CHEMISTRY THE CENTRAL SCIENCE
19 CHEMICAL THERMODYNAMICS
EXERCISES
VISUALIZING CONCEPTS
19.1 Two different gases occupy the two bulbs shown here. Consider the process that occurs when the stopcock is opened, assuming the gases behave ideally. (a) Draw the final (equilibrium) state. (b) Predict the signs of ΔH and ΔS for the process. (c) Is the process that occurs when the stopcock is opened a reversible one? (d) How does the process affect the entropy of the surroundings? [Sections 19.1 and 19.2]

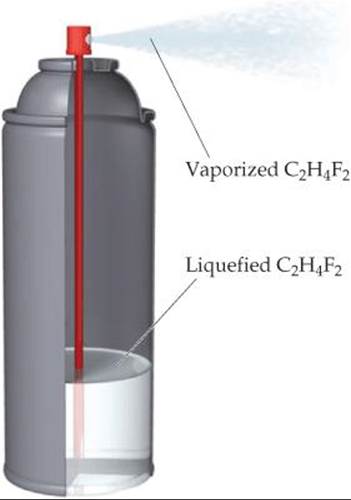
19.2 As shown here, one type of computer keyboard cleaner contains liquefied 1,1-difluoroethane (C2H4F2), which is a gas at atmospheric pressure. When the nozzle is squeezed, the 1,1-difluoroethane vaporizes out of the nozzle at high pressure, blowing dust out of objects. (a) Based on your experience, is the vaporization a spontaneous process at room temperature? (b) Defining the 1,1-difluoroethane as the system, do you expect qsys for the process to be positive or negative? Explain. (c) Predict whether ΔS is positive or negative for this process. (d) Given your answers to (a), (b), and (c), do you think the operation of this product depends more on heat flow or more on entropy change?
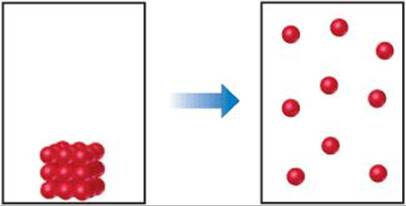
19.3 (a) What are the signs of ΔS and ΔH for the process depicted here? (b) How might temperature affect the sign of ΔG? (c) If energy can flow in and out of the system to maintain a constant temperature during the process, what can you say about the entropy change of the surroundings as a result of this process? [Sections 19.2 and 19.5]
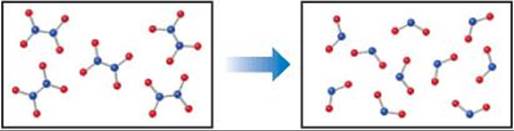
19.4 Predict the sign of ΔS accompanying this reaction. Explain your choice. [Section 19.3]
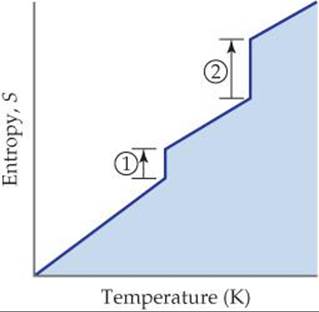
19.5 The accompanying diagram shows how entropy varies with temperature for a substance that is a gas at the highest temperature shown. (a) What processes correspond to the entropy increases along the vertical lines labeled 1 and 2? (b) Why is the entropy change for 2 larger than that for 1? [Section 19.3]
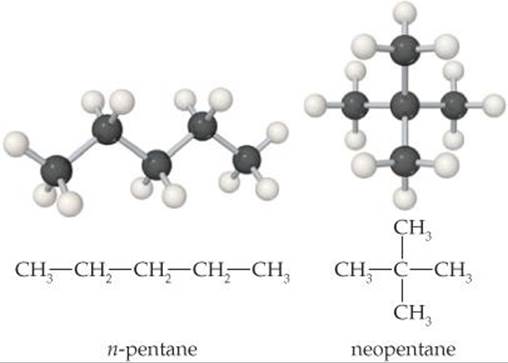
19.6 Isomers are molecules that have the same chemical formula but different arrangements of atoms, as shown here for two isomers of pentane, C5H12. (a) Do you expect a significant difference in the enthalpy of combustion of the two isomers? Explain. (b) Which isomer do you expect to have the higher standard molar entropy? Explain. [Section 19.4]
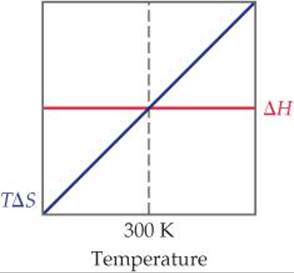
19.7 The accompanying diagram shows how ΔH (red line) and TΔS (blue line) change with temperature for a hypothetical reaction. (a) What is the significance of the point at 300 K, where ΔH and TΔS are equal? (b) In what temperature range is this reaction spontaneous? [Section 19.6]
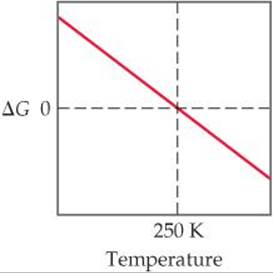
19.8 The accompanying diagram shows how ΔG for a hypothetical reaction changes as temperature changes. (a) At what temperature is the system at equilibrium? (b) In what temperature range is the reaction spontaneous? (c) Is ΔH positive or negative? (d) Is ΔS positive or negative? [Sections 19.5 and 19.6]
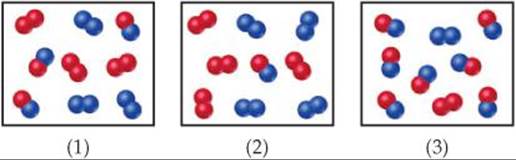
19.9 Consider a reaction ![]() with atoms of A shown in red in the diagram and atoms of B shown in blue. (a) If Kc = 1, which box represents the system at equilibrium? (b) What is the sign of ΔG for any process in which the contents of a reaction vessel move to equilibrium? (c) Rank the boxes in order of increasing magnitude of ΔG for the reaction. [Sections 19.5 and 19.7]
with atoms of A shown in red in the diagram and atoms of B shown in blue. (a) If Kc = 1, which box represents the system at equilibrium? (b) What is the sign of ΔG for any process in which the contents of a reaction vessel move to equilibrium? (c) Rank the boxes in order of increasing magnitude of ΔG for the reaction. [Sections 19.5 and 19.7]
19.10 The accompanying diagram shows how the free energy, G, changes during a hypothetical reaction A(g) + B(g) → C(g). On the left are pure reactants, each at 1 atm, and on the right is the pure product, also at 1 atm. (a) What is the significance of the minimum in the plot? (b)What does the quantity x, shown on the right side of the diagram, represent? [Section 19.7]
SPONTANEOUS PROCESSES (section 19.1)
19.11 Which of the following processes are spontaneous and which are nonspontaneous: (a) the ripening of a banana, (b) dissolution of sugar in a cup of hot coffee, (c) the reaction of nitrogen atoms to form N2 molecules at 25 °C and 1 atm, (d) lightning, (e) formation of CH4 and O2molecules from CO2 and H2O at room temperature and 1 atm of pressure?
