CHEMISTRY THE CENTRAL SCIENCE
5 THERMOCHEMISTRY
EXERCISES
VISUALIZING CONCEPTS
5.1 Imagine a book that is falling from a shelf. At a particular moment during its fall, the book has a kinetic energy of 24 J and a potential energy with respect to the floor of 47 J. (a) How does the book's kinetic energy and its potential energy change as it continues to fall? (b) What is its total kinetic energy at the instant just before it strikes the floor? (c) If a heavier book fell from the same shelf, would it have the same kinetic energy when it strikes the floor? [Section 5.1]
5.2 The accompanying photo shows a pipevine swallowtail caterpillar climbing up a twig. (a) As the caterpillar climbs, its potential energy is increasing. What source of energy has been used to effect this change in potential energy? (b) If the caterpillar is the system, can you predict the sign of q as the caterpillar climbs? (c) Does the caterpillar do work in climbing the twig? Explain. (d) Does the amount of work done in climbing a 12-inch section of the twig depend on the speed of the caterpillar's climb? (e) Does the change in potential energy depend on the caterpillar's speed of climb? [Section 5.1]

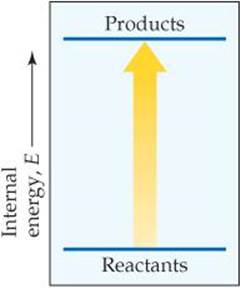
5.3 Consider the accompanying energy diagram. (a) Does this diagram represent an increase or decrease in the internal energy of the system? (b) What sign is given to Δ E for this process? (c) If there is no work associated with the process, is it exothermic or endothermic? [Section 5.2]
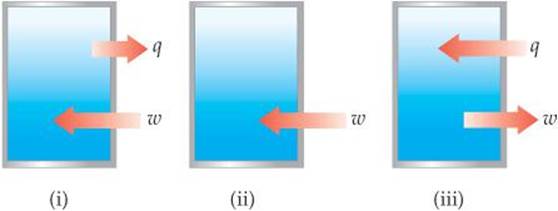
5.4 The contents of the closed box in each of the following illustrations represent a system, and the arrows show the changes to the system during some process. The lengths of the arrows represent the relative magnitudes of q and w. (a) Which of these processes is endothermic? (b) For which of these processes, if any, is Δ E < 0? (c) For which process, if any, does the system experience a net gain in internal energy? [Section 5.2]
5.5 Imagine that you are climbing a mountain. (a) Is the distance you travel to the top a state function? Why or why not? (b) Is the change in elevation between your base camp and the peak a state function? Why or why not? [Section 5.2]
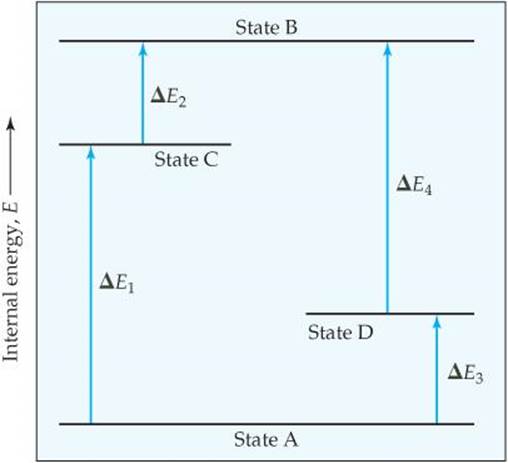
5.6 The diagram shows four states of a system, each with different internal energy, E. (a) Which of the states of the system has the greatest internal energy? (b) In terms of the ΔE values, write two expressions for the difference in internal energy between State A and State B. (c) Write an expression for the difference in energy between State C and State D. (d) Suppose there is another state of the system, State E, and that its energy relative to State A is ΔE = ΔE1 + ΔE4. Where would State E be on the diagram? [Section 5.2]
5.7 You may have noticed that when you compress the air in a bicycle pump, the body of the pump gets warmer. (a) Assuming the pump and the air in it comprise the system, what is the sign of w when you compress the air? (b) What is the sign of q for this process? (c) Based on your answers to parts (a) and (b), can you determine the sign of Δ E for compressing the air in the pump? If not, what would you expect for the sign of Δ E? What is your reasoning? [Section 5.2]
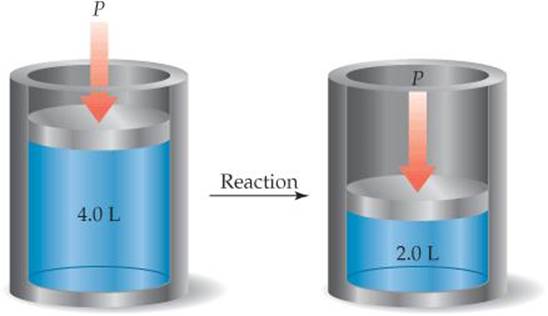
5.8 In the accompanying cylinder diagram a chemical process occurs at constant temperature and pressure. (a) Is the sign of w indicated by this change positive or negative? (b) If the process is endothermic, does the internal energy of the system within the cylinder increase or decrease during the change and is Δ E positive or negative? [Sections 5.2 and 5.3]
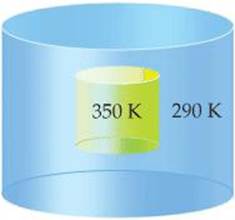
5.9 Imagine a container placed in a tub of water, as depicted in the accompanying diagram. (a) If the contents of the container are the system and heat is able to flow through the container walls, what qualitative changes will occur in the temperatures of the system and in its surroundings? What is the sign of q associated with each change? From the system's perspective, is the process exothermic or endothermic? (b) If neither the volume nor the pressure of the system changes during the process, how is the change in internal energy related to the change in enthalpy? [Sections 5.2 and 5.3]
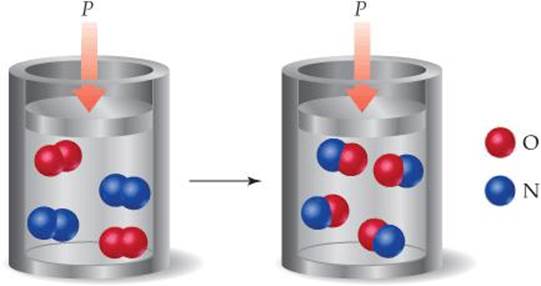
5.10 The gas-phase reaction shown, between N2 and O2, was run in an apparatus designed to maintain a constant pressure. (a) Write a balanced chemical equation for the reaction depicted and predict whether w is positive, negative, or zero. (b) Using data from Appendix C, determine ΔH for the formation of one mole of the product. Why is this enthalpy change called the enthalpy of formation of the involved product? [Sections 5.3 and 5.7]
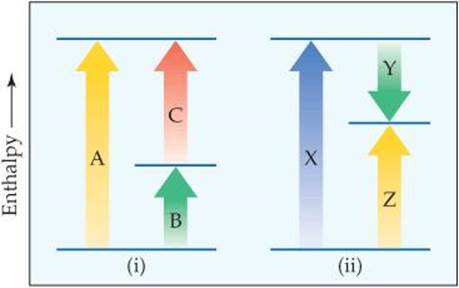
5.11 Consider the two diagrams that follow. (a) Based on (i), write an equation showing how ΔHA is related to ΔHB and ΔHC. How do both diagram (i) and your equation relate to the fact that enthalpy is a state function? (b) Based on (ii), write an equation relating ΔHZ to the other enthalpy changes in the diagram. (c) How do these diagrams relate to Hess's law? [Section 5.6]
