CHEMISTRY THE CENTRAL SCIENCE
23 TRANSITION METALS AND COORDINATION CHEMISTRY
23.3 COMMON LIGANDS IN COORDINATION CHEMISTRY
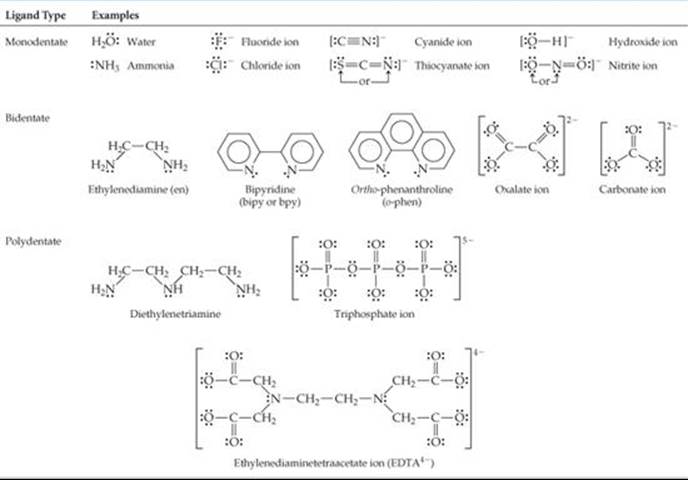
The ligand atom that binds to the central metal ion in a coordination complex is called the donor atom of the ligand. Ligands having only one donor atom are called monodentate ligands (from the Latin, meaning “one-toothed”). These ligands are able to occupy only one site in a coordination sphere. Ligands having two donor atoms are bidentate ligands (“two-toothed”), and those having three or more donor atoms are polydentate ligands (“many-toothed”). In both bidentate and polydentate species, the multiple donor atoms can simultaneously bond to the metal ion, thereby occupying two or more sites in a coordination sphere. ![]() TABLE 23.4 gives examples of all three types of ligands.
TABLE 23.4 gives examples of all three types of ligands.
Because they appear to grasp the metal between two or more donor atoms, bidentate and polydentate ligands are also known as chelating agents (pronounced “KEE-lay-ting”; from the Greek chele, “claw”).
TABLE 23.4 • Some Common Ligands
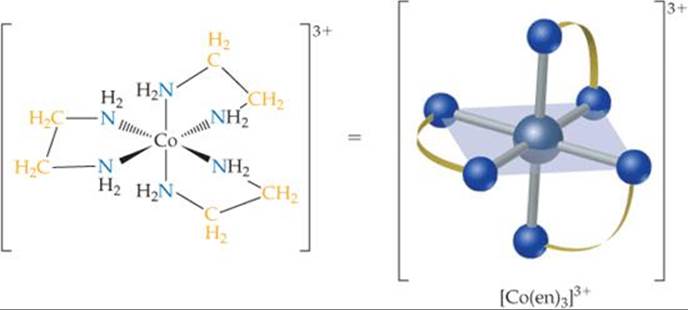
![]() FIGURE 23.11 The [Co(en)3]3+ ion. The ligand is ethylenediamine.
FIGURE 23.11 The [Co(en)3]3+ ion. The ligand is ethylenediamine.
One common chelating agent is the bidentate ligand ethylenediamine, en:
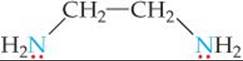
in which each donor nitrogen atom has one nonbonding electron pair. These donor atoms are sufficiently far apart to allow both of them to bond to the metal ion in adjacent positions. The [Co(en)3]3+ complex ion, which contains three ethylenediamine ligands in the octahedral coordination sphere of cobalt(III), is shown in ![]() FIGURE 23.11. Notice that in the image on the right the en is written in a shorthand notation as two nitrogen atoms connected by an arc.
FIGURE 23.11. Notice that in the image on the right the en is written in a shorthand notation as two nitrogen atoms connected by an arc.
The ethylenediaminetetraacetate ion, [EDTA]4–, is an important polydentate ligand that has six donor atoms. It can wrap around a metal ion using all six donor atoms, as shown in ![]() FIGURE 23.12, although it sometimes binds to a metal using only five of its donor atoms.
FIGURE 23.12, although it sometimes binds to a metal using only five of its donor atoms.