19.12 Which of the following processes are spontaneous: (a) the melting of ice cubes at –10 °C and 1 atm pressure; (b) separating a mixture of N2 and O2 into two separate samples, one that is pure N2 and one that is pure O2; (c) alignment of iron filings in a magnetic field; (d) the reaction of hydrogen gas with oxygen gas to form water vapor; (e) the dissolution of HCl(g) in water to form concentrated hydrochloric acid?
______
19.13 (a) Give two examples of endothermic processes that are spontaneous. (b) Give an example of a process that is spontaneous at one temperature but nonspontaneous at a different temperature.
19.14 The crystalline hydrate Cd(NO3)2 · 4 H2O(s) loses water when placed in a large, closed, dry vessel:
![]()
This process is spontaneous and ΔH is positive. Is this process an exception to Bertholet's generalization that all spontaneous changes are exothermic? Explain.
______
19.15 Consider the vaporization of liquid water to steam at a pres sure of 1 atm. (a) Is this process endothermic or exothermic? (b) In what temperature range is it a spontaneous process? (c) In what temperature range is it a nonspontaneous process? (d) At what temperature are the two phases in equilibrium?
19.16 The normal freezing point of n-octane (C8H18) is –57 °C. (a) Is the freezing of n-octane an endothermic or exothermic process? (b) In what temperature range is the freezing of n-octane a spontaneous process? (c) In what temperature range is it a nonspontaneous process? (d) Is there any temperature at which liquid n-octane and solid n-octane are in equilibrium? Explain.
______
19.17(a) What is special about a reversible process? (b) Suppose a reversible process is reversed, restoring the system to its original state. What can be said about the surroundings after the process is reversed? (c) Under what circumstances will the vaporization of water to steam be a reversible process? (d) Are any of the processes that occur in the world around us reversible in nature? Explain.
19.18 (a) What is meant by calling a process irreversible? (b) After a particular irreversible process, the system is restored to its original state. What can be said about the condition of the surroundings after the system is restored to its original state? (c) Under what conditions will the condensation of a liquid be an irreversible process?
______
19.19 Consider a process in which an ideal gas changes from state 1 to state 2 in such a way that its temperature changes from 300 K to 200 K. (a) Describe how this change might be carried out while keeping the volume of the gas constant. (b) Describe how it might be carried out while keeping the pressure of the gas constant. (c) Does the change in ΔE depend on the particular pathway taken to carry out this change of state? Explain.
19.20 A system goes from state 1 to state 2 and back to state 1. (a) What is the relationship between the value of ΔE for going from state 1 to state 2 to that for going from state 2 back to state 1? (b) Without further information, can you conclude anything about the amount of heat transferred to the system as it goes from state 1 to state 2 as compared to that upon going from state 2 back to state 1? (c) Suppose the changes in state are reversible processes. Can you conclude anything about the work done by the system upon going from state 1 to state 2 as compared to that upon going from state 2 back to state 1?
______
19.21 Consider a system consisting of an ice cube. (a) Under what conditions can the ice cube melt reversibly? (b) If the ice cube melts reversibly, is ΔE zero for the process? Explain.
19.22 Consider what happens when a sample of the explosive TNT (Section 8.8: “Chemistry Put to Work: Explosives and Alfred Nobel”) is detonated under atmospheric pressure. (a) Is the detonation a spontaneous process? (b) What is the sign of q for this process? (c) Can you determine whether w is positive, negative, or zero for the process? Explain. (d) Can you determine the sign of ΔE for the process? Explain.
ENTROPY AND THE SECOND LAW OF THERMODYNAMICS (section 19.2)
19.23 (a) How can we calculate ΔS for an isothermal process? (b) Does ΔS for a process depend on the path taken from the initial state to the final state of the system? Explain.
19.24 Suppose we vaporize a mole of liquid water at 25 °C and another mole of water at 100 °C. (a) Assuming that the enthalpy of vaporization of water does not change much between 25 °C and 100 °C, which process involves the larger change in entropy? (b) Does the entropy change in either process depend on whether we carry out the process reversibly or not? Explain.
______
19.25 The normal boiling point of Br2(l) is 58.8 °C, and its molar enthalpy of vaporization is ΔHvap = 29.6 kJ/mol. (a) When Br2(l) boils at its normal boiling point, does its entropy increase or decrease? (b) Calculate the value of ΔS when 1.00 mol of Br2(l) is vaporized at 58.8 °C.