5.12 Consider the conversion of compound A into compound B: A → B. For both compounds A and B, ![]() . (a) Sketch an enthalpy diagram for the reaction that is analogous to Figure 5.23. (b) Suppose the overall reaction is exothermic. What can you conclude? [Section 5.7]
. (a) Sketch an enthalpy diagram for the reaction that is analogous to Figure 5.23. (b) Suppose the overall reaction is exothermic. What can you conclude? [Section 5.7]
THE NATURE OF ENERGY (section 5.1)
5.13 In what two ways can an object possess energy? How do these two ways differ from one another?
5.14 Suppose you toss a tennis ball upward. (a) Does the kinetic energy of the ball increase or decrease as it moves higher? (b) What happens to the potential energy of the ball as it moves higher? (c) If the same amount of energy were imparted to a ball the same size as a tennis ball but of twice the mass, how high would it go in comparison to the tennis ball? Explain your answers.
______
5.15 (a) Calculate the kinetic energy in joules of a 1200-kg automobile moving at 18 m/s. (b) Convert this energy to calories. (c) What happens to this energy when the automobile brakes to a stop?
5.16 (a) A baseball weighs 5.13 oz. What is the kinetic energy in joules of this baseball when it is thrown by a major-league pitcher at 95.0 mph? (b) By what factor will the kinetic energy change if the speed of the baseball is decreased to 55.0 mph? (c) What happens to the kinetic energy when the baseball is caught by the catcher? (d) What careful experimental measurement could (in principle) be made to confirm your answer to (c)?
______
5.17 The use of the British thermal unit (Btu) is common in much engineering work. A Btu is the amount of heat required to raise the temperature of 1 lb of water by 1 °F. Calculate the number of joules in a Btu.
5.18 A watt is a measure of power (the rate of energy change) equal to 1 J/s. (a) Calculate the number of joules in a kilowatt-hour. (b) An adult person radiates heat to the surroundings at about the same rate as a 100-watt electric incandescent lightbulb. What is the total amount of energy in kcal radiated to the surroundings by an adult in 24 hours?
______
5.19 (a) What is meant by the term system in thermodynamics? (b) What is a closed system? (c) What do we call the part of the universe that is not part of the system?
5.20 In a thermodynamic study a scientist focuses on the properties of a solution in an apparatus as illustrated. A solution is continuously flowing into the apparatus at the top and out at the bottom, such that the amount of solution in the apparatus is constant with time. (a) Is the solution in the apparatus a closed system, open system, or isolated system? Explain your choice. (b) If it is not a closed system, what could be done to make it a closed system?

______
5.21 (a) What is work? (b) How do we determine the amount of work done, given the force associated with the work?
5.22 (a) What is heat? (b) Under what conditions is heat transferred from one object to another?
______
5.23 Identify the force present and explain whether work is being performed in the following cases: (a) You lift a pencil off the top of a desk. (b) A spring is compressed to half its normal length.
5.24 Identify the force present and explain whether work is done when (a) a positively charged particle moves in a circle at a fixed distance from a negatively charged particle; (b) an iron nail is pulled off a magnet.
THE FIRST LAW OF THERMODYNAMICS (section 5.2)
5.25 (a) State the first law of thermodynamics. (b) What is meant by the internal energy of a system? (c) By what means can the internal energy of a closed system increase?
5.26 (a) Write an equation that expresses the first law of thermodynamics in terms of heat and work. (b) Under what conditions will the quantities q and w be negative numbers?
______
5.27 Calculate Δ E and determine whether the process is endother-mic or exothermic for the following cases: (a) q = 0.763 kJ and w = –840 J; (b) a system releases 66.1 kJ of heat to its surroundings while the surroundings do 44.0 kJ of work on the system; (c) the system absorbs 7.25 kJ of heat from the surroundings while its volume remains constant (assume that only P-V work can be done).
5.28 For the following processes, calculate the change in internal energy of the system and determine whether the process is en-dothermic or exothermic: (a) A balloon is cooled by removing 0.655 kJ of heat. It shrinks on cooling, and the atmosphere does 382 J of work on the balloon.(b) A 100.0-g bar of gold is heated from 25 °C to 50 °C during which it absorbs 322 J of heat. Assume the volume of the gold bar remains constant. (c) The surroundings do 1.44 kJ of work compressing gas in a perfectly insulated cylinder.
______
5.29 A gas is confined to a cylinder fitted with a piston and an electrical heater, as shown here:

Suppose that current is supplied to the heater so that 100 J of energy is added. Consider two different situations. In case (1) the piston is allowed to move as the energy is added. In case (2) the piston is fixed so that it cannot move. (a) In which case does the gas have the higher temperature after addition of the electrical energy? Explain. (b) What can you say about the values of q and w in each case? (c) What can you say about the relative values of Δ E for the system (the gas in the cylinder) in the two cases?