In general, the complexes formed by chelating ligands (that is, bidentate and polydentate ligands) are more stable than the complexes formed by related monodentate ligands. The equilibrium formation constants for [Ni(NH3)6]2+ and [Ni(en)3]2+ illustrate this observation:
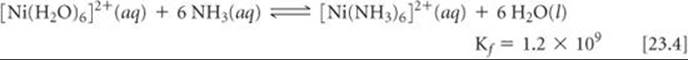
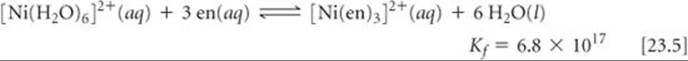
Although the donor atom is nitrogen in both instances, [Ni(en)3]2+ has a formation constant that is more than 108 times larger than that of [Ni(NH3)6]2+. This trend of generally larger formation constants for bidentate and polydentate ligands, known as the chelate effect, is examined in the “A Closer Look” essay on page 977.
Chelating agents are often used to prevent one or more of the customary reactions of a metal ion without removing the ion from solution. For example, a metal ion that interferes with a chemical analysis can often be complexed and its interference thereby removed. In a sense, the chelating agent hides the metal ion. For this reason, scientists sometimes refer to these ligands as sequestering agents.
Phosphate ligands, such as sodium tripolyphosphate, Na5[OPO2OPO2OPO3], are used to sequester Ca2+ and Mg2+ ions in hard water so that these ions cannot interfere with the action of soap or detergents.
Chelating agents are used in many prepared foods, such as salad dressings and frozen desserts, to complex trace metal ions that catalyze decomposition reactions. Chelating agents are used in medicine to remove toxic heavy metal ions that have been ingested, such as Hg2+, Pb2+, and Cd2+. One method of treating lead poisoning, for example, is to administer Na2[Ca(EDTA)]. The EDTA chelates the lead, allowing it to be removed from the body via urine.
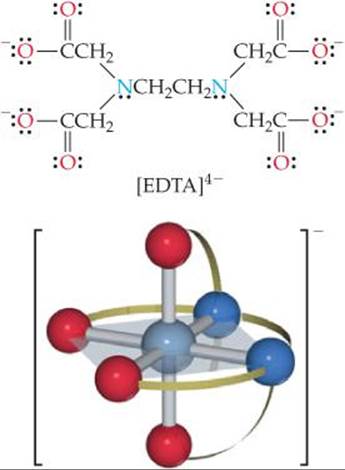
![]() FIGURE 23.12 The EDTA4– ligand (top) and the complex ion [Co(EDTA)]–(bottom). The ligand is the polydentate ethylenediaminetetraacetate ion, which has six donor atoms, two N and four O.
FIGURE 23.12 The EDTA4– ligand (top) and the complex ion [Co(EDTA)]–(bottom). The ligand is the polydentate ethylenediaminetetraacetate ion, which has six donor atoms, two N and four O.
![]() GO FIGURE
GO FIGURE
What is the coordination number of the metal ion in heme b? In chlorophyll a?
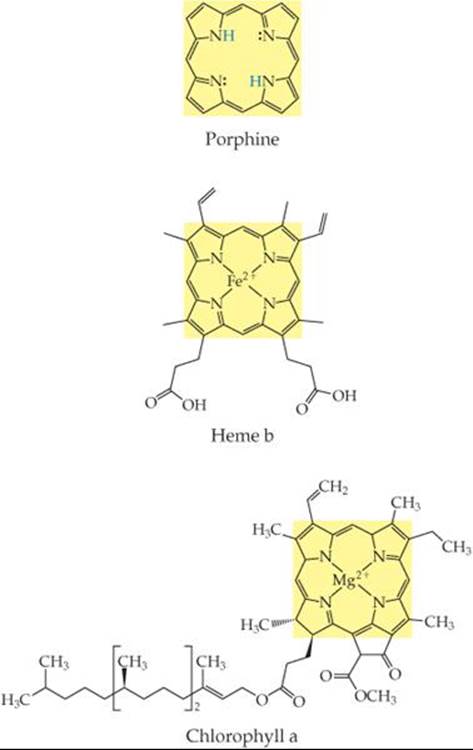
![]() FIGURE 23.13 Porphine and two porphyrins, heme b and chlorophyll a. Fe(II) and Mg(II) ions replace the two blue H atoms in porphine and bond with all four nitrogens in heme b and chlorophyll a, respectively.