19.26 The element gallium (Ga) freezes at 29.8 °C, and its molar enthalpy of fusion is ΔHfus = 5.59 kJ/mol. (a) When molten gallium solidifies to Ga(s) at its normal melting point, is ΔS positive or negative? (b) Calculate the value of ΔS when 60.0 g of Ga(l) solidifies at 29.8 °C.
______
19.27 (a) Express the second law of thermodynamics in words. (b) If the entropy of the system increases during a reversible process, what can you say about the entropy change of the surroundings? (c) In a certain spontaneous process the system undergoes an entropy change, ΔS = 42 J/K. What can you conclude about ΔSsurr?
19.28 (a) Express the second law of thermodynamics as a mathematical equation. (b) In a particular spontaneous process the entropy of the system decreases. What can you conclude about the sign and magnitude of ΔSsurr? (c) During a certain reversible process, the surroundings undergo an entropy change, ΔSsurr = –78 J/K. What is the entropy change of the system for this process?
______
19.29 (a) What sign for ΔS do you expect when the volume of 0.200 mol of an ideal gas at 27 °C is increased isothermally from an initial volume of 10.0 L? (b) If the final volume is 18.5 L, calculate the entropy change for the process. (c) Do you need to specify the temperature to calculate the entropy change? Explain.
19.30 (a) What sign for ΔS do you expect when the pressure on 0.600 mol of an ideal gas at 350 K is increased isothermally from an initial pressure of 0.750 atm? (b) If the final pressure on the gas is 1.20 atm, calculate the entropy change for the process. (c) Do you need to specify the temperature to calculate the entropy change? Explain.
THE MOLECULAR INTERPRETATION OF ENTROPY (section 19.3)
19.31 For the isothermal expansion of a gas into a vacuum, ΔE = 0, q = 0, and w = 0. (a) Is this a spontaneous process? (b) Explain why no work is done by the system during this process. (c) In thermodynamics, what is the “driving force” for the expansion of the gas?
19.32 (a) What is the difference between a state and a microstate of a system? (b) As a system goes from state A to state B, its entropy decreases. What can you say about the number of microstates corresponding to each state? (c) In a particular spontaneous process, the number of microstates available to the system decreases. What can you conclude about the sign of ΔSsurr?
______
19.33 How would each of the following changes affect the number of microstates available to a system: (a) increase in temperature, (b) decrease in volume, (c) change of state from liquid to gas?
19.34 (a) Using the heat of vaporization in Appendix B, calculate the entropy change for the vaporization of water at 25 °C and at 100 °C. (b) From your knowledge of microstates and the structure of liquid water, explain the difference in these two values.
______
19.35 (a) What do you expect for the sign of ΔS in a chemical reaction in which two moles of gaseous reactants are converted to three moles of gaseous products? (b) For which of the processes in Exercise 19.11 does the entropy of the system increase?
19.36 (a) In a chemical reaction two gases combine to form a solid. What do you expect for the sign of ΔS? (b) How does the entropy of the system change in the processes described in Exercise 19.12?
______
19.37 How does the entropy of the system change when (a) a solid melts, (b) a gas liquefies, (c) a solid sublimes?
19.38 How does the entropy of the system change when (a) the temperature of the system increases, (b) the volume of a gas increases, (c) equal volumes of ethanol and water are mixed to form a solution?
______
19.39 (a) State the third law of thermodynamics. (b) Distinguish between translational motion, vibrational motion, and rotational motion of a molecule. (c) Illustrate these three kinds of motion with sketches for the HCl molecule.
19.40 (a) If you are told that the entropy of a certain system is zero, what do you know about the system and the temperature? (b) The energy of a gas is increased by heating it. Using CO2 as an example, illustrate the different ways in which additional energy can be distributed among the molecules of the gas. (c) CO2(g) and Ar(g) have nearly the same molar mass. At a given temperature, will they have the same number of microstates? Explain.
______
19.41 For each of the following pairs, choose the substance with the higher entropy per mole at a given temperature: (a) Ar(l) or Ar(g), (b) He(g) at 3 atm pressure or He(g) at 1.5 atm pressure, (c) 1 mol of Ne(g) in 15.0 L or 1 mol of Ne(g) in 1.50 L, (d) CO2(g) or CO2(s).
19.42 For each of the following pairs, indicate which substance possesses the larger standard entropy: (a) 1 mol of P4(g) at 300 °C, 0.01 atm, or 1 mol of As4(g) at 300 °C, 0.01 atm; (b) 1 mol of H2O(g) at 100 °C, 1 atm, or 1 mol of H2O(l) at 100 °C, 1 atm; (c) 0.5 mol of N2(g) at 298 K, 20-L volume, or 0.5 mol CH4(g) at 298 K, 20-L volume; (d) 100 g Na2O4(s) at 30 °C or 100 g Na2SO4(aq) at 30 °C.
______
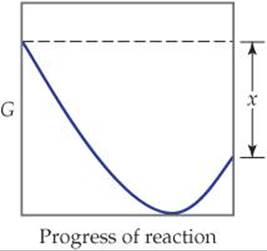
19.43 Predict the sign of the entropy change of the system for each of the following reactions:
19.44 Predict the sign of ΔSsys for each of the following processes: (a) Molten gold solidifies. (b) Gaseous Cl2 dissociates in the stratosphere to form gaseous Cl atoms. (c) Gaseous CO reacts with gaseous H2 to form liquid methanol, CH3OH. (d) Calcium phosphate precipitates upon mixing Ca(NO3)2(aq) and (NH4)3PO4(aq).
