The Handy Chemistry Answer Book (2014)
ATOMS AND MOLECULES
PROPERTIES OF ATOMS AND ELECTRONS IN ATOMS
How large is an atom relative to things we can see?
The smallest object a human eye can see is approximately 0.1 mm, or 10−4 m. Atoms have sizes on the order of 10−10 m, or approximately one million times smaller than something the human eye can possibly see.
Is it possible to split an atom?
It is possible to split an atom. When people refer to splitting an atom, it’s the nucleus of the atom that is being split. One process that splits the nucleus of an atom is called fission, which can happen spontaneously in heavier elements. Spontaneous fission basically involves a nucleus emitting a particle containing one or more protons or neutrons. One of the most commonly emitted particles is called an alpha particle, which consists of two neutrons and two protons. Whenever the number of protons in a nucleus changes, it becomes a different element.
Nuclei can also be split intentionally in laboratories. The nucleus is held together very tightly, so it usually takes a high-energy particle colliding with an atom to break it apart. Typically a high-energy neutron is used to initiate the process of splitting a nucleus. This process results in an overall release of energy so that once one nucleus is split, its products can cause the reaction to happen again. This is called a chain reaction, and it can be used to produce energy in a nuclear reactor (if it happens somewhat slowly), or an explosion (if it happens quickly).
Can elements be converted into one another?
It is possible for atoms of one element to become atoms of another element. One way this can happen is any fission process that results in the loss of one or more protons from a nucleus. The joining of two nuclei to form a single, heavier nucleus is also possible, and this process is known as fusion. Both fission and fusion can result in the creation of new atoms with different numbers of protons than were present before the reaction. These processes are often difficult to control in a laboratory, however, so it’s mostly only in specific cases, such as energy production, that chemists and scientists in related fields devote a lot of time to these nuclear reactions.
What is an atomic orbital?
Atomic orbitals are mathematical or pictorial descriptions of the locations of electrons in an atom. Electrons are tricky particles to understand because their location isn’t easy to define. They can be thought of as clouds of negative charge surrounding a nucleus, and atomic orbitals describe the shapes of these clouds. Atomic orbitals can take on different shapes and sizes, but are essentially very similar from one element to the next. The number and type of orbitals that contain electrons play a central role in determining the properties of that atom.
How many electrons can fit in each orbital?
Each atomic orbital can contain up to two electrons. Electrons have a property called spin angular momentum, which can take on two different values of opposite sign. It turns out that electrons residing in the same atomic orbital must have opposite spin angular momenta. This is a consequence of a physical principle known as the Pauli Exclusion Principle.
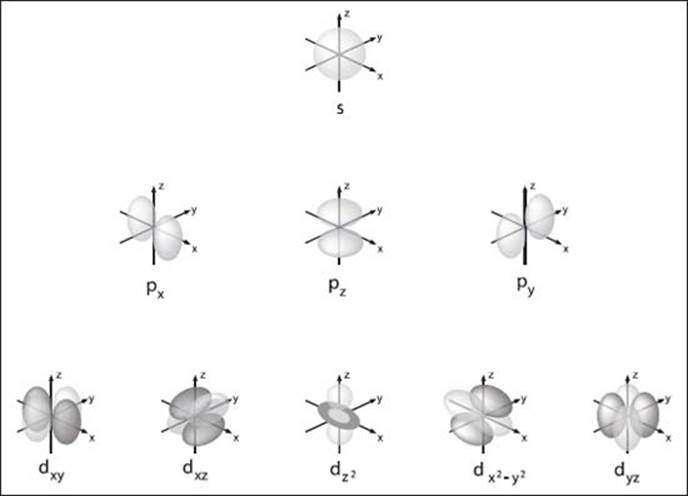
The four main types of atomic orbitals (s, p, and d) and their variants.
What do atomic orbitals look like?
There are three main shapes of orbitals relevant to most of chemistry, and these are referred to as s, p, and d orbitals. The designations s, p, and d are abbreviations for sharp, principle, diffuse, and fundamental, which have historical significance describing results of early experiments to probe the electronic structure of atoms. You can see what these orbitals look like in the graphic on the preceding page.
The shapes of these orbitals are determined by their orbital angular momentum, which is a property that describes the motion of the electron around the nucleus.
What is the valence shell of electrons?