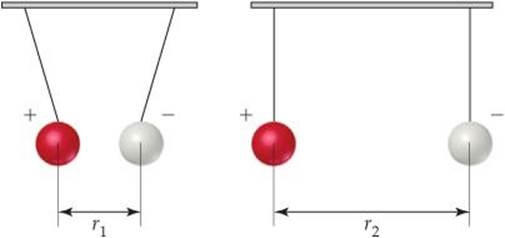
5.30 Consider a system consisting of two oppositely charged spheres hanging by strings and separated by a distance r1, as shown in the accompanying illustration. Suppose they are separated to a larger distance r2, by moving them apart along a track. (a) What change, if any, has occurred in the potential energy of the system? (b) What effect, if any, does this process have on the value of Δ E? (c) What can you say about q and w for this process?
______
5.31 (a) What is meant by the term state function? (b) Give an example of a quantity that is a state function and one that is not. (c) Is the volume of the system a state function? Why or why not?
5.32 Indicate which of the following is independent of the path by which a change occurs: (a) the change in potential energy when a book is transferred from table to shelf, (b) the heat evolved when a cube of sugar is oxidized to CO2(g) and H2O(g), (c) the work accomplished in burning a gallon of gasoline.
ENTHALPY (sections 5.3 and 5.4)
5.33 (a) Why is the change in enthalpy usually easier to measure than the change in internal energy? (b) if is a state function, but q is not a state function. Explain. (c) For a given process at constant pressure, ΔH is positive. Is the process endothermic or exothermic?
5.34 (a) Under what condition will the enthalpy change of a process equal the amount of heat transferred into or out of the system? (b) During a constant-pressure process, the system releases heat to the surroundings. Does the enthalpy of the system increase or decrease during the process? (c) In a constant-pressure process, ΔH = 0. What can you conclude about ΔE, q, and w?
______
5.35 You are given ΔH for a process that occurs at constant pressure. What additional information do you need to determine ΔE for the process?
5.36 Suppose that the gas-phase reaction 2 NO(g) + O2(g) → 2 NO2(g) were carried out in a constant-volume container at constant temperature. Would the measured heat change represent ΔH or ΔE? If there is a difference, which quantity is larger for this reaction? Explain.
______
5.37 A gas is confined to a cylinder under constant atmospheric pressure, as illustrated in Figure 5.4. When the gas undergoes a particular chemical reaction, it absorbs 824 J of heat from its surroundings and has 0.65 kJ of P-V work done on it by its surroundings. What are the values of ΔH and ΔE for this process?
5.38 A gas is confined to a cylinder under constant atmospheric pressure, as illustrated in Figure 5.4. When 0.49 kJ of heat is added to the gas, it expands and does 214 J of work on the surroundings. What are the values of ΔH and ΔE for this process?
______
5.39 The complete combustion of ethanol, C2H5OH(l), to form H2O(g) and CO2(g) at constant pressure releases 1235 kJ of heat per mole of C2H5OH. (a) Write a balanced thermochem-ical equation for this reaction. (b) Draw an enthalpy diagram for the reaction.
5.40 The decomposition of slaked lime, Ca(OH)2(s), into lime, CaO(s), and H2O(g) at constant pressure requires the addition of 109 kJ of heat per mole of Ca(OH)2. (a) Write a balanced thermochemical equation for the reaction. (b) Draw an enthalpy diagram for the reaction.
______
5.41 Ozone, O3(g), is a form of elemental oxygen that is important in the absorption of ultraviolet radiation in the stratosphere. It decomposes to O2(g) at room temperature and pressure according to the following reaction:
2 O3(g) → 3 O2(g) ΔH = –284.6 kJ
(a) What is the enthalpy change for this reaction per mole of O3(g)? (b) Which has the higher enthalpy under these conditions, 2 O3(g) or 3 O2(g)?
5.42 Without referring to tables, predict which of the following has the higher enthalpy in each case: (a) 1 mol CO2(s) or 1 mol CO2(g) at the same temperature, (b) 2 mol of hydrogen atoms or 1 mol of H2, (c) 1 mol H2(g) and 0.5 mol O2(g) at 25 °C or 1 mol H2O(g) at 25 °C, (d) 1 mol N2(g) at 100 °C or 1 mol N2(g) at 300 °C.
______
5.43 Consider the following reaction:
![]()
(a) Is this reaction exothermic or endothermic? (b) Calculate the amount of heat transferred when 3.55 g of Mg(s) reacts at constant pressure. (c) How many grams of MgO are produced during an enthalpy change of –234 kJ? (d) How many kilo-joules of heat are absorbed when 40.3 g of MgO(s) is decomposed into Mg(s) and O2(g) at constant pressure?
5.44 Consider the following reaction:
![]()
(a) Is this reaction exothermic or endothermic? (b) Calculate the amount of heat transferred when 24.0 g of CH3OH(g) is decomposed by this reaction at constant pressure. (c) For a given sample of CH3OH, the enthalpy change during the reaction is 82.1 kJ. How many grams of methane gas are produced? (d) How many kilojoules of heat are released when 38.5 g of CH4(g) reacts completely with O2(g) to form CH3OH(g) at constant pressure?
5.45 When solutions containing silver ions and chloride ions are mixed, silver chloride precipitates:
![]()
(a) Calculate ΔH for production of 0.450 mol of AgCl by this reaction. (b) Calculate ΔH for the production of 9.00 g of AgCl. (c) Calculate ΔH when 9.25 × 10−4 mol of AgCl dissolves in water.
5.46 At one time, a common means of forming small quantities of oxygen gas in the laboratory was to heat KClO3:
![]()
For this reaction, calculate ΔH for the formation of (a) 1.36 mol of O2 and (b) 10.4 g of KCl. (c) The decomposition of KClO3 proceeds spontaneously when it is heated. Do you think that the reverse reaction, the formation of KClO3 from KCl and O2, is likely to be feasible under ordinary conditions? Explain your answer.