ENTROPY CHANGES IN CHEMICAL REACTIONS (section 19.4)
19.45 (a) Using Figure 19.13 as a model, sketch how the entropy of water changes as it is heated from –50 °C to 110 °C at sea level. Show the temperatures at which there are vertical increases in entropy. (b) Which process has the larger entropy change: melting ice or boiling water? Explain.
19.46 Propanol (C3H7OH) melts at –126.5 °C and boils at 97.4 °C. Draw a qualitative sketch of how the entropy changes as propanol vapor at 150 °C and 1 atm is cooled to solid propanol at –150 °C and 1 atm.
______
19.47 In each of the following pairs, which compound would you expect to have the higher standard molar entropy: (a) C2H2(g) or C2H6(g), (b) CO2(g) or CO(g)? Explain.
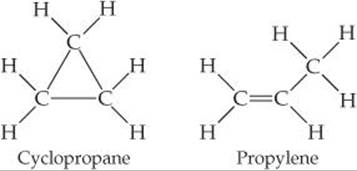
19.48 Cyclopropane and propylene are isomers (see Exercise 19.6) that both have the formula C3H6. Based on the molecular structures shown, which of these isomers would you expect to have the higher standard molar entropy at 25 °C?
______
19.49 Use Appendix C to compare the standard entropies at 25 °C for the following pairs of substances: (a) Sc(s) and Sc(g), (b) NH3(g) and NH3(aq), (c) 1 mol P4(g) and 2 mol P2(g), (d) C(graphite) and C(diamond). In each case explain the difference in the entropy values.
19.50 Using Appendix C, compare the standard entropies at 25 °C for the following pairs of substances: (a) CuO(s) and Cu2O(s), (b) 1 mol N2O4(g) and 2 mol NO2(g), (c) SiO2(s) and CO2(g), (d) CO(g) and CO2(g). For each pair, explain the difference in the entropy values.
______
[19.51] The standard entropies at 298 K for certain of the group 4A elements are as follows: C(s, diamond) = 2.43 J/mol-K, Si(s) = 18.81 J/mol-K, Ge(s) = 31.09 J/mol-K, and Sn(s) = 51.818 J/mol-K. All but Sn have the diamond structure. How do you account for the trend in the S°values?
[19.52] Three of the forms of elemental carbon are graphite, diamond, and buckminsterfullerene. The entropies at 298 K for graphite and diamond are listed in Appendix C. (a) Account for the difference in the S° values of graphite and diamond in light of their structures (Figure 12.30).(b) What would you expect for the S° value of buckminsterfullerene (Figure 12.47) relative to the values for graphite and diamond? Explain.
______
19.53 Using S° values from Appendix C, calculate ΔS° values for the following reactions. In each case account for the sign of ΔS°.
19.54 Calculate ΔS° values for the following reactions by using tabulated S° values from Appendix C. In each case explain the sign of ΔS°
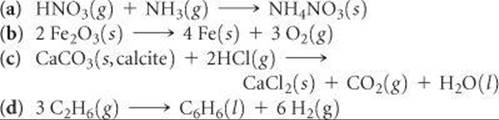
GIBBS FREE ENERGY (sections 19.5 and 19.6)
19.55 (a) For a process that occurs at constant temperature, express the change in Gibbs free energy in terms of changes in the enthalpy and entropy of the system. (b) For a certain process that occurs at constant T and P, the value of ΔG is positive. What can you conclude? (c) What is the relationship between ΔG for a process and the rate at which it occurs?
19.56 (a) What is the meaning of the standard free-energy change, ΔG°, as compared with ΔG? (b) For any process that occurs at constant temperature and pressure, what is the significance of ΔG = 0? (c) For a certain process, ΔG is large and negative. Does this mean that the process necessarily occurs rapidly?
______
19.57 For a certain chemical reaction, ΔH° = –35.4 kJ and ΔS° = –85.5 J/K. (a) Is the reaction exothermic or endother-mic? (b) Does the reaction lead to an increase or decrease in the randomness or disorder of the system? (c) Calculate ΔG° for the reaction at 298 K. (d) Is the reaction spontaneous at 298 K under standard conditions?
19.58 A certain reaction has ΔH° = +23.7 kJ and ΔS° = + 52.4J/K. (a) Is the reaction exothermic or endothermic? (b) Does the reaction lead to an increase or decrease in the randomness or disorder of the system? (c) Calculate ΔG° for the reaction at 298 K. (d) Is the reaction spontaneous at 298 K under standard conditions?
______
19.59 Using data in Appendix C, calculate ΔH°, ΔS°, and ΔG° at 298 K for each of the following reactions. In each case show that ΔG° = ΔH ° – TΔS°.

19.60 Use data in Appendix C to calculate ΔH°, ΔS°, and ΔG° at 25 °C for each of the following reactions. In each case show that ΔG° = ΔH ° – TΔS°.
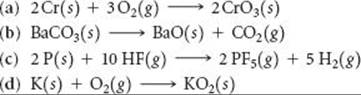
______
19.61 Using data from Appendix C, calculate ΔG° for the following reactions. Indicate whether each reaction is spontaneous at 298 K under standard conditions.
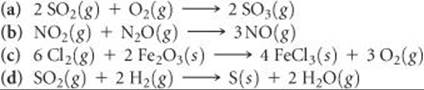
19.62 Using data from Appendix C, calculate the change in Gibbs free energy for each of the following reactions. In each case indicate whether the reaction is spontaneous at 298 K under standard conditions.
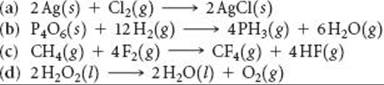
______
19.63 Octane (C8H18) is a liquid hydrocarbon at room temperature that is the primary constituent of gasoline. (a) Write a balanced equation for the combustion of C8H18(l) to form CO2(g) and H2O(l). (b) Without using thermochemical data, predict whether ΔG° for this reaction is more negative or less negative than ΔH°.
