THE LIVING WORLD
Unit Eight. The Living Environment
35. Populations and Communities
35.9. The Niche and Competition
Within a community, each organism occupies a particular biological role, or niche. The niche that an organism occupies is the sum total of all the ways it utilizes the resources of its environment. A niche may be described in terms of space utilization, food consumption, temperature range, appropriate conditions for mating, requirements for moisture, and other factors. Niche is not synonymous with habitat, the place where an organism lives. Habitat is a place, and niche is a pattern of living. Many species can share a habitat, but as we shall see, no two species can long occupy exactly the same niche.
Sometimes species are not able to occupy their entire niche because of the presence or absence of other species. Species can interact with each other in a number of ways, and these interactions can either have positive or negative effects. Competition describes the interaction when two organisms attempt to use the same resource when there is not enough of the resource to satisfy both.
Competition between individuals of different species is called interspecific competition. Interspecific competition is often greatest between organisms that obtain their food in similar ways and between organisms that are more similar. Another type of competition, called intraspecific competition, is competition between individuals of the same species.
The Realized Niche
Because of competition, organisms may not be able to occupy the entire niche they are theoretically capable of using, called the fundamental niche (or theoretical niche). The actual niche the organism is able to occupy in the presence of competitors is called its realized niche.
In a classic study, J. H. Connell of the University of California, Santa Barbara, investigated competitive interactions between two species of barnacles that grow together on rocks along the coast of Scotland. Barnacles are marine animals (crustaceans) that have free-swimming larvae. The larvae eventually settle down, cementing themselves to rocks and remaining attached for the rest of their lives. Of the two species Connell studied, Chthamalus stellatus (the smaller barnacle in figure 35.17) lives in shallower water, where tidal action often exposes it to air, and Semibalanus balanoides (the larger barnacle) lives at lower depths, where it is rarely exposed to the atmosphere. In the deeper zone, Semibalanus could always outcompete Chthamalus by crowding it off the rocks, undercutting it, and replacing it even where it had begun to grow. When Connell removed Semibalanus from the area, however, Chthamalus was easily able to occupy the deeper zone, indicating that no physiological or other general obstacles prevented it from becoming established there. In contrast, Semibalanus could not survive in the shallow-water habitats where Chthamalus normally occurs; it evidently does not have the special physiological and morphological adaptations that allow Chthamalus to occupy this zone. Thus, the fundamental niche of the barnacle Chthamalus in Connell’s experiments in Scotland included that of Semibalanus (the red dashed arrow), but its realized niche was much narrower (the red solid arrow) because Chthamalus was outcompeted by Semibalanus in its fundamental niche.
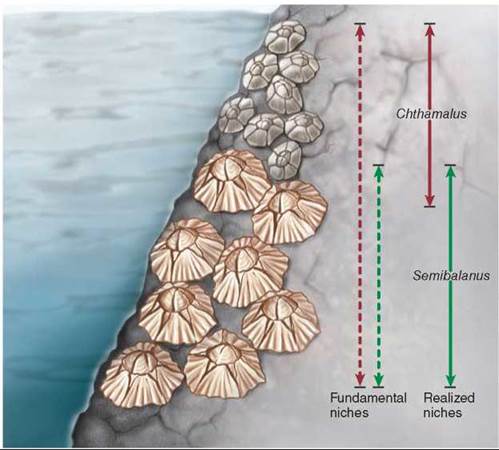
Figure 35.17. Competition among two species of barnacles limits niche use.
Chthamalus can live in both deep and shallow zones (its fundamental niche), but Semibalanus forces Chthamalus out of the part of its fundamental niche that overlaps the realized niche of Semibalanus.
Predators, as well as competitors, can limit the realized niche of a species. In the previous example, Chthamalus was able to fully occupy its fundamental niche when there was no competition. This is often the case when a species first enters a new, very favorable habitat that presents it with adequate resources, little or no competition, and no predators. However, once resources are limited, the population approaches carrying capacity, and other species begin to compete for the same resources. Also, predators may begin to more frequently recognize the species, and the population will be forced into its realized niche. For example, a plant called the St. John’s wort was introduced and became widespread in open rangeland habitats in California. It occupied all of its fundamental niche until a species of beetle that feeds on the plant was introduced into the habitat. Populations of the plant then quickly decreased, and it is now only found in shady sites where the beetle cannot thrive.
