MCAT General Chemistry Review
Chapter 7: Thermochemistry
7.6 Gibbs Free Energy
The final state function that we will examine in this chapter is Gibbs free energy, G. This state function is a combination of the three that we’ve just examined: enthalpy, temperature, and entropy. The change in Gibbs free energy, ΔG, is a measure of the change in the enthalpy and the change in entropy as a system undergoes a process, and it indicates whether a reaction is spontaneous or nonspontaneous. The change in the free energy is the maximum amount of energy released by a process—occurring at constant temperature and pressure—that is available to perform useful work. The change in Gibbs free energy is defined as follows:
ΔG = ΔH – TΔS
Equation 7.10
where T is the temperature in kelvin and TΔS represents the total amount of energy that is absorbed by a system when its entropy increases reversibly.
MNEMONIC
Gibbs free energy: ΔG = ΔH – TΔS. Goldfish are (equals sign) Horrible without (minus sign) Tartar Sauce.
OVERVIEW
A helpful visual aid for conceptualizing Gibbs free energy is to think of it as a valley between two hills. Just as a ball would tend to roll down the hill into the valley and eventually come to rest at the lowest point in the valley, any system—including chemical reactions—will move in whichever direction results in a reduction of the free energy of the system. The bottom of the valley represents equilibrium, and the sides of the hill represent the various points in the pathway toward or away from equilibrium. This is shown graphically in Figure 7.11, which was also discussed in the previous chapter.
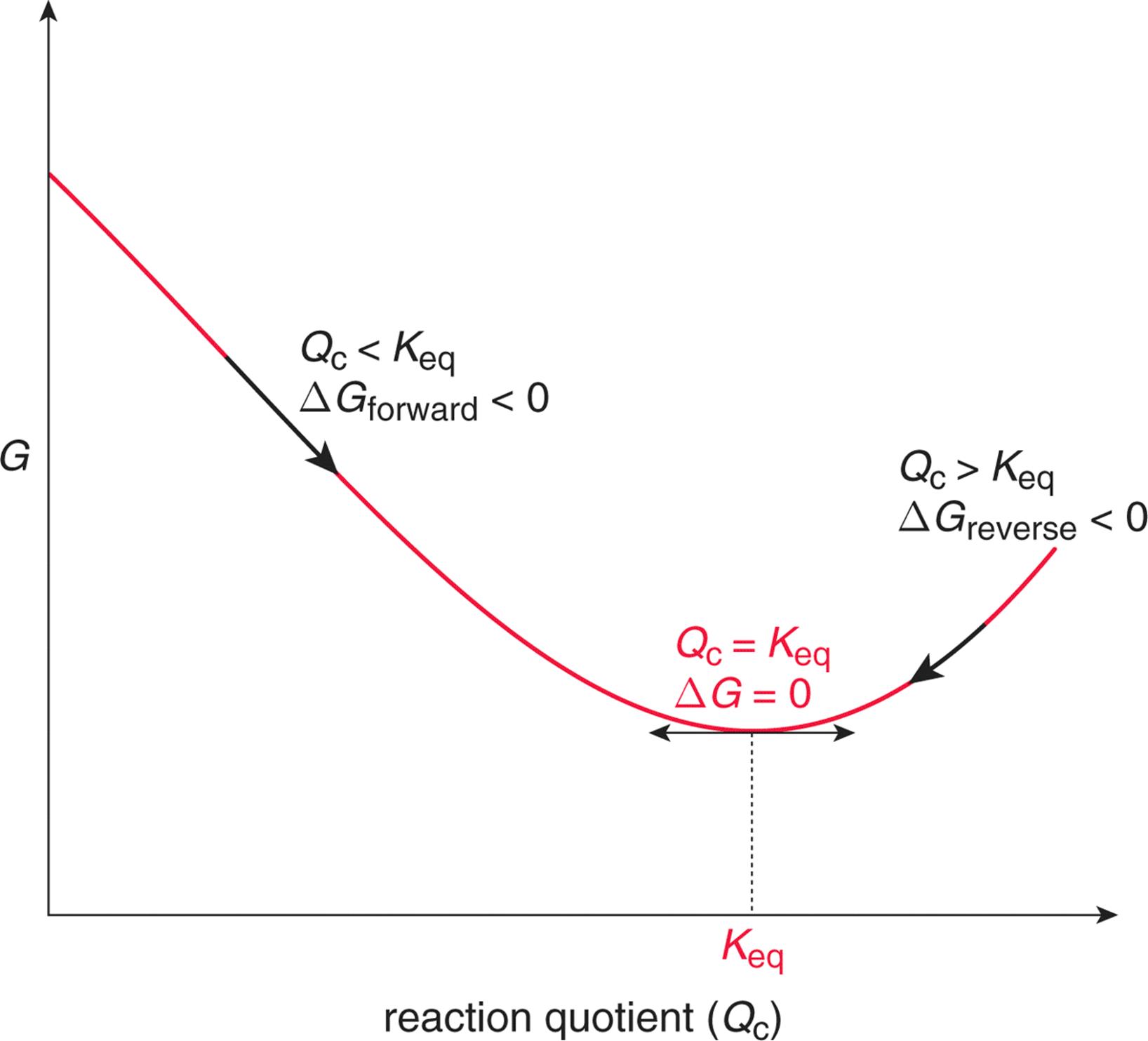 Figure 7.11. Gibbs Free Energy and Spontaneity A decrease in Gibbs free energy indicates that a reaction is spontaneous. When an equilibrated system is disturbed, it will spontaneously act to restore equilibrium.