Electrons fill up orbitals in “shells.” The innermost shell consists of just one s-type orbital and can hold just two electrons. The next shell consists of one s-type and three p-type orbitals, and can hold eight electrons. Higher shells consist of more and more orbitals and can thus hold more and more electrons. The valence shell of electrons is the highest occupied, or partially occupied, set of orbitals.
What is the atomic radius of an atom?
The atomic radius of an atom is defined as half of the distance between two atoms of the same element held together in a chemical bond. Not surprisingly, these are very small distances! For hydrogen, the smallest atom, the atomic radius is 0.37 Ångströms, or 3.7 × 10−11 meters.
How do the atomic radii of atoms change across the table?
The atomic radii of atoms generally decrease going from left to right across a period, and increase going top to bottom down a group (see graphic on next page).
The increase in atomic radius going down a group is fairly straightforward to understand: additional shells of electrons are added and they must surround the inner shells, resulting in an increased atomic radius. Though the number of protons in the nucleus increases going down a group, the inner shells of electrons serve to shield the valence shell from the attractive force of the nucleus, resulting in an overall increase in atomic radius.
Moving to the right across a period, the number of protons increases, increasing the attractive force on electrons in the valence shell. Within a period, additional electrons go into the same valence shell, and an increasing attractive pull from the nucleus results in a more contracted valence shell, resulting in a smaller atomic radius. The situation is complicated by the rightmost group (known as the Noble gases), but the atomic radius of these elements is typically not important as they are rarely involved in chemical bonds to other atoms.
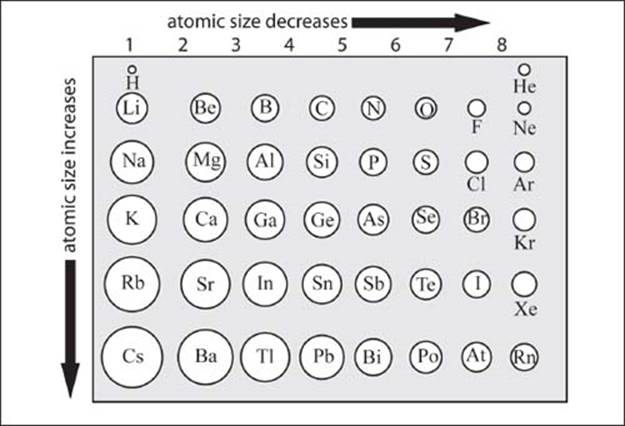
The atomic radii of atoms generally decrease going from left to right across a period, and increase going top to bottom down a group. (Atomic sizes are not to precise ratios and are for illustrative purposes only.)
What is the ionization energy of an atom?
The ionization energy of an atom is the amount of energy it takes to remove an electron from the atom. The process of removing an electron leaves the atom with an extra proton, relative to the number of electrons, and thus creates a positively charged ion, known as a cation. The ionization energy can be thought of as a measure of how strongly an atom holds on to its electrons. In general, ionization energies increase from left to right across a period (though there are exceptions) due to an increasing number of protons to attract electrons in the valence shell. Ionization energies decrease going down a group in the periodic table, due to the valence electrons being farther from the nucleus, and thus more shielded from its positive charge. Note that the trends in atomic radii and ionization energy go in the same direction—larger atoms tend to have lower ionization energies.
What keeps an electron from crashing into the nucleus?
Opposites attract, so electrons and protons are attracted to each other, making it somewhat difficult to understand why an electron wouldn’t just get as close as possible to the nucleus and crash into it. The key to answering this question has to do with the fact that electrons are very, very small particles, so they are governed by rules that don’t apply to larger objects. As we’ve talked about a little already, electrons are best thought of as clouds of negative charge surrounding the nucleus. Their properties are governed by rules that describe the cloud as a whole, rather than as a single particle. It turns out that there is something favorable about the electron being spread out, or delocalized, around the nucleus. For reasons we won’t go into in detail, when the electron’s cloud gets packed closer to the nucleus, the energy associated with its motion (its kinetic energy) begins to rise, which makes the situation unstable. There’s a balance between the stability associated with placing the electron close to the nucleus (the favorable positive-negative charge attraction) and that associated with spreading out the electron’s cloud (to keep its kinetic energy low). This prevents the electron’s cloud from getting too close to the nucleus or the electron just crashing into the nucleus.