Competitive Exclusion
In classic experiments carried out between 1934 and 1935, Russian ecologist G. F. Gause studied competition among three species of Paramecium, a tiny protist. All three species grew well alone in culture tubes (figure 35.18a), preying on bacteria and yeasts that fed on oatmeal suspended in the culture fluid. However, when Gause grew P. aurelia together with P. caudatum in the same culture tube (figure 35.18b), the numbers of P. caudatum (the green line) always declined to extinction, leaving P. aurelia the only survivor. Why? Gause found P. aurelia was able to grow six times faster than its competitor, P. caudatum, because it was able to better use the limited available resources.
From experiments such as this, Gause formulated what is now called the principle of competitive exclusion. This principle states that if two species are competing for a resource, the species that uses the resource more efficiently will eventually eliminate the other locally—no two species with the same niche can coexist.
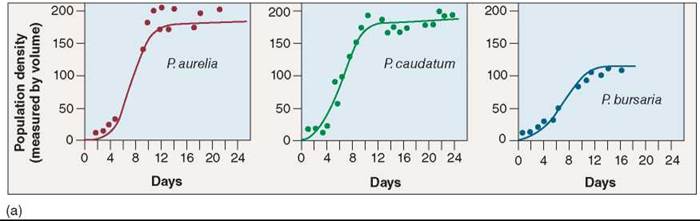
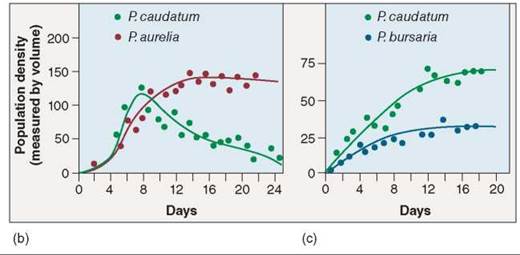
Figure 35.18. Competitive exclusion among three species of Paramecium.
In the microscopic world, Paramecium is a ferocious predator. Paramecia eat by ingesting their prey; their plasma membranes surround bacterial or yeast cells, forming a food vacuole containing the prey cell. In his experiments, (a) Gause found that three species of Paramecium grew well alone in culture tubes. (b) However, P. caudatum declined to extinction when grown with P aurelia because they shared the same realized niche, and P aurelia outcompeted P caudatum for food resources. (c) P caudatum and P bursaria were able to coexist, although in smaller populations, because the two have different realized niches and thus avoid competition.
Niche Overlap
In a revealing experiment, Gause challenged P caudatum— the defeated species in his earlier experiments—with a third species, P. bursaria. Because he expected these two species to also compete for the limited bacterial food supply, Gause thought one would win out, as had happened in his previous experiments. But that’s not what happened. Instead, both species survived in the culture tubes (figure 35.18c); the paramecia found a way to divide the food resources. How did they do it? In the upper part of the culture tubes, where the oxygen concentration and bacterial density were high, P. caudatum dominated because it was better able to feed on bacteria. However, in the lower part of the tubes, the lower oxygen concentration favored the growth of a different potential food, yeast, and P. bursaria was better able to eat this food. The fundamental niche of each species was the whole culture tube, but the realized niche of each species was only a portion of the tube. This graph also demonstrates the negative effect competition had on the participants: Competition was always detrimental to both species involved. Both species more than doubled their densities when grown without a competitor as when grown together.
Gause’s principle of competitive exclusion can be restated to say that no two species can occupy the same niche indefinitely when resources are limiting. Certainly species can and do coexist while competing for the same resources; we have seen many examples of such relationships. Nevertheless, Gause’s theory predicts that when two species are able to coexist on a long-term basis, either resources must not be limited or their niches will always differ in one or more features; otherwise, one species outcompetes the other and the extinction of the second species inevitably results through competitive exclusion.
