THE NONMETALS
22.3 GROUP 8A: THE NOBLE GASES
The elements of group 8A are chemically unreactive. Indeed, most of our references to these elements have been in relation to their physical properties, as when we discussed intermolecular forces. ![]() (Section 11.2) The relative inertness of these elements is due to the presence of a completed octet of valence-shell electrons (except He, which only has a filled 1s shell). The stability of such an arrangement is reflected in the high ionization energies of the group 8A elements.
(Section 11.2) The relative inertness of these elements is due to the presence of a completed octet of valence-shell electrons (except He, which only has a filled 1s shell). The stability of such an arrangement is reflected in the high ionization energies of the group 8A elements. ![]() (Section 7.4)
(Section 7.4)
The group 8A elements are all gases at room temperature. They are components of Earth's atmosphere, except for radon, which exists only as a short-lived radioisotope. ![]() (Section 21.9) Only argon is relatively abundant.
(Section 21.9) Only argon is relatively abundant. ![]() (Table 18.1) Neon, argon, krypton, and xenon are recovered from liquid air by distillation. All four of these noble gases are used in lighting, display, and laser applications in which the atoms are excited electrically and electrons that are in a higher energy state emit light as they fall to the ground state.
(Table 18.1) Neon, argon, krypton, and xenon are recovered from liquid air by distillation. All four of these noble gases are used in lighting, display, and laser applications in which the atoms are excited electrically and electrons that are in a higher energy state emit light as they fall to the ground state. ![]() (Section 6.2) Argon is used as a blanketing atmosphere in electric lightbulbs. The gas conducts heat away from the filament but does not react with it. Argon is also used as a protective atmosphere to prevent oxidation in welding and certain high-temperature metallurgical processes.
(Section 6.2) Argon is used as a blanketing atmosphere in electric lightbulbs. The gas conducts heat away from the filament but does not react with it. Argon is also used as a protective atmosphere to prevent oxidation in welding and certain high-temperature metallurgical processes.
Helium is in many ways the most important noble gas. Liquid helium is used as a coolant to conduct experiments at very low temperatures. Helium boils at 4.2 K and 1 atm, the lowest boiling point of any substance. It is found in relatively high concentrations in many natural-gas wells and can be obtained from them.
Noble-Gas Compounds
Because the noble gases are exceedingly stable, they react only under rigorous conditions. We expect the heavier ones to be most likely to form compounds because their ionization energies are lower. ![]() (Figure 7.9) A lower ionization energy suggests the possibility of sharing an electron with another atom, leading to a chemical bond. In addition, because the group 8A elements (except helium) already contain eight electrons in their valence shell, formation of covalent bonds will require an expanded valence shell. Valence-shell expansion occurs most readily with larger atoms.
(Figure 7.9) A lower ionization energy suggests the possibility of sharing an electron with another atom, leading to a chemical bond. In addition, because the group 8A elements (except helium) already contain eight electrons in their valence shell, formation of covalent bonds will require an expanded valence shell. Valence-shell expansion occurs most readily with larger atoms. ![]() (Section 8.7)
(Section 8.7)
The first noble-gas compound was reported in 1962. This discovery caused a sensation because it undercut the belief that the noble-gas elements were inert. The initial study involved xenon in combination with fluorine, the element we would expect to be most reactive in pulling electron density from another atom. Since that time chemists have prepared several xenon compounds of fluorine and oxygen (![]() TABLE 22.1). The fluorides XeF2, XeF4, and XeF6 are made by direct reaction of the elements. By varying the ratio of reactants and altering reaction conditions, one of the three compounds can be obtained. The oxygen-containing compounds are formed when the fluorides react with water as, for example,
TABLE 22.1). The fluorides XeF2, XeF4, and XeF6 are made by direct reaction of the elements. By varying the ratio of reactants and altering reaction conditions, one of the three compounds can be obtained. The oxygen-containing compounds are formed when the fluorides react with water as, for example,
![]()
SAMPLE EXERCISE 22.3 Predicting a Molecular Structure
Use the VSEPR model to predict the structure of XeF4.
SOLUTION
Analyze We must predict the geometrical structure given only the molecular formula.
Plan We must first write the Lewis structure for the molecule. We then count the number of electron pairs (domains) around the Xe atom and use that number and the number of bonds to predict the geometry.
Solve There are 36 valence-shell electrons (8 from xenon and 7 from each fluorine). If we make four single Xe—F bonds, each fluorine has its octet satisfied. Xe then has 12 electrons in its valence shell, so we expect an octahedral arrangement of six electron pairs. Two of these are nonbonded pairs. Because nonbonded pairs require more volume than bonded pairs ![]() (Section 9.2), it is reasonable to expect these nonbonded pairs to be opposite each other. The expected structure is square planar, as shown in
(Section 9.2), it is reasonable to expect these nonbonded pairs to be opposite each other. The expected structure is square planar, as shown in ![]() FIGURE 22.7.
FIGURE 22.7.
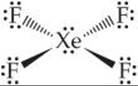
![]() FIGURE 22.7 Xenon tetrafluoride.
FIGURE 22.7 Xenon tetrafluoride.
Comment The experimentally determined structure agrees with this prediction.
PRACTICE EXERCISE
Describe the electron-domain geometry and molecular geometry of XeF2.
Answer: trigonal bipyramidal, linear
The other noble-gas elements form compounds much less readily than xenon. For many years, only one binary krypton compound, KrF2, was known with certainty, and it decomposes to its elements at –10 °C. Other compounds of krypton have been isolated at very low temperatures (40 K).
TABLE 22.1 • Properties of Xenon Compounds