FIGURE 23.13 Porphine and two porphyrins, heme b and chlorophyll a. Fe(II) and Mg(II) ions replace the two blue H atoms in porphine and bond with all four nitrogens in heme b and chlorophyll a, respectively.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Cobalt(III) has a coordination number of 6 in all its complexes. Is the carbonate ion a monodentate or bidentate ligand in the [Co(NH3)4CO3]+ ion?
Metals and Chelates in Living Systems
Ten of the 29 elements known to be necessary for human life are transition metals. ![]() (Section 2.7, “Chemistry and Life: Elements Required by Living Organisms”) These ten elements—V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Mo, and Cd—form complexes with a variety of groups present in biological systems.
(Section 2.7, “Chemistry and Life: Elements Required by Living Organisms”) These ten elements—V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Mo, and Cd—form complexes with a variety of groups present in biological systems.
Although our bodies require only small quantities of metals, deficiencies can lead to serious illness. A deficiency of manganese, for example, can lead to convulsive disorders. Some epilepsy patients have been helped by the addition of manganese to their diets.
Among the most important chelating agents in nature are those derived from the porphine molecule (![]() FIGURE 23.13). This molecule can coordinate to a metal via its four nitrogen donor atoms. Once porphine bonds to a metal ion, the two H atoms on the nitrogens are displaced to form complexes called porphyrins. Two important porphyrins are hemes, in which the metal ion is Fe(II), and chlorophylls, with a Mg(II) central ion.
FIGURE 23.13). This molecule can coordinate to a metal via its four nitrogen donor atoms. Once porphine bonds to a metal ion, the two H atoms on the nitrogens are displaced to form complexes called porphyrins. Two important porphyrins are hemes, in which the metal ion is Fe(II), and chlorophylls, with a Mg(II) central ion.
![]() FIGURE 23.14 shows a schematic structure of myoglobin, a protein that contains one heme group. Myoglobin is a globular protein, one that folds into a compact, roughly spherical shape. Myoglobin is found in the cells of skeletal muscle, particularly in seals, whales, and porpoises. It stores oxygen in cells, one molecule of O2 per myoglobin, until it is needed for metabolic activities. Hemoglobin, the protein that transports oxygen in human blood, is made up of four heme-con-taining subunits, each of which is very similar to myoglobin. One hemoglobin can bind up to four O2 molecules.
FIGURE 23.14 shows a schematic structure of myoglobin, a protein that contains one heme group. Myoglobin is a globular protein, one that folds into a compact, roughly spherical shape. Myoglobin is found in the cells of skeletal muscle, particularly in seals, whales, and porpoises. It stores oxygen in cells, one molecule of O2 per myoglobin, until it is needed for metabolic activities. Hemoglobin, the protein that transports oxygen in human blood, is made up of four heme-con-taining subunits, each of which is very similar to myoglobin. One hemoglobin can bind up to four O2 molecules.
In both myoglobin and hemoglobin, the iron is coordinated to the four nitrogen atoms of a porphyrin and to a nitrogen atom from the protein chain (![]() FIGURE 23.15). In hemoglobin, the sixth position around the iron is occupied either by O2 (in oxyhemoglobin, the bright red form) or by water (in deoxyhemoglobin, the purplish red form). (The oxy form is the one shown in Figure 23.15.)
FIGURE 23.15). In hemoglobin, the sixth position around the iron is occupied either by O2 (in oxyhemoglobin, the bright red form) or by water (in deoxyhemoglobin, the purplish red form). (The oxy form is the one shown in Figure 23.15.)
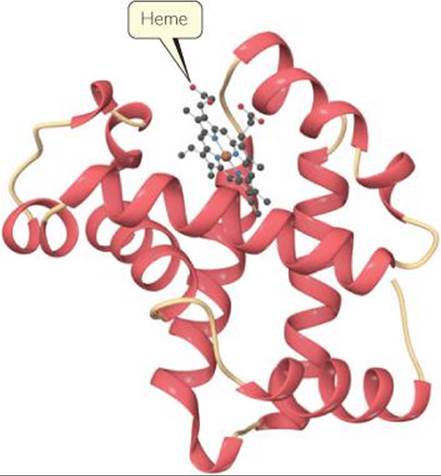
![]() FIGURE 23.14 Myoglobin. This ribbon diagram does not show most of the atoms.