19.64 Sulfur dioxide reacts with strontium oxide as follows:
![]()
(a) Without using thermochemical data, predict whether ΔG° for this reaction is more negative or less negative than ΔH°.
(b) If you had only standard enthalpy data for this reaction, how would you go about making a rough estimate of the value of ΔG° at 298 K, using data from Appendix C on other substances?
______
19.65 Classify each of the following reactions as one of the four possible types summarized in Table 19.3:

19.66 From the values given for ΔH° and ΔS°, calculate ΔG° for each of the following reactions at 298 K. If the reaction is not spontaneous under standard conditions at 298 K, at what temperature (if any) would the reaction become spontaneous?
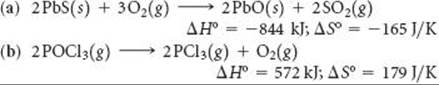
______
19.67 A particular constant-pressure reaction is spontaneous at 390 K. The enthalpy change for the reaction is +23.7 kJ. What can you conclude about the sign and magnitude of ΔS for the reaction?
19.68 A certain constant-pressure reaction is nonspontaneous at 45 °C. The entropy change for the reaction is 72 J/K. What can you conclude about the sign and magnitude of ΔH?
______
19.69 For a particular reaction, ΔH = –32 kJ and ΔS = –98 J/K. Assume that ΔH and ΔS do not vary with temperature. (a) At what temperature will the reaction have ΔG = 0? (b) If T is increased from that in part (a), will the reaction be spontaneous or nonspontaneous?
19.70 Reactions in which a substance decomposes by losing CO are called decarbonylation reactions. The decarbonylation of acetic acid proceeds as follows:
![]()
By using data from Appendix C, calculate the minimum temperature at which this process will be spontaneous under standard conditions. Assume that ΔH° and ΔS° do not vary with temperature.
______
19.71 Consider the following reaction between oxides of nitrogen:
![]()
(a) Use data in Appendix C to predict how ΔG° for the reaction varies with increasing temperature. (b) Calculate ΔG° at 800 K, assuming that ΔH° and ΔS° do not change with temperature. Under standard conditions is the reaction spontaneous at 800 K? (c) Calculate ΔG° at 1000 K. Is the reaction spontaneous under standard conditions at this temperature?
19.72 Methanol (CH3OH) can be made by the controlled oxidation of methane:
![]()
(a) Use data in Appendix C to calculate ΔH° and ΔS° for this reaction. (b) How is ΔG° for the reaction expected to vary with increasing temperature? (c) Calculate ΔG° at 298 K. Under standard conditions, is the reaction spontaneous at this temperature? (d) Is there a temperature at which the reaction would be at equilibrium under standard conditions and that is low enough so that the compounds involved are likely to be stable?
______
19.73 (a) Use data in Appendix C to estimate the boiling point of benzene, C6H6(l). (b) Use a reference source, such as the CRC Handbook of Chemistry and Physics, to find the experimental boiling point of benzene. How do you explain any deviation between your answer in part (a) and the experimental value?
19.74 (a) Using data in Appendix C, estimate the temperature at which the free-energy change for the transformation from I2(s) to I2(g) is zero. What assumptions must you make in arriving at this estimate? (b) Use a reference source, such as Web Elements (www.webelements.com), to find the experimental melting and boiling points of I2. (c) Which of the values in part (b) is closer to the value you obtained in part (a)? Can you explain why this is so?
______
19.75 Acetylene gas, C2H2(g), is used in welding. (a) Write a balanced equation for the combustion of acetylene gas to CO2(g) and H2O(l). (b) How much heat is produced in burning 1 mol of C2H2 under standard conditions if both reactants and products are brought to 298 K? (c) What is the maximum amount of useful work that can be accomplished under standard conditions by this reaction?
19.76 The fuel in high-efficiency natural gas vehicles consists primarily of methane (CH4). (a) How much heat is produced in burning 1 mol of CH4(g) under standard conditions if reactants and products are brought to 298 K and H2O(l) is formed? (b) What is the maximum amount of useful work that can be accomplished under standard conditions by this system?
FREE ENERGY AND EQUILIBRIUM (section 19.7)
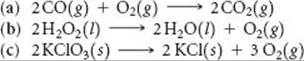
19.77 Explain qualitatively how ΔG changes for each of the follow ing reactions as the partial pressure of O2 is increased:
19.78 Indicate whether ΔG increases, decreases, or does not change when the partial pressure of H2 is increased in each of the following reactions:
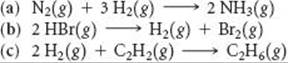
______
19.79 Consider the reaction 2 NO2(g) → N2O4(g). (a) Using data from Appendix C, calculate ΔG° at 298 K. (b) Calculate ΔG at 298 K if the partial pressures of NO2 and N2O4 are 0.40 atm and 1.60 atm, respectively.
19.80 Consider the reaction 3 CH4(g) → C3H8(g) + 2 H2(g). (a) Using data from Appendix C, calculate ΔG° at 298 K. (b) Calculate ΔG at 298 K if the reaction mixture consists of 40.0 atm of CH4, 0.0100 atm of C3H8(g), and 0.0180 atm of H2.
______
19.81 Use data from Appendix C to calculate the equilibrium constant, K, at 298 K for each of the following reactions:
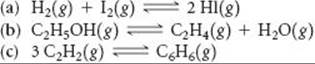
19.82 Using data from Appendix C, write the equilibrium-constant expression and calculate the value of the equilibrium constant for these reactions at 298 K:
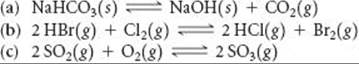
______
19.83 Consider the decomposition of barium carbonate:
![]()
Using data from Appendix C, calculate the equilibrium pressure of CO2 at (a) 298 K and (b) 1100 K.