______
5.47 Consider the combustion of liquid methanol, CH3OH(l):
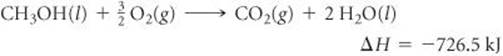
(a) What is the enthalpy change for the reverse reaction? (b) Balance the forward reaction with whole-number coefficients. What is ΔH for the reaction represented by this equation? (c) Which is more likely to be thermodynamically favored, the forward reaction or the reverse reaction? (d) If the reaction were written to produce H2O(g) instead of H2O(l), would you expect the magnitude of ΔH to increase, decrease, or stay the same? Explain.
5.48 Consider the decomposition of liquid benzene, C6H6(l), to gaseous acetylene, C2H2(g):
![]()
(a) What is the enthalpy change for the reverse reaction? (b) What is ΔH for the formation of 1 mol of acetylene? (c) Which is more likely to be thermodynamically favored, the forward reaction or the reverse reaction? (d) If C6H6(g) were consumed instead of C6H6(l), would you expect the magnitude of ΔH to increase, decrease, or stay the same? Explain.
CALORIMETRY (section 5.5)
5.49 (a) What are the units of molar heat capacity? (b) What are the units of specific heat? (c) If you know the specific heat of copper, what additional information do you need to calculate the heat capacity of a particular piece of copper pipe?
5.50 Two solid objects, A and B, are placed in boiling water and allowed to come to temperature there. Each is then lifted out and placed in separate beakers containing 1000 g water at 10.0 °C. Object A increases the water temperature by 3.50 °C; B increases the water temperature by 2.60 °C. (a) Which object has the larger heat capacity? (b) What can you say about the specific heats of A and B?
______
5.51 (a) What is the specific heat of liquid water? (b) What is the molar heat capacity of liquid water? (c) What is the heat capacity of 185 g of liquid water? (d) How many kJ of heat are needed to raise the temperature of 10.00 kg of liquid water from 24.6 °C to 46.2 °C?
5.52 (a) Which substance in Table 5.2 requires the smallest amount of energy to increase the temperature of 50.0 g of that substance by 10 K? (b) Calculate the energy needed for this temperature change.
______
5.53 The specific heat of octane, C8H18(l), is 2.22 J/g-K. (a) How many J of heat are needed to raise the temperature of 80.0 g of octane from 10.0 °C to 25.0 °C? (b) Which will require more heat, increasing the temperature of 1 mol of C8H18(l) by a certain amount or increasing the temperature of 1 mol of H2O(l) by the same amount?
5.54 Consider the data about gold metal in Exercise 5.28(b). (a) Based on the data, calculate the specific heat of Au(s). (b) Suppose that the same amount of heat is added to two 10.0-g blocks of metal, both initially at the same temperature. One block is gold metal and one is iron metal. Which block will have the greater rise in temperature after the addition of the heat? (c) What is the molar heat capacity of Au(s)?
______
5.55 When a 6.50-g sample of solid sodium hydroxide dissolves in 100.0 g of water in a coffee-cup calorimeter (Figure 5.18), the temperature rises from 21.6 °C to 37.8 °C. Calculate ΔH (in kJ/mol NaOH) for the solution process
NaOH(s)→ Na+(aq) + OH−(aq)
Assume that the specific heat of the solution is the same as that of pure water.
5.56 (a) When a 4.25-g sample of solid ammonium nitrate dis solves in 60.0 g of water in a coffee-cup calorimeter (Figure 5.18), the temperature drops from 22.0 °C to 16.9 °C. Calculate ΔH (in kJ/mol NH4NO3) for the solution process
NH4NO3(s) → NH4+(aq) + NO3−(aq)
Assume that the specific heat of the solution is the same as that of pure water. (b) Is this process endothermic or exothermic?
______
5.57 A 2.200-g sample of quinone (C6H4O2) is burned in a bomb calorimeter whose total heat capacity is 7.854 kJ/°C. The temperature of the calorimeter increases from 23.44 °C to 30.57 °C. What is the heat of combustion per gram of quinone? Per mole of quinone?
5.58 A 1.800-g sample of phenol (C6H5OH) was burned in a bomb calorimeter whose total heat capacity is 11.66 kJ/°C. The temperature of the calorimeter plus contents increased from 21.36 °C to 26.37 °C. (a) Write a balanced chemical equation for the bomb calorimeter reaction. (b)What is the heat of combustion per gram of phenol? Per mole of phenol?
______
5.59 Under constant-volume conditions, the heat of combustion of glucose (C6H12O6) is 15.57 kJ/g. A 3.500-g sample of glucose is burned in a bomb calorimeter. The temperature of the calorimeter increased from 20.94 °C to 24.72 °C. (a) What is the total heat capacity of the calorimeter? (b) If the size of the glucose sample had been exactly twice as large, what would the temperature change of the calorimeter have been?
5.60 Under constant-volume conditions, the heat of combustion of benzoic acid (C6H5COOH) is 26.38 kJ/g. A 2.760-g sample of benzoic acid is burned in a bomb calorimeter. The temperature of the calorimeter increases from 21.60 °C to 29.93 °C. (a) What is the total heat capacity of the calorimeter? (b) A 1.440-g sample of a new organic substance is combusted in the same calorimeter. The temperature of the calorimeter increases from 22.14 °C to 27.09 °C. What is the heat of combustion per gram of the new substance? (c) Suppose that in changing samples, a portion of the water in the calorimeter were lost. In what way, if any, would this change the heat capacity of the calorimeter?
HESS'S LAW (section 5.6)
5.61 What is the connection between Hess's law and the fact that H is a state function?
5.62 Consider the following hypothetical reactions:
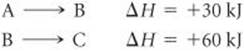
(a) Use Hess's law to calculate the enthalpy change for the reaction A → C. (b) Construct an enthalpy diagram for substances A, B, and C, and show how Hess's law applies.