Niche is a complex concept, involving all facets of the environment that are important to individual species. In recent years, a vigorous debate has arisen concerning the role of competitive exclusion, not only in determining the structure of communities but also in setting the course of evolution. When resources are abundant, species may overlap substantially in their use. However, when one or more resources suddenly become sharply limiting, as in periods of drought, the role of competition becomes much more obvious. When niches overlap, two outcomes are possible: competitive exclusion (winner takes all), or resource partitioning (dividing up resources to create two realized niches). It is only through resource partitioning, the topic of the next section, that the species can continue to coexist over long periods.
Resource Partitioning
Gause’s exclusion principle has a very important consequence: Persistent and intense competition between two species is rare in natural communities. Either one species drives the other to extinction, or natural selection reduces the competition between them, such as through resource partitioning. In resource partitioning, species that live in the same geographical area avoid competition by living in different portions of the habitat or by using different food or other resources. A clear example of this is seen in Anolis lizards (figure 35.19), where species may live in different parts of a tree habitat to avoid competition for food and space with other species that may live on the twigs, trunks, or grass.
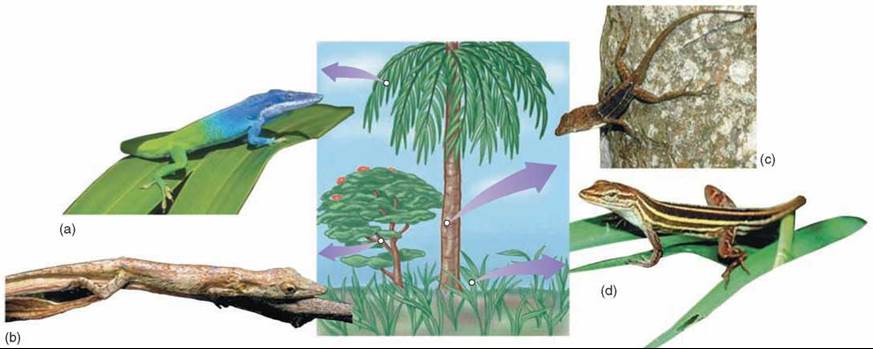
Figure 35.19. Resource partitioning among lizard species.
Species of Anolis lizards in the Caribbean partition their tree habitats in a variety of ways. Some species of anoles occupy the canopy of trees (a), others use twigs on the periphery (b), and still others are found at the base of the trunk (c). In addition, some use grassy areas in the open (d). This same pattern of resource partitioning has evolved independently on different Caribbean islands.
Resource partitioning can often be seen in closely related species that occupy the same geographical area. Called sympatric species (Greek, syn, same, and patria, country), these species avoid competition by evolving different adaptations to use different portions of the habitat, food or other resources. Closely related species that do not live in the same geographical area, called allopatric species (Greek, allos, other, and patria, country), often use the same habitat locations and food resources—because they are not in competition, natural selection does not favor evolutionary changes that subdivide their niche.
When a pair of closely related species occur in the same place, they tend to exhibit greater differences in morphology and behavior than the same two species do when living in different areas. Called character displacement, the differences evident between sympatric species are thought to have been favored by natural selection as a mechanism to facilitate resource partitioning and thus reduce competition. Character displacement can be seen clearly among Darwin’s finches. The two Galapagos finches in figure 35.20 have beaks of similar size when each is living on an island where the other does not occur. On islands where they are found living together, the two species have evolved beaks of different sizes, one adapted to larger seeds, the other to smaller ones. In essence, the two finches have subdivided the food niche, creating two new smaller niches. By partitioning the available food resources, the two species have avoided direct competition with each other, and so are able to live together in the same habitat.
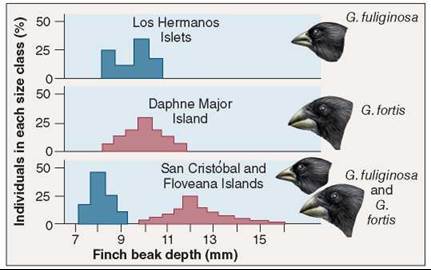
Figure 35.20. Character displacement.
These two species of Galapagos finches (genus Geospiza) have beaks of similar sizes when living apart, but different sizes when living together.