19.84 Consider the reaction
![]()
Using data in Appendix C, calculate the equilibrium pressure of CO2 in the system at (a) 400 °C and (b) 180 °C.
______
19.85 The value of Ka for nitrous acid (HNO2) at 25 °C is given in Appendix D. (a) Write the chemical equation for the equilibrium that corresponds to Ka. (b) By using the value of Ka, calculate ΔG° for the dissociation of nitrous acid in aqueous solution. (c) What is the value of ΔG at equilibrium? (d) What is the value of ΔG when [H+] = 5.0 × 10–2M, [NO2–] = 6.0 × 10–4M, and [HNO2] = 0.20 M?
19.86 The Kb for methylamine (CH3NH2) at 25 °C is given in Appendix D. (a) Write the chemical equation for the equilibrium that corresponds to Kb. (b) By using the value of Kb, calculate ΔG° for the equilibrium in part (a). (c) What is the value of ΔG at equilibrium? (d) What is the value of ΔG when [H+] = 6.7 × 10–9M, [CH3NH3+] = 2.4 × 10–3M, and [CH3NH2] = 0.098 M?
ADDITIONAL EXERCISES
19.87 (a) Which of the thermodynamic quantities T, E, q, w, and S are state functions? (b) Which depend on the path taken from one state to another? (c) How many reversible paths are there between two states of a system? (d) For a reversible isothermal process, write an expression for ΔE in terms of q and w and an expression for ΔS in terms of q and T.
19.88 Indicate whether each of the following statements is true or false. If it is false, correct it. (a) The feasibility of manufacturing NH3 from N2 and H2 depends entirely on the value of ΔH for the process N2 (g) + 3 H2(g) → 2 NH3(g). (b) The reaction of Na(s) with Cl2(g) to form NaCl(s) is a spontaneous process. (c) A spontaneous process can in principle be conducted reversibly. (d) Spontaneous processes in general require that work be done to force them to proceed. (e) Spontaneous processes are those that are exothermic and that lead to a higher degree of order in the system.
19.89 For each of the following processes, indicate whether the signs of ΔS and ΔH are expected to be positive, negative, or about zero. (a) A solid sublimes. (b) The temperature of a sample of Co(s) is lowered from 60 °C to 25 °C. (c) Ethyl alcohol evaporates from a beaker. (d) A diatomic molecule dissociates into atoms. (e) A piece of charcoal is combusted to form CO2(g) and H2O(g).
19.90 The reaction 2 Mg(s) + O2(g) →2 MgO(s) is highly spontaneous and has a negative value for ΔS°. The second law of thermodynamics states that in any spontaneous process there is always an increase in the entropy of the universe. Is there an inconsistency between this reaction and the second law?
[19.91] Suppose four gas molecules are placed in the left flask in Figure 19.6(a). Initially, the right flask is evacuated and the stopcock is closed. (a) After the stopcock is opened, how many different arrangements of the molecules are possible? (b) How many of the arrangements from part (a) have all the molecules in the left flask? (c) How does the answer to part (b) explain the spontaneous expansion of the gas?
[19.92] Consider a system that consists of two standard playing dice, with the state of the system defined by the sum of the values shown on the top faces. (a) The two arrangements of top faces shown here can be viewed as two possible microstates of the system. Explain. (b) To which state does each mi-crostate correspond? (c) How many possible states are there for the system? (d) Which state or states have the highest entropy? Explain. (e) Which state or states have the lowest entropy? Explain.

19.93 Ammonium nitrate dissolves spontaneously and endother-mally in water at room temperature. What can you deduce about the sign of ΔS for this solution process?
[19.94] A standard air conditioner involves a refrigerant that is typically now a fluorinated hydrocarbon, such as CH2F2. An air-conditioner refrigerant has the property that it readily vaporizes at atmospheric pressure and is easily compressed to its liquid phase under increased pressure. The operation of an air conditioner can be thought of as a closed system made up of the refrigerant going through the two stages shown here (the air circulation is not shown in this diagram).
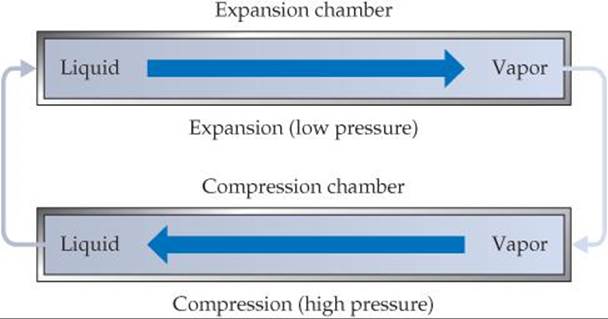
During expansion, the liquid refrigerant is released into an expansion chamber at low pressure, where it vaporizes. The vapor then undergoes compression at high pressure back to its liquid phase in a compression chamber. (a) What is the sign of q for the expansion? (b) What is the sign of q for the compression? (c) In a central air-conditioning system, one chamber is inside the home and the other is outside. Which chamber is where, and why? (d) Imagine that a sample of liquid refrigerant undergoes expansion followed by compression, so that it is back to its original state. Would you expect that to be a reversible process? (e) Suppose that a house and its exterior are both initially at 31 °C. Some time after the air conditioner is turned on, the house is cooled to 24 °C. Is this process spontaneous or nonspontaneous?
[19.95] Trouton's rule states that for many liquids at their normal boiling points, the standard molar entropy of vaporization is about 88 J/mol-K. (a) Estimate the normal boiling point of bromine, Br2, by determining ![]() for Br2 using data from Appendix C. Assume that
for Br2 using data from Appendix C. Assume that ![]() remains constant with temperature and that Trouton's rule holds. (b) Look up the normal boiling point of Br2 in a chemistry handbook or at the WebElements Web site (www.webelements.com).
remains constant with temperature and that Trouton's rule holds. (b) Look up the normal boiling point of Br2 in a chemistry handbook or at the WebElements Web site (www.webelements.com).