______
5.63 Calculate the enthalpy change for the reaction
P4O6(s) + 2 O2(g)→P4O10(s)
given the following enthalpies of reaction:
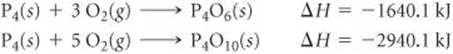
5.64 From the enthalpies of reaction
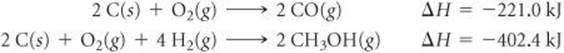
Calculate ΔH for the reaction
CO(g) + 2H2(g) → CH3OH(g)
______
5.65 From the enthalpies of reaction
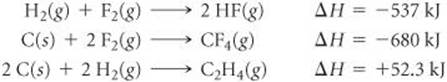
Calculate ΔH for the reaction of ethylene with F2
![]()
5.66 Given the data
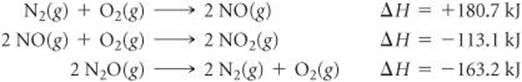
use Hess's law to calculate ΔH for the reaction
N2O(g) + NO2(g)→3 NO(g)
ENTHALPIES OF FORMATION (section 5.7)
5.67 (a) What is meant by the term standard conditions with reference to enthalpy changes? (b) What is meant by the term enthalpy of formation? (c) What is meant by the term standard enthalpy of formation?
5.68 (a) Why are tables of standard enthalpies of formation so useful? (b) What is the value of the standard enthalpy of formation of an element in its most stable form? (c) Write the chemical equation for the reaction whose enthalpy change is the standard enthalpy of formation of sucrose (table sugar), C12H22O11(s), ΔH°f[C12H22O11].
______
5.69 For each of the following compounds, write a balanced ther-mochemical equation depicting the formation of one mole of the compound from its elements in their standard states and use Appendix C to obtain the value of ![]() : (a) NO2(g), (b) SO3(g), (c) NaBr(s), (d) Pb(NO3)2(s).
: (a) NO2(g), (b) SO3(g), (c) NaBr(s), (d) Pb(NO3)2(s).
5.70 Write balanced equations that describe the formation of the following compounds from elements in their standard states, and use Appendix C to obtain the values of their standard enthalpies of formation: (a) H2O2(g), (b) CaCO3(s), (c) POCl3(l), (d) C2H5OH(l).
______
5.71 The following is known as the thermite reaction [Figure 5.8(b)]:
2 Al(s) + Fe2O3(s) → Al2O3(s) + 2 Fe(s)
This highly exothermic reaction is used for welding massive units, such as propellers for large ships. Using standard enthalpies of formation in Appendix C, calculate ΔH° for this reaction.
5.72 Many portable gas heaters and grills use propane, C3H8(g), as a fuel. Using standard enthalpies of formation, calculate the quantity of heat produced when 10.0 g of propane is completely combusted in air under standard conditions.
______
5.73 Using values from Appendix C, calculate the standard en thalpy change for each of the following reactions:
(a)![]()
(b)![]()
(c)![]()
(d)![]()
5.74 Using values from Appendix C, calculate the value of ΔH° for each of the following reactions:
(a)![]()
(b)![]()
(c)![]()
(d)![]()
______
5.75 Complete combustion of 1 mol of acetone (C3H6O) liberates 1790 kJ:
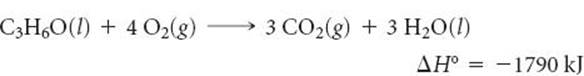
Using this information together with data from Appendix C, calculate the enthalpy of formation of acetone.
5.76 Calcium carbide (CaC2) reacts with water to form acetylene (C2H2) and Ca(OH)2. From the following enthalpy of reaction data and data in Appendix C, calculate Δ H°f for CaC2(s):
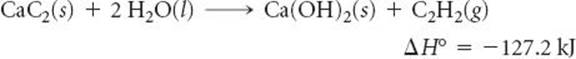
______
5.77 Gasoline is composed primarily of hydrocarbons, including many with eight carbon atoms, called octanes. One of the cleanest-burning octanes is a compound called 2, 3, 4-trimethylpentane, which has the following structural formula:
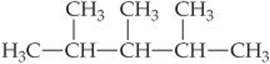
The complete combustion of one mole of this compound to CO2(g) and H2O(g) leads to ΔH° = −5064.9 kJ/mol. (a) Write a balanced equation for the combustion of 1 mol of C8H18(l). (b) Write a balanced equation for the formation of C8H18(l) from its elements. (c) By using the information in this problem and data in Table 5.3, calculate ![]() for 2, 3, 4-trimethylpentane.
for 2, 3, 4-trimethylpentane.
5.78 Diethyl ether, C4H10O(l), a flammable compound that has long been used as a surgical anesthetic, has the structure
![]()
The complete combustion of 1 mol of C4H10O(l) to CO2(g) and H2O(l) yields ΔH° = −2723.7 kJ. (a) Write a balanced equation for the combustion of 1 mol of C4H10O(l). (b) Wr i te a balanced equation for the formation of C4H10O(l) from its elements. (c) By using the information in this problem and data in Table 5.3, calculate ![]() for diethyl ether.
for diethyl ether.
______
5.79 Ethanol (C2H5OH) is currently blended with gasoline as an automobile fuel. (a) Write a balanced equation for the combustion of liquid ethanol in air. (b) Calculate the standard enthalpy change for the reaction, assuming H2O(g) as a product. (c) Calculate the heat produced per liter of ethanol by combustion of ethanol under constant pressure. Ethanol has a density of 0.789 g/mL. (d) Calculate the mass of CO2 produced per kJ of heat emitted.
5.80 Methanol (CH3OH) is used as a fuel in race cars. (a) Write a balanced equation for the combustion of liquid methanol in air. (b) Calculate the standard enthalpy change for the reaction, assuming H2O(g) as a product. (c) Calculate the heat produced by combustion per liter of methanol. Methanol has a density of 0.791 g/mL. (d) Calculate the mass of CO2 produced per kJ of heat emitted.
FOODS AND FUELS (section 5.8)
5.81 (a) What is meant by the term fuel value? (b) Which is a greater source of energy as food, 5 g of fat or 9 g of carbohydrate? (c) The metabolism of glucose produces CO2(g) and H2O(l). How does the human body expel these reaction products?
5.82 (a) Why are fats well suited for energy storage in the human body? (b) A particular chip snack food is composed of 12% protein, 14% fat, and the rest carbohydrate. What percentage of the calorie content of this food is fat? (c) How many grams of protein provide the same fuel value as 25 g of fat?
