Cracking the AP Biology Exam
6
Molecular Genetics
PROTEIN SYNTHESIS
Protein synthesis involves three basic steps: transcription, RNA processing, and translation.
TRANSCRIPTION
Transcription involves copying the genetic code directly from DNA. The initial steps in transcription are similar to the initial steps in DNA replication. The obvious difference is that whereas in replication we end up with a complete copy of the cell’s DNA, in translation we end up with only a partial copy in the form of mRNA.
Transcription involves three phases: initiation, elongation, and termination. As in DNA replication, the first initiation step in transcription is to unwind and unzip the DNA strands using helicase. Transcription begins at special sequences of the DNA strand called promoters. Because RNA is single-stranded, we have to copy only one of the two DNA strands. The strand that serves as the template is the sense strand. The other strand that lies dormant is the antisense strand.
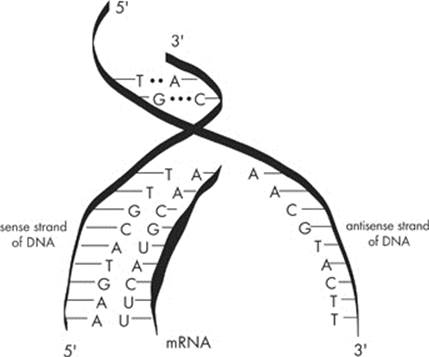
This time, RNA nucleotides line up alongside one DNA strand to form an mRNA strand. Another enzyme, RNA polymerase, brings free-floating RNA nucleotides to the DNA strand. As we saw earlier, guanines and cytosines pair up. However, the RNA molecule replaces the DNA’s thymine molecules with uracils. In other words, the exposed adenines are now paired up with uracil instead of thymine.
Once the mRNA finishes adding on nucleotides and reaches a termination point, it separates from the DNA template, completing the process of transcription. The new RNA has now transcribed, or “copied,” the sequence of nucleotide bases directly from the exposed DNA strand.
RNA PROCESSING
Now the mRNA strand is ready to move out of the nucleus. But before the mRNA molecule can leave the nucleus, it must be processed. That means the mRNA has to be modified before it exits the nucleus.
A newly made mRNA molecule (called the heterogeneous nuclear RNA, or hnRNA) contains more nucleotides than it needs to code for a protein. The mRNA consists of both coding regions and noncoding regions. The regions that express the code for the polypeptide are exons. The noncoding regions in the mRNA are introns.
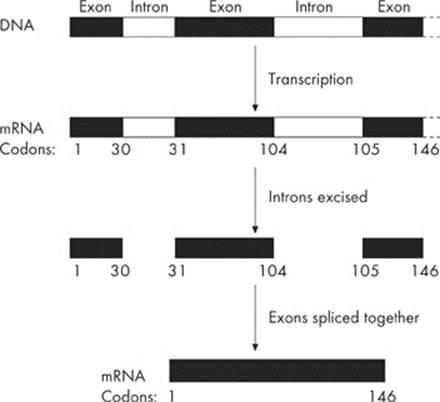
The introns—the intervening sequences—must be removed before the mRNA leaves the nucleus. The removal of introns is a complex process accomplished by an RNA-protein complex called a spliceosome. In addition, a poly(A) tail is added to the 3′ end and a 5′ cap is added to the 5′ end. This process produces a final mRNA that is shorter than the transcribed mRNA.
TRANSLATION
Now, the mRNA leaves the nucleus and searches for a ribosome. The mRNA molecule carries the message from DNA in the form of codons, a group of three bases, or “letters,” that corresponds to one of 20 amino acids. The genetic code is redundant, meaning that certain amino acids are specified by more than one codon.
The mRNA attaches to the ribosome and “waits” for the appropriate amino acids to come to the ribosome. That’s where tRNA comes in. A tRNA molecule has a unique three-dimensional structure that resembles a four-leaf clover:
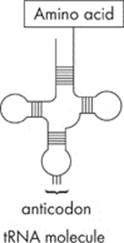
One end of the tRNA carries an amino acid. The other end, called an anticodon, has three nitrogenous bases that can base pair with the codon in the mRNA.
Transfer RNAs are the “go-betweens” in protein synthesis. Each tRNA becomes charged and enzymatically attaches to an amino acid in the cell’s cytoplasm and “shuttles” it to the ribosome. The charging enzymes involved in forming the bond between the amino acid and tRNA require ATP.