Key Learning Outcome 35.9. A niche may be defined as the way in which an organism uses its environment. No two species can occupy the same niche indefinitely without competition driving one to extinction if resources are limiting. Sympatric species partition available resources, reducing competition between them.
A Closer Look
Character Displacement Among Darwin's Finches
While evidence of character displacement can be seen in many laboratory populations, it has been difficult to observe the process in action in natural populations. Recently a particularly clear view of character displacement in action has been reported among finches on one of the smaller Galapagos islands, Daphne Major. For 33 years, researchers led by the husband-and-wife team of Peter and Rosemary Grant from Princeton University have marked each bird on the island, recording births and deaths, beak sizes, eating habits, and a wealth of other information. The seed-eating medium ground finch (Geospiza fortis) has lived on this island with essentially no competition for food from other finches. It shares the island only with the cactus finch, which uses its pointed beak to eat cactus fruit and pollen, and does not eat seeds. The absence of other seed-eating finches means that G. fortis could take maximum advantage of all seeds available on the island. While it prefers smaller, more tender seeds, when those are not available, G. fortis will eat larger, tougher seeds. During such difficult times, the G. fortis individuals with larger beaks are better able to crack open the large, tough seeds than smaller-beaked birds are, and so are more likely to survive and leave offspring.
As described in chapter 14, a severe drought in 1977 acted to weed out the G. fortis population on Daphne Major. The only food left for the medium ground finches was larger seeds. Most finches died that year on Daphne Major, but the birds with smaller beaks died in higher numbers.
The selection for larger beaks in a drought—beaks better able to break open the hard case of large, dry seeds—was precisely what evolutionary theory would predict, a clear example of directional selection.
However, a severe drought in 2003 and 2004 produced a dramatically different result! In the year immediately following that drought, the beak sizes in the population of medium ground finches showed a shift to smaller beaks, not larger ones. What had happened during the 2003/2004 drought that was different?
The answer, scientists found, was a new competitor. During the previous severe drought of 1977, the only competition that G. fortis experienced was from within its own species, between larger-beaked and smaller-beaked individuals. But in the drought of 2003 a new species of seedeating finch was present on the island, vying for the same limited food resources as G. fortis.
How did this happen? Prior to 1982, the large ground finch G. magnirostris (seen in this photo) was sometimes a visitor to Daphne Major but never stayed and bred on the island. Heavy rains in 1982 changed all that. The abundant food on the island after the rains induced two females and three males to stay and start a breeding population. Their numbers grew substantially over the next 10 years. By 2004, the large ground finch—weighing 30 grams, almost twice the size of G. fortis— made up nearly 40% of the ground finch population on the island.
Then the hammer fell. The drought that year was devastating. No small, new seeds were produced by the island's drought-stricken plants, and the two species of ground finches soon exhausted the supply of larger, dry seeds left from previous years. The shortage nearly wiped out both populations. But, unlike in previous droughts, small-beaked G. fortis individuals fared better than large-beak birds of that species—only 13% of large-billed G. fortis birds survived.
Within a year, the average beak size of the G. fortis finch population on Daphne Major was considerably smaller than in the years prior to the drought, which was exactly opposite to the results following earlier droughts. Two droughts, two different results. Why? In the drought of 1977, G. fortis experienced selective pressure favoring large beak size, as this helped them eat the only food available—large, dry seeds. In the drought of 2003/2004, G. fortis experienced this same selective pressure, but also experienced a new and even stronger selective pressure exerted by a competitor that it had never had to face on Daphne Major in the past. The large ground finch, with its stout, large beak, was better adapted than G. fortis at cracking open the bigger seeds and ate most of the remaining large seeds. As a result, although pickings were slim, this time more small seeds were available than large seeds for G. fortis, and individuals with smaller beaks were more likely to survive. When the drought ended, G. fortis finches used these smaller beaks to eat smaller seeds, leaving the large seeds to G. magnirostris.
This competition-driven shift in beak size is a clear example of character displacement. Often observed in laboratory experiments, this study is the first time it has been documented in the wild.

Geospiza magnirostris