Figure 7.11. Gibbs Free Energy and Spontaneity A decrease in Gibbs free energy indicates that a reaction is spontaneous. When an equilibrated system is disturbed, it will spontaneously act to restore equilibrium.
Movement toward the equilibrium position is associated with a decrease in Gibbs free energy (ΔG < 0) and is spontaneous. When a system releases energy, it is said to be exergonic, as shown in Figure 7.12.
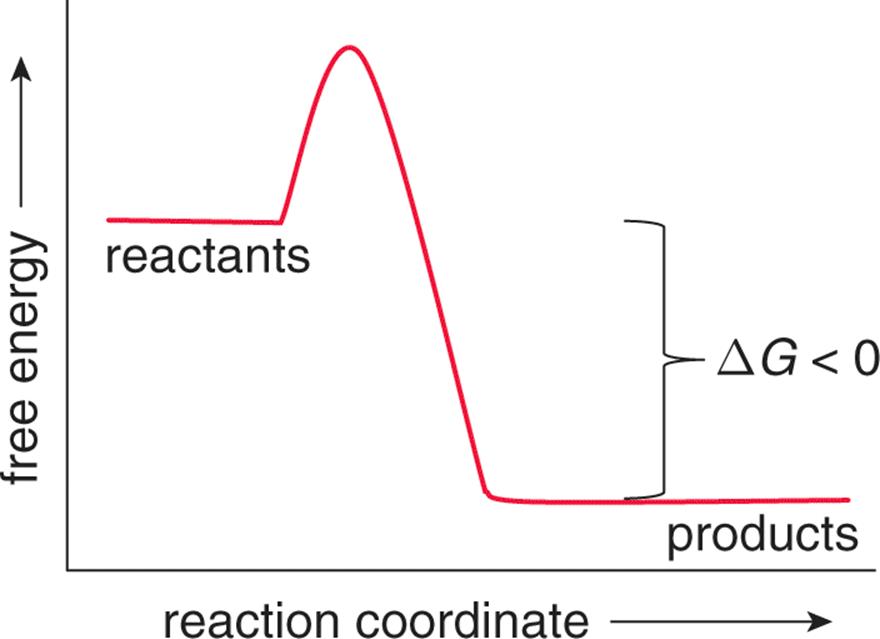 Figure 7.12. Exergonic Reaction Profile Exergonic reactions release energy and are spontaneous (ΔGrxn < 0).
Figure 7.12. Exergonic Reaction Profile Exergonic reactions release energy and are spontaneous (ΔGrxn < 0).
MCAT EXPERTISE
Be careful not to confuse endergonic/exergonic (describing Gibbs free energy) with endothermic/exothermic (describing enthalpy).
On the other hand, movement away from the equilibrium position is associated with an increase in Gibbs free energy (ΔG > 0) and is nonspontaneous. Such a reaction is said to be endergonic, as shown in Figure 7.13.