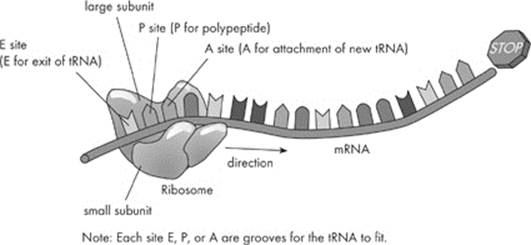
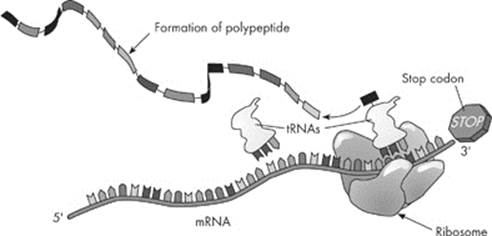
Translation also involves three phases: initiation, elongation, and termination. Initiation begins when a ribosome attaches to the mRNA.
What does the ribosome do? It helps the process along by holding everything in place while the RNAs assist in assembling polypeptides.
Initiation
Ribosomes contain three binding sites: an A site, a P site, and an E site. An initiator tRNA serves to activate translation and occupies the P site. In all organisms the codon for the initiation of protein synthesis is A–U–G, which codes for the amino acid methionine. The tRNA with the complementary anticodon, U–A–C, is methionine’s personal shuttle. Once the methionine tRNA is attached to the P site, the A site can be filled by the appropriate tRNA that corresponds to the next codon. The E site binds a free tRNA before it exits the ribosome.
Elongation
The addition of amino acids is called elongation. Remember that the mRNA contains many codons, or “triplets,” of nucleotide bases. As each amino acid is brought to the mRNA, it is linked to its neighboring amino acid by a peptide bond. When many amino acids link up, a polypeptide is formed.
Termination
How does this process know when to stop? The synthesis of a polypeptide is ended by stop codons. A codon doesn’t always code for an amino acid; there are three that serve as a stop codon. For example, U–A–A, U–A–G, and U–G–A are all stop codons. Termination occurs when the ribosome runs into one of these three stop codons.
Higher Protein Structure
The polypeptide has to go through several changes before it can officially be called a protein. Proteins can have four levels of structure. The linear sequence of the amino acids is called the primary structure of a protein. Now the polypeptide begins to twist, forming either a coil (known as an alpha helix), or zigzagging patterns (known as beta-pleated sheets). These are examples of proteins’ secondary structures. Next, the polypeptide folds in a three dimensional pattern. This is called the tertiary structure. Finally, when two or more polypeptides get together, we call it a quaternary structure. Once that’s complete the protein is ready to perform its task.
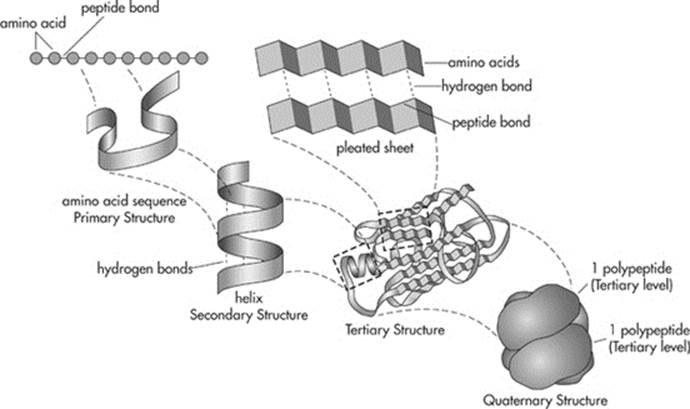
One more thing: In some case, the folding of proteins involves other proteins known as chaperon proteins (or chaperonins). They help the protein fold properly and make the process more efficient.
How about a little review?
- In transcription, mRNA copies, or “transcribes,” the code from an exposed strand of DNA in the nucleus.
- The mRNA is “processed” by having its introns, or noncoding sequences, removed.
- Now, ready to be translated, mRNA proceeds to the ribosome.
- Free-floating amino acids are picked up by tRNA and shuttled over to the ribosome, where mRNA awaits.
- In translation, the anticodon of a tRNA molecule carrying the appropriate amino acid base pairs with the codon on the mRNA.
- As new tRNA molecules match up to new codons, the ribosome holds them in place, allowing peptide bonds to form between the amino acids.
- The newly formed polypeptide grows until a stop codon is reached.
- The polypeptide or protein is released into the cell.