FIGURE 23.14 Myoglobin. This ribbon diagram does not show most of the atoms.
Carbon monoxide is poisonous because the equilibrium binding constant of human hemoglobin for CO is about 210 times greater than that for O2. As a result, a relatively small quantity of CO can inactivate a substantial fraction of the hemoglobin in the blood. For example, a person breathing air that contains only 0.1% CO takes in enough CO after a few hours to convert up to 60% of the hemoglobin (Hb) into COHb, thereby reducing the blood's normal oxygen-carrying capacity by 60%.
![]() GO FIGURE
GO FIGURE
Where would CO bind in this molecule?
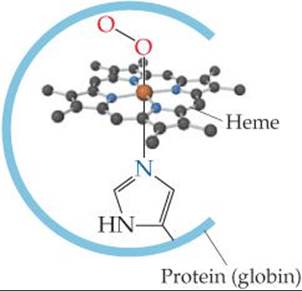
![]() FIGURE 23.15 Coordination sphere of the hemes in oxymyoglobin and oxyhemoglobin.
FIGURE 23.15 Coordination sphere of the hemes in oxymyoglobin and oxyhemoglobin.
Under normal conditions, a nonsmoker breathing unpolluted air has about 0.3 to 0.5% COHb in her or his blood. This amount arises mainly from the production of small quantities of CO in the course of normal body chemistry and from the small amount of CO present in clean air. Exposure to higher concentrations of CO causes the COHb level to increase, which in turn leaves fewer Hb sites to which O2 can bind. If the level of COHb becomes too high, oxygen transport is effectively shut down and death occurs. Because CO is colorless and odorless, CO poisoning occurs with very little warning. Improperly ventilated combustion devices, such as kerosene lanterns and stoves, thus pose a potential health hazard.
 A CLOSER LOOK
A CLOSER LOOK
ENTROPY AND THE CHELATE EFFECT
We learned in Section 19.5 that chemical processes are favored by positive entropy changes and by negative enthalpy changes. The special stability associated with the formation of chelates, called the chelate effect, can be explained by comparing the entropy changes that occur with monodentate ligands with the entropy changes that occur with polydentate ligands.
We begin with the reaction in which two H2O ligands of the square-planar Cu(II) complex [Cu(H2O)4]2+ are replaced by monodentate NH3 ligands at 27 °C:
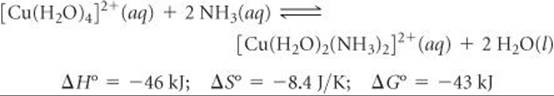
The thermodynamic data tell us about the relative abilities of H2O and NH3 to serve as ligands in this reaction. In general, NH3 binds more tightly to metal ions than does H2O, so this substitution reaction is exothermic (ΔH < 0). The stronger bonding of the NH3 ligands also causes the [Cu(H2O)2(NH3)2]2+ ion to be more rigid, which is probably the reason ΔS° is slightly negative.
We can use Equation 19.20, ΔG° = –RT ln K, to calculate the equilibrium constant of the reaction at 27 °C. The result, K = 3.1 × 107, tells us that the equilibrium lies far to the right, favoring replacement of H2O by NH3. For this equilibrium, therefore, the enthalpy change, ΔH° = –46 kJ, is large enough and negative enough to overcome the entropy change, ΔS° = –8.4 J/K.
Now let's use a single bidentate ethylenediamine ligand in our substitution reaction:
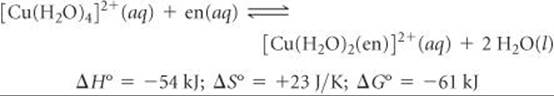
The en ligand binds slightly more strongly to the Cu2+ ion than two NH3 ligands, so the enthalpy change here (–54 kJ) is slightly more negative than for [Cu(H2O)2(NH3)2]2+ (–46 kJ). There is a big difference in the entropy change, however: ΔS° is –8.4 J/K for the NH3 reaction but +23 J/K for the en reaction. We can explain the positive ΔS° value by using concepts discussed in Section 19.3. Because a single en ligand occupies two coordination sites, two molecules of H2O are released when one en ligand bonds. Thus, there are three product molecules in the reaction but only two reactant molecules. The greater number of product molecules leads to the positive entropy change for the equilibrium.