[19.96] For the majority of the compounds listed in Appendix C, the value of ![]() is more positive (or less negative) than the value of
is more positive (or less negative) than the value of ![]() . (a) Explain this observation, using NH3(g), CCl4(l), and KNO3(s) as examples. (b) An exception to this observation is CO(g). Explain the trend in the
. (a) Explain this observation, using NH3(g), CCl4(l), and KNO3(s) as examples. (b) An exception to this observation is CO(g). Explain the trend in the ![]() and
and ![]() values for this molecule.
values for this molecule.
19.97 Consider the following three reactions:

(a) For each of the reactions, use data in Appendix C to calculate ΔH°, ΔG°, and ΔS° at 25 °C. (b) Which of these reactions are spontaneous under standard conditions at 25 °C? (c) For each of the reactions, predict the manner in which the change in free energy varies with an increase in temperature.
19.98 Using the data in Appendix C and given the pressures listed, calculate ΔG° for each of the following reactions:
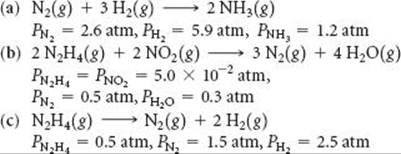
19.99 (a) For each of the following reactions, predict the sign of ΔH° and ΔS° and discuss briefly how these factors determine the magnitude of K. (b) Based on your general chemical knowledge, predict which of these reactions will have K > 0. (c) In each case indicate whether Kshould increase or decrease with increasing temperature.
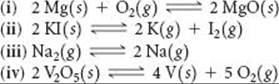
19.100 Acetic acid can be manufactured by combining methanol with carbon monoxide, an example of a carbonylation reaction:
![]()
(a) Calculate the equilibrium constant for the reaction at 25 °C. (b) Industrially, this reaction is run at temperatures above 25 °C. Will an increase in temperature produce an increase or decrease in the mole fraction of acetic acid at equilibrium? Why are elevated temperatures used? (c)At what temperature will this reaction have an equilibrium constant equal to 1? (You may assume that ΔH° and ΔS° are temperature independent, and you may ignore any phase changes that might occur.)
19.101 The oxidation of glucose (C6H12O6) in body tissue produces CO2 and H2O. In contrast, anaerobic decomposition, which occurs during fermentation, produces ethanol (C2H5OH) and CO2. (a) Using data given in Appendix C, compare the equilibrium constants for the following reactions:
![]()
(b) Compare the maximum work that can be obtained from these processes under standard conditions.
[19.102] The conversion of natural gas, which is mostly methane, into products that contain two or more carbon atoms, such as ethane (C2H6), is a very important industrial chemical process. In principle, methane can be converted into ethane and hydrogen:
![]()
In practice, this reaction is carried out in the presence of oxygen:
![]()
(a) Using the data in Appendix C, calculate K for these reactions at 25 °C and 500 °C. (b) Is the difference in ΔG° for the two reactions due primarily to the enthalpy term (ΔH) or the entropy term (–TΔS)? (c) Explain how the preceding reactions are an example of driving a nonspontaneous reaction, as discussed in the “Chemistry and Life” box in Section 19.7. (d) The reaction of CH4 and O2 to form C2H6 and H2O must be carried out carefully to avoid a competing reaction. What is the most likely competing reaction?
[19.103] Cells use the hydrolysis of adenosine triphosphate (ATP) as a source of energy (Figure 19.19). The conversion of ATP to ADP has a standard free-energy change of –30.5 kJ/mol. If all the free energy from the metabolism of glucose,
![]()
goes into the conversion of ADP to ATP, how many moles of ATP can be produced for each mole of glucose?
[19.104] The potassium-ion concentration in blood plasma is about 5.0 × 10–3M, whereas the concentration in muscle-cell fluid is much greater (0.15 M). The plasma and intracellular fluid are separated by the cell membrane, which we assume is permeable only to K+. (a) What is ΔGfor the transfer of 1 mol of K+ from blood plasma to the cellular fluid at body temperature 37 °C? (b) What is the minimum amount of work that must be used to transfer this K+?
[19.105] The relationship between the temperature of a reaction, its standard enthalpy change, and the equilibrium constant at that temperature can be expressed as the following linear equation:
![]()
(a) Explain how this equation can be used to determine ΔH° experimentally from the equilibrium constants at several different temperatures. (b) Derive the preceding equation using relationships given in this chapter. To what is the constant equal?
[19.106] One way to derive Equation 19.3 depends on the observation that at constant T the number of ways, W, of arranging m ideal-gas particles in a volume V is proportional to the volume raised to the m power:
![]()
Use this relationship and Boltzmann's relationship between entropy and number of arrangements (Equation 19.5) to derive the equation for the entropy change for the isothermal expansion or compression of n moles of an ideal gas.
[19.107] About 86% of the world's electrical energy is produced by using steam turbines, a form of heat engine. In his analysis of an ideal heat engine, Sadi Carnot concluded that the maximum possible efficiency is defined by the total work that could be done by the engine, divided by the quantity of heat available to do the work (for example, from hot steam produced by combustion of a fuel such as coal or methane). This efficiency is given by the ratio (Thigh – Tlow)/Thigh, where Thigh is the temperature of the heat going into the engine and Tlow is that of the heat leaving the engine. (a) What is the maximum possible efficiency of a heat engine operating between an input temperature of 700 K and an exit temperature of 288 K? (b) Why is it important that electrical power plants be located near bodies of relatively cool water? (c)Under what conditions could a heat engine operate at or near 100% efficiency? (d) It is often said that if the energy of combustion of a fuel such as methane were captured in an electrical fuel cell instead of by burning the fuel in a heat engine, a greater fraction of the energy could be put to useful work. Make a qualitative drawing like that in Figure 5.10 that illustrates the fact that in principle the fuel cell route will produce more useful work than the heat engine route from combustion of methane.