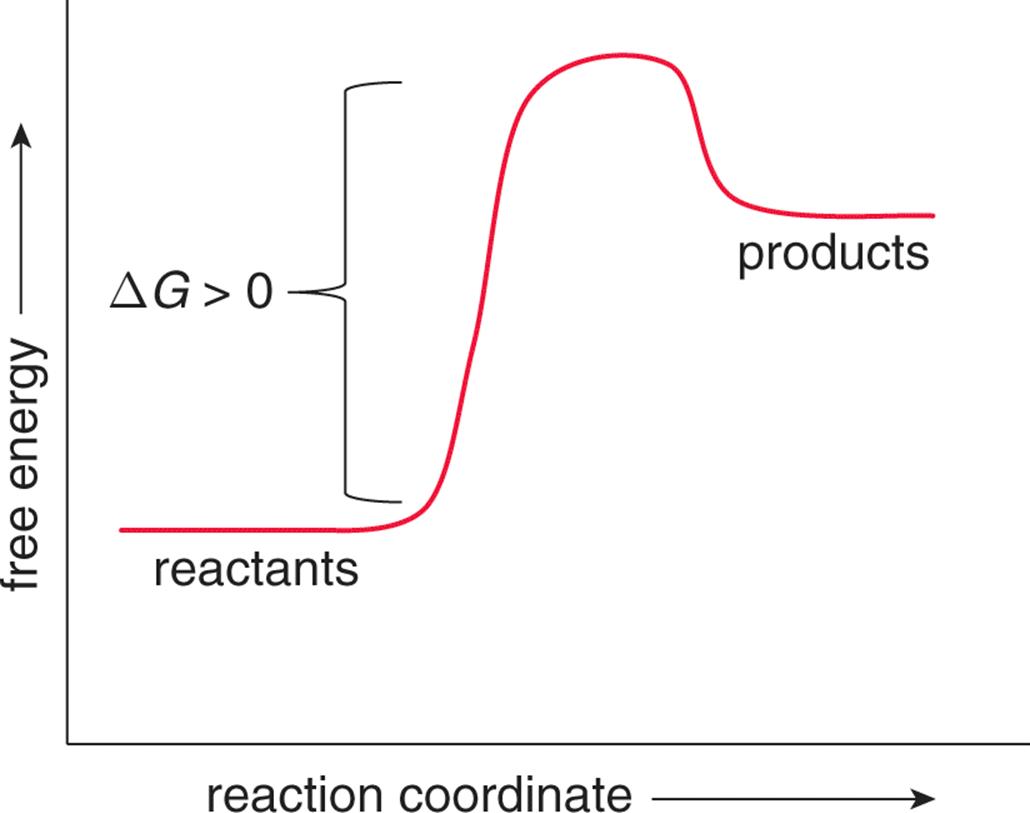 Figure 7.13. Endergonic Reaction Profile Endergonic reactions absorb energy and are nonspontaneous (ΔGrxn > 0).
Figure 7.13. Endergonic Reaction Profile Endergonic reactions absorb energy and are nonspontaneous (ΔGrxn > 0).
Once at the energy minimum state—equilibrium—the system will resist any changes to its state, and the change in free energy is zero. To summarize:
1. If ΔG is negative, the reaction is spontaneous.
2. If ΔG is positive, the reaction is nonspontaneous.
3. If ΔG is zero, the system is in a state of equilibrium; ΔH = TΔS.
You should recall that phase equilibria are states in which more than one phase exists. As with all equilibria, the change in Gibbs free energy must be equal to zero (ΔG = 0). For an equilibrium between a gas and a solid,
ΔG = G (g) – G (s) = 0
Therefore,
G (g) = G (s)
KEY CONCEPT
Recall that thermodynamics and kinetics are separate topics. When a reaction is thermodynamically spontaneous, it has no bearing on how fast it goes. It only means that it will proceed eventually without external energy input.
Because the temperature in Gibbs free energy is in kelvin, it is always positive. Therefore, the effects of the signs of ΔH and ΔS on the spontaneity of a process can be summarized as in Table 7.2.
|
ΔH |
ΔS |
Outcome |
|
+ |
+ |
Spontaneous at high T |
|
+ |
− |
Nonspontaneous at all T |
|
− |
+ |
Spontaneous at all T |
|
− |
− |
Spontaneous at low T |
|
Table 7.2 Effects of ΔH, ΔS, and T on Spontaneity |
||
KEY CONCEPT
ΔG is temperature dependent when ΔH and ΔS have the same sign.
Phase changes are examples of temperature-dependent processes. The phase changes of water should be familiar to you; have you ever wondered why water doesn’t boil at, say, 20°C instead of 100°C? When water boils, hydrogen bonds are broken, and the water molecules gain sufficient energy to escape into the gas phase. Thus, boiling is an endothermic process, and ΔH is positive. As thermal energy is transferred to the water molecules, energy is distributed through the molecules entering the gas phase. Thus, entropy is positive and the term TΔS is positive. If both ΔH andTΔS are positive, the reaction will only be spontaneous if TΔS is greater than ΔH, resulting in a negative ΔG. These conditions are met only when the temperature of the system is greater than 373 K (100°C). Below 100°C, the free energy change is positive, and boiling is nonspontaneous; the water remains a liquid. At 100°C, ΔH – TΔS = 0, and an equilibrium is established between the liquid and gas phases in such a way that the water’s vapor pressure equals the ambient pressure. This is the definition of the boiling point: the temperature at which the vapor pressure equals the ambient pressure.