______
5.83 (a) A serving of a particular ready-to-serve chicken noodle soup contains 2.5 g fat, 14 g carbohydrate, and 7 g protein. Estimate the number of Calories in a serving. (b) According to its nutrition label, the same soup also contains 690 mg of sodium. Do you think the sodium contributes to the caloric content of the soup?
5.84 A pound of plain M & M® candies contains 96 g fat, 320 g carbohydrate, and 21 g protein. What is the fuel value in kJ in a 42-g (about 1.5 oz) serving? How many Calories does it provide?
______
5.85 The heat of combustion of fructose, C6H12O6, is –2812 kJ/mol. If a fresh golden delicious apple weighing 4.23 oz (120 g) contains 16.0 g of fructose, what caloric content does the fructose contribute to the apple?
5.86 The heat of combustion of ethanol, C2H5OH(l), is –1367 kJ/mol. A batch of Sauvignon Blanc wine contains 10.6% ethanol by mass. Assuming the density of the wine to be 1.0 g/mL, what is the caloric content due to the alcohol (ethanol) in a 6-oz glass of wine (177 mL)?
______
5.87 The standard enthalpies of formation of gaseous propyne (C3H4), propylene (C3H6), and propane (C3H8) are +185.4, +20.4, and −103.8 kJ/mol, respectively. (a) Calculate the heat evolved per mole on combustion of each substance to yield CO2(g) and H2O(g). (b) Calculate the heat evolved on combustion of 1 kg of each substance. (c) Which is the most efficient fuel in terms of heat evolved per unit mass?
5.88 It is interesting to compare the “fuel value” of a hydrocarbon in a world where fluorine rather than oxygen is the combustion agent. The enthalpy of formation of CF4(g) is –679.9 kJ/mol. Which of the following two reactions is the more exothermic?
![]()
______
[5.89] At the end of 2009, global population was about 6.8 billion people. What mass of glucose in kg would be needed to provide 1500 Cal/person/day of nourishment to the global population for one year? Assume that glucose is metabolized entirely to CO2(g) and H2O(l) according to the following ther-mochemical equation:
![]()
[5.90] The automobile fuel called E85 consists of 85% ethanol and 15% gasoline. E85 can be used in so-called “flex-fuel” vehicles (FFVs), which can use gasoline, ethanol, or a mix as fuels. Assume that gasoline consists of a mixture of octanes (different isomers of C8H18), that the average heat of combustion of C8H18(l) is 5400 kJ/mol, and that gasoline has an average density of 0.70 g/mL. The density of ethanol is 0.79 g/mL. (a) By using the information given as well as data in Appendix C, compare the energy produced by combustion of 1.0 L of gasoline and of 1.0 L of ethanol. (b) Assume that the density and heat of combustion of E85 can be obtained by using 85% of the values for ethanol and 15% of the values for gasoline. How much energy could be released by the combustion of 1.0 L of E85? (c) How many gallons of E85 would be needed to provide the same energy as 10 gal of gasoline? (d) If gasoline costs $3.10 per gallon in the United States, what is the break-even price per gallon of E85 if the same amount of energy is to be delivered?
ADDITIONAL EXERCISES
5.91 At 20 °C (approximately room temperature) the average velocity of N2 molecules in air is 1050 mph. (a) What is the average speed in m/s? (b) What is the kinetic energy (in J) of an N2 molecule moving at this speed? (c) What is the total kinetic energy of 1 mol of N2 molecules moving at this speed?
5.92 Suppose an Olympic diver who weighs 52.0 kg executes a straight dive from a 10-m platform. At the apex of the dive, the diver is 10.8 m above the surface of the water. (a) What is the potential energy of the diver at the apex of the dive, relative to the surface of the water? (b)Assuming that all the potential energy of the diver is converted into kinetic energy at the surface of the water, at what speed in m/s will the diver enter the water? (c) Does the diver do work on entering the water? Explain.
5.93 The air bags that provide protection in autos in the event of an accident expand because of a rapid chemical reaction. From the viewpoint of the chemical reactants as the system, what do you expect for the signs of q and w in this process?
[5.94] An aluminum can of a soft drink is placed in a freezer. Later, you find that the can is split open and its contents frozen. Work was done on the can in splitting it open. Where did the energy for this work come from?
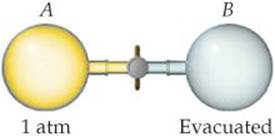
[5.95] Consider a system consisting of the following apparatus, in which gas is confined in one flask and there is a vacuum in the other flask. The flasks are separated by a valve that, when opened, connects the two flasks. Assume that the flasks are perfectly insulated and will not allow the flow of heat into or out of the flasks to the surroundings. (a) When the valve is opened, gas flows from the filled flask to the evacuated one. Does that surprise you? (b) Is work performed during the expansion of the gas? Why or why not? (c) Can you determine the value of ΔE for the process? Does the answer surprise you? (We will talk about this system more in Chapter 19.)
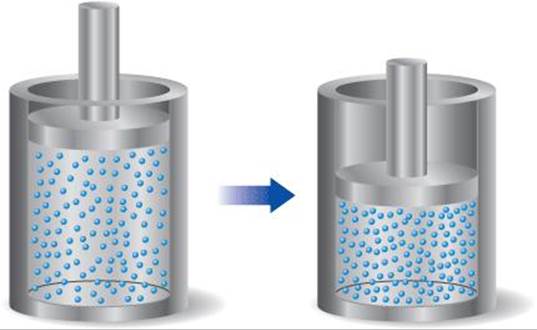
[5.96] A sample of gas is contained in a cylinder-and-piston arrangement. It undergoes the change in state shown in the drawing. (a) Assume first that the cylinder and piston are perfect thermal insulators that do not allow heat to be transferred. What is the value of q for the state change? What is the sign of w for the state change? What can be said about Δ E for the state change? (b) Now assume that the cylinder and piston are made up of a thermal conductor such as a metal. During the state change, the cylinder gets warmer to the touch. What is the sign of q for the state change in this case? Describe the difference in the state of the system at the end of the process in the two cases. What can you say about the relative values of Δ E?