INTEGRATIVE EXERCISES
19.108 Most liquids follow Trouton's rule, which states that the molar entropy of vaporization lies in the range of 88 ± 5 J/mol-K. The normal boiling points and enthalpies of vaporization of several organic liquids are as follows:
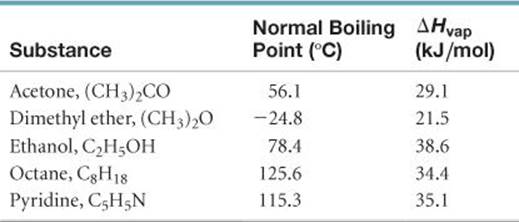
(a) Calculate ΔSvap for each of the liquids. Do all the liquids obey Trouton's rule? (b) With reference to intermolecular forces (Section 11.2), can you explain any exceptions to the rule? (c) Would you expect water to obey Trouton's rule? By using data in Appendix B, check the accuracy of your conclusion. (d) Chlorobenzene (C6H5Cl) boils at 131.8 °C. Use Trouton's rule to estimate ΔHvap for this substance.
19.109 In chemical kinetics the entropy of activation is the entropy change for the process in which the reactants reach the activated complex. The entropy of activation for bimolecular processes is usually negative. Explain this observation with reference to Figure 14.17.
19.110 The following processes were all discussed in Chapter 18, “Chemistry of the Environment.” Estimate whether the entropy of the system increases or decreases during each process: (a) photodissociation of O2(g), (b) formation of ozone from oxygen molecules and oxygen atoms,(c) diffusion of CFCs into the stratosphere, (d) desalination of water by reverse osmosis.
19.111 Carbon disulfide (CS2) is a toxic, highly flammable substance. The following thermodynamic data are available for CS2(l) and CS2(g) at 298 K:

(a) Draw the Lewis structure of the molecule. What do you predict for the bond order of the C — S bonds? (b) Use the VSEPR method to predict the structure of the CS2 molecule. (c) Liquid CS2 burns in O2 with a blue flame, forming CO2(g) and SO2(g). Write a balanced equation for this reaction. (d) Using the data in the preceding table and in Appendix C, calculate ΔH° and ΔG° for the reaction in part (c). Is the reaction exothermic? Is it spontaneous at 298 K? (e) Use the data in the table to calculate ΔS° at 298 K for the vaporization of CS2(l). Is the sign of ΔS° as you would expect for a vaporization? (f) Using data in the table and your answer to part (e), estimate the boiling point of CS2(l). Do you predict that the substance will be a liquid or a gas at 298 K and 1 atm?
[19.112] The following data compare the standard enthalpies and free energies of formation of some crystalline ionic substances and aqueous solutions of the substances:

(a) Write the formation reaction for AgNO3(s). Based on this reaction, do you expect the entropy of the system to increase or decrease upon the formation of AgNO3(s)? (b) Use ![]() and
and ![]() of AgNO3(s) to determine the entropy change upon formation of the substance. Is your answer consistent with your reasoning in part (a)? (c) Is dissolving AgNO3 in water an exothermic or endothermic process? What about dissolving MgSO4 in water? (d) For both AgNO3 and MgSO4, use the data to calculate the entropy change when the solid is dissolved in water. (e)Discuss the results from part (d) with reference to material presented in this chapter and in the “A Closer Look” box on page 540.
of AgNO3(s) to determine the entropy change upon formation of the substance. Is your answer consistent with your reasoning in part (a)? (c) Is dissolving AgNO3 in water an exothermic or endothermic process? What about dissolving MgSO4 in water? (d) For both AgNO3 and MgSO4, use the data to calculate the entropy change when the solid is dissolved in water. (e)Discuss the results from part (d) with reference to material presented in this chapter and in the “A Closer Look” box on page 540.
[19.113] Consider the following equilibrium:
![]()
Thermodynamic data on these gases are given in Appendix C. You may assume that ΔH° and ΔS° do not vary with temperature. (a) At what temperature will an equilibrium mixture contain equal amounts of the two gases? (b) At what temperature will an equilibrium mixture of 1 atm total pressure contain twice as much NO2 as N2O4? (c) At what temperature will an equilibrium mixture of 10 atm total pressure contain twice as much NO2 as N2O4? (d) Rationalize the results from parts (b) and (c) by using Le Châtelier's principle. [Section 15.7]
[19.114] The reaction
![]()
is the basis of a suggested method for removal of SO2 from power-plant stack gases. The standard free energy of each substance is given in Appendix C. (a) What is the equilibrium constant for the reaction at 298 K? (b) In principle, is this reaction a feasible method of removing SO2?(c) If PSO2 = PH2S and the vapor pressure of water is 25 torr, calculate the equilibrium SO2 pressure in the system at 298 K. (d) Would you expect the process to be more or less effective at higher temperatures?
19.115 When most elastomeric polymers (e.g., a rubber band) are stretched, the molecules become more ordered, as illustrated here:

Suppose you stretch a rubber band. (a) Do you expect the entropy of the system to increase or decrease? (b) If the rubber band were stretched isothermally, would heat need to be absorbed or emitted to maintain constant temperature? (c) Try this experiment: Stretch a rubber band and wait a moment. Then place the stretched rubber band on your upper lip, and let it return suddenly to its unstretched state (remember to keep holding on). What do you observe? Are your observations consistent with your answer to part (b)?