The slightly more negative value of ΔH° for the en reaction (–54 kJ versus –46 kJ) coupled with the positive entropy change leads to a much more negative value of ΔG° (–61 kJ for en, –43 for NH3) and a correspondingly larger equilibrium constant: K = 4.2 × 1010.
We can combine our two equations using Hess's law ![]() (Section 5.6) to calculate the enthalpy, entropy, and free-energy changes that occur for en to replace ammonia as ligands on Cu(II):
(Section 5.6) to calculate the enthalpy, entropy, and free-energy changes that occur for en to replace ammonia as ligands on Cu(II):
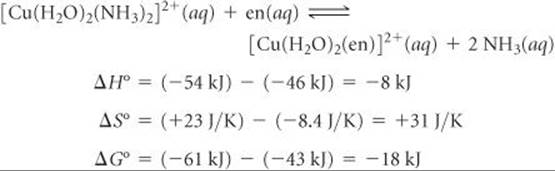
Notice that at 27 °C, the entropic contribution (–TΔS°) to the free-energy change, ΔG° = ΔH° – TΔS° (Equation 19.12), is negative and greater in magnitude than the enthalpic contribution (ΔH°). The equilibrium constant for the NH3–en reaction, 1.4 × 103, shows that the replacement of NH3 by en is thermodynamically favorable.
The chelate effect is important in biochemistry and molecular biology. The additional thermodynamic stabilization provided by entropy effects helps stabilize biological metal–chelate complexes, such as porphyrins, and can allow changes in the oxidation state of the metal ion while retaining the structural integrity of the complex.
RELATED EXERCISES: 23.31, 23.32, 23.33, 23.98
The chlorophylls, which are porphyrins that contain Mg(II) (Figure 23.13), are the key components in the conversion of solar energy into forms that can be used by living organisms. This process, called photosynthesis, occurs in the leaves of green plants:
![]()
The formation of 1 mol of glucose, C6H12O6, requires the absorption of 48 mol of photons from sunlight or other sources of light. Chlorophyll-containing pigments in the leaves of plants absorb the photons. Figure 23.13 shows that the chlorophyll molecule has a series of alternating, orconjugated, double bonds in the ring surrounding the metal ion. This system of conjugated double bonds makes it possible for chlorophyll to absorb light strongly in the visible region of the spectrum. As ![]() FIGURE 23.16 shows, chlorophyll is green because it absorbs red light (maximum absorption at 655 nm) and blue light (maximum absorption at 430 nm) and transmits green light.
FIGURE 23.16 shows, chlorophyll is green because it absorbs red light (maximum absorption at 655 nm) and blue light (maximum absorption at 430 nm) and transmits green light.
Photosynthesis is nature's solar-energy–conversion machine, and thus all living systems on Earth depend on photosynthesis for continued existence.
![]() GO FIGURE
GO FIGURE
Which peak in this curve corresponds to the lowest-energy transition by an electron in a chlorophyll molecule?
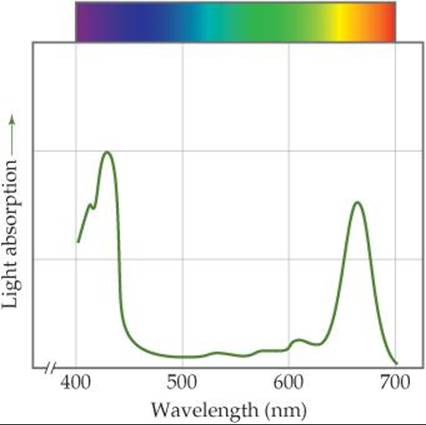
![]() FIGURE 23.16 The absorption of sunlight by chlorophyll.
FIGURE 23.16 The absorption of sunlight by chlorophyll.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What property of the porphine ligand makes it possible for chlorophyll to play a role in plant photosynthesis?
 CHEMISTRY AND LIFE
CHEMISTRY AND LIFE
THE BATTLE FOR IRON IN LIVING SYSTEMS
Because living systems have difficulty assimilating enough iron to satisfy their nutritional needs, irondeficiency anemia is a common problem in humans. Chlorosis, an iron deficiency in plants that makes leaves turn yellow, is also commonplace.
Living systems have difficulty assimilating iron because most iron compounds found in nature are not very soluble in water. Microorganisms have adapted to this problem by secreting an iron-binding compound, called a siderophore, that forms an extremely stable water-soluble complex with iron(III). One such complex is ferrichrome (![]() FIGURE 23.17). The iron-binding strength of a siderophore is so great that it can extract iron from glass cooking pots and the iron in iron oxides.