[5.97] Limestone stalactites and stalagmites are formed in caves by the following reaction:
![]()
If 1 mol of CaCO3 forms at 298 K under 1 atm pressure, the reaction performs 2.47 kJ of P-V work, pushing back the atmosphere as the gaseous CO2 forms. At the same time, 38.95 kJ of heat is absorbed from the environment. What are the values of ΔH and of Δ E for this reaction?
[5.98] Consider the systems shown in Figure 5.10. In one case the battery becomes completely discharged by running the current through a heater and in the other case by running a fan. Both processes occur at constant pressure. In both cases the change in state of the system is the same: The battery goes from being fully charged to being fully discharged. Yet in one case the heat evolved is large, and in the other it is small. Is the enthalpy change the same in the two cases? If not, how can enthalpy be considered a state function? If it is, what can you say about the relationship between enthalpy change and q in this case, as compared with others that we have considered?
5.99 A house is designed to have passive solar energy features. Brickwork incorporated into the interior of the house acts as a heat absorber. Each brick weighs approximately 1.8 kg. The specific heat of the brick is 0.85 J/g-K. How many bricks must be incorporated into the interior of the house to provide the same total heat capacity as 1.7 × 103 gal of water?
[5.100] A coffee-cup calorimeter of the type shown in Figure 5.18 contains 150.0 g of water at 25.1 °C. A 121.0-g block of copper metal is heated to 100.4 °C by putting it in a beaker of boiling water. The specific heat of Cu(s) is 0.385 J/g-K. The Cu is added to the calorimeter, and after a time the contents of the cup reach a constant temperature of 30.1 °C. (a) Determine the amount of heat, in J, lost by the copper block. (b) Determine the amount of heat gained by the water. The specific heat of water is 4.18 J/g-K. (c) The difference between your answers for (a) and (b) is due to heat loss through the Styrofoam® cups and the heat necessary to raise the temperature of the inner wall of the apparatus. The heat capacity of the calorimeter is the amount of heat necessary to raise the temperature of the apparatus (the cups and the stopper) by 1 K. Calculate the heat capacity of the calorimeter in J/K. (d) What would be the final temperature of the system if all the heat lost by the copper block were absorbed by the water in the calorimeter?
[5.101] (a) When a 0.235-g sample of benzoic acid is combusted in a bomb calorimeter (Figure 5.19), the temperature rises 1.642 °C. When a 0.265-g sample of caffeine, C8H10O2N4, is burned, the temperature rises 1.525 °C. Using the value 26.38 kJ/g for the heat of combustion of benzoic acid, calculate the heat of combustion per mole of caffeine at constant volume. (b) Assuming that there is an uncertainty of 0.002 °C in each temperature reading and that the masses of samples are measured to 0.001 g, what is the estimated uncertainty in the value calculated for the heat of combustion per mole of caffeine?
5.102 Meals-ready-to-eat (MREs) are military meals that can be heated on a flameless heater. The heat is produced by the following reaction: Mg(s) + 2 H2O(l) → Mg(OH)2(s) + H2(g). (a) Calculate the standard enthalpy change for this reaction. (b) Calculate the number of grams of Mg needed for this reaction to release enough energy to increase the temperature of 75 mL of water from 21 °C to 79 °C.
5.103 Burning methane in oxygen can produce three different carbon-containing products: soot (very fine particles of graphite), CO(g), and CO2(g). (a) Write three balanced equations for the reaction of methane gas with oxygen to produce these three products. In each case assume that H2O(l) is the only other product. (b) Determine the standard enthalpies for the reactions in part (a). (c) Why, when the oxygen supply is adequate, is CO2(g) the predominant carbon-containing product of the combustion of methane?
5.104 (a) Calculate the standard enthalpy of formation of gaseous di-borane (B2H6) using the following thermochemical information:
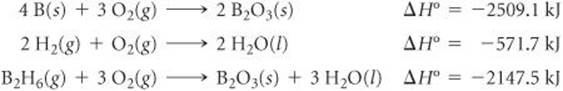
(b) Pentaborane (B5H9) is another boron hydride. What experiment or experiments would you need to perform to yield the data necessary to calculate the heat of formation of B5H9(l)? Explain by writing out and summing any applicable chemical reactions.
5.105 From the following data for three prospective fuels, calculate which could provide the most energy per unit volume:
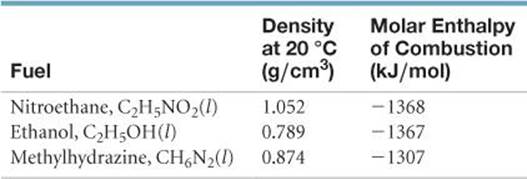
5.106 The hydrocarbons acetylene (C2H2) and benzene (C6H6) have the same empirical formula. Benzene is an “aromatic” hydrocarbon, one that is unusually stable because of its structure. (a) By using the data in Appendix C, determine the standard enthalpy change for the reaction 3 C2H2(g) → C6H6(l). (b) Which has greater enthalpy, 3 mol of acetylene gas or 1 mol of liquid benzene? (c) Determine the fuel value in kJ/g for acetylene and benzene.
[5.107] Ammonia (NH3) boils at –33 °C; at this temperature it has a density of 0.81 g/cm3. The enthalpy of formation of NH3(g) is –46.2 kJ/mol, and the enthalpy of vaporization of NH3(l) is 23.2 kJ/mol. Calculate the enthalpy change when 1 L of liquid NH3 is burned in air to give N2(g) and H2O(g). How does this compare with ΔH for the complete combustion of 1 L of liquid methanol, CH3OH(l)? For CH3OH(l), the density at 25 °C is 0.792 g/cm3, and ![]() .
.
[5.108] Three common hydrocarbons that contain four carbons are listed here, along with their standard enthalpies of formation:

(a) For each of these substances, calculate the molar enthalpy of combustion to CO2(g) and H2O(l). (b) Calculate the fuel value in kJ/g for each of these compounds. (c) For each hydrocarbon, determine the percentage of hydrogen by mass. (d) By comparing your answers for parts (b) and (c), propose a relationship between hydrogen content and fuel value in hydrocarbons.