It is important to remember that the rate of a reaction depends on the activation energy Ea, not ΔG. Spontaneous reactions may be fast or slow. Sometimes a reversible reaction may produce two products that differ both in their stability (as measured by the change in the free energy associated with their production) and in their kinetics (as measured by their respective activation energies). Sometimes, the thermodynamically more stable product will have the slower kinetics due to higher activation energy. In this situation, we talk about kinetic vs. thermodynamic reaction control, which is discussed in Chapter 6 of MCAT General Chemistry Review. For a period of time after the reaction begins, the major product will be the one that is produced more quickly as a result of its lower activation energy. The reaction can be said to be under kinetic control at this time. Given enough time, however, and assuming a reversible reaction, the dominant product will be the thermodynamically more stable product as a result of its lower free energy value. The reaction can then be said to be under thermodynamic control. Eventually, the reaction will reach equilibrium, as defined by its Keq.
STANDARD GIBBS FREE ENERGY
The free energy change of reactions can be measured under standard state conditions to yield the standard free energy, ΔG°rxn. For standard free energy determinations, the concentrations of any solutions in the reaction are 1 M. The standard free energy of formation of a compound, ΔG°f, is the free energy change that occurs when 1 mole of a compound in its standard state is produced from its respective elements in their standard states under standard state conditions. The standard free energy of formation for any element under standard state conditions is, by definition, zero. The standard free energy of a reaction, ΔG°rxn, is the free energy change that occurs when that reaction is carried out under standard state conditions; that is, when the reactants are converted to the products at standard conditions of temperature (298 K) and pressure (1 atm). Like enthalpy and entropy, the free energy of the reaction can be calculated from the free energies of formation of the reactants and products:
![]()
Equation 7.11
FREE ENERGY, Keq, AND Q
We can derive the standard free energy change for a reaction from the equilibrium constant Keq for the reaction using the equation
ΔG°rxn = –RT ln Keq
Equation 7.12
where R is the ideal gas constant, T is the temperature in kelvin, and Keq is the equilibrium constant. This equation allows us to make not only quantitative evaluations of the free energy change of a reaction, but also qualitative assessments of the spontaneity of the reaction. The greater the value of Keq, the more positive the value of its natural logarithm. The more positive the natural logarithm, the more negative the standard free energy change. The more negative the standard free energy change, the more spontaneous the reaction
Once a reaction begins, however, the standard state conditions (specifically 1 M solutions) no longer apply. The value of the equilibrium constant must be replaced with another number that is reflective of where the reaction is in its path toward equilibrium. To determine the free energy change for a reaction that is in progress, we relate ΔGrxn (not ΔG°rxn) to the reaction quotient, Q:
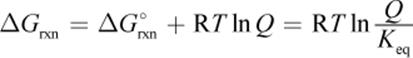
Equation 7.13
KEY CONCEPT
Note that the right side of this equation is similar to Equation 7.12. The use of the Q indicates that the system is not at equilibrium.
As described in Chapter 6 of MCAT General Chemistry Review, if the ratio of ![]() is less than one (Q < Keq), then the natural logarithm will be negative, and the free energy change will be negative, so the reaction will spontaneously proceed forward until equilibrium is reached. If the ratio of
is less than one (Q < Keq), then the natural logarithm will be negative, and the free energy change will be negative, so the reaction will spontaneously proceed forward until equilibrium is reached. If the ratio of ![]() is greater than one (Q > Keq), then the natural logarithm will be positive, and the free energy change will be positive. In that case, the reaction will spontaneously move in the reverse direction until equilibrium is reached. Of course, if the ratio is equal to one, the reaction quotient is equal to the equilibrium constant; the reaction is at equilibrium, and the free energy change is zero (ln 1 = 0).
is greater than one (Q > Keq), then the natural logarithm will be positive, and the free energy change will be positive. In that case, the reaction will spontaneously move in the reverse direction until equilibrium is reached. Of course, if the ratio is equal to one, the reaction quotient is equal to the equilibrium constant; the reaction is at equilibrium, and the free energy change is zero (ln 1 = 0).
Reaction profiles of free energy can be altered by the presence of catalysts. While the overall free energy change of the reaction is not altered, the activation energy required to accomplish the reaction is reduced significantly in the presence of a catalyst, as shown in Figure 7.14.