FIGURE 23.17). The iron-binding strength of a siderophore is so great that it can extract iron from glass cooking pots and the iron in iron oxides.
When ferrichrome enters a living cell, the iron it carries is removed through an enzyme-catalyzed reaction that reduces the strongly bonding iron(III) to iron(II), which is only weakly com-plexed by the siderophore (![]() FIGURE 23.18). Microorganisms thus acquire iron by excreting a siderophore into their immediate environment and then taking the resulting iron complex into the cell.
FIGURE 23.18). Microorganisms thus acquire iron by excreting a siderophore into their immediate environment and then taking the resulting iron complex into the cell.
In humans, iron is assimilated from food in the intestine. A protein called transferrin binds iron and transports it across the intestinal wall to distribute it to other tissues in the body. The normal adult body contains about 4 g of iron. At any one time, about 3 g of this iron is in the blood, mostly in the form of hemoglobin. Most of the remainder is carried by transferrin.
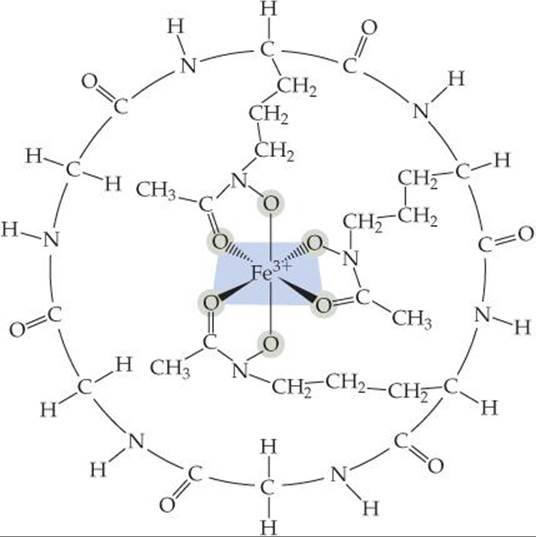
![]() FIGURE 23.17 Ferrichrome.
FIGURE 23.17 Ferrichrome.
A bacterium that infects the blood requires a source of iron if it is to grow and reproduce. The bacterium excretes a siderophore into the blood to compete with transferrin for iron. The equilibrium constants for forming the iron complex are about the same for transferrin and siderophores. The more iron available to the bacterium, the more rapidly it can reproduce and thus the more harm it can do.
Several years ago, New Zealand clinics regularly gave iron supplements to infants soon after birth. However, the incidence of certain bacterial infections was eight times higher in treated than in untreated infants. Presumably, the presence of more iron in the blood than absolutely necessary makes it easier for bacteria to obtain the iron needed for growth and reproduction.
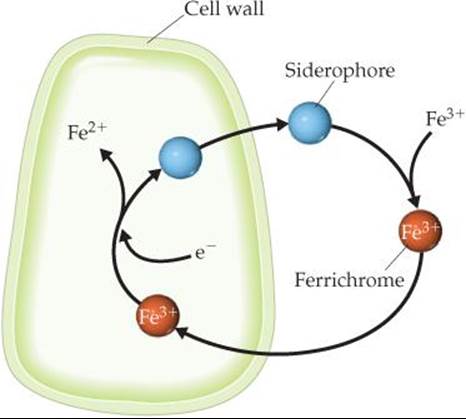
![]() FIGURE 23.18 The iron-transport system of a bacterial cell.
FIGURE 23.18 The iron-transport system of a bacterial cell.
In the United States it is common medical practice to supplement infant formula with iron sometime during the first year of life. However, iron supplements are not necessary for infants who breastfeed because breast milk contains two specialized proteins, lactoferrin and transferrin, which provide sufficient iron while denying its availability to bacteria. Even for infants fed with infant formulas, supplementing with iron during the first several months of life may be ill-advised.
For bacteria to continue to multiply in the blood, they must synthesize new supplies of siderophores. Synthesis of siderophores in bacteria slows, however, as the temperature is increased above the normal body temperature of 37 °C and stops completely at 40 °C. This suggests that fever in the presence of an invading microbe is a mechanism used by the body to deprive bacteria of iron.
RELATED EXERCISE: 23.74