5.109 A 200-lb man decides to add to his exercise routine by walking up three flights of stairs (45 ft) 20 times per day. He figures that the work required to increase his potential energy in this way will permit him to eat an extra order of French fries, at 245 Cal, without adding to his weight. Is he correct in this assumption?
5.110 The Sun supplies about 1.0 kilowatt of energy for each square meter of surface area (1.0 kW/m2, where a watt = 1 J/s). Plants produce the equivalent of about 0.20 g of sucrose (C12H22O11) per hour per square meter. Assuming that the sucrose is produced as follows, calculate the percentage of sunlight used to produce sucrose.
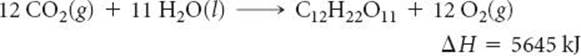
[5.111] It is estimated that the net amount of carbon dioxide fixed by photosynthesis on the landmass of Earth is 5.5 × 10 g/yrof CO2. Assume that all this carbon is converted into glucose. (a) Calculate the energy stored by photosynthesis on land per year in kJ. (b) Calculate the average rate of conversion of solar energy into plant energy in MW (1W = 1 J/s). A large nuclear power plant produces about 103 MW. The energy of how many such nuclear power plants is equivalent to the solar energy conversion?
INTEGRATIVE EXERCISES
5.112 Consider the combustion of a single molecule of CH4(g) forming H2O(l) as a product. (a) How much energy, in J, is produced during this reaction? (b) A typical X-ray light source has an energy of 8 keV. How does the energy of combustion compare to the energy of the X-ray?
5.113 Consider the following unbalanced oxidation-reduction reactions in aqueous solution:

(a) Balance each of the reactions. (b) By using data in Appendix C, calculate ΔH° for each of the reactions. (c) Based on the values you obtain for ΔH°, which of the reactions would you expect to be thermodynamically favored? (d) Use the activity series to predict which of these reactions should occur.![]() (Section 4.4) Are these results in accord with your conclusion in part (c) of this problem?
(Section 4.4) Are these results in accord with your conclusion in part (c) of this problem?
[5.114] Consider the following acid-neutralization reactions involving the strong base NaOH(aq):
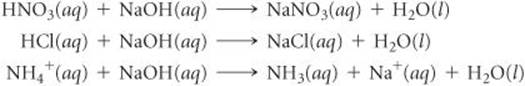
(a) By using data in Appendix C, calculate ΔH° for each of the reactions. (b) As we saw in Section 4.3, nitric acid and hydrochloric acid are strong acids. Write net ionic equations for the neutralization of these acids. (c) Compare the values of ΔH° for the first two reactions. What can you conclude? (d) In the third equation NH4+(aq) is acting as an acid. Based on the value of ΔH° for this reaction, do you think it is a strong or a weak acid? Explain.
5.115 Consider two solutions, the first being 50.0 mL of 1.00 M CuSO4 and the second 50.0 mL of 2.00 M KOH. When the two solutions are mixed in a constant-pressure calorimeter, a precipitate forms and the temperature of the mixture rises from 21.5 °C to 27.7 °C. (a) Before mixing, how many grams of Cu are present in the solution of CuSO4? (b) Predict the identity of the precipitate in the reaction. (c) Write complete and net ionic equations for the reaction that occurs when the two solutions are mixed. (d) From the calorimetric data, calculate ΔHfor the reaction that occurs on mixing. Assume that the calorimeter absorbs only a negligible quantity of heat, that the total volume of the solution is 100.0 mL, and that the specific heat and density of the solution after mixing are the same as that of pure water.
5.116 The precipitation reaction between AgNO3(aq) and NaCl(aq) proceeds as follows:
AgNO3(aq) + NaCl(aq) → NaNO3(aq) + AgCl(s)
(a) By using Appendix C, calculate ΔH° for the net ionic equation of this reaction. (b) What would you expect for the value of ΔH° of the overall molecular equation compared to that for the net ionic equation? Explain. (c) Use the results from (a) and (b) along with data in Appendix C to determine the value of ![]() for AgNO3(aq).
for AgNO3(aq).
[5.117] A sample of a hydrocarbon is combusted completely in O2(g) to produce 21.83 g CO2(g), 4.47 g H2O(g), and 311 kJ of heat. (a) What is the mass of the hydrocarbon sample that was combusted? (b) What is the empirical formula of the hydrocarbon? (c) Calculate the value of ![]() per empirical-formula unit of the hydrocarbon. (d) Do you think that the hydrocarbon is one of those listed in Appendix C? Explain your answer.
per empirical-formula unit of the hydrocarbon. (d) Do you think that the hydrocarbon is one of those listed in Appendix C? Explain your answer.
5.118 The methane molecule, CH4, has the geometry shown in Figure 2.19. Imagine a hypothetical process in which the methane molecule is “expanded,” by simultaneously extending all four C—H bonds to infinity. We then have the process
CH4(g) → C(g) + 4 H(g)
(a) Compare this process with the reverse of the reaction that represents the standard enthalpy of formation of CH4(g). (b) Calculate the enthalpy change in each case. Which is the more endothermic process? What accounts for the difference in ΔH° values? (c) Suppose that 3.45 g CH4(g) reacts with 1.22 g F2(g), forming CF4(g) and HF(g) as sole products. What is the limiting reagent in this reaction? If the reaction occurs at constant pressure, what amount of heat is evolved?
5.119 World energy supplies are often measured in the unit of quadrillion British thermal units (1012 Btu), generally called a “quad.” In 2015, world energy consumption is projected to be 5.81 × 1017 kJ. (a) With reference to Exercise 5.17, how many quads of energy does this quantity represent? (b) Current annual energy consumption in the United States is 99.5 quads. Assume that all this energy is to be generated by burning CH4(g) in the form of natural gas. If the combustion of the CH4(g) were complete and 100% efficient, how many moles of CH4(g) would need to be combusted in order to provide the U.S. energy demand? (c) How many kilograms of CO2(g) would be generated in the combustion in part (b)? (d) Compare your answer to part (c) with information given in Exercise 5.111. Do you think that photosynthesis is an adequate means to maintain a stable level of CO2 in the atmosphere?