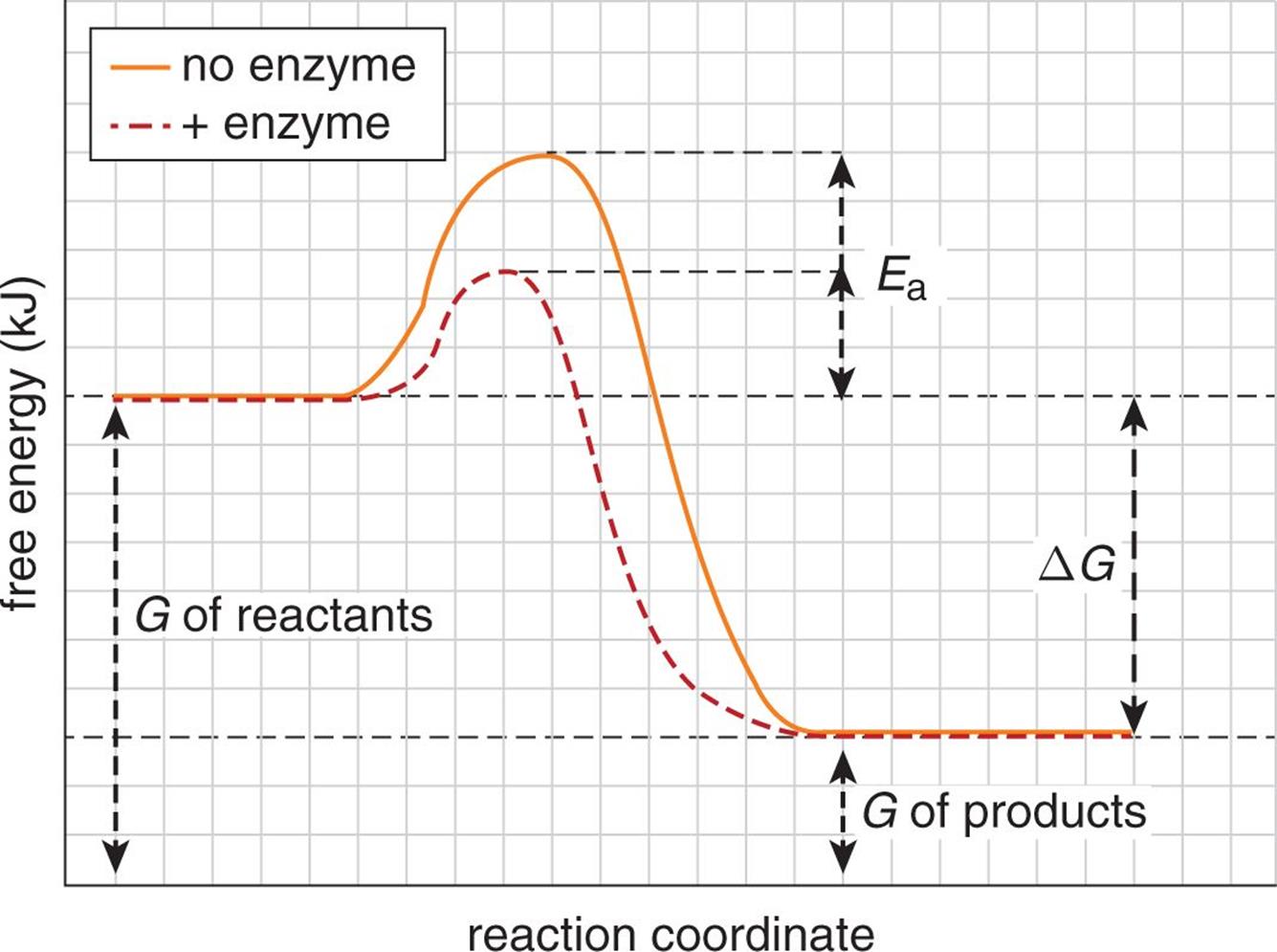 Figure 7.14. Catalysts Alter Kinetics but Not Equilibrium or Free Energy Change
Figure 7.14. Catalysts Alter Kinetics but Not Equilibrium or Free Energy Change
MCAT Concept Check 7.6:
Before you move on, assess your understanding of the material with these questions.
1. The Haber–Bosch process creates ammonia through several reactions, the final step of which is

Determine what the Gibbs free energy of this reaction is at standard conditions and at 500 K:
· At standard conditions:
· At 500 K:
2. At what temperature would the reaction described above be at equilibrium?
3. If you were to suddenly flood the reaction vessel with significant amounts of ammonia, what would occur?